Chemical Characterization and Seasonality of Ambient Particles (PM2.5) in the City Centre of Addis Ababa
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Setting
2.2. Sampling Approach
2.3. Laboratory Analysis
2.4. Data QA/QC
2.5. Chemical Mass Closure
2.6. Data Analysis
3. Results
3.1. PM2.5 Concentration
3.2. PM2.5 Bulk Composition
3.3. Organic and Elemental Carbon
3.4. Dust Contributions and Components
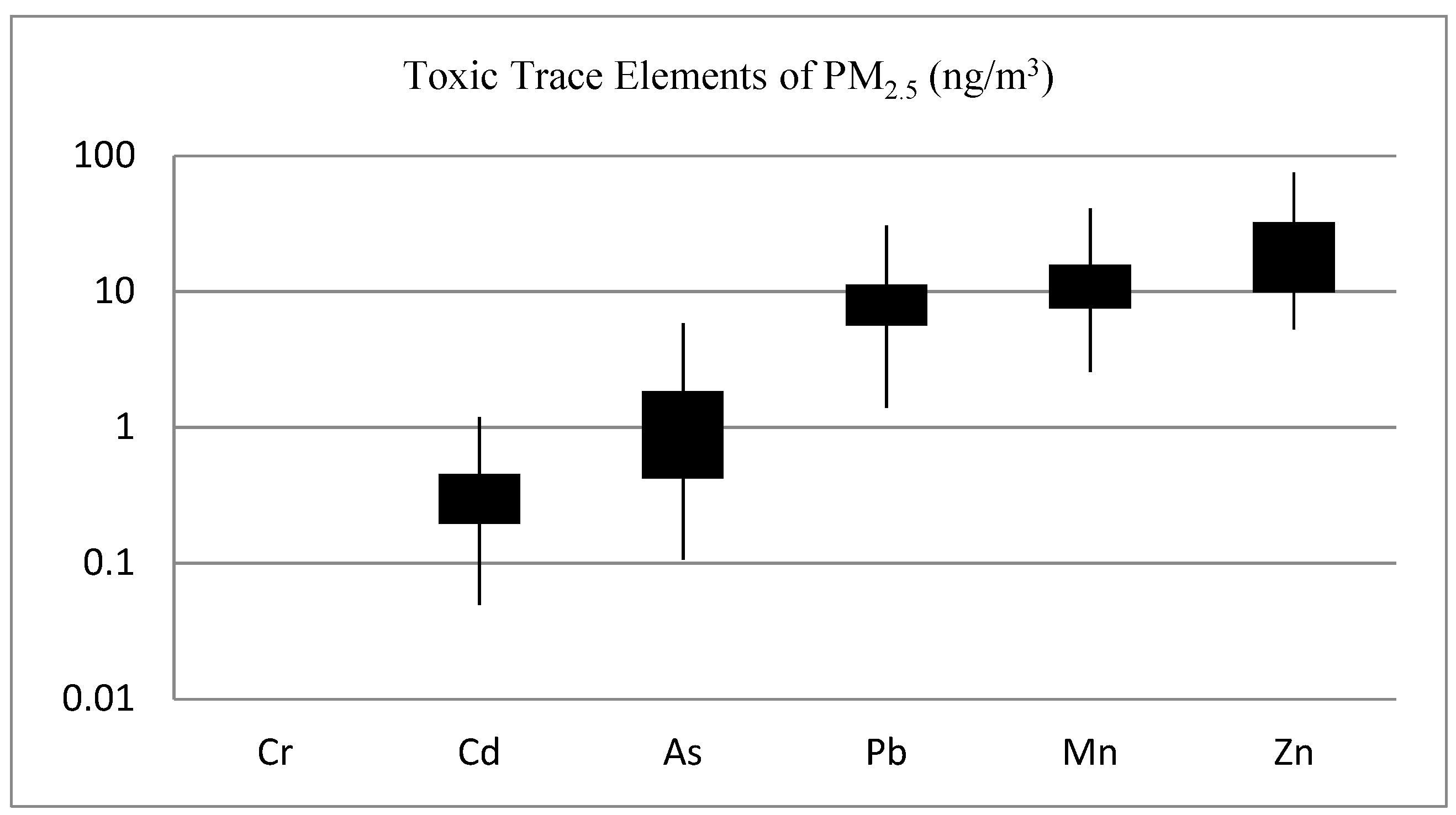
3.5. Trace Elements and Heavy Metals
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Forouzanfar, M.H.; Afshin, A.; Alexander, L.T.; Anderson, H.R.; Bhutta, Z.A.; Biryukov, S.; Brauer, M.; Burnett, R.; Cercy, K.; Charlson, F.J.; et al. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2017, 388, 1990–2015. [Google Scholar] [CrossRef]
- EPA. NAAQS Table. United States Environmental Protection Agency. 2016. Available online: https://www.epa.gov/criteria-air-pollutants/naaqs-table (accessed on 10 September 2020).
