Cardiac Troponin T Release after Football 7 in Healthy Children and Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Procedures
2.3. Statistical Analysis
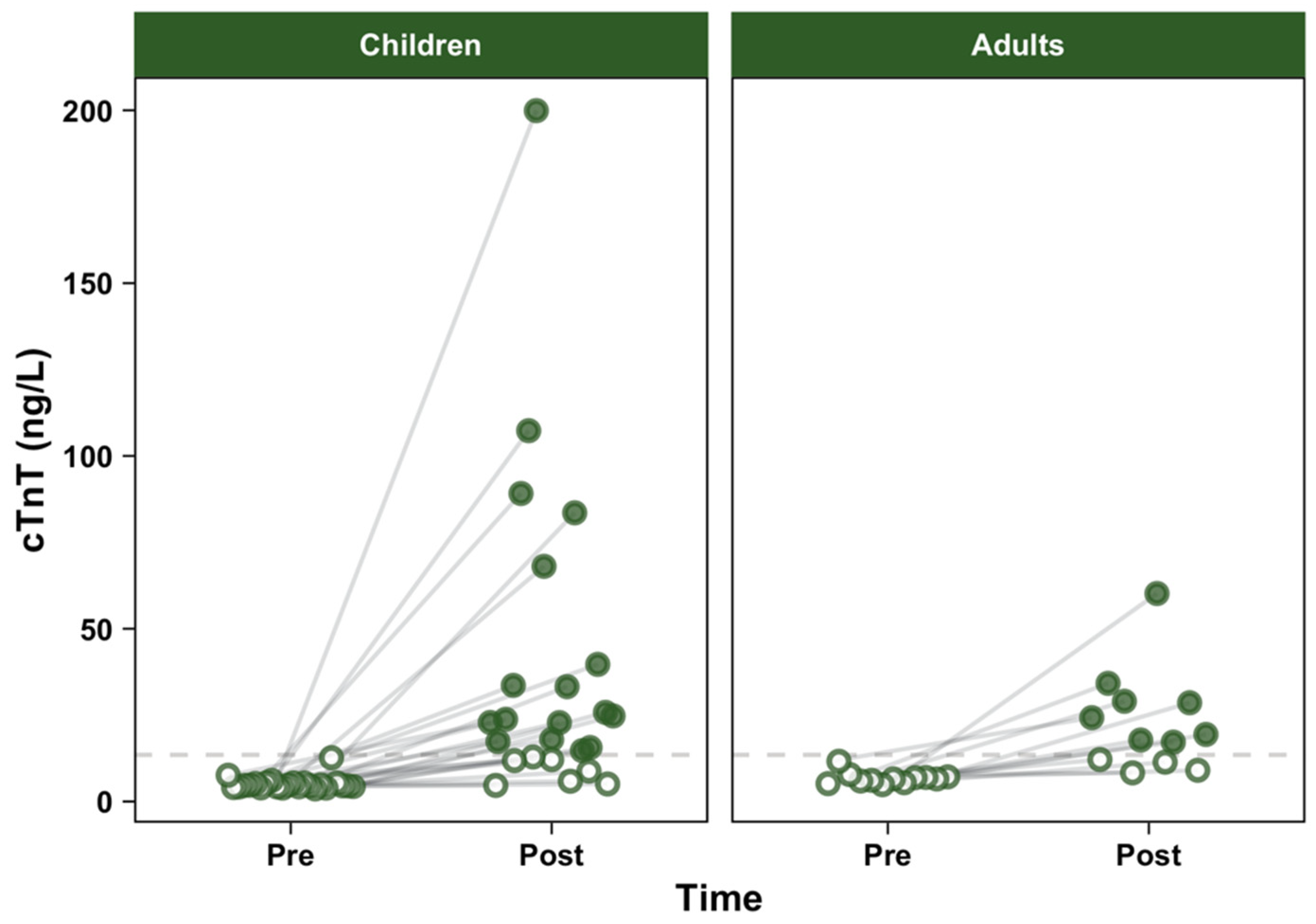
3. Results
3.1. Participants
3.2. Cardiac Troponins
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Thygesen, K.; Alpert, J.S.; Jaffe, A.S.; Chaitman, B.R.; Bax, J.J.; Morrow, D.A.; White, H.D.; Mickley, H.; Crea, F.; Van de Werf, F.; et al. Fourth Universal Definition of Myocardial Infarction. J. Am. Coll. Cardiol. 2018, 33, 2551–2567. [Google Scholar] [CrossRef]
- Baker, P.; Leckie, T.; Harrington, D.; Richardson, A. Exercise-induced cardiac troponin elevation: An update on the evidence, mechanism and implications. IJC Heart Vasc. 2019, 22, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Legaz-Arrese, A.; Carranza-García, L.E.; Navarro-Orocio, R.; Valadez-Lira, A.; Mayolas-Pi, C.; Munguía-Izquierdo, D.; Reverter-Masía, J.; George, K. Cardiac Biomarker Release after Endurance Exercise in Male and Female Adults and Adolescents. J. Pediatr. 2017, 191, 96–102. [Google Scholar] [CrossRef] [PubMed]
- López-Laval, I.; Legaz-Arrese, A.; George, K.; Serveto-Galindo, O.; González-Rave, J.M.; Reverter-Masia, J.; Munguía-Izquierdo, D. Cardiac troponin I release after a basketball match in elite, amateur and junior players. Clin. Chem. Lab. Med. CCLM 2016, 54, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Nie, J.; Huang, C.; George, K.P. The kinetics of highly sensitive cardiac troponin T release after prolonged treadmill exercise in adolescent and adult athletes. J. Appl. Physiol. 2012, 113, 418–425. [Google Scholar] [CrossRef]
