Predictors of Plasma Fluoride Concentrations in Children and Adolescents
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Population Characteristics
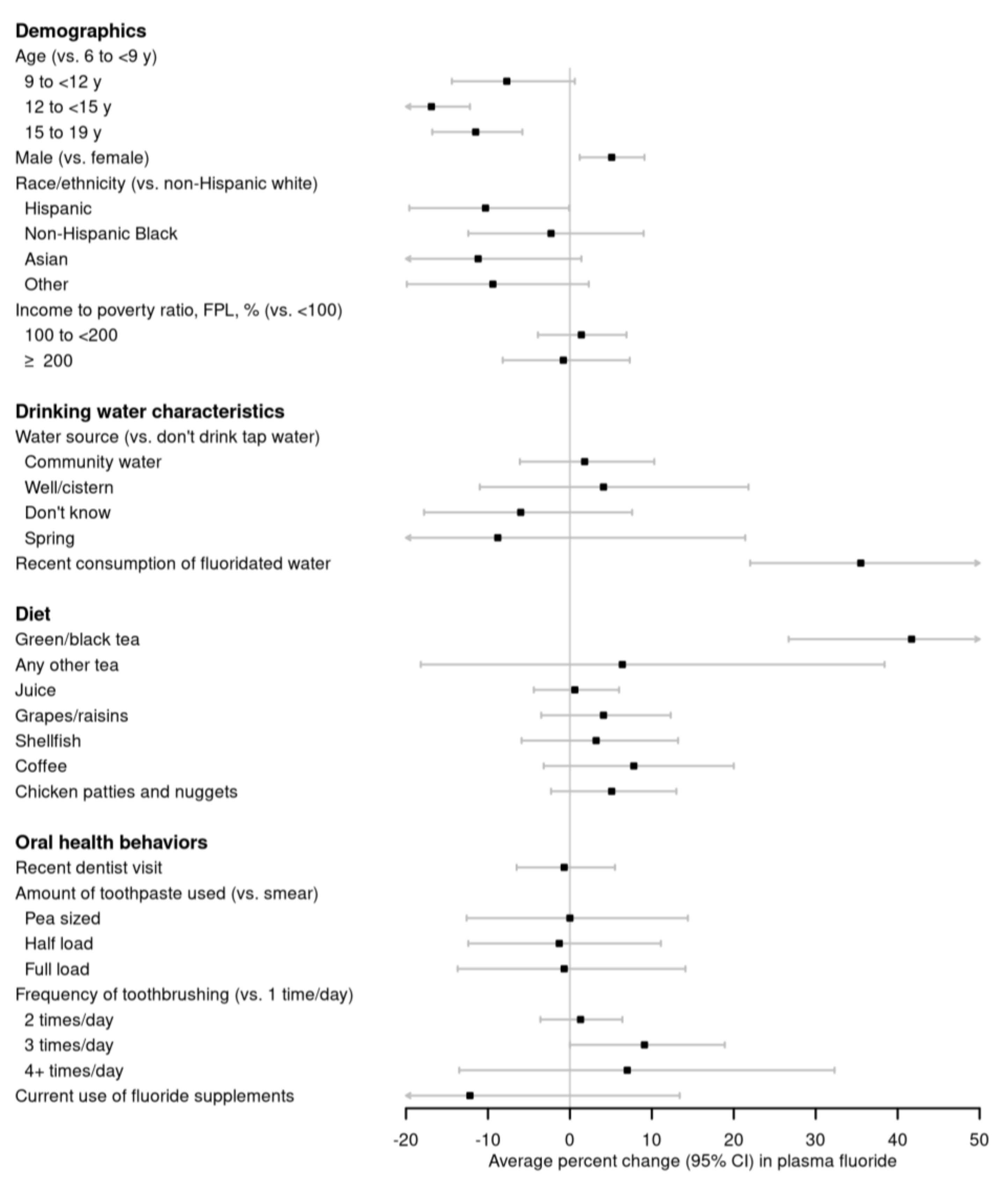
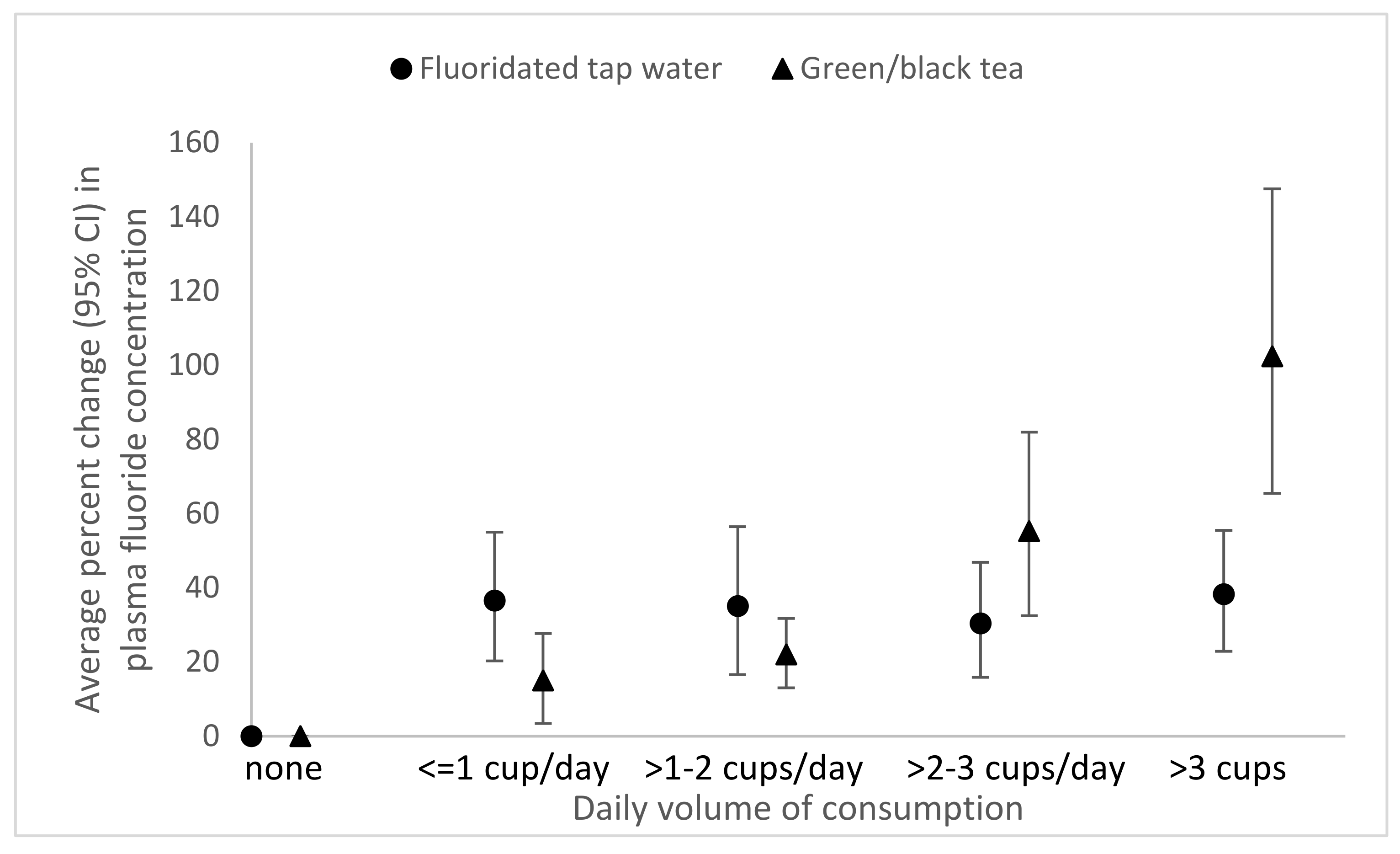
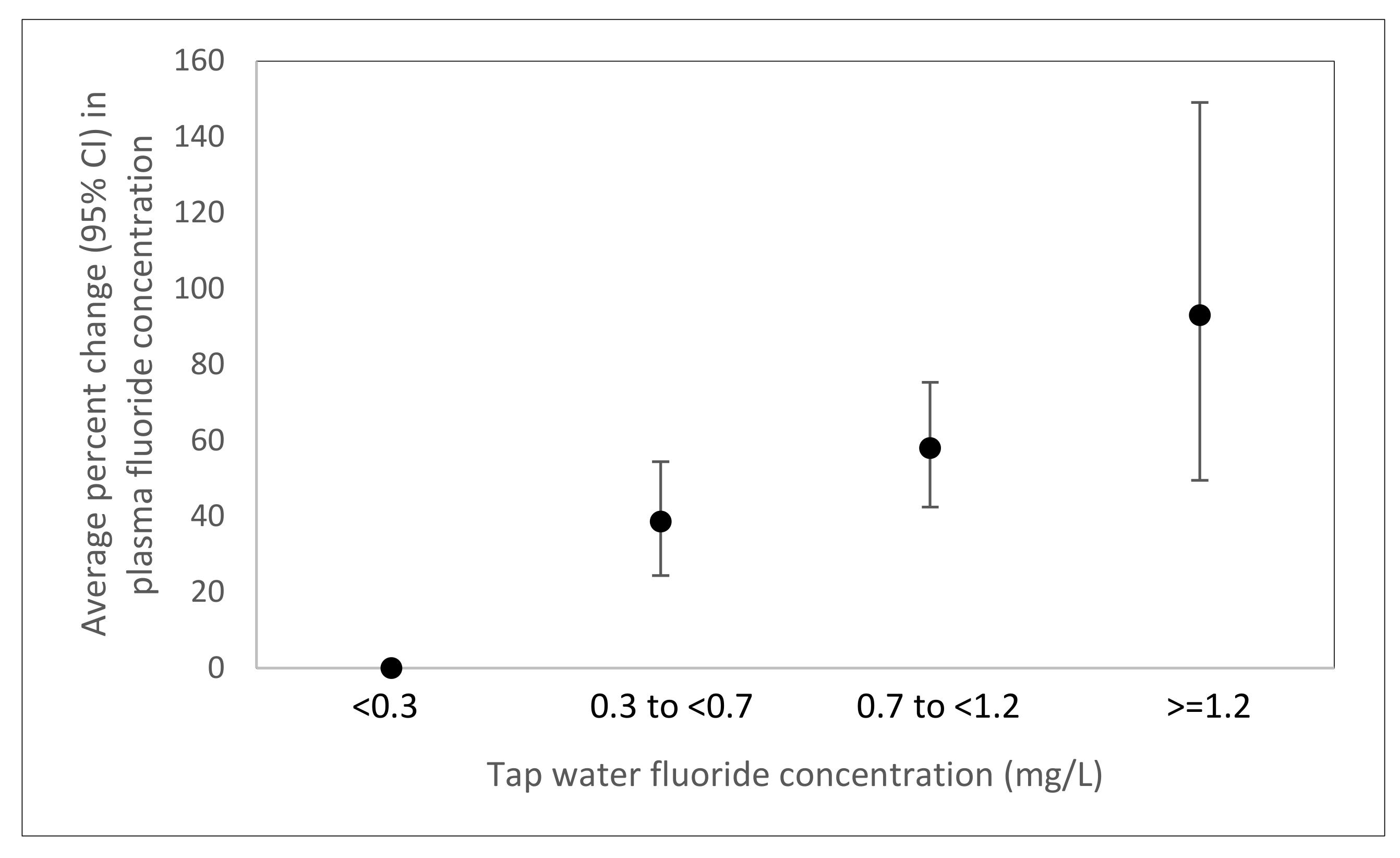
3.2. Associations of Demographics, Drinking Water Characteristics, Diet, and Oral Health Behaviors with Plasma Fluoride Concentration
3.3. Sensitivity Analyses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Percent Change (95% CI) in Plasma Fluoride | ||
|---|---|---|
| Characteristics | Unadjusted | Adjusted for Demographics, Fluoridated Tap Water Consumption, and Green/Black Tea Consumption 1 |
| Demographic Characteristics | ||
| Age categories | ||
| 6 to <9 years | Reference | Reference |
| 9 to <12 years | −5.1% (−11.5, 1.8) | −7.7% (−14.4, 0.6) |
| 12 to <15 years | −13.3% (−18.0, −8.3) | −16.9% (−21.4, −12.2) |
| 15 to <19 years | −7.9% (−15.0, −0.2) | −11.5% (−16.8, −5.8) |
| Sex | ||
| Girls | Reference | Reference |
| Boys | 5.8% (1.6, 10.1) | 5.1% (1.2, 9.1) |
| Race/ethnicity | ||
| Non-Hispanic white | Reference | Reference |
| Hispanic | −12.3% (−22.1, −1.4) | −10.3% (−19.6, −0.1) |
| Non-Hispanic Black | −4.0% (−14.5, 7.7) | −2.3% (−12.4, 9.0) |
| Asian | −10.8% (−22.4, 2.5) | −11.2% (−22.3, 1.4) |
| Other | −8.3% (−18.4, 3.1) | −9.4% (−19.9, 2.3) |
| Income to poverty ratio, FPL, % | ||
| <100 | Reference | Reference |
| 100 to <200 | 2.9% (−3.3, 9.4) | 1.4% (−3.9, 6.9) |
| ≥200 | 3.9% (−5.3, 14.0) | −0.8% (−8.2, 7.3) |
| Drinking Water Characteristics | ||
| Tap water source | ||
| Don’t drink tap water | Reference | Reference |
