Prevalence and Subtype Distribution of Blastocystis Infection in Patients with Diabetes Mellitus in Thailand
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Study Population and Study Design
2.3. Stool Collection and Processing
2.4. DNA Extraction and Nested PCR Amplification
2.5. Sequencing and Phylogenetic Analysis
2.6. Statistical Analysis
3. Results
3.1. Basic Characteristics of the Individuals
3.2. The Prevalence and Subtype of Blastocystis sp.
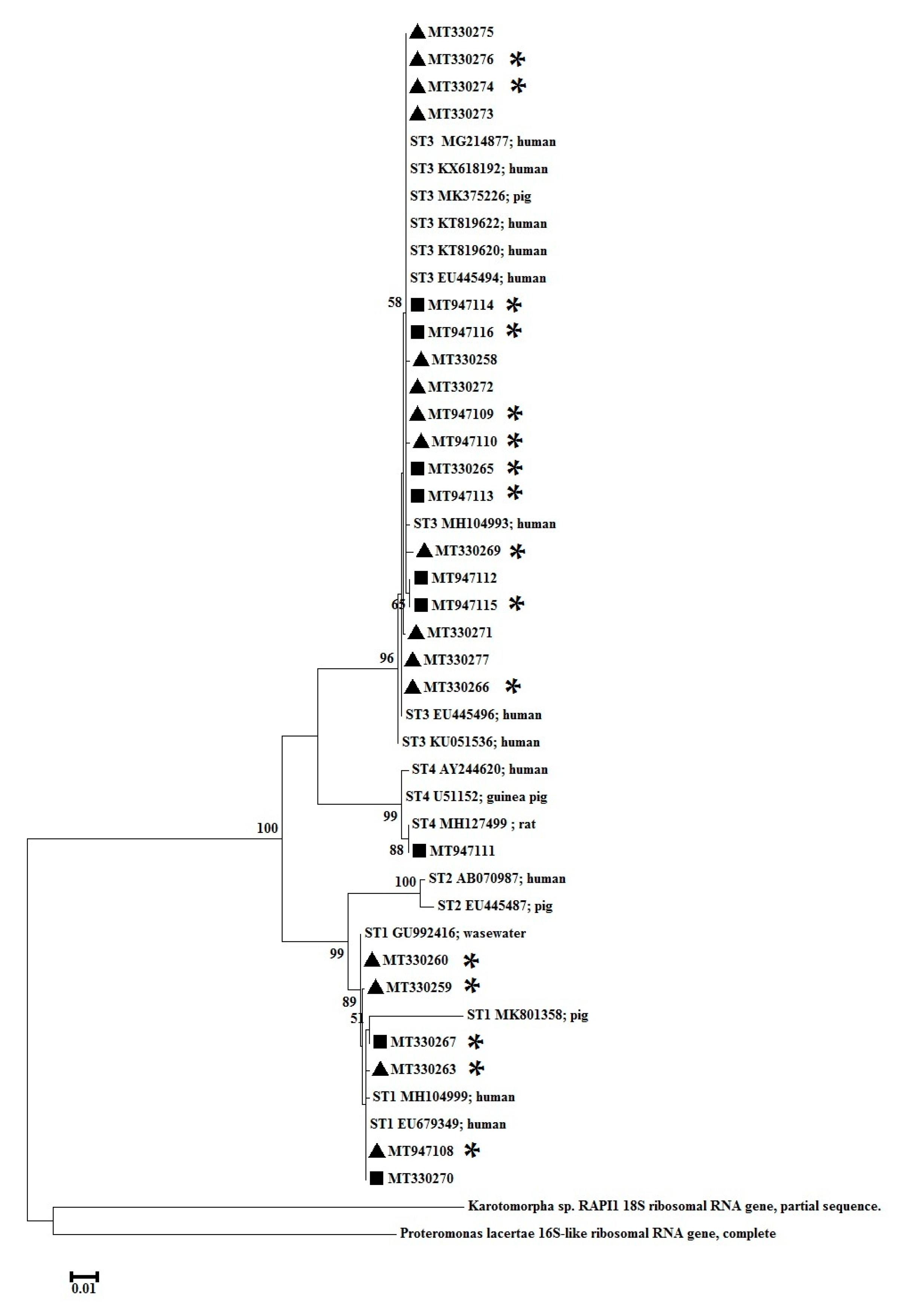
3.3. Phylogenetic Analysis
4. Discussion
5. Conclusions
6. Limitations
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Status Report on Noncommunicable Diseases 2014; WHO: Geneva, Switzerland, 2014; pp. 1–298. [Google Scholar]
- Berbudi, A.; Rahmadika, N.; Tjahjadi, A.; Ruslami, R. Type 2 Diabetes and its Impact on the Immune System. Curr Diabetes Rev. 2020, 16, 442–449. [Google Scholar] [CrossRef] [PubMed]
- Shah, B.R.; Hux, J.E. Quantifying the risk of infectious diseases for people with diabetes. Diabetes Care 2003, 26, 510–513. [Google Scholar] [CrossRef] [PubMed]
- Carey, I.M.; Critchley, J.A.; DeWilde, S.; Harris, T.; Hosking, F.J.; Cook, D.G. Risk of Infection in Type 1 and Type 2 Diabetes Compared with the General Population: A Matched Cohort Study. Diabetes Care 2018, 41, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Casqueiro, J.; Casqueiro, J.; Alves, C. Infections in patients with diabetes mellitus: A review of pathogenesis. Indian J. Endrocrinol. Metab. 2012, 16, S27–S36. [Google Scholar]
- Akinbo, F.O.; Olujobi, S.O.; Omoregie, R.; Egbe, C. Intestinal parasitic infections among diabetes mellitus patients. Biomarkers Genomic Med. 2013, 5, 44–47. [Google Scholar] [CrossRef]
- Machado, E.R.; Matos, N.O.; Rezende, S.M.; Carlos, D.; Silva, T.C.; Rodrigues, L.; Almeida, M.J.R.; de Oliveira, M.R.F.; Muniz-Junqueira, M.I.; Gurgel-Gonçalves, R. Host-Parasite Interactions in Individuals with Type 1 and 2 Diabetes Result in Higher Frequency of Ascaris lumbricoides and Giardia lamblia in Type 2 Diabetic Individuals. J. Diabetes Res. 2018, 2018, 4238435. [Google Scholar] [CrossRef]
- Htun, N.S.N.; Odermatt, P.; Paboriboune, P.; Sayasone, S.; Vongsakid, M.; Phimolsarn-Nusith, V.; Tran, X.D.; Ounnavong, P.S.; Andriama-Hefasoa, N.; Senvanpan, N.D.; et al. Association between helminth infections and diabetes mellitus in adults from the Lao People’s Democratic Republic: A cross-sectional study. Infect. Dis. Poverty 2018, 7, 105. [Google Scholar] [CrossRef]
- Nuchprayoon, S.; Siriyasatien, P.; Kraivichian, K.; Porksakorn, C.; Nuchprayoon, I. Prevalence of parasitic infections among Thai patients at the King Chulalongkorn Memorial Hospital, Bangkok, Thailand. J. Med. Assoc. Thai. 2002, 85, S415–S423. [Google Scholar]