- Londahl, J.; Massling, A.; Pagels, J.; Swietlicki, E.; Vaclavik, E.; Loft, S. Size-Resolved Respiratory-Tract Deposition of Fine and Ultrafine Hydrophobic and Hygroscopic Aerosol Particles During Rest and Exercise. Inhal. Toxicol. 2007, 19, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Löndahl, J.; Pagels, J.; Swietlicki, E.; Zhou, J.; Ketzel, M.; Massling, A.; Bohgard, M. A set-up for field studies of respiratory tract deposition of fine and ultrafine particles in humans. Aerosol Sci. 2006, 37, 1152–1163. [Google Scholar]
- Fu, M.; Zheng, F.; Xu, X.; Nui, L. Advances of study on monitoring and evaluation of PM2.5 pollution. Meteorol. Disaster Reduc. Res 2011, 34, 1–6. [Google Scholar]
- Kim, K.-H.; Kabir, E.; Kabir, S. A review on the human health impact of airborne particulate matter. Environ. Int. 2015, 74, 136–143. [Google Scholar]
- Anderson, J.O.; Thundiyil, J.G.; Stolbach, A. Clearing the Air: A Review of the Effects of Particulate Matter Air Pollution on Human Health. J. Med. Toxicol. 2012, 8, 166–175. [Google Scholar] [CrossRef]
- Bae, M.S.; Schauer, J.J.; Lee, T.; Jeong, J.H.; Kim, Y.K.; Ro, C.U.; Song, S.-K.; Shon, Z.-H. Relationship between reactive oxygen species and water-soluble organic compounds: Time-resolved benzene carboxylic acids measurement in the coastal area during the KORUS-AQ campaign. Environ. Pollut. 2017, 231, 1–2. [Google Scholar]
- Velali, E.; Papachristou, E.; Pantazaki, A.; Choli-Papadopoulou, T.; Planou, S.; Kouras, A.; Manoli, E.; Besis, A.; Voutsa, D.; Samara, C. Redox activity and in vitro bioactivity of the water-soluble fraction of urban particulate matter in relation to particle size and chemical composition. Environ. Pollut. 2016, 208, 774–786. [Google Scholar] [CrossRef]
- WHO. Global Urban Ambient Air Pollution Database (updated 2016b). World Health Organization. 2014 [cited 2003 Aug 20]. Available online: http://www.who.int/entity/phe/health_topics/outdoorair/databases/who-aap-database-may2016.xlsx (accessed on 8 July 2019).
- Tefera, W.; Asfaw, A.; Gilliland, F.; Worku, A.; Wondimagegn, M.; Kumie, A.; Samet, J.; Berhane, K.; Ababa, A.; Change, C. Indoor and Outdoor Air Pollution- related Health Problem in Ethiopia: Review of Related Literature. Ethiop. J. Heal. Dev. 2016, 30, 5–16. [Google Scholar]
- Etyemezian, V.; Tesfaye, M.; Yimer, A.; Chow, J.C.; Mesfin, D.; Nega, T.; Nikolich, G.; Watson, J.G.; Wondmagegn, M. Results from a pilot-scale air quality study in Addis Ababa, Ethiopia. Atmos. Environ. 2005, 39, 7849–7860. [Google Scholar] [CrossRef]
- World Bank Group. Worldwide Total Motor Vehicles (per 1000 People); ChartsBin Statistics Collector Team, 2011. Available online: http://chartsbin.com/view/1114 (accessed on 23 June 2019).
- UN. Addis Ababa, Ethiopia—Population 1950–2020. World Population Prospects 2019; Department of Economic and Social Affairs: New York, NY, USA, 2019; Available online: https://population.un.org/wpp/ (accessed on 10 September 2020).
- NMA. Climate of City: Addis Ababa. National Meteorological Agency of Ethiopia; National Meteorological Agency: Addis Ababa, Ethiopia, 2016. [Google Scholar]
- NMA. Long-Term Weather Forecast Addis Ababa, Ethiopia. Ethiopia National Meteorological Agency (Weather Atlas). 2020. Available online: https://www.weather-atlas.com/en/ethiopia/addis-ababa-long-term-weather-forecast (accessed on 16 August 2020).