| Community supply | 8.4% (−1.1, 18.8) | 1.8% (−6.1, 10.3) |
| Well/cistern | 10.3% (−7.0, 30.9) | 4.1% (−11.0, 21.8) |
| Don’t know | 1.7% (−11.3, 16.8) | −6.0% (−17.8, 7.6) |
| Spring | −8.2% (−30.1, 20.6) | −8.8% (−31.4, 21.4) |
| Recent consumption of fluoridated home tap water (≥0.7 mg/L) | ||
| No | Reference | Reference |
| Yes | 34.8% (22.0, 49.0) | 35.5% (22.0, 50.5) |
| Recent Diet | ||
| Green/black tea | ||
| No consumption | Reference | Reference |
| Any consumption | 37.7% (22.1, 55.2) | 41.7% (26.7, 58.4) |
| Other tea | ||
| No consumption | Reference | Reference |
| Any consumption | 0.5% (−22.7, 30.7) | 6.4% (−18.2, 38.4) |
| Fruit juice | ||
| No consumption | Reference | Reference |
| Any consumption | −0.1% (−6.1, 6.3) | 0.6% (−4.4, 6.0) |
| Grapes/raisins | ||
| No consumption | Reference | Reference |
| Any consumption | 7.9% (−1.7, 18.4) | 4.1% (−3.5, 12.3) |
| Shellfish | ||
| No consumption | Reference | Reference |
| Any consumption | −2.9% (−11.4, 6.3) | 3.2% (−5.9, 13.2) |
| Coffee beverages | ||
| No consumption | Reference | Reference |
| Any consumption | 4.8% (−6.7, 17.7) | 7.8% (−3.2, 20.0) |
| Chicken nuggets, tenders, and patties | ||
| No consumption | Reference | Reference |
| Any consumption | 4.4% (−3.2, 12.7) | 5.1% (−2.3, 13.0) |
| Oral Health | ||
| Recent dentist visit (<6 months) | ||
| No | Reference | Reference |
| Yes | 1.0% (−5.5, 7.9) | −0.7% (−6.5, 5.5) |
| Amount of toothpaste used | ||
| Smear | Reference | Reference |
| Pea size | −2.5% (−14.9, 11.7) | 0.0% (−12.6, 14.4) |
| Half load | −4.5% (−16.2, 8.8) | −1.3% (−12.4, 11.1) |
| Full load | −4.6% (−17.2, 9.9) | −0.7% (−13.7, 14.1) |
