Health Risk, Functional Markers and Cognitive Status in Institutionalized Older Adults: A Longitudinal Study
Abstract
1. Introduction
2. Materials and Methods
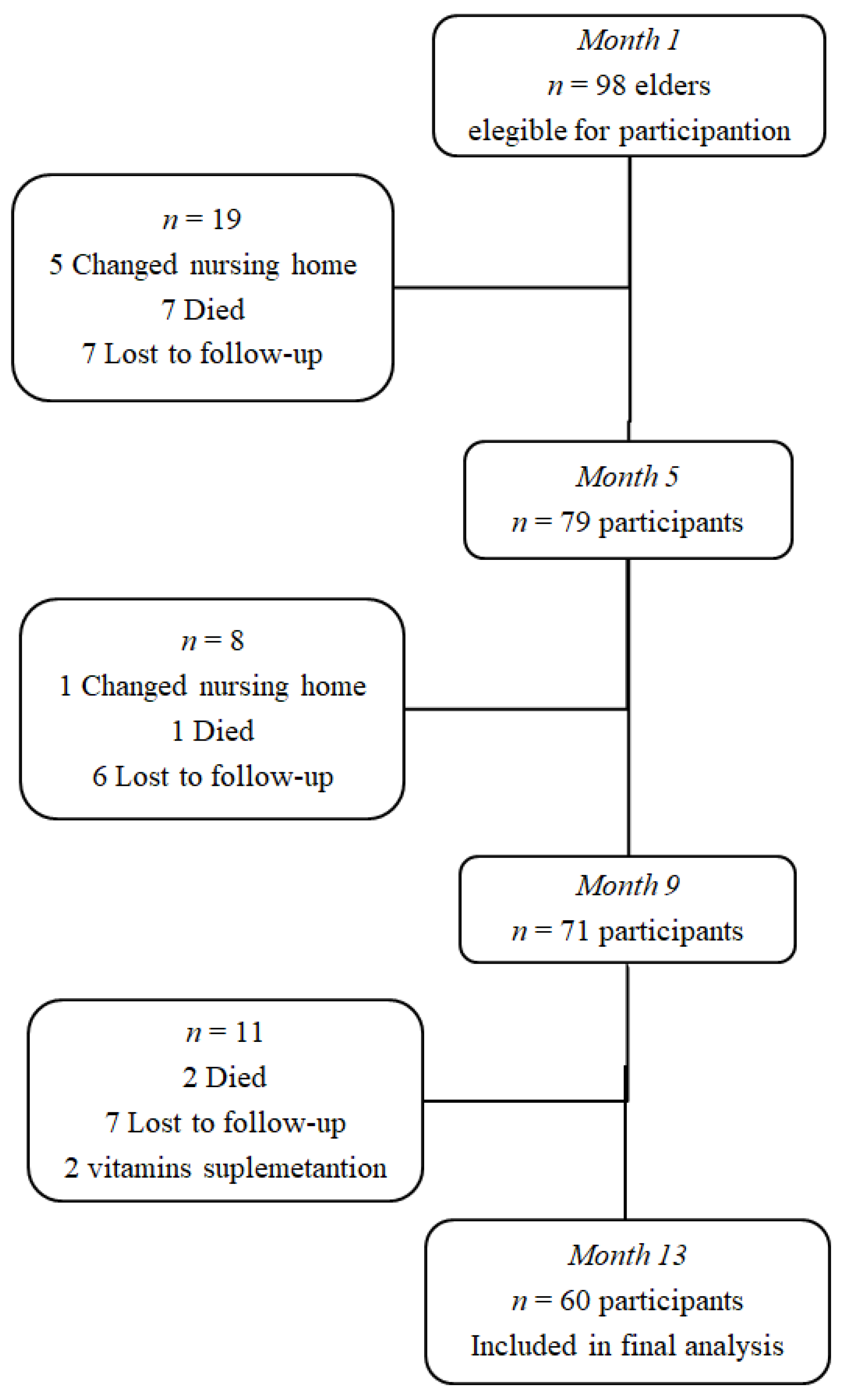
2.1. Study Population
2.2. Blood Sampling
2.3. Biomarker Measurements
2.4. Functional Capacity
2.5. Cognitive Status
2.6. Confounders
2.7. Statistical Analysis
3. Results
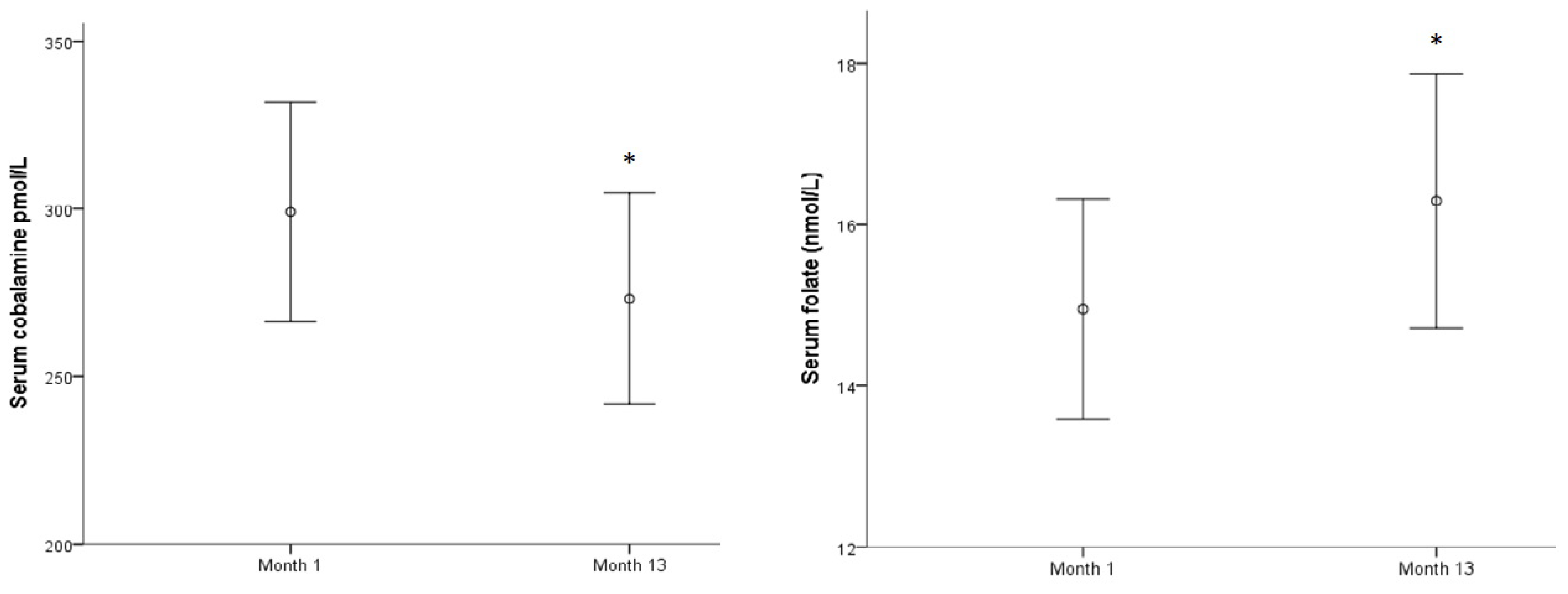
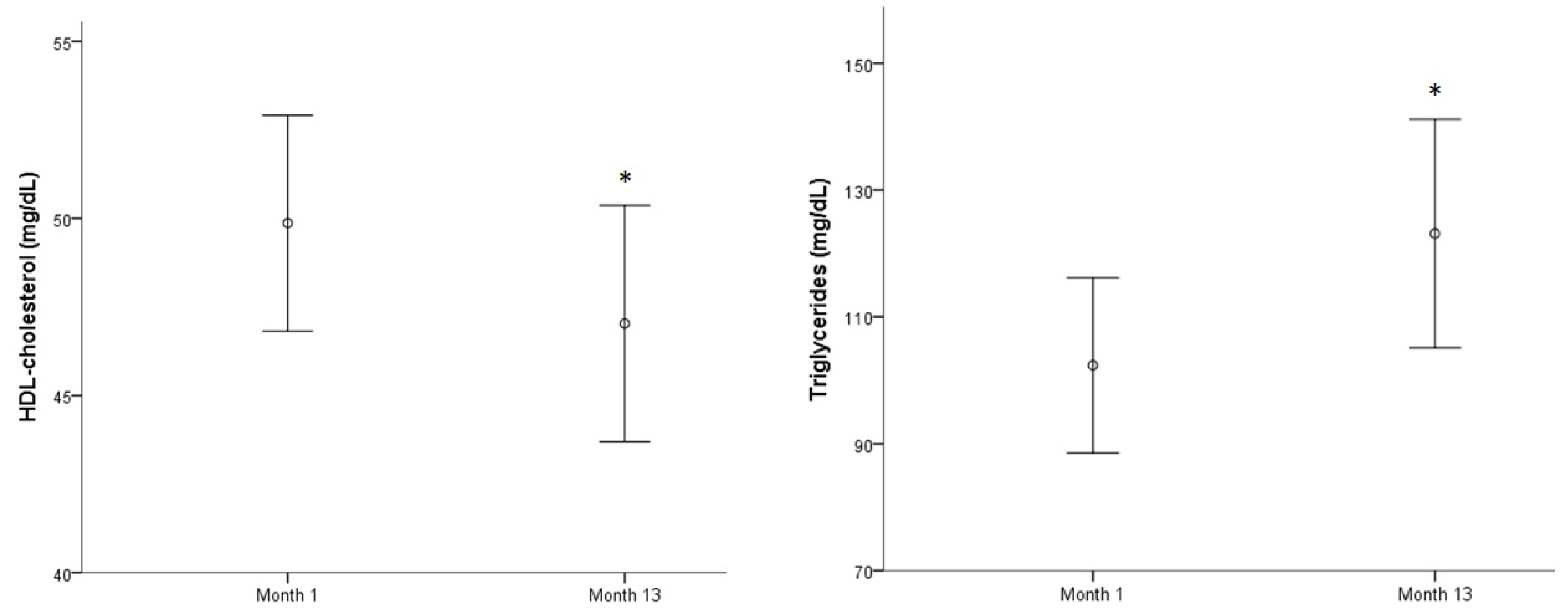
3.1. Evolution of Biomarkers and Cognitive and Physical Tests over a Year
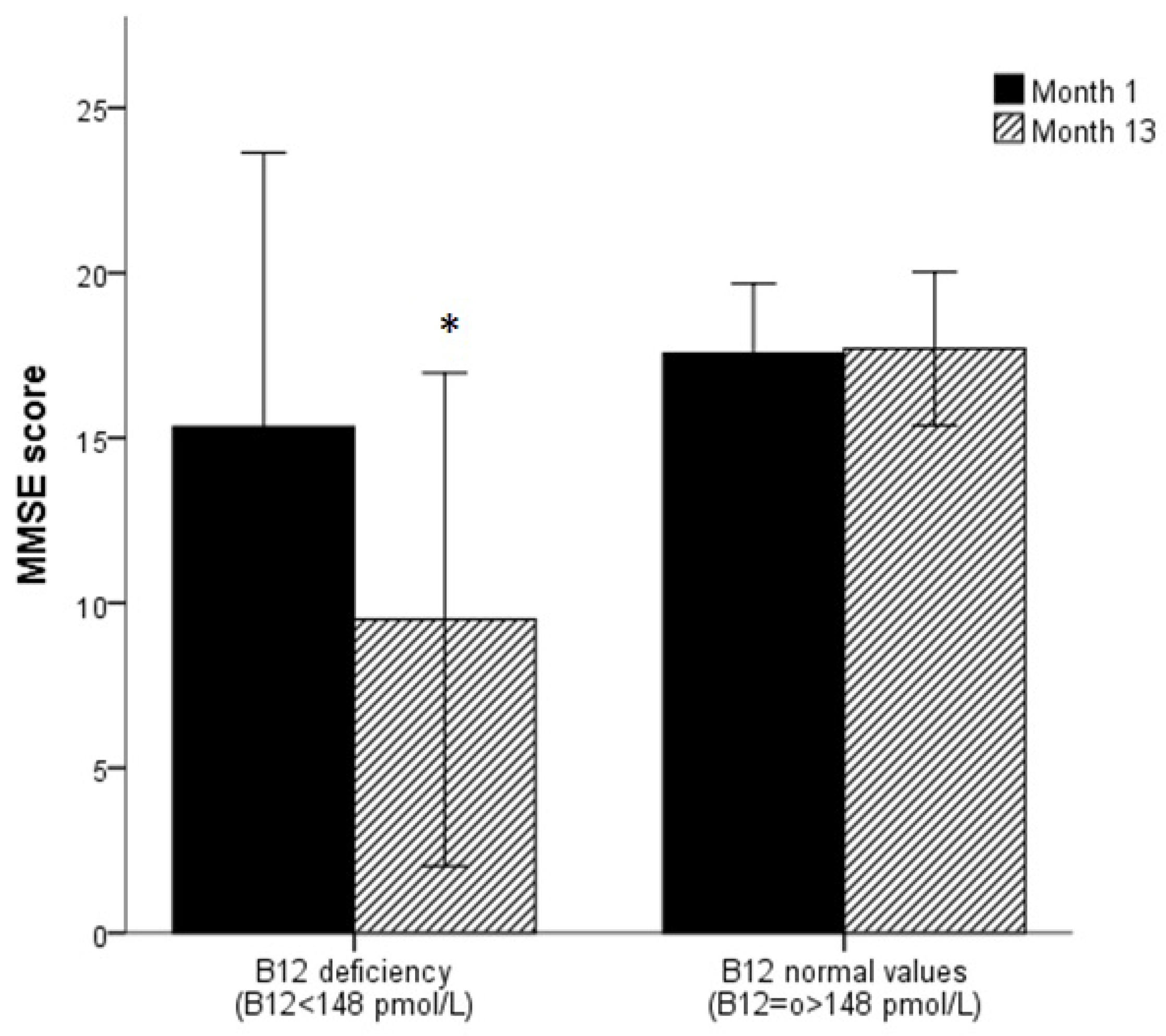
