The Relationship between Simple Snoring and Sleep Bruxism: A Polysomnographic Study
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Counter, P.; Wilson, J.A. The Management of Simple Snoring. Sleep Med. Rev. 2004, 8, 433–441. [Google Scholar] [CrossRef]
- De Meyer, M.M.D.; Jacquet, W.; Vanderveken, O.M.; Marks, L.A.M. Systematic Review of the Different Aspects of Primary Snoring. Sleep Med. Rev. 2019, 45, 88–94. [Google Scholar] [CrossRef]
- Deary, V.; Ellis, J.G.; Wilson, J.A.; Coulter, C.; Barclay, N.L. Simple Snoring: Not Quite so Simple after All? Sleep Med. Rev. 2014, 18, 453–462. [Google Scholar] [CrossRef]
- Kalchiem-Dekel, O.; Westreich, R.; Regev, A.; Novack, V.; Goldberg, M.; Maimon, N. Snoring Intensity and Excessive Daytime Sleepiness in Subjects Without Obstructive Sleep Apnea. Laryngoscope 2016, 126, 1696–1701. [Google Scholar] [CrossRef]
- Lee, S.A.; Amis, T.C.; Byth, K.; Larcos, G.; Kairaitis, K.; Robinson, T.D.; Wheatley, J.R. Heavy snoring as a cause of carotid artery atherosclerosis. Sleep 2008, 31, 1207–1213. [Google Scholar]
- Sands, M.; Loucks, E.B.; Lu, B.; Carskadon, M.A.; Sharkey, K.; Stefanick, M.; Ockene, J.; Shah, N.; Hairston, K.G.; Robinson, J.; et al. Self-reported Snoring and Risk of Cardiovascular Disease Among Postmenopausal Women (from the Women’s Health Initiative). Am. J. Cardiol. 2013, 111, 540–546. [Google Scholar] [CrossRef]
- Telakivi, T. Snoring as a Risk Factor for Ischaemic Heart Disease and Stroke in Men. Br. Med. J. (Clin. Res. Ed.) 1987, 294, 643. [Google Scholar] [CrossRef][Green Version]
- Zou, J.; Song, F.; Xu, H.; Fu, Y.; Xia, Y.; Qian, Y.; Zou, J.; Liu, S.; Fang, F.; Meng, L.; et al. The Relationship between Simple Snoring and Metabolic Syndrome: A Cross-Sectional Study. J. Diabetes Res. 2019, 2019, 9578391. [Google Scholar] [CrossRef]
- Rich, J.; Raviv, A.; Raviv, N.; Brietzke, S.E. An Epidemiologic Study of Snoring and All-cause Mortality. Otolaryngol. Head Neck Surg. 2011, 145, 341–346. [Google Scholar] [CrossRef]
- Meira e Cruz, M.; Soca, R.; Kryger, M. How Much is Too Much After All? Primary Snoring as a Remaining Unsolved Issue. J. Clin. Sleep Med. 2020, 8442. [Google Scholar] [CrossRef]
- American Academy of Sleep Medicine. International Classification of Sleep Disorders, 3rd ed.; American Academy of Sleep Medicine: Darien, IL, USA, 2014. [Google Scholar]
- Guzman, M.A.; Sgambati, F.P.; Pho, H.; Guzman, M.A.; Sgambati, F.P.; Pho, H.; Arias, R.S.; Hawks, E.M.; Wolfe, E.M.; Ötvös, T.; et al. The Efficacy of Low-level Continuous Positive Airway Pressure for the Treatment of Snoring. J. Clin. Sleep Med. 2017, 13, 703–711. [Google Scholar] [CrossRef][Green Version]
- Snoring Frequency Evaluation and Reporting. Available online: https://support.noxmedical.com/hc/en-us/articles/115000068594-Snoring-Frequency-Evaluation-and-Reporting (accessed on 20 November 2020).
