Adsorption of Strontium onto Adaxial and Abaxial Cuticle of Photinia serrulata Leaf
Abstract
:1. Introduction
2. Material and Methods
2.1. Samples Collection
2.2. The Characterization of Abaxial and Adaxial Cuticles
2.3. Batch Adsorption Experiment
2.4. Data Analysis
3. Results and Discussion
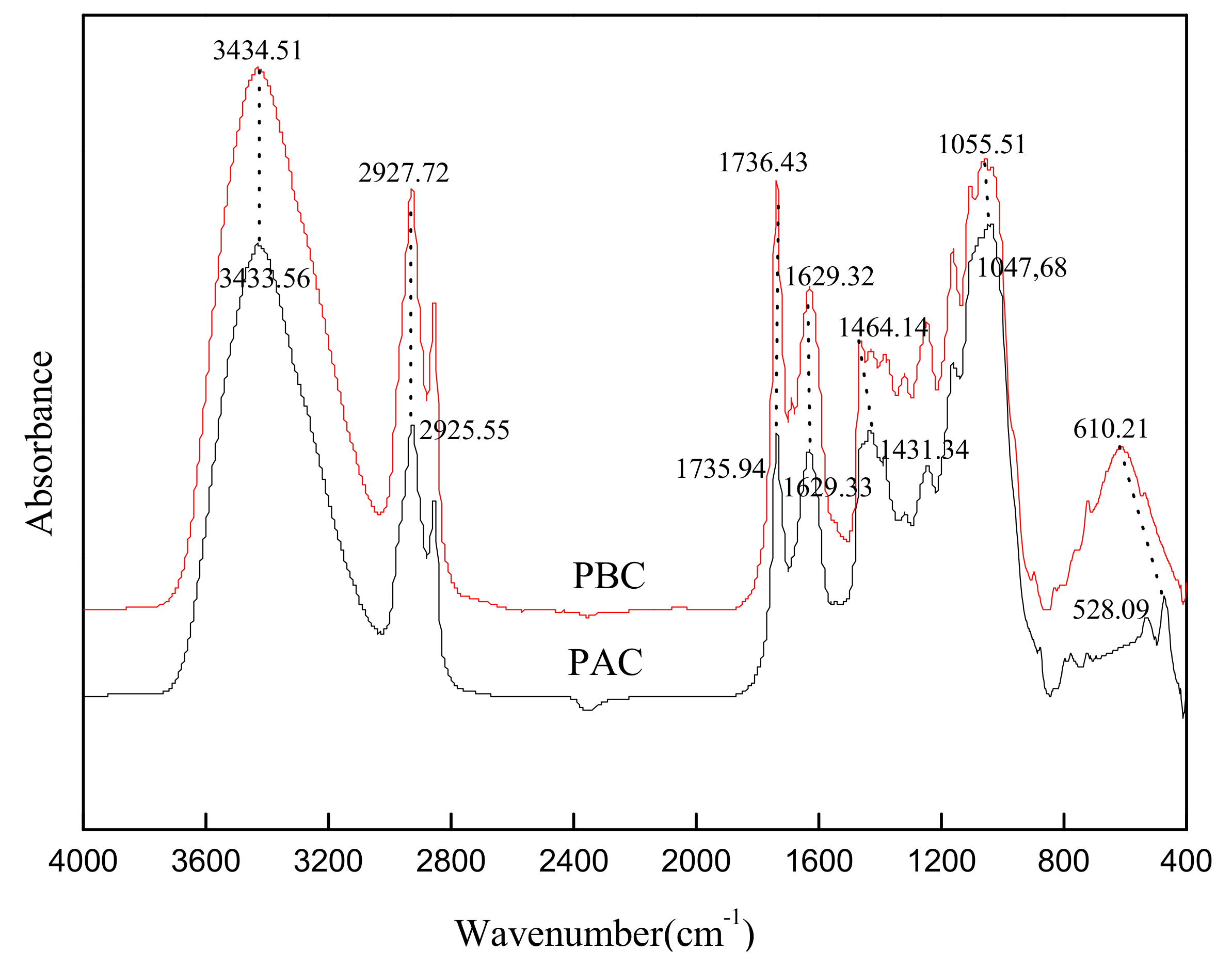
3.1. The Characterizations of Cuticles
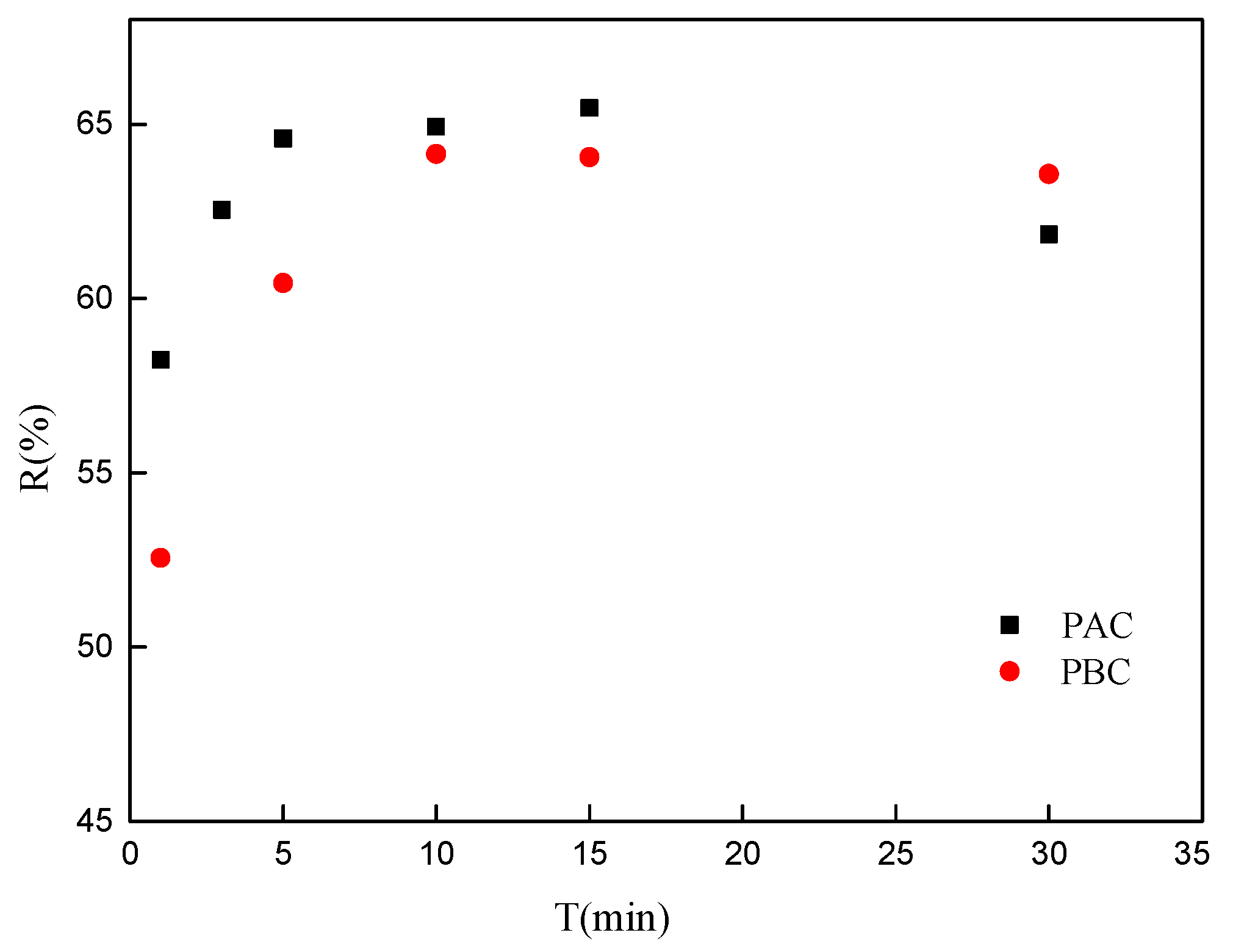
3.2. Adsorption Kinetics
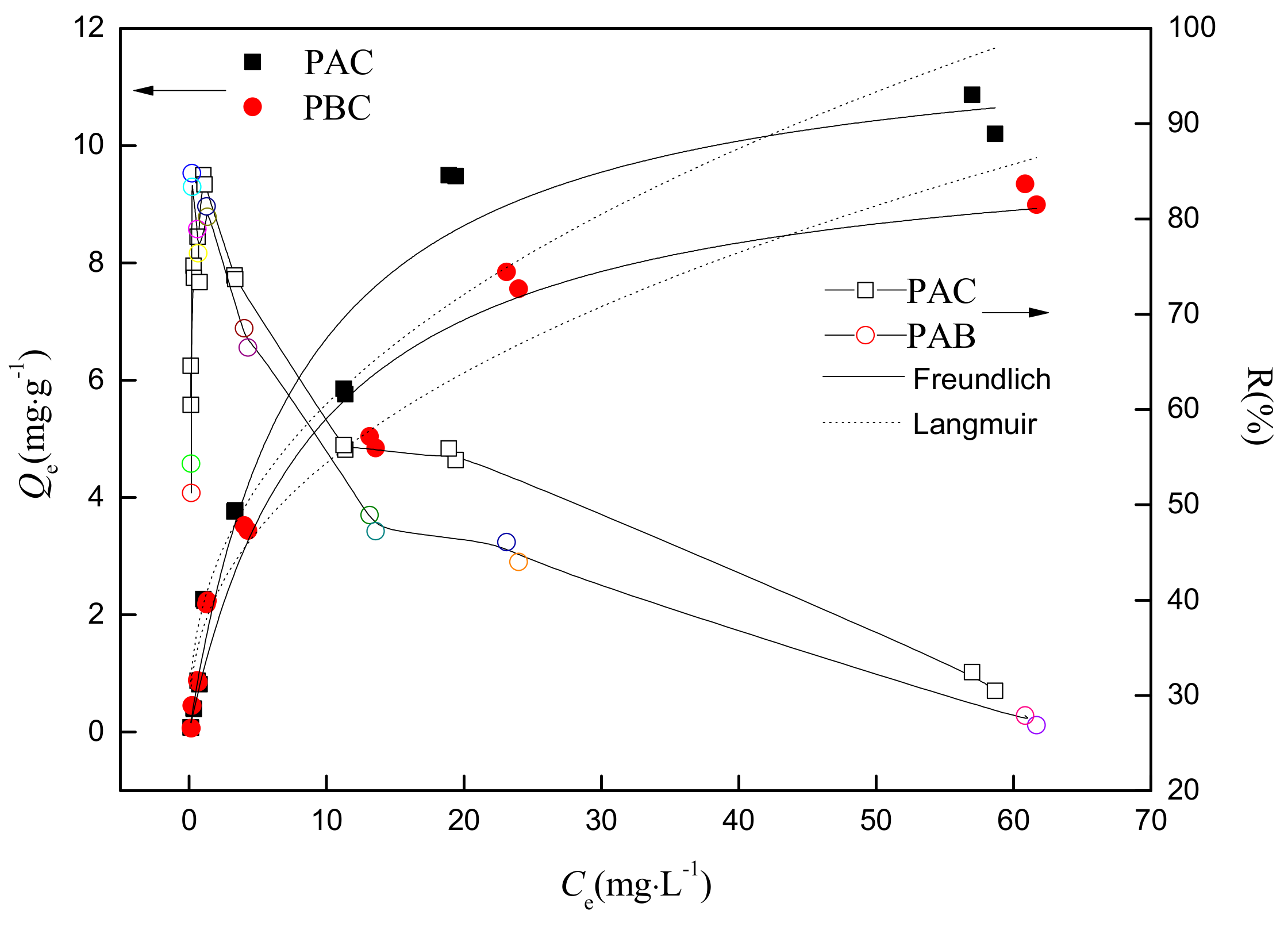
3.3. Adsorption Isotherm
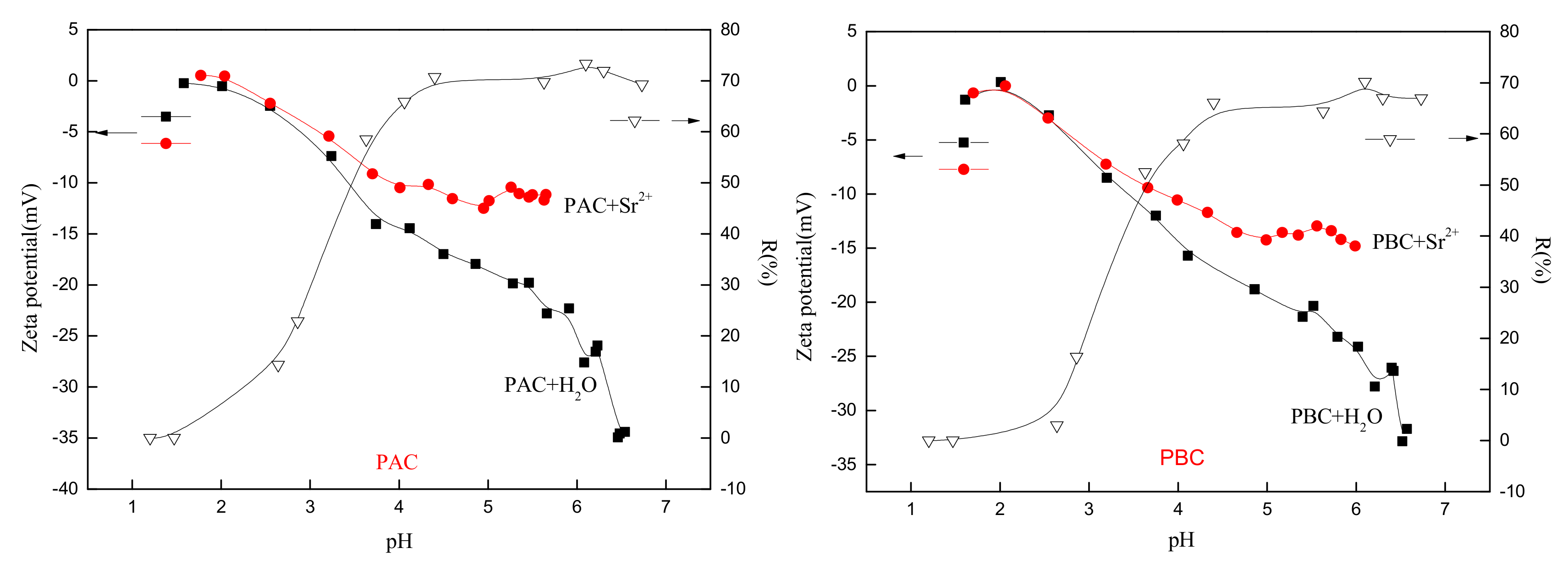
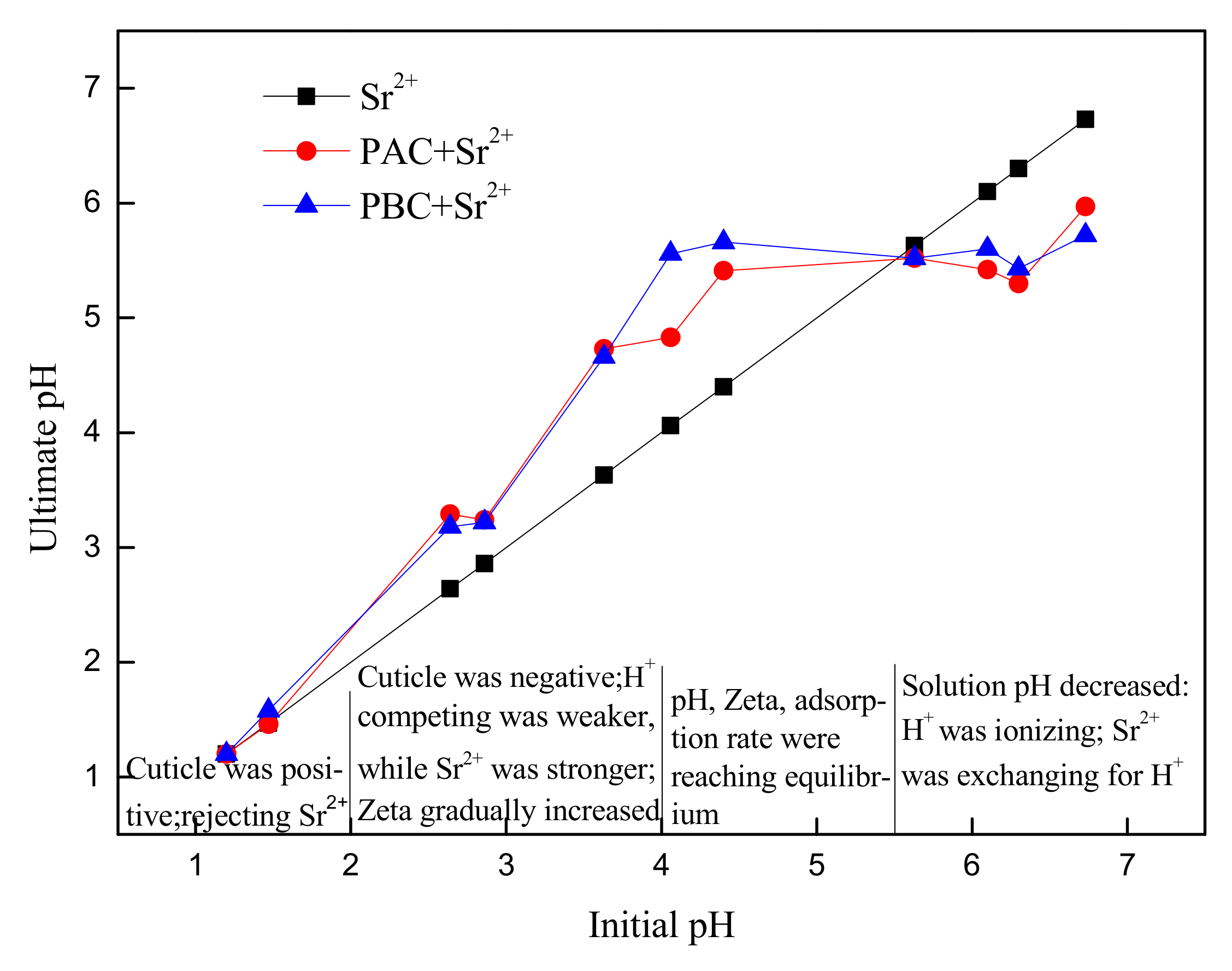
3.4. Effect of pH on Sr Adsorption
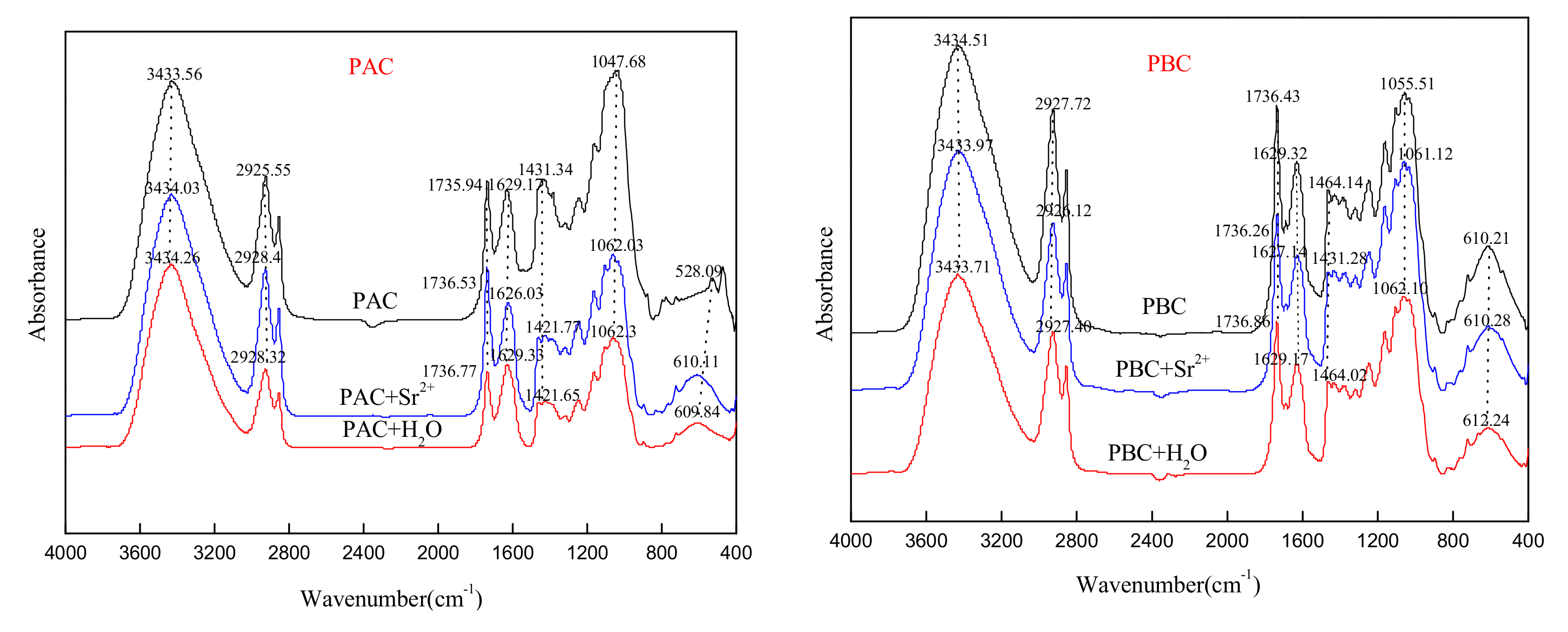
3.5. FTIR Spectra of the Cuticles before and after Loading with Sr
4. Conclusions
- The elemental composition and FTIR spectra for both adaxial and abaxial cuticles were quite similar and both show high affinity (H/C, 1.59 and 1.65) and polarity ((O + N)/C, 0.470 and 0.499). Adsorption of Sr onto adaxial and abaxial cuticles was up to equilibrium with 10 min, and both adsorption isotherms fit well with Langmuir model (R2, 0.97 and 0.97).
