The Epidemiology of Skin Cancer and Public Health Strategies for Its Prevention in Southern Africa
Abstract
1. Introduction
2. Geography and Population
3. Meteorology and Solar UVR Environment
4. Introduction to Keratinocyte Cancer (KC) and Cutaneous Melanoma (CM)
5. The Incidence of Skin Cancer in Southern Africa
6. Mortality due to Skin Cancer in Southern Africa
7. Risk Factors for the Development of Skin Cancer in Southern Africa
8. Campaigns to Prevent Skin Cancer in Southern Africa
9. Climate Change and Skin Cancer in Southern Africa
10. Sun Protection Strategies
11. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Green, A.; Whiteman, D.; Frost, C.; Battistutta, D. Sun Exposure, Skin Cancers and Related Skin Conditions. J. Epidemiol. 1999, 9, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Diepgen, T.L.; Mahler, V. The Epidemiology of Skin Cancer. Br. J. Dermatol. 2002, 146, 1–6. [Google Scholar] [CrossRef] [PubMed]
- World Cancer Research Fund. Skin Cancer. 27 April 2018. Available online: https://www.wcrf.org/dietandcancer/skin-cancer (accessed on 17 December 2019).
- Rogers, H.W.; Weinstock, M.A.; Feldman, S.R.; Coldiron, B.M. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the U.S. Population, 2012. JAMA Dermatol. 2015, 151, 1081–1086. [Google Scholar] [CrossRef] [PubMed]
- Welcome to the SACU Website. Available online: https://www.sacu.int/index.php (accessed on 5 December 2019).
- World Life Expectancy. World Life Expectancy. Available online: https://www.worldlifeexpectancy.com/ (accessed on 5 December 2019).
- Africa Internet Users, 2019 Population and Facebook Statistics. Available online: https://www.internetworldstats.com/stats1.htm (accessed on 10 December 2019).
- Reason, C.J.C.; Landman, W.A.; Tennant, W. Seasonal to Decadal Prediction of Southern African Climate and Its Links with Variability of the Atlantic Ocean. Bull. Amer. Meteor. Soc. 2006, 87, 941–956. [Google Scholar] [CrossRef]
- World Atlas. 15 Biggest Cities in Africa. Available online: https://www.worldatlas.com/articles/15-biggest-cities-in-africa.html (accessed on 5 December 2019).
- Hovila, J.; Arola, A.; Tamminen, J. OMI/Aura Surface UVB Irradiance and Erythemal Dose Daily L3 Global Gridded 1.0 degree × 1.0 degree V3, N.G.S.F. Center, Editor. 2013, Goddard Earth Sciences Data and Information Services Center (GES DISC). Available online: https://catalog.data.gov/dataset/omi-aura-surface-uvb-irradiance-and-erythemal-dose-daily-l3-global-gridded-1-0-degree-x-1- (accessed on 17 December 2019).
- Venkataraman, S.; Ogunniyi, J. Ozone Climatology and Variability over Irene, South Africa Determined by Ground Based and Satellite Observations. Part 1: Vertical Variations in the Troposphere and Stratosphere. Atmósfera 2017, 30, 337–353. [Google Scholar]
- Diab, R.; Barsby, J.; Bodeker, G.; Scourfield, M.; Salter, L. Satellite Observations of Total Ozone above South Africa. S. Afr. Geogr. J. 1992, 74, 13–18. [Google Scholar] [CrossRef]
- Chahal, H.S.; Rieger, K.E.; Sarin, K.Y. Incidence Ratio of Basal Cell Carcinoma to Squamous Cell Carcinoma Equalizes with Age. J. Am. Acad. Dermatol. 2017, 76, 353–354. [Google Scholar] [CrossRef]
- Pandeya, N.; Olsen, C.M.; Whiteman, C.C. The incidence and multiplicity of keratinocyte cancers in Australia. Med. J. Aust. 2017, 207, 339–343. [Google Scholar] [CrossRef]
- Lucas, R.M.; Yazar, S.; Young, A.R.; Norval, M.; de Gruijl, F.R.; Takizawa, Y.; Rhodes, L.E.; Sinclair, C.A.; Neale, R.E. Human Health in Relation to Exposure to Solar Ultraviolet Radiation under Changing Stratospheric Ozone and Climate. Photochem. Photobiol. Sci. 2019, 18, 641–680. [Google Scholar] [CrossRef]
- Cancer Research UK. Melanoma Skin Cancer Incidence Statistics. Available online: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/melanoma-skin-cancer/incidence (accessed on 15 May 2015).