- Torzsa, P.; Keszei, A.; Kalabay, L.; Vamos, E.P.; Zoller, R.; Mucsi, I.; Novak, M.; Kopp, M.S. Socio-demographic Characteristics, Health Behaviour, Co-morbidity and Accidents in Snorers: A Population Survey. Sleep Breath. 2011, 15, 809–818. [Google Scholar] [CrossRef]
- Lobbezoo, F.; Ahlberg, J.; Raphael, K.G.; Wetselaar, P.; Glaros, A.G.; Kato, T.; Santiago, V.; Winocur, E.; De Laat, A.; De Leeuw, R.; et al. International Consensus on the Assessment of Bruxism: Report of a Work in Progress. J. Oral Rehabil. 2018, 45, 837–844. [Google Scholar] [CrossRef]
- Beddis, H.; Pemberton, M.; Davies, S. Sleep Bruxism: An Overview for Clinicians. Br. Dent. J. 2018, 225, 497–501. [Google Scholar] [CrossRef]
- Wieckiewicz, M.; Bogunia-Kubik, K.; Mazur, G.; Danel, D.; Smardz, J.; Wojakowska, A.; Poreba, R.; Dratwa, M.; Chaszczewska-Markowska, M.; Winocur, E.; et al. Genetic Basis of Sleep Bruxism and Sleep Apnea—Response to a Medical Puzzle. Sci. Rep. 2020, 10, 7497. [Google Scholar] [CrossRef]
- Jokubauskas, L.; Baltrušaitytė, A. Relationship between Obstructive Sleep Apnoea Syndrome and Sleep Bruxism: A Systematic Review. J. Oral. Rehabil. 2017, 44, 144–153. [Google Scholar] [CrossRef]
- Martynowicz, H.; Gac, P.; Brzecka, A.; Poreba, R.; Wojakowska, A.; Mazur, G.; Smardz, J.; Wieckiewicz, M. The Relationship between Sleep Bruxism and Obstructive Sleep Apnea Based on Polysomnographic Findings. J. Clin. Med. 2019, 8, 1653. [Google Scholar] [CrossRef]
- Ohayon, M.M.; Li, K.K.; Guilleminault, C. Risk Factors for Sleep Bruxism in the General Population. Chest 2001, 119, 53–61. [Google Scholar] [CrossRef]
- Miyawaki, S.; Tanimoto, Y.; Araki, Y.; Katayama, A.; Imai, M.; Takano-Yamamoto, T. Relationships among Nocturnal Jaw Muscle Activities, Decreased Esophageal pH, and Sleep Positions. Am. J. Orthod. Dentofac. Orthop. 2004, 126, 615–619. [Google Scholar] [CrossRef]
- Miyawaki, S.; Tanimoto, Y.; Araki, Y.; Katayama, A.; Fujii, A.; Takano-Yamamoto, T. Association between Nocturnal Bruxism and Gastroesophageal Reflux. Sleep 2003, 26, 888–892. [Google Scholar] [CrossRef]
- Martynowicz, H.; Dymczyk, P.; Dominiak, M.; Martynowicz, H.; Dymczyk, P.; Dominiak, M.; Kazubowska, K.; Skomro, R.; Poreba, R.; Gac, P.; et al. Evaluation of Intensity of Sleep Bruxism in Arterial Hypertension. J. Clin. Med. 2018, 7, 327. [Google Scholar] [CrossRef]
- Nashed, A.; Lanfranchi, P.; Rompré, P.; Carra, M.C.; Mayer, P.; Colombo, R.; Huynh, N.; Lavigne, G. Sleep Bruxism Is Associated with a Rise in Arterial Blood Pressure. Sleep 2012, 35, 529–536. [Google Scholar] [CrossRef]
- Benjafield, A.V.; Ayas, N.T.; Eastwood, P.R.; Heinzer, R.; Ip, M.; Morrell, M.J.; Nunez, C.M.; Patel, S.R.; Penzel, T.; Pépin, J.L.; et al. Estimation of the Global Prevalence and Burden of Obstructive Sleep Apnoea: A Literature-based Analysis. Lancet Respir. Med. 2019, 7, 687–698. [Google Scholar] [CrossRef]
