Antithrombotic Preventive Medication Prescription Redemption and Socioeconomic Status in Hungary in 2016: A Cross-Sectional Study
Abstract
1. Introduction
- Deprivation and mortality caused by thrombotic diseases;
- Deprivation and the prescription/redemption of selected oral antiplatelet and anticoagulant drugs, with special focus on the comparison between areas with high mortality, although socioeconomically well developed and areas with relatively favorable mortality although socioeconomically deprived, and
- The level of deprivation and the prescribing pattern of antithrombotic drugs.
2. Materials and Methods
2.1. Data
2.2. Deprivation Index Calculation
2.3. Study at the District Level
2.4. Statistics
3. Results
3.1. The Spatial Distribution of Mortality Caused by Different Thromboembolic Diseases in Connection with Deprivation
3.2. Prescription and Redemption Rates of Antithrombotic Drugs and Their Relationships with Deprivation
3.3. Prescription and Redemption Patterns of Antithrombotic Drugs and Their Relationships with Deprivation Levels
3.4. Identification of Anomalies in Preventive Medication in a Cluster with High Mortality in the Less Deprived Region of the Country
4. Discussion
4.1. Implications
4.2. Strengths and Weaknesses of the Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OECD/European Union. Health at a Glance: Europe 2018: State of Health in the EU Cycle; OECD Publishing: Paris, France, 2018. [Google Scholar] [CrossRef]
- Mackenbach, J.P. Nordic paradox, southern miracle, eastern disaster: Persistence of inequalities in mortality in Europe. Eur. J. Public Health 2017, 27, 14–17. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Health Estimates 2016: Disease Burden by Cause, Age, Sex, by Country and by Region, 2000–2016. Available online: https://www.who.int/healthinfo/global_burden_disease/estimates/en/index1.html (accessed on 4 June 2020).
- Roth, G.A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S.F.; Abyu, G.; Ahmed, M.; Aksut, B.; Alam, T.; Alam, K.; et al. Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017, 70, 1–25. [Google Scholar] [CrossRef]
- Mackenbach, J.P.; Cavelaars, A.E.J.M.; Kunst, A.E.; Groenhof, F.; Andersen, O.; Borgan, J.K.; Costa, G.; Crialesi, R.; Desplanques, G.; Faggiano, F.; et al. Socioeconomic inequalities in cardiovascular disease mortality. An international study. Eur. Heart J. 2000, 21, 1141–1151. [Google Scholar] [CrossRef] [PubMed]
- Karanikolos, M.; Adany, R.; McKee, M. The epidemiological transition in Eastern and Western Europe: A historic natural experiment. Eur. J. Public Health 2017, 27, 4–8. [Google Scholar] [CrossRef]
- Institute for Health Metrics and Evaluation (IHME). GBD Compare. Available online: https://vizhub.healthdata.org/gbd-compare/ (accessed on 4 June 2020).
- Di Girolamo, C.; Nusselder, W.J.; Bopp, M.; Brønnum-Hansen, H.; Costa, G.; Kovács, K.; Leinsalu, M.; Martikainen, P.; Pacelli, B.; Rubio Valverde, J.; et al. Progress in reducing inequalities in cardiovascular disease mortality in Europe. Heart 2020, 106, 40–49. [Google Scholar] [CrossRef]
- Lewer, D.; Jayatunga, W.; Aldridge, R.W.; Edge, C.; Marmot, M.; Story, A.; Hayward, A. Premature mortality attributable to socioeconomic inequality in England between 2003 and 2018: An observational study. Lancet Public Health 2020, 5, e33–e41. [Google Scholar] [CrossRef]
- Kort, D.; van Rein, N.; van der Meer, F.J.M.; Vermaas, H.W.; Wiersma, N.; Cannegieter, S.C.; Lijfering, W.M. Relationship between neighborhood socioeconomic status and venous thromboembolism: Results from a population-based study. J. Thromb. Haemost. 2017, 15, 2352–2360. [Google Scholar] [CrossRef]
