Influence of Ambient Air Pollution on Rheumatoid Arthritis Disease Activity Score Index
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients Data Source (Kuwait Registry for Rheumatic Diseases—KRRD)
2.2. Calculating RA Indices
2.3. Ambient Air Pollutants Data (Environmental Public Authority of Kuwait—K-EPA)
2.4. Air Pollution Data Processing and Treatment
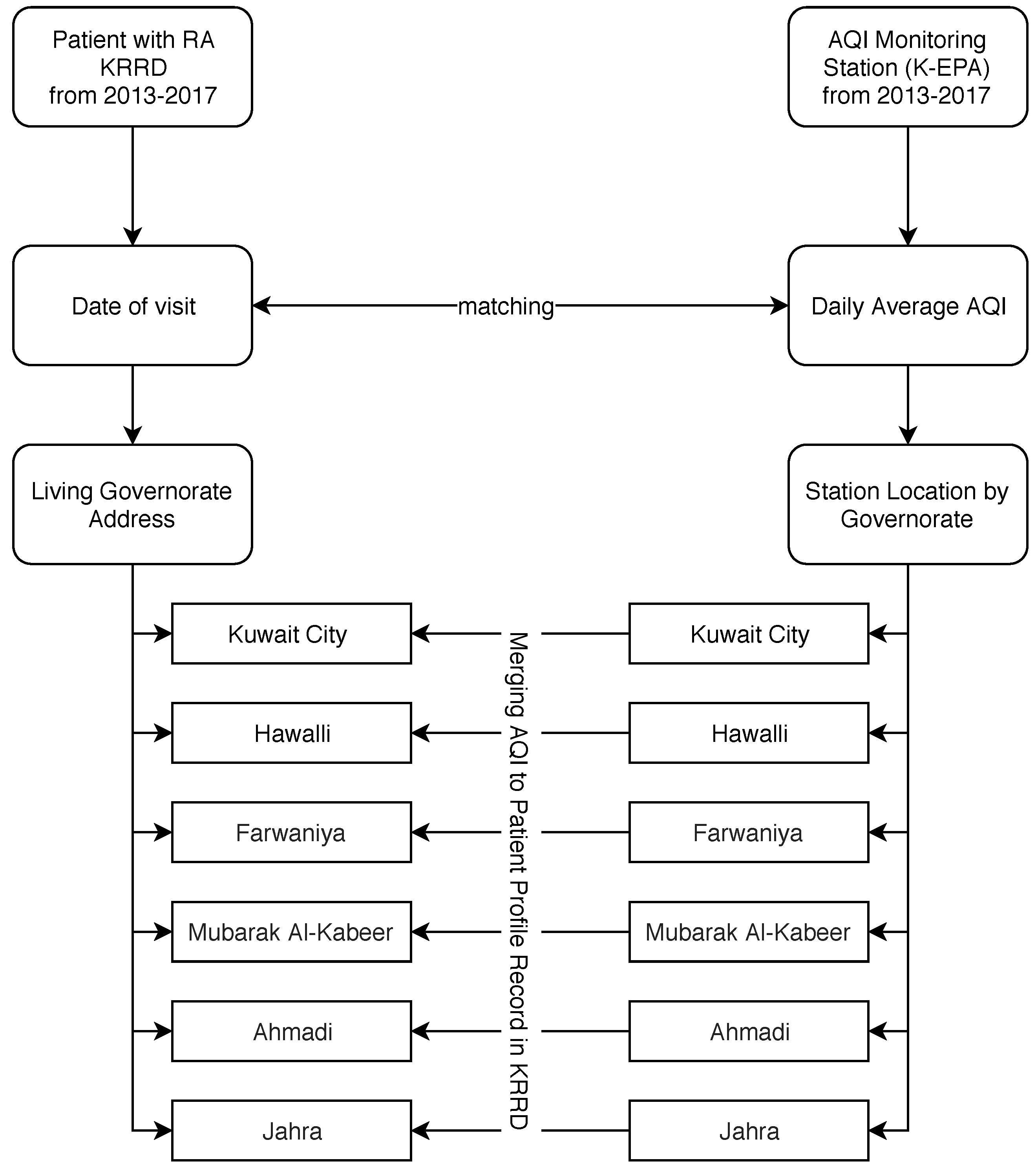
2.5. Matching Procedure between Patients and AQI
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
6. Ethical Approval
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chang, K.H.; Hsu, C.C.; Muo, C.H.; Hsu, C.Y.; Liu, H.C.; Kao, C.H.; Chen, C.Y.; Chang, M.Y.; Hsu, Y.C. Air pollution exposure increases the risk of rheumatoid arthritis: A longitudinal and nationwide study. Environ. Int. 2016, 94, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Zhang, H.; Zhou, X.; Liu, R. Association between polymorphisms of interleukin 12 and rheumatoid arthritis associated biomarkers in a Chinese population. Cytokine 2015, 28, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Solus, J.F.; Chung, C.P.; Oeser, A.; Li, C.; Rho, Y.H.; Bradley, K.M.; Kawai, V.K.; Smith, J.R.; Stein, C.M. Genetics of serum concentration of IL-6 and TNFα in systemic lupus erythematosus and rheumatoid arthritis: A candidate gene analysis. Clin. Rheumatol. 2015, 34, 1375–1382. [Google Scholar] [CrossRef] [PubMed]
- Hart, J.E.; Laden, F.; Puett, R.C.; Costenbader, K.H.; Karlson, E.W. Exposure to traffic pollution and increased risk of rheumatoid arthritis. Environ. Health Perspect. 2009, 117, 1065–1069. [Google Scholar] [CrossRef]
- De Roos, A.J.; Koehoorn, M.; Tamburic, L.; Davies, H.W.; Brauer, M. Proximity to traffic, ambient air pollution, and community noise in relation to incident rheumatoid arthritis. Environ. Health Perspect. 2014, 122, 1075–1080. [Google Scholar] [CrossRef]
- Kobayashi, S.; Okamoto, H.; Iwamoto, T.; Toyama, Y.; Tomatsu, T.; Yamanaka, H.; Momohara, S. A role for the aryl hydrocarbon receptor and the dioxin TCDD in rheumatoid arthritis. Rheumatology 2008, 47, 1317–1322. [Google Scholar] [CrossRef]
- Organization, W.H. Air Quality Management: Air Quality Guidelines; World Health Organization: Geneva, Switzerland, 1999; Available online: www.who.int/peh/air/airqualitygd.htm (accessed on 16 April 2019).
- Sun, G.; Hazlewood, G.; Bernatsky, S.; Kaplan, G.G.; Eksteen, B.; Barnabe, C. Association between air pollution and the development of rheumatic disease: A systematic review. Int. J. Rheumatol. 2016, 2016, 5356307. [Google Scholar] [CrossRef]