- Hollowell, D.E.; Bhandary, P.R.; Funsten, A.W.; Suratt, P.M. Respiratory-related Recruitment of the Masseter: Response to Hypercapnia and Loading. J. Appl. Physiol. 1991, 70, 2508–2513. [Google Scholar] [CrossRef]
- Phillips, B.A.; Okeson, J.; Paesani, D.; Gilmore, R. Effect of Sleep Position on Sleep Apnea and Parafunctional Activity. Chest 1986, 90, 424–429. [Google Scholar] [CrossRef]
- Hosoya, H.; Kitaura, H.; Hashimoto, T.; Ito, M.; Kinbara, M.; Deguchi, T.; Irokawa, T.; Ohisa, N.; Ogawa, H.; Takano-Yamamoto, T. Relationship between Sleep Bruxism and Sleep Respiratory Events in Patients with Obstructive Sleep Apnea Syndrome. Sleep Breath. 2014, 18, 837–844. [Google Scholar] [CrossRef]
- Costa Lopes, A.J.; Cunha, T.C.A.; Monteiro, M.C.M.; Serra-Negra, J.M.; Cabral, L.C.; Júnior, P.C.S. Is there an Association between Sleep Bruxism and Obstructive Sleep Apnea Syndrome? A systematic review. Sleep Breath. Schlaf Atm. 2020, 24, 913–921. [Google Scholar] [CrossRef]
- Saito, M.; Yamaguchi, T.; Mikami, S.; Watanabe, K.; Gotouda, A.; Okada, K.; Hishikawa, R.; Shibuya, E.; Shibuya, Y.; Lavigne, G. Weak Association between Sleep Bruxism and Obstructive Sleep Apnea. A Sleep Laboratory Study. Sleep Breath. 2016, 20, 703–709. [Google Scholar] [CrossRef]
- Okeson, J.P.; Phillips, B.A.; Berry, D.T.; Cook, Y.R.; Cabelka, J.F. Nocturnal Bruxing Events in Subjects with Sleep-disordered Breathing and Control Subjects. J. Craniomandib. Disord. 1991, 5, 258–264. [Google Scholar]
- McNicholas, W.T. Diagnosis of Obstructive Sleep Apnea in Adults. Proc. Am. Thorac. Soc. 2008, 5, 154–160. [Google Scholar] [CrossRef]
- Reutrakul, S.; Mokhlesi, B. Obstructive Sleep Apnea and Diabetes: A State of the Art Review. Chest 2017, 152, 1070–1086. [Google Scholar] [CrossRef]
- Hou, H.; Zhao, Y.; Yu, W.; Dong, H.; Xue, X.; Ding, J.; Xing, W.; Wang, W. Association of Obstructive Sleep Apnea with Hypertension: A Systematic Review and Meta-analysis. J. Glob. Health 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Munoz, R.; Duran-Cantolla, J.; Martínez-Vila, E.; Gallego, J.; Rubio, R.; Aizpuru, F.; De La Torre, G. Severe Sleep Apnea and Risk of Ischemic Stroke in the Elderly. Stroke 2006, 37, 2317–2321. [Google Scholar] [CrossRef]
- Vasheghani-Farahani, A.; Kazemnejad, F.; Sadeghniiat-Haghighi, K.; Saadat, S.; Tavakolipoor, P.; Yazdani, T.; Alidoosti, M.; Ghasem-Amooeian, V.; Ashraf, H. Obstructive Sleep Apnea and Severity of Coronary Artery Disease. Casp. J. Intern. Med. 2018, 9, 276–282. [Google Scholar] [CrossRef]
- Raghuram, A.; Clay, R.; Kumbam, A.; Tereshchenko, L.G.; Khan, A. A Systematic Review of the Association Between Obstructive Sleep Apnea and Ventricular Arrhythmias. J. Clin. Sleep Med. 2014, 10, 1155–1160. [Google Scholar] [CrossRef]