- Neither adaxial nor abaxial cuticles play a negative role on the absorption of nuclide which was usually considered to be a barrier for the foliar uptake of pollutants, but showed a strong retention capacity for the nuclides in the environment. The maximum adsorption capacity of PAC was 12.1 mg·g−1, little higher than that of PBC (10.3 mg·g−1).
- The adsorption of Sr increased with the increase of pH, and the maximum was attained when pH ≥ 4. Electrostatic attraction was demonstrated to be the main mechanism of Sr adsorption onto PAC and PBC, and the similar adsorption of adaxial and abaxial cuticles was consistent with their similar isoelectric point (≈ 2).
- Importantly, the characteristic functional groups in the abaxial and adaxial foliar cuticles did not change before and after the adsorption for Sr, suggesting that there was no chemical precipitation in the adsorption of cuticles for Sr.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Buesseler, K.; Aoyama, M.; Fukasawa, M. Impacts of the Fukushima nuclear power plants on marine radioactivity. Environ. Sci. Technol. 2011, 45, 9931–9935. [Google Scholar] [CrossRef] [PubMed]
- Garnierlaplace, J.; Beaugelinseiller, K.; Hinton, T.G. Fukushima wildlife dose reconstruction signals ecological consequences. Environ. Sci. Technol. 2011, 45, 5077–5078. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Onda, Y.; Hisadome, K.; Loffredo, N.; Kawamori, A. Temporal changes in radiocesium deposition in various forest stands following the Fukushima Dai-ichi Nuclear Power Plant accident. J. Environ. Radioact. 2017, 166, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Parache, V.; Pourcelot, L.; Rousseldebet, S.; Orjollet, D.; Leblanc, F.; Soria, C.; Gurriaran, R.; Renaud, P.; Masson, O. Transfer of 131I from Fukushima to the vegetation and milk in France. Environ. Sci. Technol. 2011, 45, 9998–10003. [Google Scholar] [CrossRef] [PubMed]
- Shozugawa, K.; Nogawa, N.; Matsuo, M. Deposition of fission and activation products after the Fukushima Dai-ichi nuclear power plant accident. Environ. Pollut. 2012, 163, 243–247. [Google Scholar] [CrossRef]
- Kanasashi, T.; Sugiura, Y.; Takenaka, C.; Hijii, N.; Umemura, M. Radiocesium distribution in sugi (Cryptomeria japonica) in Eastern Japan: Translocation from needles to pollen. J. Environ. Radioact. 2015, 139, 398–406. [Google Scholar] [CrossRef]
- Madoz-Escande, C.; Henner, P.; Bonhomme, T. Foliar contamination of Phaseolus vulgaris with aerosols of 137Cs, 85Sr, 133Ba and 123mTe: Influence of plant development stage upon contamination and rain. J. Environ. Radioact. 2004, 73, 49–71. [Google Scholar] [CrossRef]