- Baldauf, R.; Watkins, N.; Heist, D.; Bailey, C.; Rowley, P.; Shores, R. Near-road air quality monitoring: Factors affecting network design and interpretation of data. Air Qual. Atmos. Health 2009, 2, 1–9. [Google Scholar]
- Yanosky, J.; Maclntosh, D. A Comparison of Four Gravimetric Fine Particle Sampling Methods. J. Air Waste Manag. Assoc. 2001, 51, 878–884. [Google Scholar]
- Wang, Y.; Zhuang, G.; Tang, A.; Yuan, H.; Sun, Y.; Chen, S.; Zheng, A. The ion chemistry and the source of PM2.5 aerosol in Beijing. Atmos. Environ. 2005, 39, 3771–3784. [Google Scholar]
- Schauer, J.J. Evaluation of elemental carbon as a marker for diesel particulate matter. J. Expo. Anal. Environ. Epidemiol. 2003, 13, 443–453. [Google Scholar] [PubMed]
- Birch, M.E.; Cary, C.R.A. Elemental carbon-based method for monitoring occupational exposures to particulate diesel exhaust. Aerosol Sci. Technol. 1996, 25, 221–241. [Google Scholar]
- Bae, M.; Schauer, J.J.; Turner, J.R. Estimation of the Monthly Average Ratios of Organic Mass to Organic Carbon for Fine Particulate Matter at an Urban Site. Aerosol Sci. Technol. 2006, 40, 1123–1139. [Google Scholar]
- Villalobos, A.M.; Barraza, F.; Jorquera, H.; Schauer, J.J. Chemical speciation and source apportionment of fine particulate matter. Sci. Total Environ. 2015, 512–513, 133–142. [Google Scholar]
- Brehmer, C.; Norris, C.; Barkjohn, K.K.; Bergin, M.H.; Zhang, J.; Cui, X.; Zhang, Y.; Black, M.; Li, Z.; Shafer, M.; et al. The impact of household air cleaners on the chemical composition and children’s exposure to PM2.5 metal sources in suburban Shanghai. Environ. Pollut. 2019, 253, 190–198. [Google Scholar]
- Mkoma, S.L.; Chi, X.; Maenhaut, W. Characteristics of carbonaceous aerosols in ambient PM10 and PM2.5 particles in Dar es Salaam, Tanzania. Sci. Total Environ. 2010, 408, 1308–1314. [Google Scholar]
- Arhami, M.; Sillanpää, M.; Hu, S.; Olson, M.R.; Schauer, J.J.; Sioutas, C. Size-segregated inorganic and organic components of PM in the communities of the Los Angeles Harbor. Aerosol Sci. Technol. 2009, 43, 145–160. [Google Scholar]
- Brook, J.; Dann, T.; Burnett, R. The relationship among TSP, PM10, PM2.5, and inorganic constituents of atmospheric participate matter at multiple Canadian locations. J. Air Waste Manag. Assoc. 1997, 47, 2–19. [Google Scholar] [CrossRef]
- Russell, L.M. Aerosol Organic-Mass-to-Organic-Carbon Ratio Measurements. Environ. Sci. Technol. 2003, 37, 2982–2987. [Google Scholar] [CrossRef]
- Sardar, S.B.; Fine, P.M.; Mayo, P.R.; Sioutas, C. Size-Fractionated Measurements of Ambient Ultrafine Particle Chemical Composition in Los Angeles Using the NanoMOUDI. Environ. Sci. Technol. 2005, 39, 932–944. [Google Scholar] [CrossRef]
- Turpin, B.J.; Lim, H. Species Contributions to PM2.5 Mass Concentrations: Revisiting Common Assumptions for Estimating Organic Mass. Aerosol Sci. Technol. 2001, 35, 602–610. [Google Scholar] [CrossRef]
- Arhami, M.; Kamali, N.; Rajabi, M.M. Predicting hourly air pollutant levels using artificial neural networks coupled with uncertainty analysis by Monte Carlo simulations. Environ. Sci. Pollut. Res. 2013, 20, 4777–4789. [Google Scholar] [CrossRef] [PubMed]
- Chow, J.C.; Lowenthal, D.H.; Chen, L.A.; Wang, X.; Watson, J.G. Mass reconstruction methods for PM 2.5: A review. Air Qual. Atmos. Health 2015, 8, 243–263. [Google Scholar] [CrossRef] [PubMed]
- WHO. Air Quality Guidelines—Global Update 2005. Particulate Matter, Ozone, Nitrogen Dioxide and Sulfur Dioxide; WHO: Copenhagen, Denmark, 2006. [Google Scholar]
- EPA. Ethiopia Guidelines Ambient Environment Standards for Ethiopia 2003, The Environmental Protection Agency and The United Nations Industrial Development Organization prepared under the Ecologically Sustainable Industrial Development (ESID) Project, August, Addis Aba; EPA: Addis Ababa, Ethiopia, 2003. [Google Scholar]
- Gebre, G.; Feleke, Z.; Sahle-Demissie, E. Mass concentrations and elemental composition of urban. Bull. Chem. Soc. Ethiop. 2010, 24, 361–373. [Google Scholar]
- Tarekegn, M.M.; Gulilat, T.Y. Trends of Ambient Air Pollution and the Corresponding Respiratory Diseases in Addis Ababa. Res. Rep. Clin. Pharmacol. Toxicol. J. 2018, 2, 1–14. [Google Scholar]
- OICA. Deloitte Africa Automotive Insights Navigating the African Automotive Sector: Ethiopia, Kenya and Nigeria; OICA: Johannesburg, South Africa, 2016. [Google Scholar]