- Salama, A.; Abdullah, A.; Wahab, A.; Eigbire, G.; Hoefen, R.; Kouides, R.; Ritter, N.; Mieszczanska, H.; Alweis, R. Is Obstructive Sleep Apnea Associated with Ventricular Tachycardia? A Retrospective Study from the National Inpatient Sample and a Literature Review on the Pathogenesis of Obstructive Sleep Apnea. Clin. Cardiol. 2018, 41, 1543–1547. [Google Scholar] [CrossRef]
- Mehra, R.; Benjamin, E.J.; Shahar, E.; Gottlieb, D.J.; Nawabit, R.; Kirchner, H.L.; Sahadevan, J.; Redline, S.; Sleep Heart Health Study. Association of Nocturnal Arrhythmias with Sleep-Disordered Breathing: The Sleep Heart Health Study. Am. J. Respir. Crit. Care Med. 2006, 173, 910–916. [Google Scholar] [CrossRef]
- Jones, S. Sleep Disordered Breathing and Mortality: Eighteen-Year Follow-up of the Wisconsin Sleep Cohort. Yearb. Pulm. Dis. 2009, 2009, 291–292. [Google Scholar] [CrossRef]
- Sateia, M.J. International Classification of Sleep Disorders-Third Edition Highlights and Modifications. Chest 2014, 146, 1387–1394. [Google Scholar] [CrossRef]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for Scoring Respiratory Events in Sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. J. Clin. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef]
- Thetakala, R.; Chandrashekar, B.; Sunitha, S.; Maurya, M.; Sharma, P.; Shubhi, G. Bruxism and Oral Health-related Quality of Life among Male Inmates in a Penal Institution, Mysore: A Cross-sectional Study. Indian J. Dent. Res. 2018, 29, 275–279. [Google Scholar] [CrossRef]
- Câmara-Souza, M.B.; de Figueredo, O.M.C.; Rodrigues Garcia, R.C.M. Association of Sleep Bruxism with Oral Health-related Quality of Life and Sleep Quality. Clin. Oral Investig. 2019, 23, 245–251. [Google Scholar] [CrossRef]
- Manfredini, D.; Ahlberg, J.; Wetselaar, P.; Svensson, P.; Lobbezoo, F. The Bruxism Construct: From Cut-off Points to a Continuum Spectrum. J. Oral Rehabil. 2019, 46, 991–997. [Google Scholar] [CrossRef]
- Lobbezoo, F.; Ahlberg, J.; Glaros, A.G.; Kato, T.; Koyano, K.; Lavigne, G.J.; de Leeuw, R.; Manfredini, D.; Svensson, P.; Winocur, E. Bruxism Defined and Graded: An International Consensus. J. Oral Rehabil. 2013, 40, 2–4. [Google Scholar] [CrossRef]
- Ohmure, H.; Oikawa, K.; Kanematsu, K.; Saito, Y.; Yamamoto, T.; Nagahama, H.; Tsubouchi, H.; Miyawaki, S. Influence of Experimental Esophageal Acidification on Sleep Bruxism: A Randomized Trial. J. Dent. Res. 2011, 90, 665–671. [Google Scholar] [CrossRef]
- Palinkas, M.; Marrara, J.; Bataglion, C.; Hallak, J.; Canto, G.D.; Scalize, P.H.; Regalo, I.; Siessere, S.; Regalo, S. Analysis of the Sleep Period and the Amount of Habitual Snoring in Individuals with Sleep Bruxism. Med. Oral Patol. Oral Cir. Bucal 2019, 24, 782–786. [Google Scholar] [CrossRef]
- Nakano, H.; Ikeda, T.; Hayashi, M.; Ohshima, E.; Onizuka, A. Effects of Body Position on Snoring in Apneic and Nonapneic Snorers. Sleep 2003, 26, 169–172. [Google Scholar] [CrossRef][Green Version]