| Frequency of tooth brushing, times per day | ||
| 1 | Reference | Reference |
| 2 | −1.4% (−6.5, 4.0) | 1.3% (−3.6, 6.4) |
| 3 | 3.0% (−8.0, 15.2) | 9.1% (0.0, 18.9) |
| ≥4 | −3.1% (−19.5, 16.6) | 7.0% (−13.5, 32.3) |
| Current use of fluoride drops or tablets 2 | ||
| No | Reference | Reference |
| Yes | −13.7% (−29.7, 5.9) | −12.2% (−32.0, 13.4) |
| Unweighted n (Weighted %) | ||
|---|---|---|
| Characteristics | Recent Black/Green Tea Consumption (n = 503) | No Recent Black/Green Tea Consumption (n = 3425) |
| Demographic Characteristics | ||
| Age categories | ||
| 6 to <9 years | 73 (12) | 781 (21) |
| 9 to <12 years | 99 (17) | 771 (21) |
| 12 to <15 years | 117 (23) | 736 (21) |
| 15 to <19 years | 214 (48) | 1137 (37) |
| Sex | ||
| Girls | 263 (51) | 1664 (47) |
| Boys | 240 (49) | 1761 (53) |
| Race/ethnicity 1 | ||
| Non-Hispanic white | 148 (52) | 887 (50) |
| Hispanic | 173 (25) | 1241 (26) |
| Non-Hispanic Black | 100 (11) | 808 (15) |
| Asian | 51 (5) | 287 (5) |
| Other | 31 (7) | 202 (5) |
| Income to poverty ratio, FPL, % 1 | ||
| <100 | 154 (22) | 1048 (23) |
| 100 to <200 | 119 (26) | 907 (23) |
| ≥200 | 230 (53) | 1470 (54) |
| Drinking Water Characteristics | ||
| Tap water source 1 | ||
| Don’t drink tap water | 74 (11) | 503 (12) |
| Community supply | 314 (65) | 2345 (72) |
| Well/cistern | 52 (13) | 193 (8) |
| Don’t know | 57 (11) | 326 (8) |
| Spring | 6 (1) | 58 (1) |
| Recent consumption of fluoridated home tap water (≥7 mg/L) | ||
| No | 454 (88) | 2914 (84) |
| Yes | 49 (12) | 511 (16) |
| Recent Diet | ||
| Other tea | ||
| No recent consumption | 498 (100) | 3394 (99) |
| Any recent consumption | 5 (0) | 31 (1) |
| Fruit juice | ||