- Bernatsky, S.; Smargiassi, A.; Barnabe, C.; Svenson, L.W.; Brand, A.; Martin, R.V.; Hudson, M.; Clarke, A.E.; Fortin, P.R.; van Donkelaar, A.; et al. Fine particulate air pollution and systemic autoimmune rheumatic disease in two Canadian provinces. Environ. Res. 2016, 146, 85–91. [Google Scholar] [CrossRef]
- Tobón, G.J.; Youinou, P.; Saraux, A. The environment, geo-epidemiology, and autoimmune disease: Rheumatoid arthritis. Autoimmun. Rev. 2010, 9, A288–A292. [Google Scholar] [CrossRef]
- Seinfeld, J.H. Air Pollution: Physical and Chemical Fundamentals; McGraw-Hill: New York, NY, USA, 1975. [Google Scholar]
- Scott, H.M.; Soskolne, C.L.; Martin, S.W.; Ellehoj, E.A.; Coppock, R.W.; Guidotti, T.L.; Lissemore, K.D. Comparison of two atmospheric-dispersion models to assess farm-site exposure to sour-gas processing-plant emissions. Prev. Vet. Med. 2003, 57, 15–34. [Google Scholar] [CrossRef]
- Prevoo, M.; Van’T Hof, M.; Kuper, H.; Van Leeuwen, M.; Van De Putte, L.; Van Riel, P. Modified disease activity scores that include twenty-eight-joint counts development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 1995, 38, 44–48. [Google Scholar] [CrossRef] [PubMed]
- van Gestel, A.M.; Haagsma, C.J.; van Riel, P.L. Validation of rheumatoid arthritis improvement criteria that include simplified joint counts. Arthritis Rheum. 1998, 41, 1845–1850. [Google Scholar] [CrossRef]
- Fransen, J.; Creemers, M.; Van Riel, P. Remission in rheumatoid arthritis: Agreement of the disease activity score (DAS28) with the ARA preliminary remission criteria. Rheumatology 2004, 43, 1252–1255. [Google Scholar] [CrossRef] [PubMed]
- Van Riel, P. The development of the disease activity score (DAS) and the disease activity score using 28 joint counts (DAS28). Clin. Exp. Rheumatol. 2014, 32, S65–S74. [Google Scholar]
- Smolen, J.; Breedveld, F.; Schiff, M.; Kalden, J.; Emery, P.; Eberl, G.; Van Riel, P.; Tugwell, P. A simplified disease activity index for rheumatoid arthritis for use in clinical practice. Rheumatology 2003, 42, 244–257. [Google Scholar] [CrossRef]
- Martins, F.M.; da Silva, J.A.P.; Santos, M.J.; Vieira-Sousa, E.; Duarte, C.; Santos, H.; Costa, J.A.; Pimentel-Santos, F.M.; Cunha, I.; Cunha Miranda, L.; et al. DAS28, CDAI and SDAI cut-offs do not translate the same information: Results from the Rheumatic Diseases Portuguese Register Reuma. pt. Rheumatology 2014, 54, 286–291. [Google Scholar] [CrossRef][Green Version]
- For Civil Information (PACI). Directory of Population and Labor Force. Available online: https://www.paci.gov.kw/Home.aspx (accessed on 23 May 2019).