- Menon, A.; Kumar, M. Influence of Body Position on Severity of Obstructive Sleep Apnea: A Systematic Review. ISRN Otolaryngol. 2013, 2013, 670381. [Google Scholar] [CrossRef]
- Miyawaki, S.; Lavigne, G.J.; Mayer, P.; Guitard, F.; Montplaisir, J.Y.; Kato, T. Association between Sleep Bruxism, Swallowing-Related Laryngeal Movement, and Sleep Positions. Sleep 2003, 26, 461–465. [Google Scholar] [CrossRef]
- Tan, M.; Yap, A.; Chua, A.; Wong, J.; Parot, M.; Tan, K. Prevalence of Sleep Bruxism and Its Association with Obstructive Sleep Apnea in Adult Patients: A Retrospective Polysomnographic Investigation. J. Oral Facial Pain Headache 2019, 33, 269–277. [Google Scholar] [CrossRef]
- Smardz, J.; Martynowicz, H.; Wojakowska, A.; Michalek-Zrabkowska, M.; Mazur, G.; Wieczorek, T.; Wieckiewicz, M. The Meaning of the Masticatory Muscle Tonic-type Electromyographic Pathway Correlated with Sleep Bruxism and Sleep-related Breathing Disorders—A Polysomnographic Study. Sleep Med. 2020, 68, 131–137. [Google Scholar] [CrossRef]
- Dumais, I.E.; Lavigne, G.J.; Carra, M.C.; Rompré, P.H.; Huynh, N.T. Could Transient Hypoxia Be Associated with Rhythmic Masticatory Muscle Activity in Sleep Bruxism in the Absence of Sleep-disordered Breathing? A Preliminary Report. J. Oral Rehabil. 2015, 42, 810–818. [Google Scholar] [CrossRef]
- Roth, T. Insomnia: Definition, Prevalence, Etiology, and Consequences. J. Clin. Sleep Med. 2007, 3 (Suppl. 5), S7. [Google Scholar] [CrossRef]
- Macaluso, G.M.; Guerra, P.; Di Giovanni, G.; Boselli, M.; Parrino, L.; Terzano, M.G. Sleep Bruxism is a Disorder Related to Periodic Arousals During Sleep. J. Dent. Res. 1998, 77, 565–573. [Google Scholar] [CrossRef]
- Lavigne, G.J.; Khoury, S.; Abe, S.; Yamaguchi, T.; Raphael, K. Bruxism Physiology and Pathology: An Overview for Clinicians. J. Oral Rehabil. 2008, 35, 476–494. [Google Scholar] [CrossRef]
- Maluly, M.; Andersen, M.L.; Dal-Fabbro, C.; Garbuio, S.; Bittencourt, L.; de Siqueira, J.T.; Tufik, S. Polysomnographic Study of the Prevalence of Sleep Bruxism in a Population Sample. J. Dent. Res. 2013, 92, 97–103. [Google Scholar] [CrossRef]
- Hertig-Godeschalk, A.; Skorucak, J.; Malafeev, A.; Achermann, P.; Mathis, J.; Schreier, D.R. Microsleep Episodes in the Borderland between Wakefulness and Sleep. Sleep 2020, 43, 163. [Google Scholar] [CrossRef]
- Ahlberg, K.; Jahkola, A.; Savolainen, A.; Könönen, M.; Partinen, M.; Hublin, C.; Sinisalo, J.; Lindholm, H.; Sarna, S.; Ahlberg, J. Associations of Reported Bruxism with Insomnia and Insufficient Sleep Symptoms among Media Personnel with or without Irregular Shift Work. Head Face Med. 2008, 4, 4. [Google Scholar] [CrossRef]