- Nanditha, A.; Ma, R.C.; Ramachandran, A.; Snehalatha, C.; Chan, J.C.; Chia, K.S.; Shaw, J.E.; Zimmet, P.Z. Diabetes in Asia and the Pacific: Implications for the Global Epidemic. Diabetes Care 2016, 3, 472–485. [Google Scholar] [CrossRef]
- Aekplakorn, A.; Chariyalertsak, S.; Kessomboon, P.; Assanangkornchai, S.; Taneepanichskul, S.; Putwatana, P. Prevalence of Diabetes and Relationship with Socioeconomic Status in the Thai Population: National Health Examination Survey, 2004–2014. J. Diabetes Res. 2018, 2018, 1654530. [Google Scholar] [CrossRef]
- Kitvatanachai, S.; Rhongbutsri, P. Intestinal parasitic infections in suburban government schools, Lak Hok subdistrict, Muang Pathum Thani, Thailand. Asian Pac. J. Trop. Med. 2013, 6, 699–702. [Google Scholar] [CrossRef]
- Wongsaroj, T.; Nithikathkul, C.; Rojkitikul, W.; Nakai, W.; Royal, L.; Rammasut, P. Brief communication (Original). National survey of helminthiasis in Thailand. Asian Biomed. 2014, 8, 779–783. [Google Scholar] [CrossRef]
- Punsawad, C.; Phasuk, N.; Bunratsami, S.; Thongtup, K.; Siripakonuaong, N.; Nongnaul, S. Prevalence of intestinal parasitic infection and associated risk factors among village health volunteers in rural communities of southern Thailand. BMC Public Health 2017, 17, 564. [Google Scholar] [CrossRef] [PubMed]
- Tan, K.S. Blastocystis in humans and animals: New insights using modern methodologies. Vet Parasitol. 2004, 126, 121–144. [Google Scholar] [CrossRef] [PubMed]
- Stensvold, C.R.; Suresh, G.K.; Tan, K.S.; Thompson, R.C.; Traub, R.J.; Viscogliosi, E.; Yoshikawa, H.; Clark, C.G. Terminology for Blastocystis subtypes—A consensus. Trends Parasitol. 2007, 23, 93–96. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Taner-Mulla, D.; Jacob, A.S.; Imeede, C.A.; Yoshikawa, H.; Stensvold, C.R.; Clark, C.G. Genetic Diversity of Blastocystis in Livestock and Zoo Animals. Protist 2013, 164, 497–509. [Google Scholar] [CrossRef]
- Tan, K.S. New insights on classification, identification, and clinical relevance of Blastocystis spp. Clin. Microbiol. Rev. 2008, 21, 639–665. [Google Scholar] [CrossRef]
- Katsarou-Katsari, A.; Vassalos, C.M.; Tzanetou, K.; Spanakos, G.; Papadopoulou, C.; Vakalis, N. Acute urticaria associated with amoeboid forms of Blastocystis sp. subtype 3. Acta Derm. Venereol. 2008, 88, 80–81. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Lewis, H.C.; Hammerum, A.M.; Porsbo, L.J.; Nielsen, S.S.; Olsen, K.E.; Arendrup, M.C.; Nielsen, H.V.; Mølbak, K. Blastocystis: Unravelling potential risk factors and clinical significance of a common but neglected parasite. Epidemiol. Infect. 2009, 137, 1655–1663. [Google Scholar] [CrossRef]