- Australian Institute of Health and Welfare. Available online: https://www.aihw.gov.au/reports/cancer/skin-cancer-in-australia/contents/table-of-contents (accessed on 17 December 2019).
- Parkin, D.M.; Hämmer, L.; Ferlay, J.; Kantelhardt, E.J. Cancer in Africa 2018: The Role of Infections. Int. J. Cancer 2019. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Norval, M.; Kellett, P.; Wright, C.Y. The Incidence and Body Site of Skin Cancers in the Population Groups of South Africa. Photodermatol. Photoimmunol. Photomed. 2014, 30, 262–265. [Google Scholar] [CrossRef]
- Made, F.; Wilson, K.; Jina, R.; Tlotleng, N.; Jack, S.; Ntlebi, V.; Kootbodien, T. Distribution of Cancer Mortality Rates by Province in South Africa. Cancer Epidemiol. 2017, 51, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Stats, S.A. Mortality and Causes of Death in South Africa, 2016: Findings from Death Notification. Statistical Release P0309.3, 1–146. Available online: http://www.statssa.gov.za/publications/P03093/P030932016.pdf (accessed on 17 December 2019).
- Wright, C.Y.; Kapwata, K.; Singh, E.; Green, A.C.; Baade, P.; Kellett, P.; Norval, M. Trends in Melanoma Mortality in the Population Groups of South Africa. Dermatology 2019, 235, 396–399. [Google Scholar] [CrossRef]
- Gloster, H.M.; Neal, K. Skin Cancer in Skin of Color. J. Am. Acad. Dermatol. 2006, 55, 741–760. [Google Scholar] [CrossRef]
- Hudson, D.A.; Krige, J.E. Melanoma in black South Africans. J. Am. Coll. Surg. 1995, 180, 65–71. [Google Scholar]
- Lodder, J.V.; Simson, W.; Becker, P.J. Malignant melanoma of the skin in Black South Africans: A 15-year experience. S. Afr. J. Surg. 2010, 48, 76–79. [Google Scholar]
- Olsen, C.M.; Wilson, L.F.; Green, A.C.; Bain, C.J.; Fritschi, L.; Neale, R.E.; Whiteman, D.C. Cancers in Australia Attributable to Exposure to Solar Ultraviolet Radiation and Prevented by Regular Sunscreen Use. Aust. N. Z. J. Public Health 2015, 39, 471–476. [Google Scholar] [CrossRef]
- Modenese, A.; Korpinen, L.; Gobba, F. Solar Radiation Exposure and Outdoor Work: An Underestimated Occupational Risk. Int. J. Environ. Res. Public Health 2018, 15, 2063. [Google Scholar] [CrossRef]
- Armstrong, B.K.; Cust, A.E. Sun Exposure and Skin Cancer, and the Puzzle of Cutaneous Melanoma. Cancer Epidemiol. 2017, 48, 147–156. [Google Scholar] [CrossRef]
- Halder, R.M.; Bridgeman-Shah, S. Skin cancer in African Americans. Cancer 1995, 75, 667–673. [Google Scholar] [CrossRef]