| No recent consumption | 401 (81) | 2506 (75) |
| Any recent consumption | 102 (19) | 919 (25) |
| Grapes/raisins | ||
| No recent consumption | 483 (96) | 3213 (94) |
| Any recent consumption | 20 (4) | 212 (6) |
| Shellfish | ||
| No recent consumption | 488 (98) | 3348 (98) |
| Any recent consumption | 15 (2) | 77 (2) |
| Coffee beverages | ||
| No recent consumption | 470 (94) | 3239 (94) |
| Any recent consumption | 33 (6) | 186 (6) |
| Chicken nuggets, tenders, and patties | ||
| No recent consumption | 439 (85) | 3007 (88) |
| Any recent consumption | 64 (15) | 418 (12) |
| Oral Health | ||
| Recent dentist visit (<6 months) | ||
| No | 214 (43) | 1246 (35) |
| Yes | 286 (57) | 2171 (65) |
| Amount of toothpaste used | ||
| Smear | 11 (3) | 114 (4) |
| Pea size | 82 (17) | 742 (25) |
| Half load | 147 (32) | 1028 (29) |
| Full load | 258 (48) | 1496 (42) |
| Frequency of toothbrushing, times per day 1 | ||
| 1 | 170 (32) | 1040 (33) |
| 2 | 289 (62) | 2064 (62) |
| 3 | 32 (5) | 209 (5) |
| ≥4 | 2 (0) | 23 (1) |
| Current use of prescription fluoride drops or tablet 2 | ||
| No | 314 (98) | 2463 (98) |
| Yes | 4 (2) | 40 (2) |
References
- US Department of Health and Human Services Federal Panel on Community Water Fluoridation. U.S. Public Health Service Recommendation for Fluoride Concentration in Drinking Water for the Prevention of Dental Caries. Public Health Rep. 2015, 130, 318–331. [Google Scholar] [CrossRef]