- Zöller, B.; Li, X.; Sundquist, J.; Sundquist, K. Socioeconomic and occupational risk factors for venous thromboembolism in Sweden: A nationwide epidemiological study. Thromb. Res. 2012, 129, 577–582. [Google Scholar] [CrossRef]
- Ettehad, D.; Emdin, C.A.; Kiran, A.; Anderson, S.G.; Callender, T.; Emberson, J.; Chalmers, J.; Rodgers, A.; Rahimi, K. Blood pressure lowering for prevention of cardiovascular disease and death: A systematic review and meta-analysis. Lancet 2016, 387, 957–967. [Google Scholar] [CrossRef]
- Arnett, D.K.; Blumenthal, R.S.; Albert, M.A.; Buroker, A.B.; Goldberger, Z.D.; Hahn, E.J.; Himmelfarb, C.D.; Khera, A.; Lloyd-Jones, D.; McEvoy, J.W.; et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: A report of the American college of cardiology/American heart association task force on clinical practice guidelines. J. Am. Coll. Cardiol. 2019, 74, e177–e232. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, P.; Connock, M.; Gurung, T.; Freeman, K.; Johnson, S.; Ngianga-Bakwin, K.; Grove, A.; Gurung, B.; Morrow, S.; Stranges, S.; et al. Aspirin in primary prevention of cardiovascular disease and cancer: A systematic review of the balance of evidence from reviews of randomized trials. PLoS ONE 2013, 8, e81970. [Google Scholar] [CrossRef]
- Chou, R.; Dana, T.; Blazina, I.; Daeges, M.; Jeanne, T.L. Statins for prevention of cardiovascular disease in adults: Evidence report and systematic review for the US preventive services task force. JAMA 2016, 316, 2008–2024. [Google Scholar] [CrossRef] [PubMed]
- Kapil, N.; Datta, Y.H.; Alakbarova, N.; Bershad, E.; Selim, M.; Liebeskind, D.S.; Bachour, O.; Rao, G.H.R.; Divani, A.A. Antiplatelet and anticoagulant therapies for prevention of ischemic stroke. Clin. Appl. Thromb. Hemost. 2016, 23, 301–318. [Google Scholar] [CrossRef] [PubMed]
- Patrono, C.; Morais, J.; Baigent, C.; Collet, J.P.; Fitzgerald, D.; Halvorsen, S.; Rocca, B.; Siegbahn, A.; Storey, R.F.; Vilahur, G. Antiplatelet agents for the treatment and prevention of coronary atherothrombosis. J. Am. Coll. Cardiol. 2017, 70, 1760–1776. [Google Scholar] [CrossRef] [PubMed]
- Zerah, L.; Bun, R.-S.; Guillo, S.; Collet, J.-P.; Bonnet-Zamponi, D.; Tubach, F. A prescription support-tool for chronic management of oral antithrombotic combinations in adults based on a systematic review of international guidelines. PLoS ONE 2019, 14, e0211695. [Google Scholar] [CrossRef]
- Yusuf, S.; Islam, S.; Chow, C.K.; Rangarajan, S.; Dagenais, G.; Diaz, R.; Gupta, R.; Kelishadi, R.; Iqbal, R.; Avezum, A.; et al. Use of secondary prevention drugs for cardiovascular disease in the community in high-income, middle-income, and low-income countries (the PURE Study): A prospective epidemiological survey. Lancet 2011, 378, 1231–1243. [Google Scholar] [CrossRef]
- Carlsson, A.C.; Wändell, P.; Gasevic, D.; Sundquist, J.; Sundquist, K. Neighborhood deprivation and warfarin, aspirin and statin prescription A cohort study of men and women treated for atrial fibrillation in Swedish primary care. Int. J. Cardiol. 2015, 187, 547–552. [Google Scholar] [CrossRef]
- King, W.; Lacey, A.; White, J.; Farewell, D.; Dunstan, F.; Fone, D. Socioeconomic inequality in medication persistence in primary and secondary prevention of coronary heart disease—A population-wide electronic cohort study. PLoS ONE 2018, 13, e0194081. [Google Scholar] [CrossRef]
- Di Nisio, M.; van Es, N.; Büller, H.R. Deep vein thrombosis and pulmonary embolism. Lancet 2016, 388, 3060–3073. [Google Scholar] [CrossRef]
- Mauskop, A.; Borden, W.B. Predictors of statin adherence. Curr. Cardiol. Rep. 2011, 13, 553–558. [Google Scholar] [CrossRef]