- Cirer-Sastre, R.; Legaz-Arrese, A.; Corbi, F.; George, K.; Nie, J.; Carranza-García, L.E.; Reverter-Masià, J. Cardiac Biomarker Release After Exercise in Healthy Children and Adolescents: A Systematic Review and Meta-Analysis. Pediatr. Exerc. Sci. 2019, 31, 28–36. [Google Scholar] [CrossRef]
- Rausa, J.; Shetty, I.; Loomba, R.S. Troponin elevation in the setting of exercise-induced rhabdomyolysis in an athletic teenager. Cardiol Young 2019, 3, 1–4. [Google Scholar] [CrossRef]
- Donaldson, J.A.; Wiles, J.D.; Coleman, D.A.; Papadakis, M.; Sharma, R.; O’Driscoll, J.M. Left Ventricular Function and Cardiac Biomarker Release—The Influence of Exercise Intensity, Duration and Mode: A Systematic Review and Meta-Analysis. Sports Med. 2019, 49, 1275–1289. [Google Scholar] [CrossRef]
- Giza, E.; Micheli, L.J. Soccer injuries. Scand. J. Med. Sci. Sport 2005, 49, 140–169. [Google Scholar]
- Cirer-Sastre, R.; Legaz-Arrese, A.; Corbi, F.; López-Laval, I.; Puente-Lanzarote, J.; Hernández-González, V.; Reverter-Masià, J. Effect of Training Load on Post-Exercise Cardiac Troponin T Elevations in Young Soccer Players. Int. J. Environ. Res. Public Health 2019, 16, 4853. [Google Scholar] [CrossRef]
- Hosseini, S.M.; Azizi, M.; Samadi, A.; Talebi, N.; Hannes, G.; Burtscher, M. Impact of a Soccer Game on Cardiac Biomarkers in Adolescent Players. Pediatr. Exerc. Sci. 2017, 29, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Sanchez, J.; Sanchez, M.; Hernández, D.; Gonzalo-Skok, O.; Casamichana, D.; Ramirez-Campillo, R.; Nakamura, F.Y. Physical Performance During Soccer-7 Competition and Small-Sided Games in U12 Players. J. Hum. Kinet. 2019, 67, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Fédération Internationale de Football 7. Football 7 Worldwide: Book of Rules. Available online: https://www.f7federation.com/sport (accessed on 4 December 2019).