- Taamasri, P.; Leelayoova, S.; Rangsin, R.; Naaglor, T.; Ketupanya, A.; Mungthin, M. Prevalence of Blastocystis hominis carriage in Thai army personnel based in Chonburi, Thailand. Mil. Med. 2002, 167, 643–646. [Google Scholar] [CrossRef]
- Popruk, S.; Udonsom, R.; Koompapong, K.; Mahittikorn, A.; Kusolsuk, T.; Ruangsittichai, J.; Palasuwan, A. Subtype distribution of Blastocystis in Thai-Myanmar border, Thailand. Korean J. Parasitol. 2015, 53, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Pipatsatitpong, D.; Leelayoova, S.; Mungthin, M.; Aunpad, R.; Naaglor, T.; Rangsin, R. Prevalence and Risk Factors for Blastocystis Infection among Children and Caregivers in a Child Care Center, Bangkok, Thailand. Am. J. Trop. Med. Hyg. 2015, 93, 310–315. [Google Scholar] [CrossRef]
- Saksirisampant, W.; Nuchprayoon, S.; Wiwanitkit, V.; Yenthakam, S.; Ampavasiri, A. Intestinal parasitic infestations among children in an orphanage in Pathum Thani province. J. Med. Assoc. Thai. 2003, 86, S263–S270. [Google Scholar] [PubMed]
- Jongudomsuk, P.; Srithamrongsawat, S.; Patcharanarumol, W.; Limwattananon, S.; Pannarunothai, S.; Vapatanavong, P.; Sawaengdee, K.; Fahamnuaypol, P. The Kingdom of Thailand Health System Review; World Health Organization, Regional Office for the Western Pacific: Manila, Philippines, 2015. [Google Scholar]
- Clark, C.G. Extensive genetic diversity in Blastocystis hominis. Mol. Biochem. Parasitol. 1997, 87, 79–83. [Google Scholar] [CrossRef]
- Wong, K.H.; Ng, G.C.; Lin, R.T.; Yoshikawa, H.; Taylor, M.B.; Tan, K.S. Predominance of subtype 3 among Blastocystis isolates from a major hospital in Singapore. Parasitol. Res. 2008, 102, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Whipps, C.M.; Boorom, K.; Bermudez, L.E.; Kent, M.L. Molecular characterization of Blastocystis species in Oregon identifies multiple subtypes. Parasitol. Res. 2010, 106, 827–832. [Google Scholar] [CrossRef]
- Banaticla, J.E.; Rivera, W.L. Detection and subtype identification of Blastocystis isolates from wastewater samples in the Philippines. J. Water Health 2011, 9, 128–137. [Google Scholar] [CrossRef]
- Udonsom, R.; Prasertbun, R.; Mahittikorn, A.; Mori, H.; Changbunjong, T.; Komalamisra, C.; Pintong, A.R.; Sukthana, Y.; Popruk, S. Blastocystis infection and subtype distribution in humans, cattle, goats, and pigs in central and western Thailand. Infect. Genet. Evol. 2018, 65, 107–111. [Google Scholar] [CrossRef]
- Wylezich, C.; Belka, A.; Hanke, D.; Beer, M.; Blome, S.; Höper, D. Metagenomics for broad and improved parasite detection: A proof-of-concept study using swine faecal samples. Int. J. Parasitol. 2019, 49, 769–777. [Google Scholar] [CrossRef]