- Chen, B.L.; Li, Y.G. Sorption of 1-naphthol by plant cuticular fractions. J. Environ. Sci. 2007, 19, 1214–1220. [Google Scholar] [CrossRef]
- Li, Q.Q.; Chen, B.L. Organic pollutant clustered in the plant cuticular membranes: Visualizing the distribution of phenanthrene in leaf cuticle using two-photon confocal scanning laser microscopy. Environ. Sci. Technol. 2014, 48, 4774–4781. [Google Scholar] [CrossRef]
- Nishikiori, T.; Watanabe, M.; Koshikawa, M.K.; Takamatsu, T.; Ishii, Y.; Ito, S.; Takenaka, A.; Watanabe, K.; Hayashi, S. Uptake and translocation of radiocesium in cedar leaves following the Fukushima nuclear accident. Sci. Total Environ. 2015, 502, 611–615. [Google Scholar] [CrossRef]
- Chefetz, B. Sorption of phenathrene and atrazine by plant cuticular fractions. Environ. Toxicol. Chem. 2003, 22, 2492–2498. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.L.; Li, Y.G.; Guo, Y.T.; Zhu, L.Z.; Schnoor, J.L. Role of the extractable lipids and polymeric lipids in sorption of organic contaminants onto plant cuticles. Environ. Sci. Technol. 