- Green, R.; Lanphear, B.; Hornung, R.; Flora, D.; Martinez-Mier, E.A.; Neufeld, R.; Ayotte, P.; Muckle, G.; Till, C. Association Between Maternal Fluoride Exposure During Pregnancy and IQ Scores in Offspring in Canada. JAMA Pediatr. 2019, 173, 940–948. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Review of the Draft NTP Monograph: Systematic Review of Fluoride Exposure and Neurodevelopmental and Cognitive Health Effects; The National Academies Press: Washington, DC, USA, 2020. [Google Scholar] [CrossRef]
- Till, C.; Green, R.; Flora, D.; Hornung, R.; Martinez-Mier, E.A.; Blazer, M.; Farmus, L.; Ayotte, P.; Muckle, G.; Lanphear, B. Fluoride exposure from infant formula and child IQ in a Canadian birth cohort. Environ. Int. 2020, 134, 105315. [Google Scholar] [CrossRef]
- Grandjean, P. Developmental fluoride neurotoxicity: An updated review. Environ. Health 2019, 18, 110. [Google Scholar] [CrossRef]
- Bashash, M.; Thomas, D.; Hu, H.; Martinez-Mier, E.A.; Sanchez, B.N.; Basu, N.; Peterson, K.E.; Ettinger, A.S.; Wright, R.; Zhang, Z.; et al. Prenatal Fluoride Exposure and Cognitive Outcomes in Children at 4 and 6–12 Years of Age in Mexico. Environ. Health Perspect. 2017, 125, 097017. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.D.; Brizendine, E.J.; Kelly, S.A.; Hinesley, R.; Stookey, G.K.; Dunipace, A.J. The fluoride content of foods and beverages from negligibly and optimally fluoridated communities. Community Dent. Oral Epidemiol. 2002, 30, 382–391. [Google Scholar] [CrossRef] [PubMed]
- U.S. EPA. Fluoride: Exposure and Relative Source Contribution Analysis; 820-R-10-015; Health and Ecological Criteria Division, Office of Water, U.S. Environmental Protection Agency: Washington, DC, USA, 2010. Available online: https://www.epa.gov/sites/production/files/2019-03/documents/fluoride-exposure-relative-report.pdf (accessed on 23 April 2020).