- Rodríguez, F.A. Spanish version of the Physical Activity Readiness Questionnaire (C-AAF/rPAR-Q). Apunts 1994, 31, 301–310. [Google Scholar]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Molina, I.; Gómez, C.; Bastida, A.; Pino, J. Validez del dispositivo inercial WIMU PRO para el registro de la frecuencia cardiaca en un test de campo. SPORT TK Revista Euroamericana de Ciencias del Deporte 2018, 7, 81–86. [Google Scholar] [CrossRef]
- Muñoz-Lopez, A.; Granero-Gil, P.; Pino-Ortega, J.; Hoyo, M. The validity and reliability of a 5-hz GPS device for quantifying athletes’ sprints and movement demands specific to team sports. J. Hum. Sport Exerc. 2017, 12, 156–166. [Google Scholar] [CrossRef]
- Rössler, R.; Verhagen, E.; Rommers, N.; Dvorak, J.; Junge, A.; Lichtenstein, E.; Donath, L.; Faude, O. Comparison of the 11+ Kids’ injury prevention programme and a regular warmup in children’s football (soccer): A cost effectiveness analysis. Br. J. Sports Med. 2019, 53, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Giannitsis, E.; Kurz, K.; Hallermayer, K.; Jarausch, J.; Jaffe, A.S.; Katus, H.A. Analytical validation of a high-sensitivity cardiac troponin T assay. Clin. Chem. 2010, 56, 254–261. [Google Scholar] [CrossRef]
- Shargal, E.; Kislev-Cohen, R.; Zigel, L.; Epstein, S.; Pilz-Burstein, R.; Tenenbaum, G. Age-related maximal heart rate: Examination and refinement of prediction equations. J. Sports Med. Phys. Fit. 2015, 55, 1207–1218. [Google Scholar]
- Mair, J.; Lindahl, B.; Hammarsten, O.; Müller, C.; Giannitsis, E.; Huber, K.; Möckel, M.; Plebani, M.; Thygesen, K.; Jaffe, A.S. How is cardiac troponin released from injured myocardium? Eur. Heart J. Acute Cardiovasc. Care. 2018, 7, 553–560. [Google Scholar] [CrossRef]
- Gore, M.O.; Seliger, S.L.; DeFilippi, C.R.; Nambi, V.; Christenson, R.H.; Hashim, I.A.; Hoogeveen, R.C.; Ayers, C.R.; Sun, W.; McGuire, D.K.; et al. Age- and Sex-Dependent Upper Reference Limits for the High-Sensitivity Cardiac Troponin T Assay. J. Am. Coll. Cardiol. 2014, 63, 1441–1448. [Google Scholar] [CrossRef] [PubMed]
- Schulte, C.; Zeller, T. Biomarkers in primary prevention: Meaningful diagnosis based on biomarker scores? HERZ 2019. [Google Scholar] [CrossRef] [PubMed]
- Wagner, K.-H.; Cameron-Smith, D.; Wessner, B.; Franzke, B. Biomarkers of Aging: From Function to Molecular Biology. Nutrients 2016, 8, 338. [Google Scholar] [CrossRef] [PubMed]
- Bohn, M.K.; Higgins, V.; Kavsak, P.; Hoffman, B.; Adeli, K. High-Sensitivity Generation 5 Cardiac Troponin T Sex- and Age-Specific 99th Percentiles in the CALIPER Cohort of Healthy Children and Adolescents. Clin. Chem. 2019, 65, 589–591. [Google Scholar] [CrossRef]