- Brown, S.G.; Lee, T.; Roberts, P.T.; Collett, J.L., Jr. Variations in the OM/OC ratio of urban organic aerosol next to a major roadway Variations in the OM/OC ratio of urban organic aerosol next to a major roadway. J. Air Waste Manag. Assoc. 2013, 63, 1422–1433. [Google Scholar] [CrossRef]
- Snyder, D.C.; Rutter, A.P.; Collins, R.; Worley, C.; Schauer, J.J. Insights into the Origin of Water Soluble Organic Carbon in Atmospheric Fine Particulate Matter Insights into the Origin of Water Soluble Organic Carbon in Atmospheric Fine Particulate Matter. Aerosol. Sci. Technol. 2009, 43, 1099–1107. [Google Scholar] [CrossRef]
- EDHS. Ethiopian Demographic and Health Survey; EDHS: Addis Ababa, Ethiopia, 2016. [Google Scholar]
- Roychowdhury, A.; Chandola, P.; Chattopadhyaya, V. Urban Air Quality Management in Ethiopia: A Guidance Framework; Center for Science and Environment (CSE): Delhi, India, 2016. [Google Scholar]
- Egondi, T.; Muindi, K.; Kyobutungi, C.; Gatari, M.; Rocklöv, J. Measuring exposure levels of inhalable airborne particles (PM2.5) in two socially deprived areas of Nairobi, Kenya. Environ. Res. 2016, 148, 500–506. [Google Scholar] [CrossRef]
- Aboh, I.J.K.; Henriksson, D.; Laursen, J.; Lundin, M.; Ofosu, F.G.; Pind, N.; Al, E. Identification of aerosol particle sources in semi-rural area of Kwabenya, near Accra, Ghana, by EDXRFtechniques. X-Ray Spectrom. 2009, 38, 348–353. [Google Scholar] [CrossRef]
- Boman, J.; Shaltout, A.A.; Abozied, A.M.; Hassan, S.K. On the elemental composition of PM2.5 in central Cairo, Egypt. X-Ray Spectrom. 2013, 42, 276–283. [Google Scholar] [CrossRef]
- Khodeir, M.; Shamy, M.; Alghamdi, M.; Zhong, M.; Sun, H.; Costa, M.; Chen, L.Ͳ.C.; Maciejczyk, P.; Chen, L.T.C. Atmospheric Pollution Research Source apportionment and elemental composition of PM2.5 and PM 10 in Jeddah City, Saudi Arabia. Atmos. Pollut. Res. 2012, 3, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Mandal, T. Chemical composition of fine mode particulate matter (PM2.5) in an urban area of Delhi, India and its source apportionment. Urban Clim. 2017, 21, 106–122. [Google Scholar] [CrossRef]
- Yu, L.; Wang, G.; Zhang, R.; Zhang, L.; Song, Y.; Wu, B.; Li, X.; An, K.; Chu, J. Characterization and Source Apportionment of PM 2.5 in an Urban Environment in Beijing. Aerosol. Air Qual. Res. 2013, 13, 574–583. [Google Scholar] [CrossRef]
- Boman, J.; Lindén, J.; Thorsson, S.; Holmer, B.E. A tentative study of urban and suburban fine particles (PM2.5) collected in Ouagadougou, Burkina Faso. X-Ray Spectrom. 2009, 38, 354–362. [Google Scholar] [CrossRef]
- Arhami, M.; Hosseini, V.; Shahne, M.Z.; Bigdeli, M.; Lai, A.; Schauer, J.J. Seasonal trends, chemical speciation and source apportionment of fine PM in Tehran. Atmos. Environ. 2017, 153, 70–82. [Google Scholar] [CrossRef]
- Sowlat, M.H.; Hasheminassab, S.; Sioutas, C. Source apportionment of ambient particle number concentrations in central Los Angeles using positive matrix factorization (PMF). Atmos. Chem. Phys. 2016, 16, 4849–4866. [Google Scholar] [CrossRef]
- Zhang, W.; Sun, Y.; Zhuang, G.; Xu, D. Characteristics and seasonal variations of PM2.5, PM10, and TSP aerosol in Beijing. Biomed. Environ. Sci. 2006, 19, 461–468. [Google Scholar]
- Putaud, J.-P.; Raes, F.; Van Dingenen, R.; Brüggemann, E.; Facchini, M.-C.; Decesari, S.; Fuzzi, S.; Gehrig, R.; Hüglin, C.; Laj, P.; et al. A European aerosol phenomenology e 2: Chemical characteristics of particulate matter at kerbside, urban, rural and background sites in Europe. Atmos. Environ. 2004, 38, 2579–2595. [Google Scholar]
- Zhang, Q.; Jimenez, J.L.; Canagaratna, M.R.; Allan, J.D.; Coe, H.; Ulbrich, I.; Alfarra, M.R.; Takami, A.; Middlebrook, A.M.; Sun, Y.L.; et al. Ubiquity and dominance of oxygenated species in organic aerosols in anthropogenically-influenced Northern Hemisphere midlatitudes. Geophys. Res. Lett. 2007, 34, 1–6. [Google Scholar]
- Snider, G.; Weagle, C.L.; Murdymootoo, K.K.; Ring, A.; Ritchie, Y.; Stone, E.; Walsh, A.; Akoshile, C.; Anh, N.X.; Balasubramanian, R.; et al. Variation in global chemical composition of PM2.5: Emerging results from SPARTAN. Atmos. Chem. Phys. 2016, 16, 9629–9653. [Google Scholar] [CrossRef]
- Stone, E.A.; Kundu, S. Composition and sources of fine particulate matter across urban and rural sites in the Midwestern United States. Environ. Sci. Process. Impacts 2015, 16, 1360–1370. [Google Scholar]
- WHO. Exposure to Lead: A Major Public Health Concern. World Health Organization: Geneva, Switzerland. Available online: https://www.who.int/ipcs/features/lead..pdf?ua=1 (accessed on 10 September 2020).