- Lu, Y.; Guo, W.F.; Yang, X.Q. Fluoride content in tea and its relationship with tea quality. J. Agric. Food Chem. 2004, 52, 4472–4476. [Google Scholar] [CrossRef] [PubMed]
- Stannard, J.G.; Shim, Y.S.; Kritsineli, M.; Labropoulou, P.; Tsamtsouris, A. Fluoride levels and fluoride contamination of fruit juices. J. Clin. Pediatr. Dent. 1991, 16, 38–40. [Google Scholar] [PubMed]
- Heilman, J.R.; Kiritsy, M.C.; Levy, S.M.; Wefel, J.S. Fluoride concentrations of infant foods. J. Am. Dent. Assoc. 1997, 128, 857–863. [Google Scholar] [CrossRef]
- Levy, S.M.; Warren, J.J.; Davis, C.S.; Kirchner, H.L.; Kanellis, M.J.; Wefel, J.S. Patterns of fluoride intake from birth to 36 months. J. Public Health Dent. 2001, 61, 70–77. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC), National Center for Health Statistics (NCHS). National Health and Nutrition Examination Survey Demographic Variables and Sample Weights (DEMO_I); U.S. Department of Health and Human Services, Centers for Disease Control and Prevention: Hyattsville, MD, USA, 2017. Available online: https://wwwn.cdc.gov/nchs/nhanes/2015-2016/DEMO_I.htm (accessed on 23 April 2020).
- Agricultural Research Service Beltsville Human Nutrition Research Center, Food Surveys Research Group. USDA Automated Multiple-Pass Method for Dietary Recalls. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/ampm-usda-automated-multiple-pass-method/ (accessed on 23 April 2020).
- What We Eat in America, NHANES 2015–2016 Documentation: Dietary Interview-Ndividual Foods—First Day (DR1IFF_I); U.S. Department of Agriculture Agricultural Research Service, Beltsville Human Nutrition Research Center, Food Surveys Research Group: Beltsville, MD, USA; U.S. Department of Health and Human Services, Centers for Disease Conrol and Prevention, National Center for Health Statistics: Hyattsville, MD, USA, 2020. Available online: https://wwwn.cdc.gov/Nchs/Nhanes/2015-2016/DR1IFF_I.htm#Appendix_3 (accessed on 15 July 2018).
- Nutrient Data Laboratory. USDA National Fluoride Database of Selected Beverages and Foods—Release 2; Nutrient Data Laboratory, Beltsville Human Nutrition Research Center, ARS, USDA: Beltsville, MD, USA, 2005. Available online: https://www.ars.usda.gov/ARSUserFiles/80400525/Data/Fluoride/F02.pdf (accessed on 23 April 2020).
- What We Eat in America, NHANES: Dietary Methods Research; U.S. Department of Agriculture Agricultural Research Service, Beltsville Human Nutrition Research Center, Food Surveys Research Group: Beltsville, MD, USA; U.S. Department of Health and Human Services, Centers for Disease Conrol and Prevention, National Center for Health Statistics: Hyattsville, MD, USA, 2016. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/food-surveys-research-group/docs/dmr-food-categories/ (accessed on 23 April 2020).
- Waugh, D.T.; Godfrey, M.; Limeback, H.; Potter, W. Black Tea Source, Production, and Consumption: Assessment of Health Risks of Fluoride Intake in New Zealand. J. Environ. Public Health 2017, 2017, 5120504. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC), National Center for Health Statistics (NCHS). National Health and Nutrition Examination Survey Tap Water Fluoride Laboratory Procedure Manual; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention: Hyattsville, MD, USA, 2017. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2015-2016/labmethods/FLDEW_I_MET.pdf (accessed on 2 February 2020).
- Centers for Disease Control and Prevention (CDC), National Center for Health Statistics (NCHS). National Health and Nutrition Examination Survey Plasma Fluoride Laboratory Procedure Manual; U.S. Department of Health and Human Services, Centers for Disease Control and Prevention: Hyattsville, MD, USA, 2017. Available online: https://wwwn.cdc.gov/nchs/data/nhanes/2015-2016/labmethods/FLDEP_I_MET.pdf (accessed on 2 February 2020).