- Arisue, N.; Hashimoto, T.; Yoshikawa, H. Sequence heterogeneity of the small subunit ribosomal RNA genes among blastocystis isolates. Parasitology 2003, 126, 1–9. [Google Scholar] [CrossRef]
- Rivera, W.L. Phylogenetic analysis of Blastocystis isolates from animal and human hosts in the Philippines. Vet. Parasitol. 2008, 156, 178–182. [Google Scholar] [CrossRef] [PubMed]
- Pintong, A.R.; Sunyanusin, S.; Prasertbun, R.; Mahittikorn, A.; Mori, H.; Changbunjong, T.; Komalamisra, C.; Sukthana, Y.; Popruk, S. Blastocystis subtype 5: Predominant subtype on pig farms, Thailand. Parasitol. Int. 2018, 67, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Palasuwan, A.; Palasuwan, D.; Mahittikorn, A.; Chiabchalard, R.; Combes, V.; Popruk, S. Subtype Distribution of Blastocystis in Communities along the Chao Phraya River, Thailand. Korean J. Parasitol. 2016, 54, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Chavatte, J.M.; Jureen, L. Incidental Detection of Cyclospora Cayetanensis during General Health Screening: A Case Study from Singapore. J. Trop. Dis. 2016, 4, 1–5. [Google Scholar] [CrossRef]
- Han, J.Q.; Li, Z.; Zou, Y.; Pu, L.H.; Zhu, X.Q.; Zou, F.C.; Huang, C.Q. Prevalence, Molecular Characterization and Risk Factors of Blastocystis sp. from Farmed Pigs in Yunnan Province, Southwestern China. Acta Parasitol. 2020, 65, 1005–1010. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Wu, Z.; Kimata, I.; Iseki, M.; Ali, I.K.; Hossain, M.B.; Zaman, V.; Haque, R.; Takahashi, Y. Polymerase chain reaction-based genotype classification among human Blastocystis hominis populations isolated from different countries. Parasitol. Res. 2004, 92, 22–29. [Google Scholar]
- Katsumata, M.; Yoshikawa, H.; Tokoro, M.; Mizuno, T.; Nagamoto, T.; Hendarto, J.; Asih, P.B.S.; Rozi, I.E.; Kimata, I.; Takami, K.; et al. Molecular phylogeny of Blastocystis isolates from wild rodents captured in Indonesia and Japan. Parasitol. Res. 2018, 117, 2841–2846. [Google Scholar] [CrossRef]
- Silberman, J.D.; Sogin, M.L.; Leipe, D.D.; Clark, C.G. Human parasite finds taxonomic home. Nature 1996, 380, 398. [Google Scholar] [CrossRef]
- Yaicharoen, R.; Sripochang, S.; Sermsart, B.; Pidetcha, P. Prevalence of Blastocystis hominis infection in asymptomatic individuals from Bangkok, Thailand. Southeast Asian J. Trop. Med. Public Health 2005, 36, 17–20. [Google Scholar]
- Yowang, A.; Tsaousis, A.D.; Chumphonsuk, T.; Thongsin, N.; Kullawong, N.; Popluechai, S.; Gentekaki, E. High diversity of Blastocystis subtypes isolated from asymptomatic adults living in Chiang Rai, Thailand. Infect. Genet. Evol. 2018, 65, 270–275. [Google Scholar] [CrossRef]