- Aletaha, D.; Neogi, T.; Silman, A.J.; Funovits, J.; Felson, D.T.; Bingham, C.O., III; Birnbaum, N.S.; Burmester, G.R.; Bykerk, V.P.; Cohen, M.D.; et al. 2010 rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef]
- Al-Herz, A.; Al-Awadhi, A.; Saleh, K.; Al-Kandari, W.; Hasan, E.; Ghanem, A.; Abutiban, F.; Alenizi, A.; Hussain, M.; Ali, Y.; et al. A comparison of rheumatoid arthritis patients in Kuwait with other populations: Results from the KRRD registry. J. Adv. Med. Med. Res. 2016, 14, 1–11. [Google Scholar] [CrossRef]
- Johnson, M.; Isakov, V.; Touma, J.; Mukerjee, S.; Özkaynak, H. Evaluation of land-use regression models used to predict air quality concentrations in an urban area. Atmos. Environ. 2010, 44, 3660–3668. [Google Scholar] [CrossRef]
- Johnson, D.L.; Ambrose, S.H.; Bassett, T.J.; Bowen, M.L.; Crummey, D.E.; Isaacson, J.S.; Johnson, D.N.; Lamb, P.; Saul, M.; Winter-Nelson, A.E. Meanings of environmental terms. J. Environ. Qual. 1997, 26, 581–589. [Google Scholar] [CrossRef]
- Al-Shayji, K.; Lababidi, H.; Al-Rushoud, D.; Al-Adwani, H. Development of a fuzzy air quality performance indicator. Kuwait J. Sci. Eng. 2008, 35, 101–126. [Google Scholar]
- Fitz-Simons, T. Guideline for Reporting of Daily Air Quality: Air Quality Index (AQI); Technical Report; Environmental Protection Agency, Office of Air Quality Planning: Washington, DC, USA, 1999. [Google Scholar]
- Reporting, A.Q.I.; Rule, F. Protection Agency. 1999. Available online: https://www3.epa.gov/airnow/40cfrpt58_aqi-reporting.pdf (accessed on 7 July 2019).
- Junninen, H.; Niska, H.; Tuppurainen, K.; Ruuskanen, J.; Kolehmainen, M. Methods for imputation of missing values in air quality data sets. Atmos. Environ. 2004, 38, 2895–2907. [Google Scholar] [CrossRef]
- Schafer, J.L. Analysis of Incomplete Multivariate Data; Chapman and Hall/CRC: London, UK, 1997. [Google Scholar]
- Core, R.; Team, R. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013; Available online: http://www.R-project.org (accessed on 16 May 2019).
- Wickham, H. The split-apply-combine strategy for data analysis. J. Stat. Softw. 2011, 40, 1–29. [Google Scholar] [CrossRef]
- Wickham, H.; Francois, R. dplyr: A Grammar of Data Manipulation (Version 0.8.3) [Software]. 2019. Available online: http://CRAN.R-project.org/package=dplyr (accessed on 7 June 2019).
- Wickham, H. tidyr: Easily Tidy Data with Spread () and Gather () Functions. R Package. Version 1.0.0. 2019. Available online: http://CRAN.R-project.org/package=tidyr (accessed on 7 June 2019).