2008, 42, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.G.; Deng, Q.Q.; Chen, B.L. Sorption of chlorophenols onto fruit cuticles and potato periderm. J. Environ. Sci. 2012, 24, 675–681. [Google Scholar] [CrossRef]
- Chen, B.L.; Johnson, E.J.; Chefetz, B.; Zhu, L.Z.; Xing, B.S. Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: role of polarity and accessibility. Environ. Sci. Technol. 2005, 39, 6138–6146. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.G.; Chen, B.L.; Zhu, L.Z. Single-solute and bi-solute sorption of phenanthrene and pyrene onto pine needle cuticular fractions. Environ. Pollut. 2010, 158, 2478–2484. [Google Scholar] [CrossRef] [PubMed]
- Hunsche, M.; Noga, G. Cuticular wax load and surface wettability of leaves and fruits collected from sweet cherry (Prunus avium) trees grown under field conditions or inside a polytunnel. Acta Physiol. Plant. 2011, 33, 1785–1792. [Google Scholar] [CrossRef]
- Mickle, J.E.; Lumaga, M.R.B.; Moretti, A.; Luca, P. Scanning electron microscopy studies of cuticle micromorphology in Cycas L. (Cycadaceae). Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2011, 145, 191–201. [Google Scholar] [CrossRef]
- Wild, E.; Dent, J.; Thomas, G.O.; Jones, K.C. Use of two-photon excitation microscopy and autofluorescence for visualizing the fate and behavior of semivolatile organic chemicals within living vegetation. Environ. Toxicol. Chem. 2008, 26, 2486–2493. [Google Scholar] [CrossRef]
- Ahmadpour, A.; Zabihi, M.; Tahmasbi, M.; Bastami, T.R. Effect of adsorbents and chemical treatments on the removal of strontium from aqueous solutions. J. Hazard. Mater. 2010, 182, 552–556. [Google Scholar] [CrossRef]
- Balarama Krishna, M.V.; Rao, S.V.; Arunachalam, J.; Murali, M.S.; Kumar, S.; Manchanda, V.K. Removal of 137Cs and 90Sr from actual low level radioactive waste solutions using moss as a phyto-sorbent. Sep. Purif. Technol. 2004, 38, 149–161. [Google Scholar] [CrossRef]
- Chen, J.P. Batch and continuous adsorption of strontium by plant root tissues. Bioresour. Technol. 1997, 60, 185–189. [Google Scholar] [CrossRef]
- Ai, L.; Luo, X.G.; Lin, X.Y.; Zhang, S.Z. Removal of Strontium ions from Aqueous Solution by Sunflower Straw. Adv. Mater. Res. 2013, 726, 1922–1925. [Google Scholar] [CrossRef]