- Belleza, M.L.; Reyes, J.C.; Tongol-Rivera, P.N.; Rivera, W.L. Subtype analysis of Blastocystis sp. isolates from human and canine hosts in an urban community in the Philippines. Parasitol. Int. 2016, 65, 291–294. [Google Scholar] [CrossRef] [PubMed]
- Sanpool, O.; Laymanivong, S.; Thanchomnang, T.; Rodpai, R.; Sadaow, L.; Phosuk, I.; Maleewong, W.; Intapan, P.M. Subtype identification of human Blastocystis spp. isolated from Lao People’s Democratic Republic. Acta Trop. 2017, 168, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Hirata, T.; Nakamura, H.; Kinjo, N.; Hokama, A.; Kinjo, F.; Yamane, N.; Fujita, J. Prevalence of Blastocystis hominis and Strongyloides stercoralis infection in Okinawa, Japan. Parasitol. Res. 2007, 101, 1717–1719. [Google Scholar] [CrossRef] [PubMed]
- Jantermtor, S.; Pinlaor, P.; Sawadpanich, K.; Pinlaor, S.; Sangka, A.; Wilailuckana, C.; Wongsena, W.; Yoshikawa, H. Subtype identification of Blastocystis spp. isolated from patients in a major hospital in northeastern Thailand. Parasitol. Res. 2013, 112, 1781–1786. [Google Scholar] [CrossRef] [PubMed]
- Srichaipon, N.; Nuchprayoon, S.; Charuchaibovorn, S.; Sukkapan, P.; Sanprasert, V. A Simple Genotyping Method for Rapid Differentiation of Blastocystis Subtypes and Subtype Distribution of Blastocystis spp. in Thailand. Pathogens 2019, 8, 38. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Chai, Y.; Zhou, Z.; Liu, H.; Zhong, Z.; Hu, Y.; Fu, H.; Yue, C.; Peng, G. Epidemiology of Blastocystis sp. infection in China: A systematic review. Parasite 2019, 26, 41. [Google Scholar] [CrossRef]
- Leelayoova, S.; Siripattanapipong, S.; Thathaisong, U.; Naaglor, T.; Taamasri, P.; Piyaraj, P.; Mungthin, M. Drinking Water: A Possible Source of Blastocystis spp. Subtype 1 Infection in Schoolchildren of a Rural Community in Central Thailand. Am. J. Trop. Med. Hyg. 2008, 79, 401–406. [Google Scholar] [CrossRef]
- Thathaisong, U.; Siripattanapipong, S.; Mungthin, M.; Pipatsatitpong, D.; Tan-ariya, P.; Naaglor, T.; Leelayoova, S. Identification of Blastocystis subtype 1 variants in the Home for Girls, Bangkok, Thailand. Am. J. Trop. Med. Hyg. 2013, 88, 352–358. [Google Scholar] [CrossRef]
- Alfellani, M.A.; Stensvold, C.R.; Vidal-Lapiedra, A.; Onuoha, E.S.; Fagbenro-Beyioku, A.F.; Clark, C.G. Variable geographic distribution of Blastocystis subtypes and its potential implications. Acta Trop. 2013, 126, 11–18. [Google Scholar] [CrossRef]
- Noël, C.; Dufernez, F.; Gerbod, D.; Edgcomb, V.P.; Delgado-Viscogliosi, P.; Ho, L.C.; Singh, M.; Wintjens, R.; Sogin, M.L.; Capron, M.; et al. Molecular Phylogenies of Blastocystis Isolates from Different Hosts: Implications for Genetic Diversity, Identification of Species, and Zoonosis. J. Clin. Microbiol. 2005, 43, 348–355. [Google Scholar] [CrossRef]