- Nie, J.P.; George, K.K.; Tong, T.; Gaze, D.; Tian, Y.; Lin, H.; Shi, Q. The Influence of a Half-Marathon Race Upon Cardiac Troponin T Release in Adolescent Runners. Curr. Med. Chem. 2011, 18, 3452–3456. [Google Scholar] [CrossRef]
- Tian, Y.; Nie, J.; Tong, T.K.; Cao, J.; Gao, Q.; Man, J.; Shi, Q.; Liu, W. Changes in serum cardiac troponins following a 21-km run in junior male runners. J. Sports Med. Phys. Fit. 2006, 46, 481–488. [Google Scholar]
- Nie, J.; Tong, T.K.; Shi, Q.; Lin, H.; Zhao, J.; Tian, Y. Serum cardiac troponin response in adolescents playing basketball. Int. J. Sports Med. 2008, 29, 449–452. [Google Scholar] [CrossRef]
- Fu, F.H.; Nie, J.; George, K.; Tong, T.K.; Lin, H.; Shi, Q. Impact of a 21-km Run on Cardiac Biomarkers in Adolescent Runners. J. Exerc. Sci. Fit. 2010, 8, 61–66. [Google Scholar] [CrossRef]
- Mehta, R.; Gaze, D.; Mohan, S.; Williams, K.L.; Sprung, V.; George, K.; Jeffries, R.; Hudson, Z.; Perry, M.; Shave, R. Post-Exercise Cardiac Troponin Release is Related to Exercise Training History. Int. J. Sports Med. 2012, 33, 333–337. [Google Scholar] [CrossRef]
- Peretti, A.; Mauri, L.; Masarin, A.; Annoni, G.; Corato, A.; Maloberti, A.; Giannattasio, C.; Vignati, G. Cardiac Biomarkers Release in Preadolescent Athletes After an High Intensity Exercise. High Blood Press. Cardiovasc. Prev. 2018, 25, 89–96. [Google Scholar] [CrossRef]
- Fretz, A.; McEvoy, J.W.; Rebholz, C.M.; Ndumele, C.E.; Florido, R.; Hoogeveen, R.C.; Ballantyne, C.M.; Selvin, E. Relation of Lifestyle Factors and Life’s Simple 7 Score to Temporal Reduction in Troponin Levels Measured by a High-Sensitivity Assay (from the Atherosclerosis Risk in Communities Study). Am. J. Cardiol. 2018, 121, 430–436. [Google Scholar] [CrossRef] [PubMed]
| Variable | Children (n = 24) | Adults (n = 12) | All (n = 36) | Between-Groups |
|---|---|---|---|---|
| Participant characteristics | ||||
| Age (years) | 10.7 ± 1.6 | 37.5 ± 12.7 | 19.6 ± 14.7 | p < 0.001 |
| Body height (cm) | 146 ± 14.8 | 177 ± 5.72 | 157 ± 19.1 | p < 0.001 |
| Body mass (kg) | 41.3 ± 15.4 | 79.5 ± 7 | 54.0 ± 22.4 | p < 0.001 |
| Training experience (years) | 4.6 ± 1.7 | 23.6 ± 14.5 | 9.4 ± 10.9 | p = 0.007 |
| Training frequency (days/week) | 2.9 ± 1.2 | 3.2 ± 0.8 | 3 ± 1.1 | p = 0.73 |
| Training volume (hours/week) | 4.6 ± 2.6 | 4.9 ± 2.5 | 4.7 ± 2.5 | p = 0.55 |
| Exercise load | ||||
| Distance (m) | 5970 ± 722 | 5490 ± 540 | 5810 ± 697 | p = 0.17 |
| SPpeak (km/h) | 23.5 ± 2.2 | 27.1 ± 1.9 | 24.7 ± 2.7 | p < 0.001 |
| SPmean (km/h) | 5.6 ± 0.7 | 6 ± 0.7 | 5.7 ± 0.7 | p = 0.04 |
| HRpeak (bpm) | 202 ± 6 | 188 ± 7 | 197 ± 9 | p < 0.001 |
| rHRpeak (% HRmax) | 100 ± 3 | 105 ± 6 | 102 ± 5 | p = 0.14 |
| HRmean (bpm) | 161 ± 19 | 158 ± 12 | 160 ± 16 | p = 0.5 |
| rHRmean (% HRmax) | 80 ± 9 | 88 ± 8 | 83 ± 10 | p = 0.084 |
| Pre | Post | Delta | Within-Subjects | |
|---|---|---|---|---|