3.2. Association between Biomarkers and MMSE Score
3.3. Association between Biomarkers and Functional Tests
4. Discussion
4.1. Evolution of Biomarkers and Cognitive and Functional Tests over a Year
4.2. Biomarkers and Cognitive Status
4.3. Biomarkers and Physical Strength
4.4. Biomarkers, Cognitive Status, and Physical Strength
4.5. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United Nations Department of Public Information. World Population Prospects 2019: Highlights. Available online: https://www.un.org/es/sections/issues-depth/population/index.html (accessed on 10 March 2020).
- Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S.; American College of Sports Medicine. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef] [PubMed]
- Rosa, T.E.; Benicio, M.H.; Latorre Mdo, R.; Ramos, L.R. Determinant factors of functional status among the elderly. Rev. Saude Publica 2003, 37, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Vidal Domínguez, M.J. Aspectos Sociodemográficos de la Población Mayor. In INFORME 2016: Las Personas Mayores en España: Datos Estadísticos Estatales y por Comunidades Autónomas; Fernández Muñoz, J.N., Ed.; Ministerio de Sanidad, Servicios Sociales e Igualdad. Secretaria de Estado de Servicios Sociales e Instituto de Mayores y Servicios Sociales (IMSERSO): Madrid, Spain, 2016. [Google Scholar]
- Rowe, J.W.; Kahn, R.L. Successful aging. Gerontologist 1997, 37, 433–440. [Google Scholar] [CrossRef] [PubMed]
- OMS. La Situación Demográfica en el Mundo en 2014. 2018. Available online: https://www.un.org/en/development/desa/population/publications/pdf/trends/Concise%20Report%20on%20the%20World%20Population%20Situation%202014/es.pdf (accessed on 8 February 2020).