- Gil, F.F.; Barros, M.J.; Macedo, N.A.; Júnior, C.G.; Redoan, R.; Busatti, H.; Gomes, M.A.; Santos, J.F. Prevalence of intestinal parasitism and associated symptomatology among hemodialysis patients. Rev. Inst. Med. Trop. Sao Paulo 2013, 55, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Mohtashamipour, M.; Hoseini, S.G.; Pestehchian, N.; Yousefi, H.; Fallah, E.; Hazratian, T. Intestinal parasitic infections in patients with Diabetes Mellitus: A case-control study. J. Anal. Res. Clin. Med. 2015, 3, 157–163. [Google Scholar] [CrossRef]
- Taamasri, P.; Mungthin, M.; Rangsin, R.; Tongupprakarn, B.; Areekul, W.; Leelayoova, S. Transmission of intestinal blastocystosis related to the quality of drinking water. Southeast. Asian J. Trop. Med. Public Health 2000, 31, 112–117. [Google Scholar] [PubMed]
- Abdulsalam, A.M.; Ithoi, I.; Al-Mekhlafi, H.M.; Ahmed, A.; Surin, J.; Mak, J.W. Drinking water is a significant predictor of Blastocystis infection among rural Malaysian primary schoolchildren. Parasitology 2012, 139, 1014–1020. [Google Scholar] [CrossRef] [PubMed]
- Larsen, N.; Vogensen, F.K.; van den Berg, F.W.; Nielsen, D.S.; Andreasen, A.S.; Pedersen, B.K.; Al-Soud, W.A.; Sørensen, S.J.; Hansen, L.H.; Jakobsen, M. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS ONE 2010, 5, e9085. [Google Scholar] [CrossRef]
- Qin, J.; Li, Y.; Cai, Z.; Li, S.; Zhu, J.; Zhang, F.; Liang, S.; Zhang, W.; Guan, Y.; Shen, D. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 2012, 490, 55–60. [Google Scholar] [CrossRef]
- Ajjampur, S.S.; Tan, K.S. Pathogenic mechanisms in Blastocystis spp.—Interpreting results from in vitro and in vivo studies. Parasitol. Int. 2016, 65, 772–779. [Google Scholar] [CrossRef]
- Alzate, J.F.; Toro-Londoño, M.; Cabarcas, F.; Garcia-Montoya, G.; Galvan-Diaz, A. Contrasting microbiota profiles observed in children carrying either Blastocystis spp. or the commensal amoebas Entamoeba coli or Endolimax nana. Sci. Rep. 2020, 10, 15354. [Google Scholar] [CrossRef]
- Stensvold, C.R.; Alfellani, M.A.; Nørskov-Lauritsen, S.; Prip, K.; Victory, E.L.; Maddox, C.; Nielsen, H.V.; Clark, C.G. Subtype distribution of Blastocystis isolates from synanthropic and zoo animals and identification of a new subtype. Int. J. Parasitol. 2009, 39, 473–479. [Google Scholar] [CrossRef]
| Subtype | Accession Number | Host |
|---|---|---|
| 1 | EU679349 | Human [28] |
| GU992416 | Wastewater [29] | |
| MH104999 | Human [30] | |
| MK801358 | Pig [31] | |
| 2 | AB070987 | Human [32] |
| EU445487 | Pig [33] | |
| 3 | EU445494 | Human [33] |
| EU445496 | Human [33] | |