- Teju, E.; Megersa, N.; Chandravanshi, B.S.; Zewge, F. Determination of the levels of lead in the roadside soils of Addis Ababa, Ethiopia. Ethiop. J. Sci. 2012, 35, 81–94. [Google Scholar]
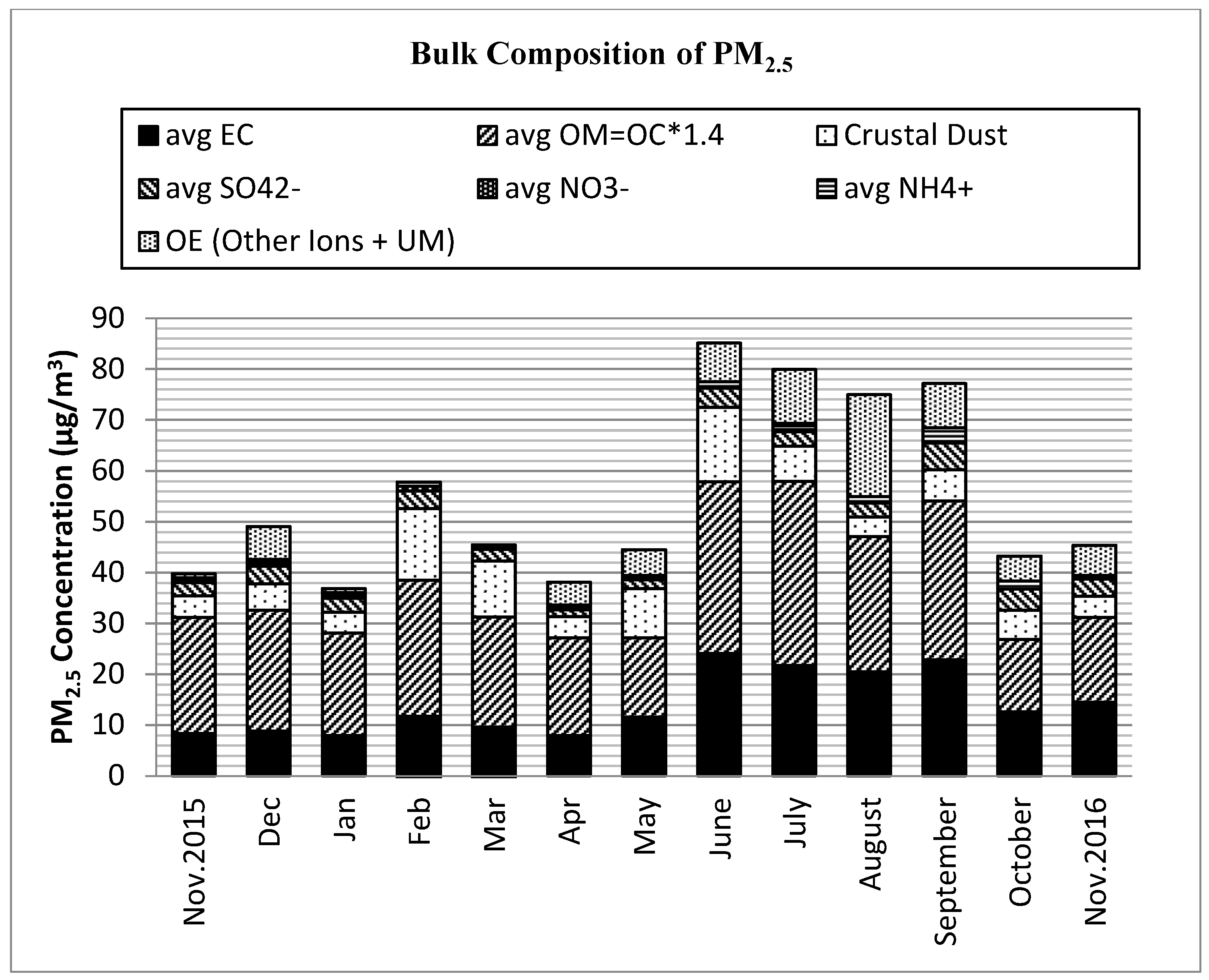
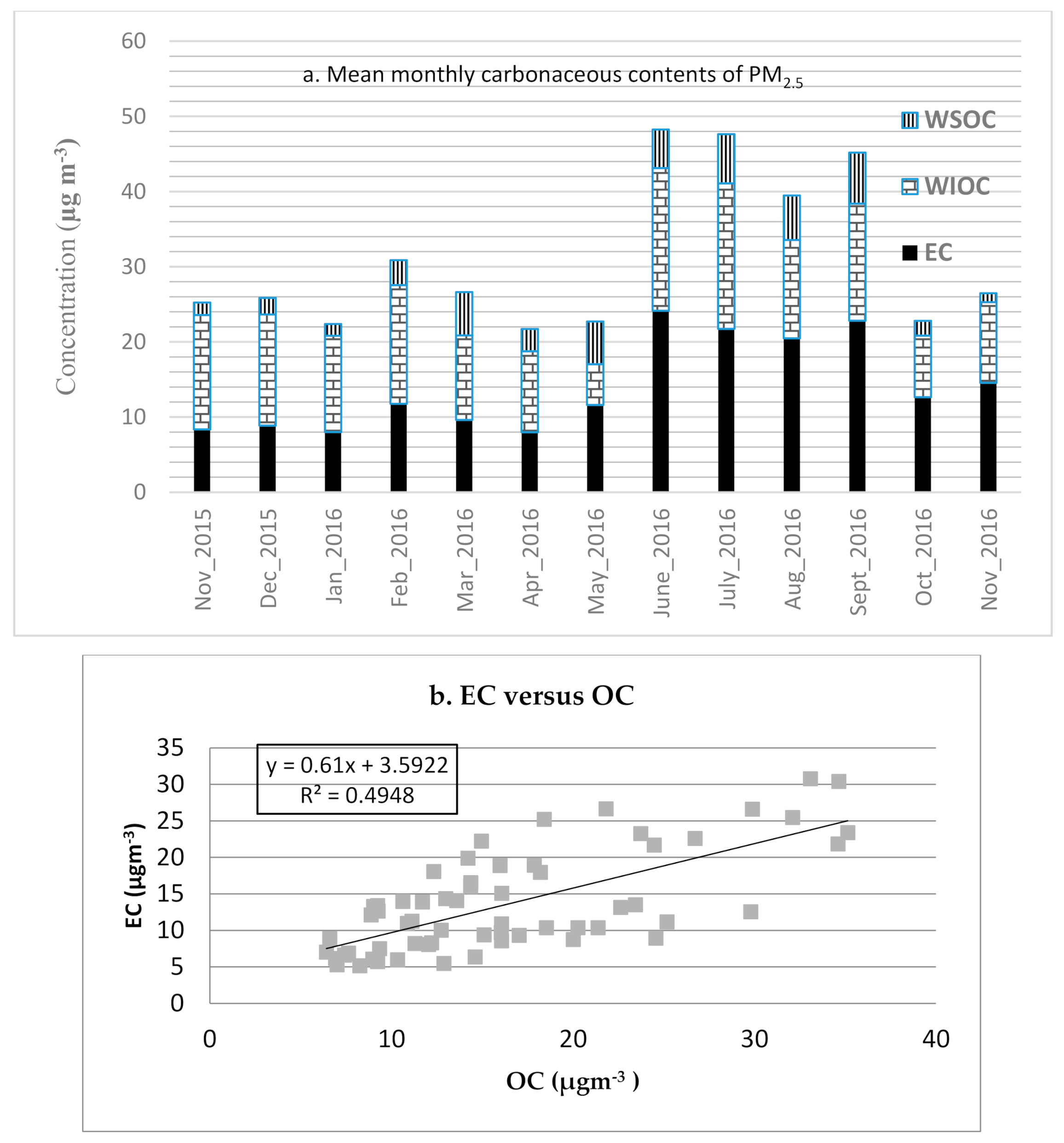
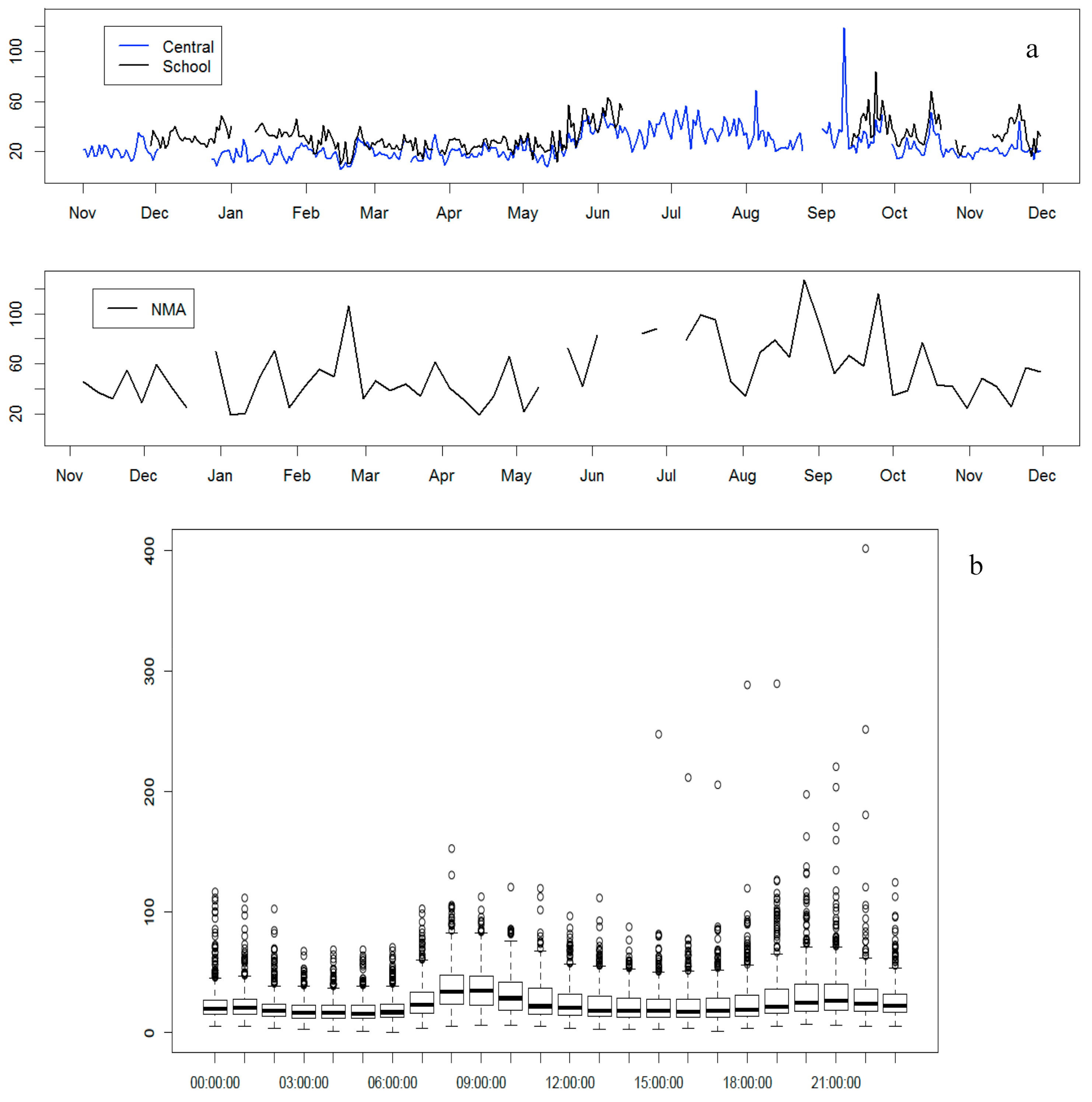
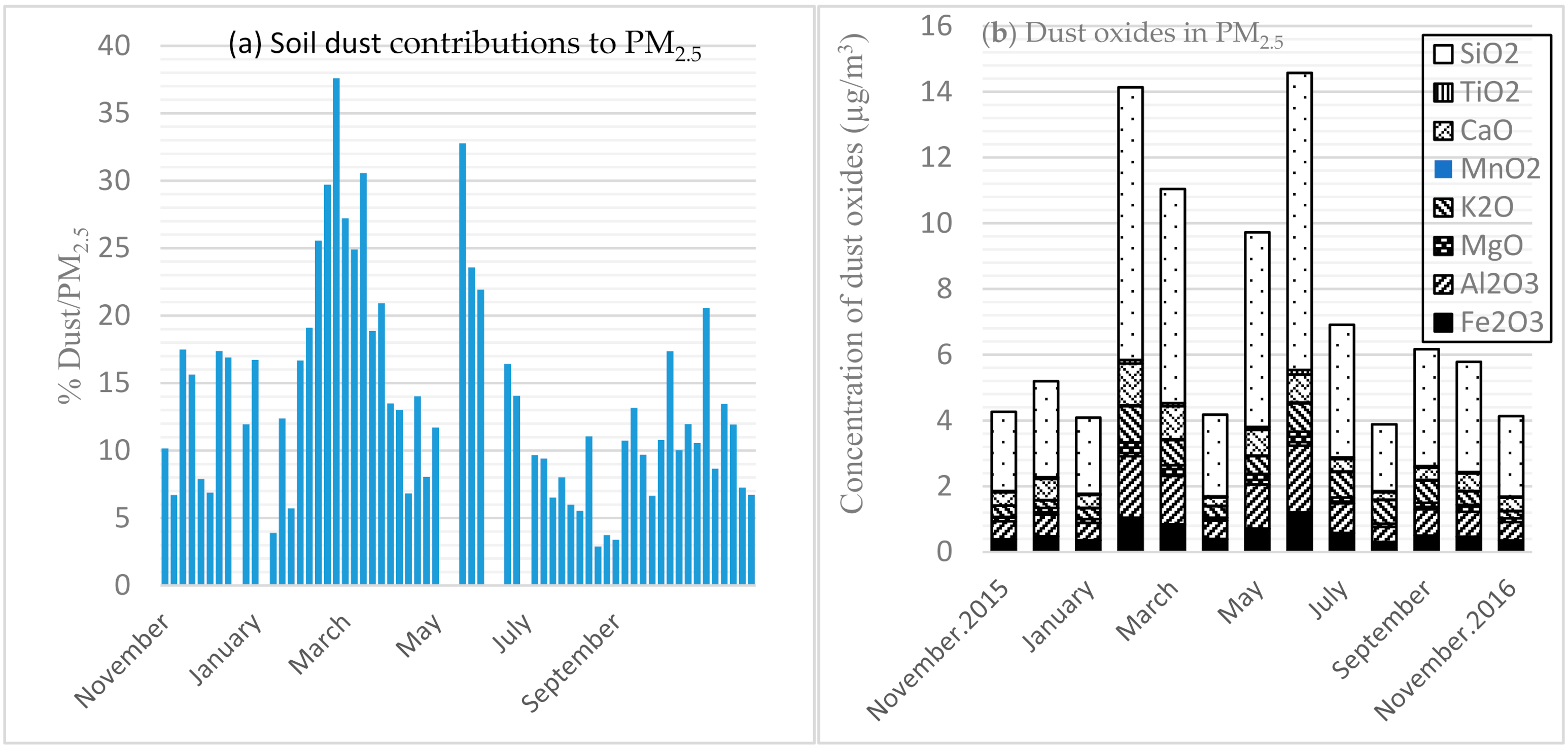
| Month (n) | Mass | EC a | OM b | SO42− | NO3− | NH4+ | OI c | Dust | UM d |
|---|---|---|---|---|---|---|---|---|---|
| Nov_15 (5) | 39.82 | 8.36 | 22.83 | 2.59 | 0.32 | 0.64 | 0.38 | 4.26 | 0.44 |
| Dec (4) | 49.1 | 8.88 | 23.75 | 3.49 | 0.54 | 0.79 | 0.93 | 5.19 | 5.53 |
| Jan (5) | 36.89 | 8 | 20.12 | 2.8 | 0.6 | 0.46 | 0.26 | 4.08 | 0.57 |