- Wickham, H. stringr: Make it easier to work with strings. R Package Vers. 0.6 2012, 2, 96–97. [Google Scholar]
- Nelder, J.A.; Wedderburn, R.W. Generalized linear models. J. R. Stat. Soc. 1972, 135, 370–384. [Google Scholar] [CrossRef]
- Dobson, A.J.; Barnett, A.G. An Introduction to Generalized Linear Models; Chapman and Hall/CRC: London, UK, 2008. [Google Scholar]
- Hart, J.E.; Källberg, H.; Laden, F.; Costenbader, K.H.; Yanosky, J.D.; Klareskog, L.; Alfredsson, L.; Karlson, E.W. Ambient air pollution exposures and risk of rheumatoid arthritis. Arthritis Care Res. 2013, 65, 1190–1196. [Google Scholar] [CrossRef]
- Gabriel, S.E.; Crowson, C.S.; O’Fallon, M. The epidemiology of rheumatoid arthritis in Rochester, Minnesota, 1955–1985. Arthritis Rheum. 1999, 42, 415–420. [Google Scholar] [CrossRef]
- Chaudhari, K.; Rizvi, S.; Syed, B.A. Rheumatoid arthritis: Current and future trends. Nat. Rev. Drug Discov. 2016, 15, 305–306. [Google Scholar] [CrossRef]
- Hart, J.E.; Källberg, H.; Laden, F.; Bellander, T.; Costenbader, K.H.; Holmqvist, M.; Klareskog, L.; Alfredsson, L.; Karlson, E.W. Ambient air pollution exposures and risk of rheumatoid arthritis: Results from the Swedish EIRA case–control study. Ann. Rheum. Dis. 2013, 72, 888–894. [Google Scholar] [CrossRef]
- Shin, J.; Lee, J.; Lee, J.; Ha, E.H. Association between Exposure to Ambient Air Pollution and Rheumatoid Arthritis in Adults. Int. J. Environ. Res. Public Health 2019, 16, 1227. [Google Scholar] [CrossRef]
- Al-Awadhi, J. Measurement of air pollution in Kuwait City using passive samplers. Atmos. Clim. Sci. 2014, 4, 253–271. [Google Scholar] [CrossRef][Green Version]
- Hankey, S.; Marshall, J.D.; Brauer, M. Health impacts of the built environment: Within-urban variability in physical inactivity, air pollution, and ischemic heart disease mortality. Environ. Health Perspect. 2011, 5, 247–253. [Google Scholar] [CrossRef]
- Al-Mutairi, N.; Koushki, P. Potential contribution of traffic to air pollution in the State of Kuwait. Am. J. Environ. Sci. 2009, 5, 218–222. [Google Scholar] [CrossRef]
- Covic, T.; Tyson, G.; Spencer, D.; Howe, G. Depression in rheumatoid arthritis patients: Demographic, clinical, and psychological predictors. J. Psychosom. Res. 2006, 60, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Godha, D.; Shi, L.; Mavronicolas, H. Association between tendency towards depression and severity of rheumatoid arthritis from a national representative sample: The Medical Expenditure Panel Survey. Curr. Med. Res. Opin. 2010, 26, 1685–1690. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Alvaro, I.; Ortiz, A.; Garcia-Vicuna, R.; Balsa, A.; Pascual-Salcedo, D.; Laffon, A. Increased serum levels of interleukin-15 in rheumatoid arthritis with long-term disease. Clin. Exp. Rheumatol. 2003, 21, 639–642. [Google Scholar]
- Jung, C.R.; Hsieh, H.Y.; Hwang, B.F. Air pollution as a potential determinant of rheumatoid arthritis: A population-based cohort study in Taiwan. Epidemiology 2017, 28, S54–S59. [Google Scholar] [CrossRef]

| Categories | AQI, Sub-Index | O (ppm), 8-h | PM (µg/m), 24-h | CO (ppm), 24-h | SO (ppm), 24-h | NO (ppm), 24-h |
|---|---|---|---|---|---|---|
| – | – | – | – | – | – | |