- Ekstrand, J.; Ehrnebo, M. The relationship between plasma fluoride, urinary excretion rate and urine fluoride concentration in man. J. Occup. Med. 1983, 25, 745–748. [Google Scholar] [CrossRef] [PubMed]
- National Research Council (NRC). Fluoride in Drinking Water: A Scientific Review of EPA’s Standards; The National Academies Press: Washington, DC, USA, 2006.
- Kleinbaum, D.G.; Kupper, L.L.; Nizam, A.; Muller, K. Applied Regression Analysis and Other Multivariable Methods, 4th ed.; Duxbury: Belmont, MA, USA, 2008. [Google Scholar]
- Malinowska, E.; Inkielewicz, I.; Czarnowski, W.; Szefer, P. Assessment of fluoride concentration and daily intake by human from tea and herbal infusions. Food Chem. Toxicol. 2008, 46, 1055–1061. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.L.; Sun, G.; Zhang, Y.; Grandjean, P. Developmental fluoride neurotoxicity: A systematic review and meta-analysis. Environ. Health Perspect. 2012, 120, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.B. Concentrations of fluoride in water and plasma for US children and adolescents: Data from NHANES 2013–2014. Environ. Toxicol. Pharmacol. 2017, 50, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Till, C.; Green, R.; Grundy, J.G.; Hornung, R.; Neufeld, R.; Martinez-Mier, E.A.; Ayotte, P.; Muckle, G.; Lanphear, B. Community Water Fluoridation and Urinary Fluoride Concentrations in a National Sample of Pregnant Women in Canada. Environ. Health Perspect. 2018, 126, 107001. [Google Scholar] [CrossRef] [PubMed]
- Waugh, D.T.; Potter, W.; Limeback, H.; Godfrey, M. Risk Assessment of Fluoride Intake from Tea in the Republic of Ireland and its Implications for Public Health and Water Fluoridation. Int. J. Environ. Res. Public Health 2016, 13, 259. [Google Scholar] [CrossRef]
- Whyte, M.P.; Essmyer, K.; Gannon, F.H.; Reinus, W.R. Skeletal fluorosis and instant tea. Am. J. Med. 2005, 118, 78–82. [Google Scholar] [CrossRef]
- Zhang, R.; Cheng, L.; Zhang, T.; Xu, T.; Li, M.; Yin, W.; Jiang, Q.; Yang, Y.; Hu, T. Brick tea consumption is a risk factor for dental caries and dental fluorosis among 12-year-old Tibetan children in Ganzi. Environ. Geochem. Health 2019, 41, 1405–1417. [Google Scholar] [CrossRef]
- Ramesh, M.; Narasimhan, M.; Krishnan, R.; Chalakkal, P.; Aruna, R.M.; Kuruvilah, S. The prevalence of dental fluorosis and its associated factors in Salem district. Contemp. Clin. Dent. 2016, 7, 203–208. [Google Scholar] [CrossRef]
- Cao, J.; Zhao, Y.; Liu, J. Brick tea consumption as the cause of dental fluorosis among children from Mongol, Kazak and Yugu populations in China. Food Chem. Toxicol. 1997, 35, 827–833. [Google Scholar] [CrossRef]
- Jin, C.; Yan, Z.; Jian-Wei, L.; Ruoden, X.; Sangbu, D.; Zeguo; Zhouma, S. Prevention and control of brick-tea type fluorosis--a 3-year observation in Dangxiong, Tibet. Ecotoxicol. Environ. Saf. 2003, 56, 222–227. [Google Scholar] [CrossRef]
- Trautner, K.; Siebert, G. An experimental study of bio-availability of fluoride from dietary sources in man. Arch. Oral. Biol. 1986, 31, 223–228. [Google Scholar] [CrossRef]
- Rozier, R.G.; Adair, S.; Graham, F.; Iafolla, T.; Kingman, A.; Kohn, W.; Krol, D.; Levy, S.; Pollick, H.; Whitford, G.; et al. Evidence-based clinical recommendations on the prescription of dietary fluoride supplements for caries prevention: A report of the American Dental Association Council on Scientific Affairs. J. Am. Dent. Assoc. 2010, 141, 1480–1489. [Google Scholar] [CrossRef]
- Levy, S.M.; Warren, J.J.; Broffitt, B. Patterns of fluoride intake from 36 to 72 months of age. J. Public Health Dent. 2003, 63, 211–220. [Google Scholar] [CrossRef]
- Bohaty, B.S.; Parker, W.A.; Seale, N.S.; Zimmerman, E.R. The prevalence of fluorosis-like lesions associated with topical and systemic fluoride usage in an area of optimal water fluoridation. Pediatr. Dent. 1989, 11, 125–128. [Google Scholar]
- Franzman, M.R.; Levy, S.M.; Warren, J.J.; Broffitt, B. Fluoride dentifrice ingestion and fluorosis of the permanent incisors. J. Am. Dent. Assoc. 2006, 137, 645–652. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention Division of Oral Health, National Center for Chronic Disease Prevention and Health Promotion. 2016 Fluoridation Statistics. Available online: https://www.cdc.gov/fluoridation/statistics/2016stats.htm (accessed on 28 April 2020).