- Upadhyay, R.K. Emerging risk biomarkers in cardiovascular diseases and disorders. J. Lipids 2015, 2015, 971453. [Google Scholar] [CrossRef]
- Mozos, I.; Stoian, D.; Luca, C.T. Crosstalk between Vitamins A, B12, D, K, C, and E Status and Arterial Stiffness. Dis. Markers 2017, 2017, 8784971. [Google Scholar] [CrossRef]
- Gonzalez-Gross, M.; Sola, R.; Albers, U.; Barrios, L.; Alder, M.; Castillo, M.J.; Pietrzik, K. B-vitamins and homocysteine in Spanish institutionalized elderly. Int. J. Vitam. Nutr. Res. 2007, 77, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Albers, U.; Palacios, G.; Pedrero-Chamizo, R.; Melendez, A.; Pietrzik, K.; Gonzalez-Gross, M. Polymedication in institutionalized elderly and its effect on vitamin B12, folate, and homocysteine levels. Nutr. Hosp. 2012, 27, 298–302. [Google Scholar] [PubMed]
- Wilson, D.H.; Yu, J.; Karian, A.; Kozlowski, J.; O’Reilly, S. Development and multisite evaluation of an automated assay for B12 on the Abbott AxSYM analyzer. Clin. Chem. 1999, 45, 428–429. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.; Wilson, L.; McGregor, L.; Valente, E.; Orning, L. Active B12: A rapid, automated assay for holotranscobalamin on the Abbott AxSYM analyzer. Clin. Chem. 2008, 54, 567–573. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.; Osikowicz, G. Abbott AxSYM random and continuous access immunoassay system for improved workflow in the clinical laboratory. Clin. Chem. 1993, 39, 2063–2069. [Google Scholar] [CrossRef]
- Pernet, P.; Lasnier, E.; Vaubourdolle, M. Evaluation of the AxSYM homocysteine assay and comparison with the IMx homocysteine assay. Clin. Chem. 2000, 46, 1440–1441. [Google Scholar] [CrossRef] [PubMed]
- Pedrero-Chamizo, R.; Albers, U.; Tobaruela, J.L.; Melendez, A.; Castillo, M.J.; Gonzalez-Gross, M. Physical strength is associated with Mini-Mental State Examination scores in Spanish institutionalized elderly. Geriatr. Gerontol. Int. 2013, 13, 1026–1034. [Google Scholar] [CrossRef] [PubMed]
- Rikli, R.E.; Jones, C. Senior Fitness Test Manual; Human Kinetics: Champaign, IL, USA, 2001. [Google Scholar]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Blesa, R.; Pujol, M.; Aguilar, M.; Santacruz, P.; Bertran-Serra, I.; Hernandez, G.; Sol, J.M.; Pena-Casanova, J.; NORMACODEM Group. Clinical validity of the ‘mini-mental state’ for Spanish speaking communities. Neuropsychologia 2001, 39, 1150–1157. [Google Scholar] [CrossRef]
- Kalmijn, S.; Launer, L.J.; Lindemans, J.; Bots, M.L.; Hofman, A.; Breteler, M.M. Total homocysteine and cognitive decline in a community-based sample of elderly subjects: The Rotterdam Study. Am. J. Epidemiol. 1999, 150, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Mooijaart, S.P.; Gussekloo, J.; Frolich, M.; Jolles, J.; Stott, D.J.; Westendorp, R.G.; De Craen, A.J. Homocysteine, vitamin B-12, and folic acid and the risk of cognitive decline in old age: The Leiden 85-Plus study. Am. J. Clin. Nutr. 2005, 82, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Tangney, C.C.; Tang, Y.; Evans, D.A.; Morris, M.C. Biochemical indicators of vitamin B12 and folate insufficiency and cognitive decline. Neurology 2009, 72, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Mendonca, N.; Granic, A.; Mathers, J.C.; Martin-Ruiz, C.; Wesnes, K.A.; Seal, C.J.; Jagger, C.; Hill, T.R. One-Carbon Metabolism Biomarkers and Cognitive Decline in the Very Old: The Newcastle 85+ Study. J. Am. Med. Dir. Assoc. 2017, 18, 806.e819–806.e827. [Google Scholar] [CrossRef]