- Hart, P.H.; Norval, M.; Byrne, S.N.; Rhodes, L.E. Exposure to Ultraviolet Radiation in the Modulation of Human Diseases. Annu. Rev. Pathol. Mech. 2019, 14, 55–81. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.A.; Young, A.R.; McGregor, J.M.; Seed, P.T.; Potten, C.S.; Walker, S.L. Sensitivity to Sunburn is Associated with Susceptibility to Ultraviolet Radiation–Induced Suppression of Cutaneous Cell–Mediated Immunity. J. Exp. Med. 2001, 191, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Wright, C.Y.; Norval, M.; Hertle, R.W. Oculocutaneous Albinism in Sub-Saharan Africa: Adverse Sun-Associated Health Effects and Photoprotection. Photochem. Photobiol. 2015, 91, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Hong, E.S.; Zeeb, H.; Repacholi, M.H. Albinism in Africa as a Public Health Issue. BMC Public Health 2006, 6, 212. [Google Scholar] [CrossRef] [PubMed]
- Kromberg, J.G.R.; Castle, D.; Zwane, E.M.; Jenkins, T. Albinism and Skin Cancer in Southern Africa. Clin. Genet. 2008, 36, 43–52. [Google Scholar] [CrossRef]
- Facciolà, A.; Rullo, E.V.; Ceccarelli, M.; D’Andrea, F.; Coco, M.; Micali, C.; Cacopardo, B.; Marino, A.; Cannavò, S.P.; Di Rosa, M.; et al. Malignant Melanoma in HIV: Epidemiology, Pathogenesis, and Management. Dermatol. Ther. 2019, 9, e13180. [Google Scholar] [CrossRef]
- Silverberg, M.J.; Leyden, W.; Warton, E.M.; Quesenberry, C.P.; Engels, E.A.; Asgari, M.M. HIV Infection Status, Immunodeficiency, and the Incidence of Non-Melanoma Skin Cancer. J. Natl. Cancer Inst. 2013, 105, 350–360. [Google Scholar] [CrossRef]
- Asgari, M.M.; Ray, G.T.; Quesenberry, C.P.; Katz, K.A.; Silverberg, M.J. Association of Multiple Primary Skin Cancers with Human Immunodeficiency Virus Infection, CD4 Count, and Viral Load. JAMA Dermatol. 2017, 153, 892–896. [Google Scholar] [CrossRef]
- Omland, S.H.; Ahlström, M.G.; Gerstoft, J.; Pedersen, G.; Mohey, R.; Pedersen, C.; Kronborg, G.; Larsen, C.S.; Kvinesdal, B.; Gniadecki, R.; et al. Risk of Skin Cancer in Patients with HIV: A Danish Nationwide Cohort Study. J. Am. Acad. Dermatol. 2018, 79, 689–695. [Google Scholar] [CrossRef]
- Yanik, E.L.; Hernández-Ramírez, R.U.; Qin, L.; Lin, H.; Leyden, W.; Neugebauer, R.S.; Horberg, M.A.; Moore, R.; Mathews, W.; Justice, A.; et al. Brief Report: Cutaneous Melanoma Risk Among People with HIV in the United States and Canada. J. Acquir. Immune Defic. Syndr. 2018, 78, 499. [Google Scholar] [CrossRef] [PubMed]
- Jaquet, A.; Odutola, M.; Ekouevi, D.K.; Tanon, A.; Oga, E.; Akakpo, J.; Charurat, M.; Zannou, M.D.; Eholie, S.P.; Sasco, A.J.; et al. Cancer and HIV Infection in Referral Hospitals from Four West African Countries. Cancer Epidemiol. 2015, 39, 1060–1065. [Google Scholar] [CrossRef] [PubMed]
- Cahoon, E.K.; Engels, E.A.; Freedman, D.M.; Norval, M.; Pfeiffer, R.M. Ultraviolet Radiation and Kaposi Sarcoma Incidence in a Nationwide US Cohort of HIV-Infected Men. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef] [PubMed]
- Life Expectancy by Country 2019. Available online: http://worldpopulationreview.com/countries/life-expectancy-by-country/ (accessed on 10 December 2019).