| Good | 0–50 | 0.0–0.03 | 0.0–90 | 0.0–4.0 | 0.0–0.03 | 0.0–0.03 |
| Moderate | 51–100 | 0.031–0.06 | 90.1–350.0 | 4.1–8.0 | 0.031–0.06 | 0.04–0.05 |
| Unhealthy (1) | 101–150 | 0.061–0.092 | 350.1–431.1 | 8.1–11.7 | 0.061–0.182 | 0.06–0.30 |
| Unhealthy (2) | 151–200 | 0.093–0.124 | 431.4–512.5 | 11.8–15.4 | 0.183–0.304 | 0.31–0.55 |
| Very Unhealthy | 201–300 | 0.125–0.374 | 512.6–675.0 | 15.5–30.4 | 0.305–0.604 | 0.56–1.04 |
| Hazardous | 301–500 | 0.375–0.504 | 675.1–1000 | 30.5–50.4 | 0.605–1.004 | 1.05–2.04 |
| [ALL] | Ahmadi | Farwaniya | Hawally | Jahra | Kuwait City | Mubarak | |
|---|---|---|---|---|---|---|---|
| n = 9875 | n = 356 | n = 4378 | n = 1272 | n = 226 | n = 3007 | Alkabeer n = 636 | |
| Gender: | |||||||
| male | 3867 (39.2%) | 73 (20.5%) | 2656 (60.7%) | 285 (22.4%) | 82 (36.3%) | 653 (21.7%) | 118 (18.6%) |
| female | 6008 (60.8%) | 283 (79.5%) | 1722 (39.3%) | 987 (77.6%) | 144 (63.7%) | 2354 (78.3%) | 518 (81.4%) |
| Nationality: | |||||||
| Kuwaitis | 5783 (58.6%) | 328 (92.1%) | 1499 (34.2%) | 773 (60.8%) | 145 (64.2%) | 2462 (81.9%) | 576 (90.6%) |
| non-Kuwaitis | 4092 (41.4%) | 28 (7.87%) | 2879 (65.8%) | 499 (39.2%) | 81 (35.8%) | 545 (18.1%) | 60 (9.43%) |
| Visited Hospital: | |||||||
| Amiri | 5051 (51.1%) | 294 (82.6%) | 364 (8.31%) | 774 (60.8%) | 120 (53.1%) | 2955 (98.3%) | 544 (85.5%) |
| Farwaniya | 3981 (40.3%) | 0 (0.00%) | 3976 (90.8%) | 5 (0.39%) | 0 (0.00%) | 0 (0.00%) | 0 (0.00%) |
| Jahra | 111 (1.12%) | 0 (0.00%) | 2 (0.05%) | 3 (0.24%) | 106 (46.9%) | 0 (0.00%) | 0 (0.00%) |
| Mubarak | 732 (7.41%) | 62 (17.4%) | 36 (0.82%) | 490 (38.5%) | 0 (0.00%) | 52 (1.73%) | 92 (14.5%) |
| Disease Duration | 9.82 (6.48) | 13.3 (9.11) | 9.39 (5.79) | 10.4 (7.03) | 10.2 (6.45) | 9.42 (5.78) | 11.4 (9.60) |
| Comorbidity : | |||||||
| Yes | 5393 (54.6%) | 226 (63.5%) | 1658 (37.9%) | 802 (63.1%) | 123 (54.4%) | 2157 (71.7%) | 427 (67.1%) |
| No | 4482 (45.4%) | 130 (36.5%) | 2720 (62.1%) | 470 (36.9%) | 103 (45.6%) | 850 (28.3%) | 209 (32.9%) |
| Treatment Class: | |||||||
| Biologics | 5214 (52.8%) | 327 (91.9%) | 1342 (30.7%) | 762 (59.9%) | 131 (58.0%) | 2124 (70.6%) | 528 (83.0%) |
| cDMARDs | 4661 (47.2%) | 29 (8.15%) | 3036 (69.3%) | 510 (40.1%) | 95 (42.0%) | 883 (29.4%) | 108 (17.0%) |
| RF : | |||||||
| Positive | 6881 (74.6%) | 235 (72.8%) | 3148 (76.3%) | 817 (71.0%) | 177 (93.2%) | 2042 (72.0%) | 462 (76.5%) |
| Negative | 2348 (25.4%) | 88 (27.2%) | 976 (23.7%) | 334 (29.0%) | 13 (6.84%) | 795 (28.0%) | 142 (23.5%) |
| ACPA: | |||||||
| Positive | 4934 (60.5%) | 102 (33.6%) | 2665 (70.3%) | 593 (63.1%) | 73 (61.3%) | 1205 (48.1%) | 296 (60.0%) |
| Negative | 3216 (39.5%) | 202 (66.4%) | 1125 (29.7%) | 347 (36.9%) | 46 (38.7%) | 1299 (51.9%) | 197 (40.0%) |
| Patient GA | 1.64 (2.36) | 1.54 (2.34) | 1.02 (1.85) | 2.60 (2.69) | 2.09 (2.52) | 1.95 (2.56) | 2.39 (2.69) |
| Physician GA | 1.05 (1.77) | 1.06 (1.82) | 0.72 (1.50) | 1.63 (2.02) | 1.58 (2.21) | 1.13 (1.78) | 1.64 (2.18) |
| DAS-28 | 2.67 (1.26) | 1.85 (1.35) | 2.70 (1.21) | 2.77 (1.29) | 3.04 (1.39) | 2.61 (1.22) | 2.79 (1.44) |
| CDAI | 6.24 (9.96) | 4.64 (8.89) | 4.83 (8.56) | 8.31(10.72) | 9.45 (14.25) | 6.78 (10.49) | 9.00 (11.53) |