- Kishi, A.; Haraki, S.; Toyota, R.; Shiraishi, Y.; Kamimura, M.; Taniike, M.; Yatani, H.; Kato, T. Sleep Stage Dynamics in Young Patients with Sleep Bruxism. Sleep 2020, 43, zsz202. [Google Scholar] [CrossRef]
- Haraki, S.; Tsujisaka, A.; Nonoue, S.; Nochino, T.; Kamimura, M.; Adachi, H.; Ishigaki, S.; Yatani, H.; Taniike, M.; Kato, T. Sleep Quality, Psychologic Profiles, Cardiac Activity, and Salivary Biomarkers in Young Subjects with Different Degrees of Rhythmic Masticatory Muscle Activity: A Polysomnography Study. J. Oral Facial Pain Headache 2019, 33, 105–113. [Google Scholar] [CrossRef]
- Huynh, N.; Kato, T.; Rompré, P.H.; Okura, K.; Saber, M.; Lanfranchi, P.A.; Montplaisir, J.Y.; Lavigne, G.J. Sleep Bruxism is Associated to Micro-arousals and an Increase in Cardiac Sympathetic Activity. J. Sleep Res. 2006, 15, 339–346. [Google Scholar] [CrossRef]
- Lavigne, G.J.; Huynh, N.; Kato, T.; Okura, K.; Adachi, K.; Yao, D.; Sessle, B. Genesis of Sleep Bruxism: Motor and Autonomic-cardiac Interactions. Arch. Oral Biol. 2007, 52, 381–384. [Google Scholar] [CrossRef]
- Marthol, H.; Reich, S.; Jacke, J.; Lechner, K.H.; Wichmann, M.; Hilz, M.J. Enhanced Sympathetic Cardiac Modulation in Bruxism Patients. Clin. Auton. Res. 2006, 16, 276–280. [Google Scholar] [CrossRef]


| Parameter | Mean | SD |
|---|---|---|
| BEI (n/h) | 4.39 | 3.33 |
| Phasic (n/h) | 2.49 | 2.74 |
| Tonic (n/h) | 1.20 | 1.01 |
| Mixed (n/h) | 0.75 | 0.66 |
| AHI (n/h) | 2.00 | 1.33 |
| ODI (n/h) | 2.38 | 1.74 |
| Snore (% of TST) | 5.01 | 9.78 |
| Snore index (n/h) | 61.64 | 105.82 |
| TST (min) | 429.68 | 56.45 |
| SL (min) | 22.83 | 21.60 |
| WASO (min) | 35.17 | 36.57 |
| SE (%) | 86.71 | 10.36 |
| N1 (% of TST) | 3.52 | 4.02 |
| N2 (% of TST) | 48.3 | 8.46 |
| N3 (% of TST) | 24.22 | 8.00 |
| REM (% of TST) | 23.97 | 6.06 |
| Arousals (n/h) | 3.28 | 2.39 |
| Mean SpO2 (%) | 94.86 | 1.58 |
| Min SpO2 (%) | 89.56 | 5.29 |
| SpO2 < 90% (%) | 0.70 | 3.20 |
| Mean desaturation drop (%) | 3.13 | 0.83 |
| Mean heart rate (n/min) | 61.26 | 9.84 |
| Max heart rate (n/min) | 99.29 | 19.11 |
| Min heart rate (n/min) | 48.69 | 7.21 |
| Parameter | Mean | SD | Parameter | Mean | SD |
|---|---|---|---|---|---|
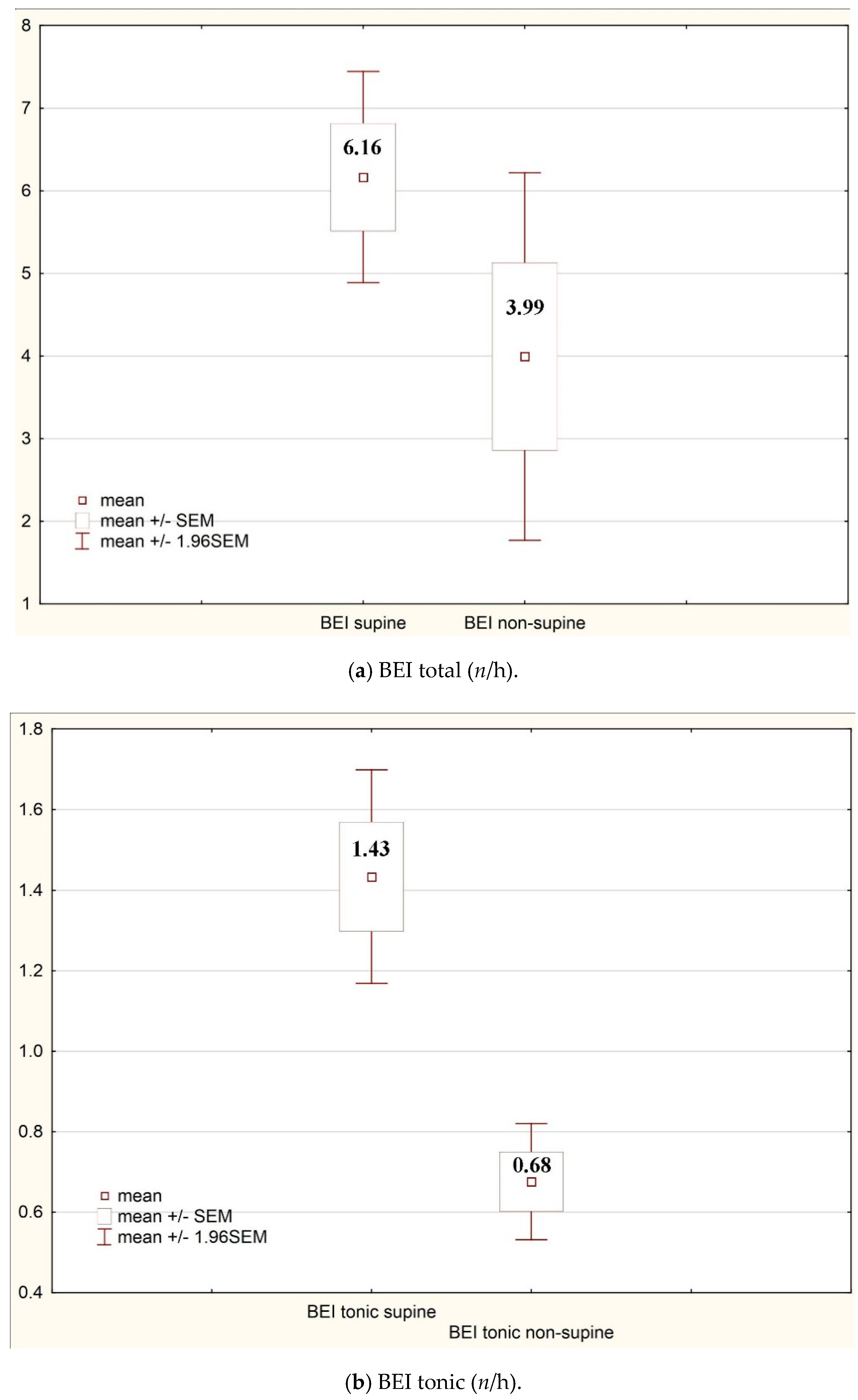
| BEI supine (n/h) | 6.16 | 7.40 | Snore index supine (n/h) | 74.21 | 138.05 |
| BEI mixed supine (n/h) | 0.85 | 0.89 | Snore index non-supine (n/h) | 35.53 | 77.18 |
| BEI phasic supine (n/h) | 3.52 | 4.92 | Snore index N1 (n/h) | 67.97 | 84.82 |
| BEI tonic supine (n/h) | 1.43 | 1.54 | Snore index N1 supine (n/h) | 67.32 | 102.80 |
| BEI non-supine (n/h) | 3.99 | 12.88 | Snore index N1 non-supine (n/h) | 58.55 | 119.32 |
| BEI mixed non-supine (n/h) | 0.72 | 2.19 | Snore index N2 (n/h) | 60.28 | 108.25 |
| BEI phasic non-supine (n/h) | 2.61 | 10.77 | Snore index N2 supine (n/h) | 68.17 | 132.57 |
| BEI tonic non-supine (n/h) | 0.68 | 0.84 | Snore index N2 non-supine (n/h) | 35.40 | 82.84 |