- Diffey, B.L.; Norval, M.; Albers, P.N.; Wright, C.Y. The Influence of HIV Infection on the Age Dependence of Squamous Cell Carcinoma of the Skin in South Africa. S. Afr. Med. J. 2017, 107, 127–129. [Google Scholar] [CrossRef]
- Read, J.; Wadt, K.A.W.; Hayward, N.K. Melanoma Genetics. J. Med. Genet. 2016, 53, 1–14. [Google Scholar] [CrossRef]
- Yakubu, A.; Mabogunje, O.A. Squamous Cell Carcinoma of the Skin in Africans. Trop. Geogr. Med. 1995, 47, 91–93. [Google Scholar]
- Tabbakh, T.; Volkov, A.; Wakefield, M.; Dobbinson, S. Implementation of the SunSmart Program and Population Sun Protection Behaviour in Melbourne, Australia: Results from Cross-Sectional Summer Surveys from 1987 to 2017. PLoS Med. 2019, 16, e1002932. [Google Scholar] [CrossRef]
- Køster, B.; Meyer, M.K.H.; Søgaard, J.; Dalum, P. Benefit–Cost Analysis of the Danish Sun Safety Campaign 2007–2015: Cost Savings from Sunburn and Sunbed Use Reduction and Derived Skin Cancer Reductions 2007–2040 in the Danish Population. Pharmaco. Econ. Open 2019, 15. [Google Scholar] [CrossRef]
- Lucas, R.M. Is the Current Public Health Message on UV Exposure Correct? Bull World Health Organ. 2006, 84, 485–491. [Google Scholar] [CrossRef]
- Wang, H.; Chen, W.; Li, D.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D and Chronic Diseases. Aging Dis. 2017, 8, 346–353. [Google Scholar] [CrossRef]
- Norval, M.; Coussens, A.K.; Wilkinson, R.J.; Bornman, L.; Lucas, R.M.; Wright, C.Y. Vitamin D Status and its Consequences for Health in South Africa. Int. J. Environ. Res. Public Health 2016, 13, 1019. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. ICD Report—Radio Broadcasting for Health: A Decision Maker’s Guide. Available online: http://www.who.int/management/programme/health_promotion/RadioBroadcastingHealth.pdf (accessed on 17 December 2019).
- Radcliffe, D. Mobile in Sub-Saharan Africa: Can World’s Fastest-Growing Mobile Region Keep It Up? ZDNet. Available online: https://www.zdnet.com/article/mobile-in-sub-saharan-africa-can-worlds-fastest-growing-mobile-region-keep-it-up/ (accessed on 6 December 2019).
- World Health Organization. Ultraviolet Index. Available online: https://www.who.int/uv/en/ (accessed on 12 December 2019).
- Engelbrecht, F.A.; Adegoke, J.; Bopape, M.-J.M.; Naidoo, M.; Garland, R.M.; Thatcher, M.; Gregor, J.; Katzfey, J.; Werner, M.; Ichoku, C.; et al. Projections of Rapidly Rising Surface Temperatures over Africa under Low Mitigation. Environ. Res. Lett. 2015, 10. [Google Scholar] [CrossRef]
- Garland, R.M.; Matooane, M.; Engelbrecht, F.A.; Bopape, M.-J.M.; Landman, W.A.; Naidoo, M.; Van der Merwe, J.; Wright, C.Y. Regional Projections of Extreme Apparent Temperature Days in Africa and the Related Potential Risk to Human Health. Int. J. Environ. Res. Public Health 2015, 12, 577–604. [Google Scholar] [CrossRef] [PubMed]
- Van der Leun, J.C.; Piacentini, R.D.; de Gruijl, F.R. Climate Change and Human Skin Cancer. Photochem. Photobiol. Sci. 2008, 7, 730–733. [Google Scholar] [CrossRef]
- Kaffenberger, B.H.; Shetlar, D.; Norton, S.A.; Rosenbach, M. The Effect of Climate Change on Skin Disease in North America. J. Am. Acad. Dermatol. 2017, 76, 140–147. [Google Scholar] [CrossRef]
- Naicker, N.; Teare, J.; Balakrishna, Y.; Wright, C.Y.; Mathee, A. Indoor Temperatures in Low Cost Housing in Johannesburg, South Africa. Int. J. Environ. Res. Public Health 2017, 14, 1410. [Google Scholar] [CrossRef]
- Bidassey-Manilal, S.; Wright, C.Y.; Engelbrecht, J.C.; Albers, P.N.; Garland, R.M.; Matooane, M. Students Perceived Heat-Health Symptoms Increased with Warmer Classroom Temperatures. Int. J. Environ. Res. Public Health 2016, 13, 566. [Google Scholar] [CrossRef]
- Schmalwieser, A.W.; Schmalwieser, V.T.; Schmalwieser, S.S. Influence of air temperature on the UV exposure on different body sites due to clothing of young women during daily errands. Photochem. Photobiol. 2019, 95, 1068–1075. [Google Scholar] [CrossRef]
- Slaper, H.; Velders, G.; Daniel, J.; de Gruijl, F.; van der Leun, J. Estimates of ozone depletion and skin cancer incidence to examine the Vienna Convention achievements. Nature 1996, 384, 256–258. [Google Scholar] [CrossRef]
- McKenzie, R.; Bernhard, G.; Liley, B.; Disterhoft, P.; Rhodes, S.; Bais, A.; Morgenstern, O.; Newman, P.; Oman, L.; Brogniez, C.; et al. Success of Montreal Protocol Demonstrated by Comparing High-Quality UV Measurements with “World Avoided” Calculations from Two Chemistry-Climate Models. Sci. Rep. 2019, 9, 12332. [Google Scholar] [CrossRef]
- World Health Organization. Sun Protection. Available online: http://www.who.int/uv/sun_protection/en/ (accessed on 5 December 2019).