| Feb (5) | 57.26 | 11.77 | 26.75 | 3.4 | 0.96 | 0.79 | 0.39 | 14.13 | 0 |
| Mar (5) | 45.1 | 9.63 | 21.66 | 2.25 | 0.19 | 0.7 | 0.31 | 11.04 | 0 |
| April (5) | 38.15 | 7.98 | 19.23 | 1.34 | 0.34 | 0.58 | 0.23 | 4.18 | 4.27 |
| May (4) | 44.53 | 11.61 | 15.55 | 1.71 | 0.39 | 0.46 | 1.22 | 9.72 | 3.87 |
| June (3) | 85.1 | 24.16 | 33.73 | 3.8 | 0.18 | 1.1 | 0.54 | 14.57 | 7.02 |
| July (4) | 79.91 | 21.75 | 36.23 | 2.89 | 0.26 | 1.25 | 0.64 | 6.91 | 9.98 |
| Aug (5) | 75.01 | 20.47 | 26.6 | 2.76 | 0.15 | 1.11 | 0.55 | 3.88 | 19.49 |
| Sep (5) | 77.17 | 22.83 | 31.28 | 5.25 | 0.27 | 2.68 | 0.98 | 6.17 | 7.71 |
| Oct (6) | 43.26 | 12.65 | 14.21 | 4.12 | 0.07 | 1.55 | 0.67 | 5.78 | 4.21 |