- Duperon, D.F.; Jedrychowski, J.R.; Kong, J. Fluoride content of Los Angeles County water. J. Calif. Dent. Assoc. 1995, 23, 45–46. [Google Scholar]
| Unweighted n (Weighted %) | |
|---|---|
| Overall | 3928 (100) |
| Demographic Characteristics | |
| Age categories | |
| 6 to <9 years | 854 (20) |
| 9 to <12 years | 870 (21) |
| 12 to <15 years | 853 (21) |
| 15 to 19 years | 1351 (38) |
| Boys | 2001 (52) |
| Race/ethnicity 1 | |
| Non-Hispanic white | 1035 (51) |
| Hispanic | 1414 (26) |
| Non-Hispanic Black | 908 (14) |
| Asian | 338 (5) |
| Other | 233 (5) |
| Income to poverty ratio, FPL, % | |
| <100 | 1202 (23) |
| 100 to <200 | 1026 (23) |
| ≥200 | 1700 (54) |
| Drinking Water Characteristics | |
| Tap water source 1 | |
| Don’t drink tap water | 577 (12) |
| Community supply | 2659 (71) |
| Well/cistern | 245 (9) |
| Don’t know | 383 (8) |
| Spring | 64 (1) |
| Recent consumption of any home tap water | 1820 (54) |
| Recent consumption of fluoridated home tap water (≥0.7 mg/L) | 560 (16) |
| Recent Diet | |
| Black or green tea beverages, hot or iced | 503 (13) |
| Other tea beverages, including herbal | 36 (1) |
| Fruit juice | 1021 (24) |
| Grapes/raisins | 232 (6) |
| Shellfish | 92 (2) |
| Coffee | 219 (6) |
| Chicken nuggets, tenders, and patties | 482 (12) |
| Oral Health Behaviors | |
| Recent dentist visit (<6 months) | 2457 (64) |
| Amount of toothpaste used 1 | |
| Smear | 125 (4) |
| Pea size | 824 (24) |
| Half load | 1175 (30) |
| Full load | 1754 (43) |
| Frequency of tooth brushing, times per day 1 | |
| 1 | 1210 (33) |
| 2 | 2353 (62) |
| 3 | 241 (5) |
| ≥4 | 25 (1) |
| Current use of prescription fluoride drops or tablet 2 | 44 (2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carwile, J.L.; Ahrens, K.A.; Seshasayee, S.M.; Lanphear, B.; Fleisch, A.F. Predictors of Plasma Fluoride Concentrations in Children and Adolescents. Int. J. Environ. Res. Public Health 2020, 17, 9205. https://doi.org/10.3390/ijerph17249205
Carwile JL, Ahrens KA, Seshasayee SM, Lanphear B, Fleisch AF. Predictors of Plasma Fluoride Concentrations in Children and Adolescents. International Journal of Environmental Research and Public Health. 2020; 17(24):9205. https://doi.org/10.3390/ijerph17249205
Chicago/Turabian StyleCarwile, Jenny L., Katherine A. Ahrens, Shravanthi M. Seshasayee, Bruce Lanphear, and Abby F. Fleisch. 2020. "Predictors of Plasma Fluoride Concentrations in Children and Adolescents" International Journal of Environmental Research and Public Health 17, no. 24: 9205. https://doi.org/10.3390/ijerph17249205
APA StyleCarwile, J. L., Ahrens, K. A., Seshasayee, S. M., Lanphear, B., & Fleisch, A. F. (2020). Predictors of Plasma Fluoride Concentrations in Children and Adolescents. International Journal of Environmental Research and Public Health, 17(24), 9205. https://doi.org/10.3390/ijerph17249205