| KT819620 | Human [34] | |
| KT819622 | Human [34] | |
| KU051536 | Human [35] | |
| KX618192 | Human [36] | |
| MG214877 | Human (unpublished) | |
| MH104993 | Human [30] | |
| MK375226 | Pig [37] | |
| 4 | AY244620 | Human [38] |
| MH127499 | Rat [39] | |
| U51152 | Guinea pig [40] |
| Characteristics | DM n = 130 (%) | Non-DM n = 100 (%) | Total n = 230 (%) | p | |
|---|---|---|---|---|---|
| Gender | Male | 43/130 (33.1%) | 34/100 (34%) | 77/230 (33.5%) | 0.883 |
| Female | 87/130 (66.9%) | 66/100 (66%) | 153/230 (66.5%) | ||
| Age | <65 years | 54/130 (41.5%) | 43/100 (43%) | 97/230 (42.2%) | 0.824 |
| ≥65 years | 76/130 (58.5%) | 57/100 (57%) | 133/230 (57.8%) | ||
| Level of education | Low (≤primary school) | 120/130 (92.3%) | 84/100 (84%) | 204/230 (88.7%) | 0.049 * |
| High (≥secondary school) | 10/130 (7.7%) | 16/100 (16%) | 26/230 (11.3%) | ||
| Employed | No | 66/130 (50.8%) | 58/100 (58%) | 124/230 (53.9%) | 0.275 |
| Yes | 64/130 (49.2%) | 42/100 (42%) | 106/230 (46.1%) | ||
| Source of drinking water | Treated water (bottled and tap water) | 118/130 (90.8%) | 96/100 (96%) | 214/230 (93%) | 0.122 |
| Untreated water (surface and rainwater) | 12/130 (9.2%) | 4/100 (4%) | 16/230 (7%) | ||
| Animals in the household | No | 53/130 (40.8%) | 44/100 (44%) | 97/230 (42.2%) | 0.623 |
| Yes | 77/130 (59.2%) | 56/100 (56%) | 133/230 (57.8%) | ||
| Characteristics | Number of Examined | % Infected | OR (95% CI) * | |
|---|---|---|---|---|
| Gender | Male | 77 | 13 (10/77) | 1.373 (0.586–3.218) |
| Female | 153 | 9.8 (15/153) | 1 | |
| Age | <65 years | 97 | 8.2 (8/97) | 1 |
| ≥65 years | 133 | 12.8 (17/133) | 1.63 (0.673–3.949) | |
| DM status | Non-DM | 100 | 9 (9/100) | 1 |
| DM | 130 | 12.3 (16/130) | 1.419 (0.599–3.36) | |
| Duration of DM | ≤10 years | 83 | 10.8 (9/83) | 1 |
| >10 years | 47 | 14.9 (7/47) | 1.439 (0.499–4.153) | |
| Education status | Low (≤primary school) | 204 | 11.3 (23/204) | 1.525 (0.338–6.877) |
| High (≥secondary school) | 26 | 7.7 (2/26) | 1 | |
| Employed | No | 124 | 12.1 (15/124) | 1.321 (0.567–3.078) |
| Yes | 106 | 9.4 (10/106) | 1 | |
| Source of drinking water | Treated water (bottled and tap water) | 214 | 11.2 (24/214) | 1.895 (0.239–14.99) |
| Untreated water (surface and rainwater) | 16 | 6.3 (1/16) | 1 | |
| Presence of animals in household | Yes | 133 | 12 (16/133) | 1.337 (0.565–3.167) |
| No | 97 | 9.3 (9/97) | 1 | |
| GenBank Accession No. | Subtype | Group (DM/Non-DM) | Query Cover | Sequence Similarity (%) | Similar GenBank Reference Sequence |
|---|---|---|---|---|---|
| MT330258 | 3 | DM | 97% | 99.53 | KT819620 |
| MT330259 | 1 | DM | 98% | 99.44 | MH104999 |
| MT330260 | 1 | DM | 97% | 99.44 | GU992416 |