| ESR | 27.19 (21.79) | 15.12 (16.82) | 30.24 (23.10) | 26.77 (20.30) | 30.06 (19.33) | 23.97 (19.54) | 27.80 (24.04) |
| CRP | 6.32 (4.85) | 4.32 (3.91) | 7.29 (4.89) | 4.53 (4.53) | 6.36 (4.42) | 6.08 (4.68) | 5.38 (4.87) |
| Swollen Joints | 0.69 (2.26) | 0.34 (1.57) | 1.08 (2.60) | 0.53 (1.97) | 0.95 (3.63) | 0.26 (1.67) | 0.57 (1.99) |
| Tender Joints | 2.87 (5.60) | 1.72 (4.32) | 2.02 (4.18) | 3.55 (6.21) | 4.82 (8.39) | 3.46 (6.36) | 4.54 (7.13) |
| Air Pollutant | Ahmadi (n = 356) | Farwaniya (n = 4378) | Hawally (n = 1272) | Jahra (n = 226) | Kuwait City (n = 3007) | Mubarak Alkabeer (n = 636) | ALL (n = 9875) |
|---|---|---|---|---|---|---|---|
| PM | |||||||
| min | 17.108 | 20.346 | 21.948 | 18.837 | 12.138 | 5.421 | 5.421 |
| 25th | 75.839 | 76.312 | 80.692 | 67.417 | 71.114 | 74.886 | 74.965 |
| median | 112.623 | 120.779 | 113.586 | 92.788 | 116.081 | 109.917 | 113.586 |
| 75th | 180.403 | 186.145 | 180.581 | 151.762 | 193.121 | 184.282 | 186.568 |
| max | 549.419 | 511.826 | 588.494 | 545.789 | 577.706 | 585.077 | 588.494 |
| mean (SD ) | 142.36 ± 99.58 | 146.47 ± 94.20 | 145.69 ± 99.23 | 123.64 ± 95.00 | 146.81 ± 102.42 | 142.40 ± 102.79 | 144.87 ± 100.64 |
| CO | |||||||
| min | 0.240 | 0.207 | 0.087 | 0.042 | 0.087 | 0.292 | 0.042 |
| 25th | 0.938 | 0.945 | 0.984 | 0.854 | 1.006 | 0.978 | 0.975 |
| median | 1.367 | 1.338 | 1.337 | 1.151 | 1.369 | 1.380 | 1.346 |
| 75th | 1.679 | 1.603 | 1.672 | 1.455 | 1.679 | 1.728 | 1.672 |
| max | 4.894 | 4.701 | 4.471 | 5.122 | 8.143 | 5.287 | 8.143 |
| mean (SD) | 1.37 ± 0.62 | 1.33 ± 0.57 | 1.40 ± 0.66 | 1.14 ± 0.64 | 1.40 ± 0.66 | 1.46 ± 0.70 | 1.39 ± 0.66 |
| NO | |||||||
| min | 9.080 | 9.080 | 5.346 | 8.229 | 5.346 | 5.346 | 5.346 |
| 25th | 25.768 | 28.200 | 27.208 | 29.127 | 27.319 | 32.037 | 27.391 |
| median | 35.762 | 36.537 | 35.810 | 52.420 | 36.795 | 44.744 | 37.355 |
| 75th | 54.218 | 53.150 | 51.409 | 67.866 | 52.054 | 68.210 | 54.552 |
| max | 137.960 | 135.878 | 134.123 | 107.279 | 208.670 | 207.557 | 208.670 |
| mean (SD) | 42.85 ± 24.13 | 42.01 ± 20.70 | 41.29 ± 20.60 | 50.92 ± 25.54 | 43.00 ± 22.76 | 52.09 ± 27.91 | 43.74 ± 23.13 |
| O | |||||||
| min | 4.051 | 4.051 | 4.051 | 5.887 | 3.476 | 4.877 | 3.476 |
| 25th | 10.965 | 11.030 | 10.851 | 12.457 | 10.581 | 11.328 | 10.851 |
| median | 15.215 | 15.372 | 15.104 | 18.192 | 14.709 | 14.164 | 14.985 |
| 75th | 20.874 | 22.240 | 20.451 | 25.172 | 20.451 | 19.507 | 20.759 |
| max | 54.262 | 69.682 | 80.656 | 37.539 | 88.623 | 57.330 | 88.623 |
| mean (SD) | 17.04 ± 8.75 | 18.53 ± 11.27 | 17.58 ± 10.52 | 18.71 ± 7.24 | 16.85 ± 10.08 | 16.26 ± 7.51 | 17.19 ± 9.90 |
| SO | |||||||
| min | 0.003 | 1.000 | 1.000 | 0.665 | 0.003 | 1.000 | 0.003 |
| 25th | 4.208 | 5.293 | 4.875 | 5.435 | 4.490 | 7.333 | 4.875 |
| median | 8.333 | 8.000 | 8.594 | 13.792 | 7.993 | 14.083 | 8.727 |
| 75th | 14.146 | 17.292 | 17.169 | 24.583 | 16.746 | 22.946 | 17.504 |
| max | 121.833 | 121.833 | 121.833 | 76.875 | 111.917 | 127.875 | 127.875 |
| mean (SD) | 13.15 ± 15.46 | 13.26 ± 14.22 | 14.18 ± 16.10 | 17.90 ± 16.52 | 13.39 ± 14.60 | 18.62 ± 17.07 | 14.26 ± 15.42 |
| DAS-28 | CDAI | NO | O | SO | CO | PM | Swollen | Tender | ESR | |