- Heckman, C.J.; Liang, K.; Riley, M. Awareness, understanding, use, and impact of the UV index: A systematic review of over two decades of international research. Prev. Med. 2019, 123, 71–83. [Google Scholar] [CrossRef]
- Harrison, S.L.; Garzón-Chavez, D.R.; Nikles, C.J. Sun Protection Policies of Australian Primary Schools in a Region of High Sun Exposure. Health Educ. Res. 2016, 31, 416–428. [Google Scholar] [CrossRef][Green Version]
- Reeder, A.I.; Jopsen, A.; Gray, A. Primary School Sun Protection Policies and Practices 4 Years after Baseline—A Follow-up Study. Health Educ. Res. 2012, 27, 844–856. [Google Scholar] [CrossRef]
- Whiteman, D.C.; Neale, R.E.; Aitken, J.; Gordon, L.; Green, A.C.; Janda, M.; Olsen, C.M.; Soyer, H.P.; on behalf of the Sunscreen Summit Policy Group. When to apply sunscreen: A consensus statement for Australia and New Zealand. Aust. N. Z. J. Public Health 2019, 43, 171–175. [Google Scholar] [CrossRef]
- Lund, P.M.; Taylor, J.S. Lack of Adequate Sun Protection for Children with Oculocutaneous Albinism in South Africa. BMC Public Health 2008, 8, 225. [Google Scholar] [CrossRef]
- Nkogatse, M.M.; Ramotsehoa, M.C.; Eloff, F.C.; Wright, C.Y. Solar ultraviolet radiation exposure and sun protection behaviours and knowledge among a high-risk and overlooked group of outdoor workers in South Africa. Photochem. Photobiol. 2019, 95, 439–445. [Google Scholar] [CrossRef]
- Makgabutlane, T.; Wright, C.Y. Real-time measurement of outdoor worker’s exposure to solar ultraviolet radiation in Pretoria. S. Afr. J. Sci. 2015, 111, 1–7. [Google Scholar] [CrossRef][Green Version]
- Modenese, A.; Farnetani, F.; Andreoli, A.; Pellacani, G.; Gobba, F. Questionnaire-based evaluation of occupational and non-occupational solar radiation exposure in a sample of Italian patients treated for actinic keratosis and other non-melanoma skin cancers. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 21–26. [Google Scholar] [CrossRef]