- Rabensteiner, J.; Hofer, E.; Fauler, G.; Fritz-Petrin, E.; Benke, T.; Dal-Bianco, P.; Ransmayr, G.; Schmidt, R.; Herrmann, M. The impact of folate and vitamin B12 status on cognitive function and brain atrophy in healthy elderly and demented Austrians, a retrospective cohort study. Aging 2017, 12, 15478–15491. [Google Scholar] [CrossRef]
- Dufouil, C.; Alperovitch, A.; Ducros, V.; Tzourio, C. Homocysteine, white matter hyperintensities, and cognition in healthy elderly people. Ann. Neurol. 2003, 53, 214–221. [Google Scholar] [CrossRef]
- Baroni, L.; Bonetto, C.; Rizzo, G.; Bertola, C.; Caberlotto, L.; Bazzerla, G. Association Between Cognitive Impairment and Vitamin B12, Folate, and Homocysteine Status in Elderly Adults: A Retrospective Study. J. Alzheimers Dis. 2019, 70, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Haron, Y.; Pulman, K.; Hua, L.; Freedman, M. Increases in homocysteine are related to worsening of stroop scores in healthy elderly persons: A prospective follow-up study. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, 1323–1327. [Google Scholar] [CrossRef] [PubMed]
- Hooshmand, B.; Solomon, A.; Kareholt, I.; Leiviska, J.; Rusanen, M.; Ahtiluoto, S.; Winblad, B.; Laatikainen, T.; Soininen, H.; Kivipelto, M. Homocysteine and holotranscobalamin and the risk of Alzheimer disease: A longitudinal study. Neurology 2010, 75, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Van Exel, E.; de Craen, A.J.; Gussekloo, J.; Houx, P.; Bootsma-van der Wiel, A.; Macfarlane, P.W.; Blauw, G.J.; Westendorp, R.G. Association between high-density lipoprotein and cognitive impairment in the oldest old. Ann. Neurol. 2002, 51, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Svensson, T.; Sawada, N.; Mimura, M.; Nozaki, S.; Shikimoto, R.; Tsugane, S. The association between midlife serum high-density lipoprotein and mild cognitive impairment and dementia after 19 years of follow-up. Transl. Psychiatry 2019, 9, 26. [Google Scholar] [CrossRef]
- Atzmon, G.; Gabriely, I.; Greiner, W.; Davidson, D.; Schechter, C.; Barzilai, N. Plasma HDL levels highly correlate with cognitive function in exceptional longevity. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, M712–M715. [Google Scholar] [CrossRef]
- Van Schoor, N.M.; Swart, K.M.; Pluijm, S.M.; Visser, M.; Simsek, S.; Smulders, Y.; Lips, P. Cross-sectional and longitudinal association between homocysteine, vitamin B12 and physical performance in older persons. Eur. J. Clin. Nutr. 2012, 66, 174–181. [Google Scholar] [CrossRef]
- Kawamoto, R.; Kohara, K.; Katoh, T.; Kusunoki, T.; Ohtsuka, N.; Abe, M.; Kumagi, T.; Miki, T. Changes in oxidized low-density lipoprotein cholesterol are associated with changes in handgrip strength in Japanese community-dwelling persons. Endocrine 2015, 48, 871–877. [Google Scholar] [CrossRef]
- Nikkila, M.; Heikkinen, J. High-density lipoprotein cholesterol and longevity. Age Ageing 1990, 19, 119–124. [Google Scholar] [CrossRef]