- Qian, L.B.; Chen, B.L. Dual role of biochars as adsorbents for aluminum: The effects of oxygen-containing organic components and the scattering of silicate particles. Environ. Sci. Technol. 2013, 47, 8759–8768. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Mei, H.; Chen, X.; Tan, X.L.; Ahmad, B.; Alsaedi, A.; Hayat, T.; Wang, X.K. Impact of environmental conditions on the sorption behavior of radionuclide 90 Sr(II) on Na-montmorillonite. J. Mol. Liq. 2015, 203, 39–46. [Google Scholar] [CrossRef]
- Gok, C.; Aytas, S. Biosorption of radiostrontium by alginate beads: Application of isotherm models and thermodynamic studies. J. Radioanal. Nucl. Chem. 2013, 295, 777–788. [Google Scholar] [CrossRef]
- Cole, T.; Bidoglio, G.; Soupioni, M.; Gorman, M.O.; Gibson, N. Diffusion mechanisms of multiple strontium species in clay. Geochim. Cosmochim. Acta 2000, 64, 385–396. [Google Scholar] [CrossRef]



| Sample | Yield/%a | C/% | H/% | N/% | O/%b | H/C | (O + N)/C |
|---|---|---|---|---|---|---|---|
| adaxial cuticle | 7.01 | 56.88 | 7.55 | 0.45 | 35.12 | 1.59 | 0.47 |
| abaxial cuticle | 5.78 | 55.60 | 7.63 | 0.45 | 36.32 | 1.65 | 0.50 |
| Sample | Langmuir Regression Parameters | Freundlich Regression Parameters | ||||
|---|---|---|---|---|---|---|
| KL/(L·mg−1) | Qmax/(mg·g−1) | R2 | Kf/(mg/g)/(mg/L)N | N | R2 | |