- Gies, P.; Glanz, K.; O’Riordan, D.; Elliott, T.; Nehl, E. Measured occupational solar UVR exposures of lifeguards in pool settings. Am. J. Indust. Med. 2009, 52, 645–653. [Google Scholar] [CrossRef]
- Hammond, V.; Reeder, A.I.; Gray, A. Patterns of real-time occupational ultraviolet radiation exposure among a sample of outdoor workers in New Zealand. Public Health 2009, 123, 182–187. [Google Scholar] [CrossRef]
- Glanz, K.; Geller, A.C.; Shigaki, D.; Maddock, J.E.; Isnec, M.R. A randomized trial of skin cancer prevention in aquatic settings: The pool cool program. Health Psychol. 2002, 21, 579–587. [Google Scholar] [CrossRef]
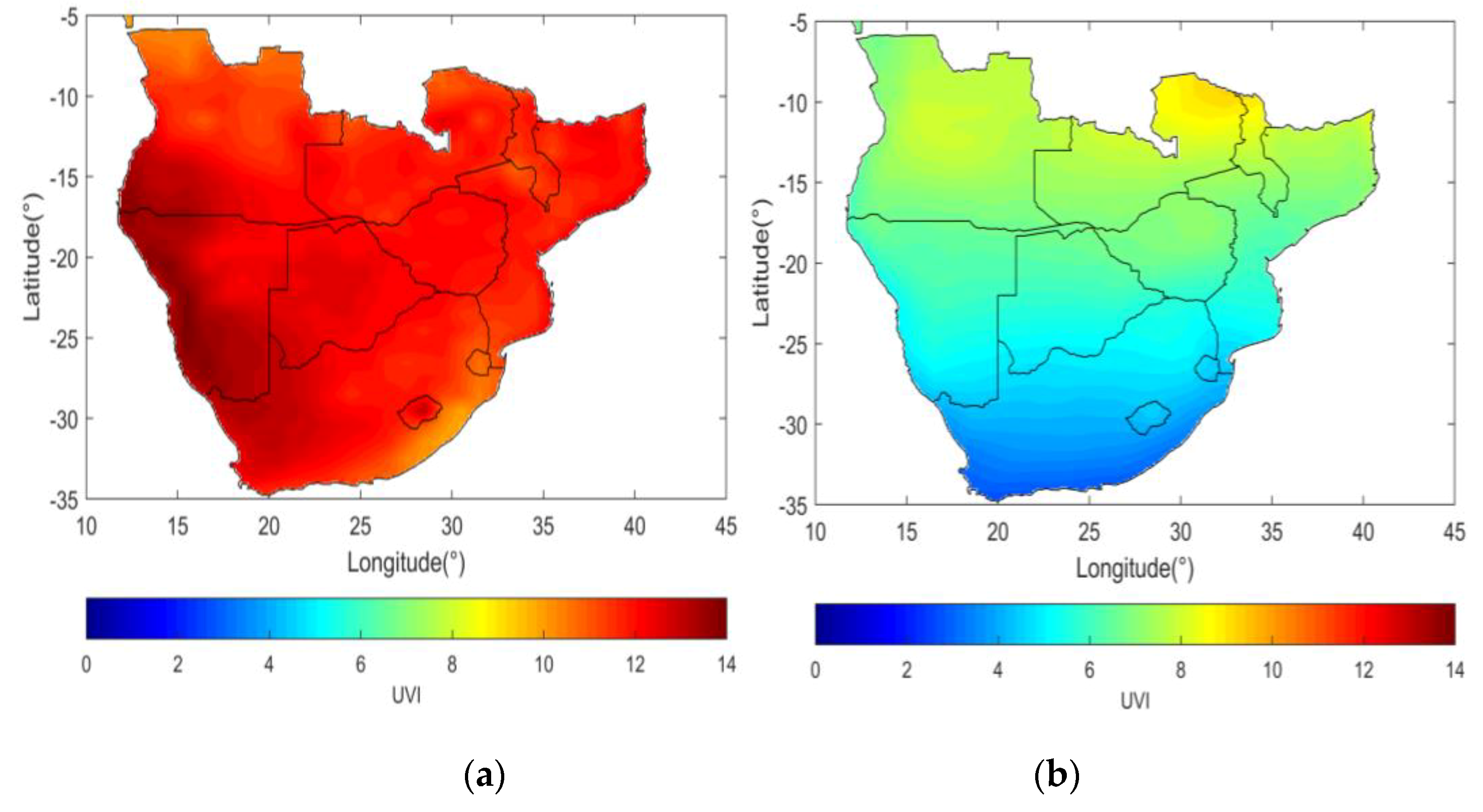
| Indicator | Botswana | Lesotho | Namibia | South Africa | Swaziland (eSwatini) |
|---|---|---|---|---|---|
| Number in population (2018) | 2.3 million | 2.1 million | 2.5 million | 58.8 million | 1.4 million |
| Percent population not Black African | 1.0% | 0.3% | 4.0% | 9.2% White; 8.8% Coloured; 2.6% Asian/Indian | 3.0% |
| Life expectancy, years (2018) | 67 | 54 | 64 | 63 | 58 |
| Prevalence HIV/AIDS in adults (2016–2018) | 25.0% | 24.0% | 12.6% | 19.0% | 27.2% |
| Death rate due to HIV/AIDS per 100,000 (2017) | 203.3 | 648.0 | 170.1 | 273.1 | 430.3 |
| Prevalence of albinism | 1 in 2243 in Southern Sotho people (in 1987) | 1 in 6000 (in 2014) | 1 in 2168 (in 2011) | 1 in 3900 (in 2006) | Not known |
| Access to and usage of Internet | 47% | 30% | 51% | 56% | 47% |
| Botswana | Lesotho | Namibia | South Africa | Swaziland (eSwatini) | |
|---|---|---|---|---|---|
| Capital city | Gaborone | Maseru | Windhoek | Pretoria | Mbabane |
| Estimated population in capital city (millions) | 0.2 | 0.1 | 0.3 | 1.6 | 0.1 |
| Latitude | 24.6° S | 29.3° S | 22.6° S | 25.7° S | 26.5° S |
| Altitude (metres) | 1010 | 1600 | 1655 | 1450 | 1200 |
| Summer mean noon UV Index | 12 | 13 | 13 | 12 | 10 |
| Winter mean noon UV Index | 5 | 4 | 7 | 5 | 3 |
| Mean day-time temperature in summer (°C) | 33 | 27 | 32 | 29 | 25 |