| All (n = 36) | 5.20 (3.65, 12.7) | 21.1 (4.67, 200) | 13.7 (0.450, 194) | V = 666, p < 0.001 |
| Children (n = 24) | 4.64 (3.65, 12.7) | 22.9 (4.67, 200) | 16.4 (0.450, 194) | V = 300, p < 0.001 |
| Adults (n = 12) | 6.54 (4.88, 11.7) | 18.6 (8.31, 60.2) | 12.4 (0.500, 53.7) | V = 78, p = 0.001 |
| Between-groups | W = 36, p < 0.001 | W = 158, p = 0.65 | W = 171, p = 0.37 |
| Variable | cTnT Pre (ng/L) | ∆cTnT (ng/L) |
|---|---|---|
| ∆cTnT (ng/L) | rs = −0.12, p = 0.523 | |
| Participant characteristics | ||
| Age (years) | rs = 0.67, p < 0.001 | rs = −0.23, p = 0.21 |
| Body height (cm) | rs = 0.64, p < 0.001 | rs = −0.23, p = 0.2 |
| Body mass (kg) | rs = 0.55, p = 0.001 | rs = −0.35, p = 0.052 |
| Training experience (years) | rs = 0.56, p = 0.001 | rs = −0.16, p = 0.37 |
| Training frequency (days/week) | rs = 0.32, p = 0.072 | rs = −0.09, p = 0.64 |
| Training volume (hours/week) | rs = 0.2, p = 0.27 | rs = −0.2, p = 0.29 |
| Exercise load | ||
| Distance (m) | rs = −0.133, p = 0.465 | rs = 0.59, p < 0.001 |
| SPpeak (km/h) | rs = 0.32, p = 0.074 | rs = 0.17, p = 0.37 |
| SPmean (km/h) | rs = 0.2, p = 0.28 | rs = 0.59, p < 0.001 |
| HRpeak (bpm) | rs = −0.18, p = 0.33 | rs = 0.43, p = 0.013 |
| rHRpeak (% HRmax) | rs = 0.36, p = 0.044 | rs = 0.21, p = 0.25 |
| HRmean (bpm) | rs = 0.29, p = 0.11 | rs = 0.42, p = 0.016 |
| rHRmean (% HRmax) | rs = 0.52, p = 0.002 | rs = 0.29, p = 0.11 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cirer-Sastre, R.; Legaz-Arrese, A.; Corbi, F.; López-Laval, I.; Puente-Lanzarote, J.J.; Hernández-González, V.; Reverter-Masia, J. Cardiac Troponin T Release after Football 7 in Healthy Children and Adults. Int. J. Environ. Res. Public Health 2020, 17, 956. https://doi.org/10.3390/ijerph17030956
Cirer-Sastre R, Legaz-Arrese A, Corbi F, López-Laval I, Puente-Lanzarote JJ, Hernández-González V, Reverter-Masia J. Cardiac Troponin T Release after Football 7 in Healthy Children and Adults. International Journal of Environmental Research and Public Health. 2020; 17(3):956. https://doi.org/10.3390/ijerph17030956
Chicago/Turabian StyleCirer-Sastre, Rafel, Alejandro Legaz-Arrese, Francisco Corbi, Isaac López-Laval, Juan José Puente-Lanzarote, Vicenç Hernández-González, and Joaquin Reverter-Masia. 2020. "Cardiac Troponin T Release after Football 7 in Healthy Children and Adults" International Journal of Environmental Research and Public Health 17, no. 3: 956. https://doi.org/10.3390/ijerph17030956
APA StyleCirer-Sastre, R., Legaz-Arrese, A., Corbi, F., López-Laval, I., Puente-Lanzarote, J. J., Hernández-González, V., & Reverter-Masia, J. (2020). Cardiac Troponin T Release after Football 7 in Healthy Children and Adults. International Journal of Environmental Research and Public Health, 17(3), 956. https://doi.org/10.3390/ijerph17030956