| MT330263 | 1 | DM | 98% | 99.07 | EU679349 |
| MT330265 | 3 | Non-DM | 99% | 98.44 | KT819620 |
| MT330266 | 3 | DM | 99% | 98.52 | KT819620 |
| MT330267 | 1 | Non-DM | 98% | 98.98 | MK801358 |
| MT330269 | 3 | DM | 98% | 99.17 | KT819620 |
| MT330270 | 1 | Non-DM | 99% | 98.99 | MK801358 |
| MT330271 | 3 | DM | 98% | 99.44 | KT819620 |
| MT330272 | 3 | DM | 98% | 99.26 | KT819620 |
| MT330273 | 3 | DM | 97% | 99.81 | KX618192 |
| MT330274 | 3 | DM | 97% | 99.81 | KT819620 |
| MT330275 | 3 | DM | 97% | 99.81 | KT819620 |
| MT330276 | 3 | DM | 97% | 99.63 | KX618192 |
| MT330277 | 3 | DM | 97% | 99.54 | MN914073 |
| MT947108 | 1 | DM | 100% | 99.5 | MH104999 |
| MT947109 | 3 | DM | 100% | 99.9 | MH104993 |
| MT947110 | 3 | DM | 100% | 99.26 | KT819622 |
| MT947111 | 4 | Non-DM | 100% | 99.8 | MH127499 |
| MT947112 | 3 | Non-DM | 100% | 99.8 | MG214877 |
| MT947113 | 3 | Non-DM | 100% | 99.9 | MK375226 |
| MT947114 | 3 | Non-DM | 100% | 100 | MK375226 |
| MT947115 | 3 | Non-DM | 100% | 99.8 | MK375226 |
| MT947116 | 3 | Non-DM | 100% | 99.9 | MK375226 |
| Group | Subtype Distribution Positive Case/Total No. (%) | Total | ||
|---|---|---|---|---|
| ST1 | ST3 | ST4 | ||
| DM | 4/130 (3%) | 12/130 (9.3%) | 0/130 (0%) | 16/130 (12.3%) |
| Non-DM | 2/100 (2%) | 6/100 (6%) | 1/100 (1%) | 9/100 (9%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popruk, N.; Prasongwattana, S.; Mahittikorn, A.; Palasuwan, A.; Popruk, S.; Palasuwan, D. Prevalence and Subtype Distribution of Blastocystis Infection in Patients with Diabetes Mellitus in Thailand. Int. J. Environ. Res. Public Health 2020, 17, 8877. https://doi.org/10.3390/ijerph17238877
Popruk N, Prasongwattana S, Mahittikorn A, Palasuwan A, Popruk S, Palasuwan D. Prevalence and Subtype Distribution of Blastocystis Infection in Patients with Diabetes Mellitus in Thailand. International Journal of Environmental Research and Public Health. 2020; 17(23):8877. https://doi.org/10.3390/ijerph17238877
Chicago/Turabian StylePopruk, Noppon, Satakamol Prasongwattana, Aongart Mahittikorn, Attakorn Palasuwan, Supaluk Popruk, and Duangdao Palasuwan. 2020. "Prevalence and Subtype Distribution of Blastocystis Infection in Patients with Diabetes Mellitus in Thailand" International Journal of Environmental Research and Public Health 17, no. 23: 8877. https://doi.org/10.3390/ijerph17238877
APA StylePopruk, N., Prasongwattana, S., Mahittikorn, A., Palasuwan, A., Popruk, S., & Palasuwan, D. (2020). Prevalence and Subtype Distribution of Blastocystis Infection in Patients with Diabetes Mellitus in Thailand. International Journal of Environmental Research and Public Health, 17(23), 8877. https://doi.org/10.3390/ijerph17238877