- Wallace, R.B.; Colsher, P.L. Blood lipid distributions in older persons. Prevalence and correlates of hyperlipidemia. Ann. Epidemiol. 1992, 2, 15–21. [Google Scholar] [CrossRef]
- An, K.O.; Kim, J. Associations between Handgrip Strength with Adverse Cardiometabolic Health among Representative Korean Adults. Int. J. Environ. Res. Public Health 2019, 17, 269. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Russo, A.; Cesari, M.; Pahor, M.; Bernabei, R.; Onder, G. HDL-cholesterol and physical performance: Results from the ageing and longevity study in the sirente geographic area (ilSIRENTE Study). Age Ageing 2007, 36, 514–520. [Google Scholar] [CrossRef] [PubMed]
- Zuliani, G.; Volpatol, S.; Romagnoni, F.; Soattin, L.; Bollini, C.; Leoci, V.; Fellin, R. Combined measurement of serum albumin and high-density lipoprotein cholesterol strongly predicts mortality in frail older nursing-home residents. Aging Clin. Exp. Res. 2004, 16, 472–475. [Google Scholar] [CrossRef] [PubMed]
- Alfaro-Acha, A.; Al Snih, S.; Raji, M.A.; Kuo, Y.F.; Markides, K.S.; Ottenbacher, K.J. Handgrip strength and cognitive decline in older Mexican Americans. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 859–865. [Google Scholar] [CrossRef] [PubMed]
- Hamer, M.; Chida, Y. Physical activity and risk of neurodegenerative disease: A systematic review of prospective evidence. Psychol. Med. 2009, 39, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Raji, M.A.; Kuo, Y.F.; Snih, S.A.; Markides, K.S.; Peek, M.K.; Ottenbacher, K.J. Cognitive status, muscle strength, and subsequent disability in older Mexican Americans. J. Am. Geriatr. Soc. 2005, 53, 1462–1468. [Google Scholar] [CrossRef]
- Fritz, N.E.; McCarthy, C.J.; Adamo, D.E. Handgrip strength as a means of monitoring progression of cognitive decline—A scoping review. Ageing Res. Rev. 2017, 35, 112–123. [Google Scholar] [CrossRef]
- Dallora, A.L.; Minku, L.; Mendes, E.; Rennemark, M.; Anderberg, P.; Sanmartin Berglund, J. Multifactorial 10-Year Prior Diagnosis Prediction Model of Dementia. Int. J. Environ. Res. Public Health 2020, 17, 6674. [Google Scholar] [CrossRef]
- Schalk, B.W.; Deeg, D.J.; Penninx, B.W.; Bouter, L.M.; Visser, M. Serum albumin and muscle strength: A longitudinal study in older men and women. J. Am. Geriatr. Soc. 2005, 53, 1331–1338. [Google Scholar] [CrossRef]
- Taniguchi, Y.; Kitamura, A.; Kaito, S.; Yokoyama, Y.; Yokota, I.; Shinozaki, T.; Seino, S.; Murayama, H.; Matsuyama, Y.; Ikeuchi, T.; et al. Albumin and Hemoglobin Trajectories and Incident Disabling Dementia in Community-Dwelling Older Japanese. Dement. Geriatr. Cogn. Disord. 2019, 47, 233–242. [Google Scholar] [CrossRef]
- Bruno, G.; Merletti, F.; Biggeri, A.; Bargero, G.; Prina-Cerai, S.; Pagano, G.; Cavallo-Perin, P. Effect of age on the association of non-high-density-lipoprotein cholesterol and apolipoprotein B with cardiovascular mortality in a Mediterranean population with type 2 diabetes: The Casale Monferrato study. Diabetologia 2006, 49, 937–944. [Google Scholar] [CrossRef] [PubMed][Green Version]
| Gender | Overall |
|---|---|
| Men | 19 |
| Women | 41 |
| Age, years | |