| PAC PBC | 0.125 ± 0.023 | 12.1 ± 0.71 | 0.97 | 2.15 ± 0.32 | 0.415 ± 0.044 | 0.92 |
| 0.109 ± 0.021 | 10.3 ± 0.64 | 0.97 | 1.76 ± 0.20 | 0.417 ± 0.032 | 0.96 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.; Luo, X.; Bai, X.; Lv, W.; Liao, Y. Adsorption of Strontium onto Adaxial and Abaxial Cuticle of Photinia serrulata Leaf. Int. J. Environ. Res. Public Health 2020, 17, 1061. https://doi.org/10.3390/ijerph17031061
Li Y, Luo X, Bai X, Lv W, Liao Y. Adsorption of Strontium onto Adaxial and Abaxial Cuticle of Photinia serrulata Leaf. International Journal of Environmental Research and Public Health. 2020; 17(3):1061. https://doi.org/10.3390/ijerph17031061
Chicago/Turabian StyleLi, Yungui, Xiang Luo, Xueying Bai, Wenxuan Lv, and Yang Liao. 2020. "Adsorption of Strontium onto Adaxial and Abaxial Cuticle of Photinia serrulata Leaf" International Journal of Environmental Research and Public Health 17, no. 3: 1061. https://doi.org/10.3390/ijerph17031061
APA StyleLi, Y., Luo, X., Bai, X., Lv, W., & Liao, Y. (2020). Adsorption of Strontium onto Adaxial and Abaxial Cuticle of Photinia serrulata Leaf. International Journal of Environmental Research and Public Health, 17(3), 1061. https://doi.org/10.3390/ijerph17031061