| Mean day-time temperature in winter (°C) | 23 | 15 | 22 | 20 | 20 |
| Botswana (in 2009–2013) | Namibia (in 2013–2015) | South Africa (in 2010–2014) | Swaziland (eSwatini) (in 2016–2017) | |
|---|---|---|---|---|
| Number of cases (KC) | ||||
| male | 176 | 391 | 59975 | 35 |
| female | 153 | 285 | 42550 | 44 |
| Number of cases (CM) | ||||
| male | 42 | 65 | 3583 | 5 |
| female | 71 | 93 | 3243 | 9 |
| Year National Cancer | 1999 | 1995 | 1986 | 2015 |
| Registry established | Population based | Pathology based plus clinical cases | Pathology based | Cases not reported centrally |
| Botswana | Lesotho | Namibia | South Africa | Swaziland (eSwatini) | |
|---|---|---|---|---|---|
| Number of skin cancer deaths | 46 | 47 | 19 | 1659 | 25 |
| Age-adjusted skin cancer deaths per 100,000 | 4.0 | 4.2 | 1.6 | 4.5 | 4.3 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wright, C.Y.; du Preez, D.J.; Millar, D.A.; Norval, M. The Epidemiology of Skin Cancer and Public Health Strategies for Its Prevention in Southern Africa. Int. J. Environ. Res. Public Health 2020, 17, 1017. https://doi.org/10.3390/ijerph17031017
Wright CY, du Preez DJ, Millar DA, Norval M. The Epidemiology of Skin Cancer and Public Health Strategies for Its Prevention in Southern Africa. International Journal of Environmental Research and Public Health. 2020; 17(3):1017. https://doi.org/10.3390/ijerph17031017
Chicago/Turabian StyleWright, Caradee Y., D. Jean du Preez, Danielle A. Millar, and Mary Norval. 2020. "The Epidemiology of Skin Cancer and Public Health Strategies for Its Prevention in Southern Africa" International Journal of Environmental Research and Public Health 17, no. 3: 1017. https://doi.org/10.3390/ijerph17031017
APA StyleWright, C. Y., du Preez, D. J., Millar, D. A., & Norval, M. (2020). The Epidemiology of Skin Cancer and Public Health Strategies for Its Prevention in Southern Africa. International Journal of Environmental Research and Public Health, 17(3), 1017. https://doi.org/10.3390/ijerph17031017