| Nov_16 (5) | 45.39 | 14.55 | 16.69 | 3.38 | 0.05 | 0.69 | 1.04 | 4.13 | 4.86 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tefera, W.; Kumie, A.; Berhane, K.; Gilliland, F.; Lai, A.; Sricharoenvech, P.; Samet, J.; Patz, J.; Schauer, J.J. Chemical Characterization and Seasonality of Ambient Particles (PM2.5) in the City Centre of Addis Ababa. Int. J. Environ. Res. Public Health 2020, 17, 6998. https://doi.org/10.3390/ijerph17196998
Tefera W, Kumie A, Berhane K, Gilliland F, Lai A, Sricharoenvech P, Samet J, Patz J, Schauer JJ. Chemical Characterization and Seasonality of Ambient Particles (PM2.5) in the City Centre of Addis Ababa. International Journal of Environmental Research and Public Health. 2020; 17(19):6998. https://doi.org/10.3390/ijerph17196998
Chicago/Turabian StyleTefera, Worku, Abera Kumie, Kiros Berhane, Frank Gilliland, Alexandra Lai, Piyaporn Sricharoenvech, Jonathan Samet, Jonathan Patz, and James J. Schauer. 2020. "Chemical Characterization and Seasonality of Ambient Particles (PM2.5) in the City Centre of Addis Ababa" International Journal of Environmental Research and Public Health 17, no. 19: 6998. https://doi.org/10.3390/ijerph17196998
APA StyleTefera, W., Kumie, A., Berhane, K., Gilliland, F., Lai, A., Sricharoenvech, P., Samet, J., Patz, J., & Schauer, J. J. (2020). Chemical Characterization and Seasonality of Ambient Particles (PM2.5) in the City Centre of Addis Ababa. International Journal of Environmental Research and Public Health, 17(19), 6998. https://doi.org/10.3390/ijerph17196998