|---|---|---|---|---|---|---|---|---|---|---|
| DAS-28 | ||||||||||
| CDAI | 0.77 **** | |||||||||
| NO | 0.07 **** | 0.11 **** | ||||||||
| O | 0.00 | 0.00 | −0.12 **** | |||||||
| SO | 0.07 **** | 0.10 **** | 0.51 **** | −0.09 **** | ||||||
| CO | −0.01 | 0.02 | 0.22 **** | 0.02 | 0.07 **** | |||||
| PM | 0.00 | −0.02 | −0.12 **** | 0.08 **** | −0.03 * | −0.05 ** | ||||
| Swollen | 0.50 **** | 0.60 **** | 0.01 | 0.01 | 0.01 | 0.00 | −0.02 | |||
| Tender | 0.72 **** | 0.93 **** | 0.13 **** | 0.01 | 0.11 **** | 0.03 | −0.01 | 0.42 **** | ||
| ESR | 0.65 **** | 0.20 **** | 0.00 | −0.02 | 0.04 * | −0.04 * | 0.02 | 0.16 **** | 0.17 **** | |
| CRP | 0.28 **** | 0.02 * | 0.01 | 0.02 | 0.01 | −0.01 | −0.01 | 0.11 **** | 0.02 * | 0.37 **** |
| Dependent Variable | ||||
|---|---|---|---|---|
| DAS-28 | ||||
| (M1) | (M2) | (M3) | (M4) | |
| Gender (male) | −0.213 *** | −0.040 *** | ||
| (−0.268, −0.157) | (−0.064, −0.017) | |||
| RA Disease Duration | −0.002 | −0.004 *** | ||
| (−0.006, 0.002) | (−0.006, −0.003) | |||
| Nationality (non-Kuwaitis) | 0.272 *** | 0.022 | ||
| (0.214, 0.331) | (−0.007, 0.051) | |||
| Governorate (Farwaniya) | 0.807 *** | 0.299 *** | ||
| (0.666, 0.947) | (0.239, 0.358) | |||
| Governorate (Hawally) | 0.852 *** | 0.277 *** | ||
| (0.704, 1.000) | (0.214, 0.341) | |||
| Governorate (Jahra) | 1.143 *** | 0.254 *** | ||
| (0.933, 1.352) | (0.150, 0.359) | |||
| Governorate (Kuwait City) | 0.744 *** | 0.290 *** | ||
| (0.606, 0.882) | (0.232, 0.348) | |||
| Governorate (Mubarak Alkabeer) | 0.955 *** | 0.175 *** | ||
| (0.793, 1.117) | (0.106, 0.244) | |||
| Comorbidity (Yes) | 0.060 ** | −0.051 *** | ||
| (0.007, 0.114) | (−0.074, −0.029) | |||
| Treatment Class (cDMARDs) | 0.064 *** | |||
| (0.036, 0.092) | ||||
| Swollen | 0.090 *** | 0.226 *** | ||
| (0.085, 0.095) | (0.208, 0.244) | |||
| Tender | 0.099 *** | |||
| (0.096, 0.101) | ||||
| RF (Positive) | 0.035 *** | 0.004 | ||
| (0.010, 0.060) | (−0.078, 0.085) | |||
| ACPA (Positive) | 0.008 | 0.007 | ||
| (−0.015, 0.031) | (−0.063, 0.078) | |||
| Patient Global Assessment | 0.097 *** | |||
| (0.088, 0.105) | ||||
| Physician Global Assessment | 0.014 ** | |||
| (0.002, 0.025) | ||||
| ESR | 0.028 *** | 0.035 *** | ||
| (0.027, 0.028) | (0.034, 0.037) | |||
| CRP | 0.017 *** | 0.001 | ||
| (0.015, 0.020) | (−0.006, 0.009) | |||
| NO | 0.003 ** | 0.003 *** | ||
| (0.001, 0.005) | (0.002, 0.005) | |||
| O | 0.002 | 0.003 | ||
| (−0.002, 0.006) | (−0.001, 0.006) | |||
| SO | 0.004 *** | 0.003 ** | ||
| (0.001, 0.007) | (0.0004, 0.005) | |||
| CO | −0.051 | −0.001 | ||
| (−0.114, 0.012) | (−0.053, 0.052) | |||
| PM | 0.0002 | 0.00003 | ||
| (−0.0002, 0.001) | (−0.0003, 0.0004) | |||
| Constant | 1.845 *** | 1.029 *** | 2.586 *** | 1.506 *** |
| (1.701, 1.988) | (0.966, 1.093) | (2.435, 2.738) | (1.358, 1.654) | |
| R | 0.034 | 0.865 | 0.007 | 0.488 |
| Adjusted R | 0.033 | 0.865 | 0.006 | 0.486 |
| Dependent Variable | ||||
|---|---|---|---|---|
| CDAI | ||||
| (M1) | (M2) | (M3) | (M4) | |
| Gender (male) | −0.748 *** | −0.078 | ||
| (−1.187, −0.309) | (−0.218, 0.061) | |||
| RA Disease Duration | −0.052 *** | 0.005 | ||
| (−0.082, −0.021) | (−0.005, 0.015) | |||
| Nationality (non-Kuwaitis) | 1.209 *** | 0.255 *** | ||