| Mean snore intensity (dB) | 54.13 | 25.45 | Snore index N3 (n/h) | 90.06 | 168.28 |
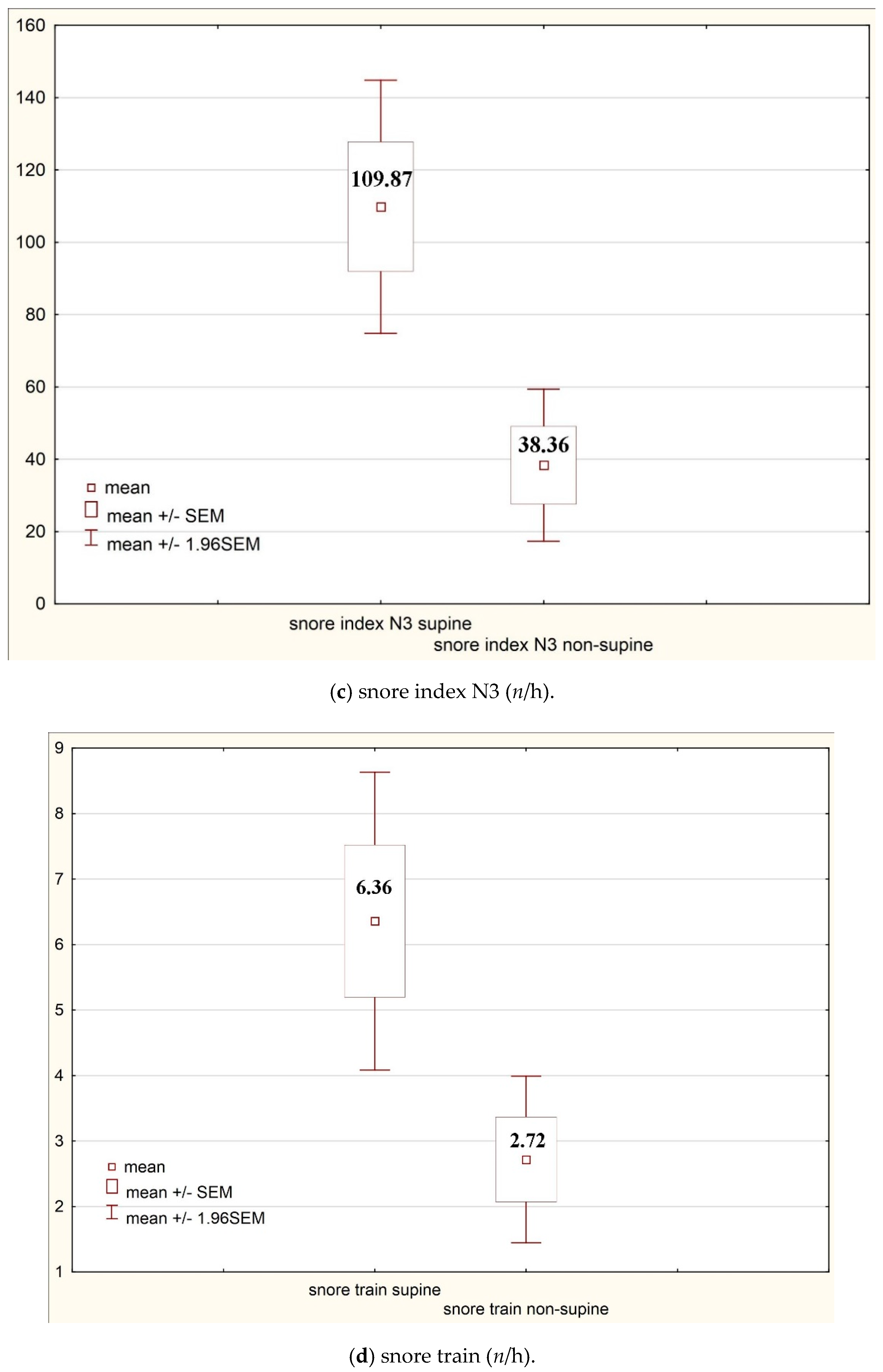
| Max snore intensity (dB) | 65.38 | 31.23 | Snore index N3 supine (n/h) | 109.87 | 202.72 |
| Min snore intensity (dB) | 43.78 | 20.97 | Snore index N3 non-supine (n/h) | 38.36 | 122.04 |
| Mean snore intensity supine (dB) | 46.20 | 30.62 | Snore index REM (n/h) | 42.86 | 108.45 |
| Max snore intensity supine(dB) | 55.00 | 36.81 | Snore index REM supine (n/h) | 44.35 | 117.10 |
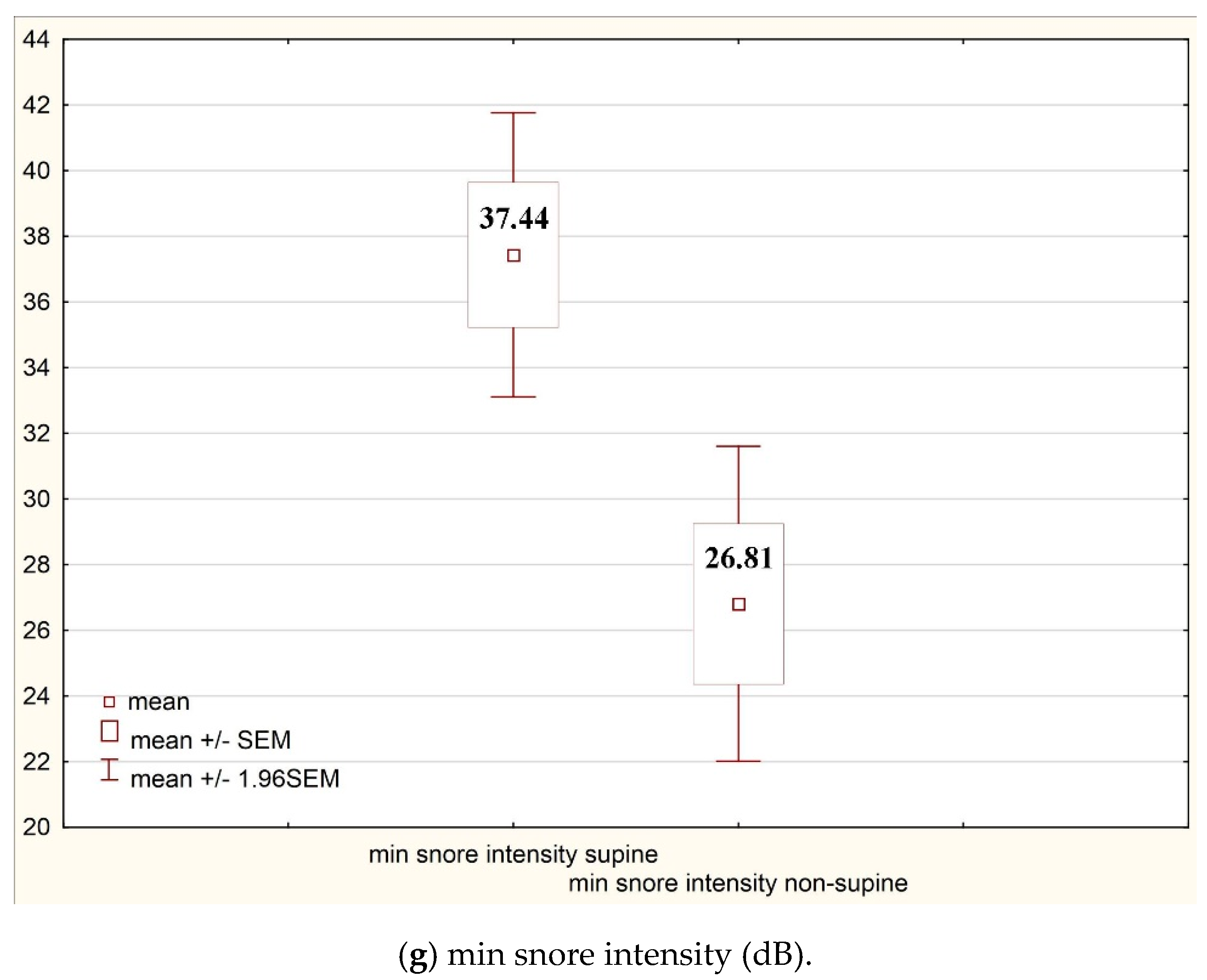
| Min snore intensity supine (dB) | 37.44 | 25.05 | Snore index REM non-supine (n/h) | 24.61 | 72.54 |
| Mean snore intensity non-supine (dB) | 31.88 | 32.81 | Snore train supine (n/h) | 6.36 | 13.18 |
| Max snore intensity non-supine (dB) | 37.99 | 39.28 | Snore train non-supine (n/h) | 2.72 | 7.36 |
| Min snore intensity non-supine (dB) | 26.81 | 27.76 |
| Body Position | Bruxism Indices | AHI (n/h) | ODI (n/h) | Snore (% of TST) | SL (min) | Arousals (n/h) | Mean Heart Rate (n/min) | Max Heart Rate (n/min) | Min Heart Rate (n/min) |
|---|---|---|---|---|---|---|---|---|---|
| Total | Average BEI (n/h) | 0.08 | 0.09 | −0.02 | −0.00 | 0.23 | −0.16 | 0.04 | −0.25 |
| Phasic (n/h) | 0.10 | 0.10 | −0.01 | 0.00 | 0.10 | −0.15 | 0.09 | −0.25 | |
| Tonic (n/h) | 0.08 | 0.07 | 0.03 | −0.05 | 0.27 | −0.06 | −0.11 | −0.03 | |
| Mixed (n/h) | −0.09 | −0.06 | −0.09 | −0.01 | 0.35 | −0.09 | 0.05 | −0.22 | |
| Supine | Average BEI (n/h) | 0.07 | 0.03 | −0.07 | −0.02 | 0.15 | −0.08 | −0.02 | −0.12 |
| Phasic (n/h) | 0.12 | 0.10 | −0.06 | 0.01 | 0.04 | −0.10 | 0.01 | −0.17 | |
| Tonic (n/h) | 0.08 | 0.10 | 0.02 | −0.03 | 0.25 | −0.03 | −0.11 | −0.04 | |
| Mixed (n/h) | 0.12 | 0.10 | −0.03 | 0.00 | 0.36 | −0.14 | −0.04 | −0.26 | |
| Non- supine | Average BEI (n/h) | 0.12 | 0.29 | −0.05 | 0.31 | −0.01 | 0.20 | 0.09 | 0.21 |
| Phasic (n/h) | 0.13 | 0.29 | −0.04 | 0.30 | −0.02 | 0.20 | 0.09 | 0.21 | |
| Tonic (n/h) | 0.05 | 0.04 | −0.09 | 0.03 | 0.09 | −0.01 | −0.05 | 0.02 | |
| Mixed (n/h) | 0.07 | 0.25 | −0.08 | 0.32 | −0.01 | 0.22 | 0.11 | 0.23 |
| Parameter | BEI < 2 (n/h) | BEI > 2 (n/h) | BEI < 4 (n/h) | BEI > 4 (n/h) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | p | Mean | SD | Mean | SD | p | |
| Mean heart rate (n/min) | 64.47 | 11.25 | 60.25 | 9.18 | 0.04 | 62.95 | 9.88 | 59.26 | 9.49 | 0.03 |
| Max heart rate (n/min) | 101.90 | 18.41 | 98.47 | 19.34 | 0.39 | 99.46 | 19.25 | 99.10 | 19.09 | 0.92 |
| Min heart rate (n/min) | 51.00 | 8.36 | 47.96 | 6.69 | 0.04 | 49.87 | 8.05 | 47.29 | 5.84 | 0.04 |
| Parameter | BEI (n/h) | Phasic (n/h) | Tonic (n/h) | Mixed (n/h) |
|---|---|---|---|---|
| Snore index (n/h) | 0.03 | 0.06 | 0.03 | −0.12 |
| Mean snore intensity (dB) | 0.17 | 0.17 | 0.02 | 0.15 |
| Max snore intensity (dB) | 0.21 | 0.22 | 0.02 | 0.15 |