| Men | 80.6 (9.9) |
| Women | 85.1 (5.5) |
| Weight, Kg | |
| Men | 70.0 (11.0) |
| Women | 59.3 (5.2) |
| BMI, kg/m2 | |
| Men | 27.9 (4.1) |
| Women | 28.3 (5.2) |
| Abbreviations: BMI = body mass index | |
| Biomarkers | MMSE Score | Handgrip Strength (kg) | Leg Strength (rep) | Arm Strength (rep) |
|---|---|---|---|---|
| Serum cobalamine, pmol/L | 0.219 ** | 0.093 | −0.065 | 0.078 |
| Holotranscobalamin | −0.070 | 0.039 | 0.060 | 0.023 |
| Serum folate, nmol/L | 0.028 | −0.048 | −0.001 | −0.001 |
| RBC folate, nmol/L | −0.012 | −0.171 * | −0.084 | −0.101 |
| Homocysteine, µmol/L | −0.077 | −0.068 | −0.042 | −0.058 |
| Creatinine, µmol/L | −0.046 | 0.085 | −0.024 | 0.053 |
| Triglycerides, mg/dL | −0.203 ** | −0.139 * | 0.067 | −0.118 |
| Total cholesterol, mg/dL | −0.132 | −0.172 * | 0.134 | −0.054 |
| LDL-cholesterol, mg/dL | −0.153 * | −0.162 * | 0.138 | −0.153 * |
| HDL-cholesterol, mg/dL | 0.182 * | 0.006 | 0.137 | 0.208 * |
| Glucose, mg/dL | −0.105 | −0.021 | −0.041 | −0.016 |
| Apolipoprotein A1, mg/dL | 0.178 * | 0.068 | 0.243 ** | 0.325 ** |
| Apolipoprotein B, mg/dL | −0.091 | −0.111 | 0.154 * | −0.072 |
| Lipoprotein A, mg/dL | 0.107 | −0.126 | 0.018 | −0.066 |
| Albumin, g/dL | 0.277 ** | 0.180 * | 0.338 ** | 0.286 * |
| Covariates | MMSE Score | Handgrip Strength (kg) | Leg Strength (kg) | Arm Strength (kg) | ||||
|---|---|---|---|---|---|---|---|---|
| Param. | p | Param. | p | Param. | p | Param. | p | |
| Serum cobalamin, pmol/L | 0.009 | 0.011 | - | - | - | - | - | - |
| HDL-cholesterol, mg/dL | 0.151 | <0.001 | 0.127 | <0.001 | - | - | - | - |
| LDL-cholesterol, mg/dL | - | - | - | - | 0.026 | 0.031 | - | - |
| Apolipoprotein A, mg/dL | - | - | - | - | - | - | 0.072 | <0.001 |
| Albumin, g/dL | - | - | - | - | 3.906 | <0.001 | 2.039 | 0.014 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pedrero-Chamizo, R.; Albers, U.; Palacios, G.; Pietrzik, K.; Meléndez, A.; González-Gross, M. Health Risk, Functional Markers and Cognitive Status in Institutionalized Older Adults: A Longitudinal Study. Int. J. Environ. Res. Public Health 2020, 17, 7303. https://doi.org/10.3390/ijerph17197303
Pedrero-Chamizo R, Albers U, Palacios G, Pietrzik K, Meléndez A, González-Gross M. Health Risk, Functional Markers and Cognitive Status in Institutionalized Older Adults: A Longitudinal Study. International Journal of Environmental Research and Public Health. 2020; 17(19):7303. https://doi.org/10.3390/ijerph17197303
Chicago/Turabian StylePedrero-Chamizo, Raquel, Ulrike Albers, Gonzalo Palacios, Klaus Pietrzik, Agustín Meléndez, and Marcela González-Gross. 2020. "Health Risk, Functional Markers and Cognitive Status in Institutionalized Older Adults: A Longitudinal Study" International Journal of Environmental Research and Public Health 17, no. 19: 7303. https://doi.org/10.3390/ijerph17197303
APA StylePedrero-Chamizo, R., Albers, U., Palacios, G., Pietrzik, K., Meléndez, A., & González-Gross, M. (2020). Health Risk, Functional Markers and Cognitive Status in Institutionalized Older Adults: A Longitudinal Study. International Journal of Environmental Research and Public Health, 17(19), 7303. https://doi.org/10.3390/ijerph17197303