| (0.747, 1.671) | (0.081, 0.429) | |||
| Governorate (Farwaniya) | 0.066 | −1.199 *** | ||
| (−1.044, 1.176) | (−1.550, −0.848) | |||
| Governorate (Hawally) | 3.408 *** | 0.526 *** | ||
| (2.241, 4.576) | (0.152, 0.900) | |||
| Governorate (Jahra) | 4.662 *** | −0.559 * | ||
| (3.008, 6.315) | (−1.179, 0.062) | |||
| Governorate (Kuwait City) | 1.947 *** | −0.135 | ||
| (0.858, 3.036) | (−0.476, 0.207) | |||
| Governorate (Mubarak Alkabeer) | 4.430 *** | −0.064 | ||
| (3.150, 5.709) | (−0.474, 0.345) | |||
| Comorbidity (Yes) | 0.984 *** | 0.147 ** | ||
| (0.564, 1.405) | (0.014, 0.281) | |||
| Treatment Class (cDMARDs) | −0.059 | |||
| (−0.225, 0.107) | ||||
| Swollen | 1.145 *** | 3.035 *** | ||
| (1.114, 1.175) | (2.848, 3.222) | |||
| Tender | 1.423 *** | |||
| (1.410, 1.435) | ||||
| RF (Positive) | 0.025 | −0.066 | ||
| (−0.123, 0.173) | (−0.901, 0.770) | |||
| ACPA (Positive) | 0.226 *** | −0.594 | ||
| (0.090, 0.363) | (−1.320, 0.132) | |||
| Patient Global Assessment | 1.067 *** | |||
| (1.059, 1.075) | ||||
| Physician Global Assessment | 0.871 *** | |||
| (0.861, 0.881) | ||||
| ESR | 0.019 *** | 0.085 *** | ||
| (0.016, 0.022) | (0.068, 0.103) | |||
| CRP | −0.050 *** | −0.171 *** | ||
| (−0.064, −0.037) | (−0.246, −0.095) | |||
| NO | 0.040 *** | 0.048 *** | ||
| (0.022, 0.058) | (0.030, 0.066) | |||
| O | 0.027 | 0.039 ** | ||
| (−0.008, 0.062) | (0.003, 0.074) | |||
| SO | 0.044 *** | 0.044 *** | ||
| (0.018, 0.070) | (0.018, 0.070) | |||
| CO | −0.062 | 0.185 | ||
| (−0.601, 0.476) | (−0.358, 0.729) | |||
| PM | 0.001 | 0.0004 | ||
| (−0.003, 0.004) | (−0.003, 0.004) | |||
| Constant | 4.537 *** | 1.322 *** | 5.306 *** | 2.540 *** |
| (3.404, 5.671) | (0.948, 1.697) | (4.006, 6.606) | (1.017, 4.064) | |
| R | 0.030 | 0.925 | 0.015 | 0.299 |
| Adjusted R | 0.029 | 0.925 | 0.014 | 0.297 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alsaber, A.; Pan, J.; Al-Herz, A.; Alkandary, D.S.; Al-Hurban, A.; Setiya, P.; on behalf of the KRRD Group. Influence of Ambient Air Pollution on Rheumatoid Arthritis Disease Activity Score Index. Int. J. Environ. Res. Public Health 2020, 17, 416. https://doi.org/10.3390/ijerph17020416
Alsaber A, Pan J, Al-Herz A, Alkandary DS, Al-Hurban A, Setiya P, on behalf of the KRRD Group. Influence of Ambient Air Pollution on Rheumatoid Arthritis Disease Activity Score Index. International Journal of Environmental Research and Public Health. 2020; 17(2):416. https://doi.org/10.3390/ijerph17020416
Chicago/Turabian StyleAlsaber, Ahmad, Jiazhu Pan, Adeeba Al-Herz, Dhary S. Alkandary, Adeeba Al-Hurban, Parul Setiya, and on behalf of the KRRD Group. 2020. "Influence of Ambient Air Pollution on Rheumatoid Arthritis Disease Activity Score Index" International Journal of Environmental Research and Public Health 17, no. 2: 416. https://doi.org/10.3390/ijerph17020416
APA StyleAlsaber, A., Pan, J., Al-Herz, A., Alkandary, D. S., Al-Hurban, A., Setiya, P., & on behalf of the KRRD Group. (2020). Influence of Ambient Air Pollution on Rheumatoid Arthritis Disease Activity Score Index. International Journal of Environmental Research and Public Health, 17(2), 416. https://doi.org/10.3390/ijerph17020416