| Min snore intensity (dB) | 0.15 | 0.13 | 0.04 | 0.18 |
| Mean snore intensity supine (dB) | 0.19 | 0.22 | 0.01 | 0.09 |
| Max snore intensity supine(dB) | 0.21 | 0.24 | 0.03 | 0.09 |
| Min snore intensity supine (dB) | 0.18 | 0.21 | 0.02 | 0.10 |
| Mean snore intensity non-supine (dB) | 0.15 | 0.21 | −0.08 | 0.03 |
| Max snore intensity non-supine (dB) | 0.17 | 0.23 | −0.09 | 0.03 |
| Min snore intensity non-supine (dB) | 0.14 | 0.19 | −0.09 | 0.04 |
| Supine | Non-Supine | ||||
|---|---|---|---|---|---|
| BEI (n/h) | Mean | SD | Mean | SD | p |
| Total | 6.16 | 7.04 | 3.99 | 12.88 | 0.04 |
| Mixed | 0.85 | 0.89 | 0.72 | 2.19 | 0.53 |
| Phasic | 3.52 | 4.92 | 2.61 | 10.77 | 0.37 |
| Tonic | 1.43 | 1.54 | 0.68 | 0.84 | 0.00 |
| Supine | Non-Supine | ||||
|---|---|---|---|---|---|
| Parameter | Mean | SD | Mean | SD | p |
| Snore index total (n/h) | 74.21 | 138.05 | 35.53 | 77.18 | 0.00 |
| Snore index N1 (n/h) | 67.32 | 102.80 | 58.55 | 119.32 | 0.52 |
| Snore index N2 (n/h) | 68.17 | 132.57 | 35.4 | 82.84 | 0.01 |
| Snore index N3 (n/h) | 109.87 | 202.72 | 38.36 | 122.04 | 0.00 |
| Snore index REM (n/h) | 44.35 | 117.10 | 24.61 | 72.54 | 0.10 |
| Snore train (n/h) | 6.36 | 13.18 | 2.72 | 7.36 | 0.00 |
| Mean snore intensity (dB) | 46.20 | 30.62 | 31.88 | 32.81 | 0.00 |
| Max snore intensity (dB) | 55.00 | 36.81 | 37.99 | 39.28 | 0.00 |
| Min snore intensity (dB) | 37.44 | 25.05 | 26.81 | 27.76 | 0.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michalek-Zrabkowska, M.; Wieckiewicz, M.; Macek, P.; Gac, P.; Smardz, J.; Wojakowska, A.; Poreba, R.; Mazur, G.; Martynowicz, H. The Relationship between Simple Snoring and Sleep Bruxism: A Polysomnographic Study. Int. J. Environ. Res. Public Health 2020, 17, 8960. https://doi.org/10.3390/ijerph17238960
Michalek-Zrabkowska M, Wieckiewicz M, Macek P, Gac P, Smardz J, Wojakowska A, Poreba R, Mazur G, Martynowicz H. The Relationship between Simple Snoring and Sleep Bruxism: A Polysomnographic Study. International Journal of Environmental Research and Public Health. 2020; 17(23):8960. https://doi.org/10.3390/ijerph17238960
Chicago/Turabian StyleMichalek-Zrabkowska, Monika, Mieszko Wieckiewicz, Piotr Macek, Pawel Gac, Joanna Smardz, Anna Wojakowska, Rafal Poreba, Grzegorz Mazur, and Helena Martynowicz. 2020. "The Relationship between Simple Snoring and Sleep Bruxism: A Polysomnographic Study" International Journal of Environmental Research and Public Health 17, no. 23: 8960. https://doi.org/10.3390/ijerph17238960
APA StyleMichalek-Zrabkowska, M., Wieckiewicz, M., Macek, P., Gac, P., Smardz, J., Wojakowska, A., Poreba, R., Mazur, G., & Martynowicz, H. (2020). The Relationship between Simple Snoring and Sleep Bruxism: A Polysomnographic Study. International Journal of Environmental Research and Public Health, 17(23), 8960. https://doi.org/10.3390/ijerph17238960