- Boruzs, K.; Juhász, A.; Nagy, C.; Ádány, R.; Bíró, K. Relationship between statin utilization and socioeconomic deprivation in Hungary. Front. Pharmacol. 2016, 7, 66. [Google Scholar] [CrossRef] [PubMed]
- Boruzs, K.; Juhász, A.; Nagy, C.; Szabó, Z.; Jakovljevic, M.; Bíró, K.; Ádány, R. High inequalities associated with socioeconomic deprivation in cardiovascular disease burden and antihypertensive medication in Hungary. Front. Pharmacol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Juhász, A.; Nagy, C.; Páldy, A.; Beale, L. Development of a Deprivation Index and its relation to premature mortality due to diseases of the circulatory system in Hungary, 1998–2004. Soc. Sci. Med. 2010, 70, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Nagy, C.; Juhász, A.; Beale, L.; Páldy, A. Mortality amenable to health care and its relation to socio-economic status in Hungary, 2004–2008. Eur. J. Public Health 2012, 22, 620–624. [Google Scholar] [CrossRef] [PubMed]
- Nagy, C.; Juhász, A.; Papp, Z.; Beale, L. Hierarchical spatio-temporal mapping of premature mortality due to alcoholic liver disease in Hungary, 2005–2010. Eur. J. Public Health 2013, 24, 827–833. [Google Scholar] [CrossRef] [PubMed]
- Jakab, Z.; Juhasz, A.; Nagy, C.; Schuler, D.; Garami, M. Trends and territorial inequalities of incidence and survival of childhood leukaemia and their relations to socioeconomic status in Hungary, 1971–2015. Eur. J. Cancer Prev. 2017. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. European Regional and Urban Statistics Reference Guide; Office for Official Publications of the European Communities: Luxembourg, 2009. [Google Scholar]
- Beale, L.; Hodgson, S.; Abellan, J.J.; Lefevre, S.; Jarup, L. Evaluation of spatial relationships between health and the environment: The rapid inquiry facility. Environ. Health Perspect. 2010, 118, 1306–1312. [Google Scholar] [CrossRef]
- Besag, J.; York, J.; Mollié, A. Bayesian image restoration, with two applications in spatial statistics. Ann. Instit. Stat. Math. 1991, 43, 1–20. [Google Scholar] [CrossRef]
- Richardson, S.; Thomson, A.; Best, N.; Elliott, P. Interpreting posterior relative risk estimates in disease-mapping studies. Environ. Health Perspect. 2004, 112, 1016–1025. [Google Scholar] [CrossRef]
- Varga, J. Out-migration and attrition of physicians and dentists before and after EU accession (2003 and 2011): The case of Hungary. Eur J. Health Econ. 2017, 18, 1079–1093. [Google Scholar] [CrossRef]
- Perchinig, B.; Horváth, V.; Molnár, D.; Tavodová, L. Cross-Border Mobilities in the Austrian-Hungarian and Austrian-Slovak Border Regions. Available online: www.reminder-project.eu (accessed on 16 June 2020).
- BVZ at Burgenland. Ungarisches Spitalspersonal im Burgenland umworben. Available online: https://www.bvz.at/burgenland/chronik-gericht/nach-grenzschliessung-ungarisches-spitalspersonal-im-burgenland-umworben-burgenland-burgenland-epidemie-viruserkrankung-corona-coronavirus-196675677 (accessed on 20 June 2020).
- Ouali, S.; Mechri, M.; Ben Ali, Z.; Boudiche, S.; Ben Halima, M.; Rejaibi, S.; Mourali, M.S.; Larbi, N.; Meghaieth, F. Factors associated to adequate time in therapeutic range with oral vitamin K antagonists in Tunisia. Tunis. Med. 2019, 97, 113–121. [Google Scholar] [PubMed]
- Sjölander, M.; Eriksson, M.; Asplund, K.; Norrving, B.; Glader, E.L. Socioeconomic inequalities in the prescription of oral anticoagulants in stroke patients with atrial fibrillation. Stroke 2015, 46, 2220–2225. [Google Scholar] [CrossRef] [PubMed]
- Raparelli, V.; Proietti, M.; Buttà, C.; Di Giosia, P.; Sirico, D.; Gobbi, P.; Corrao, S.; Davì, G.; Vestri, A.R.; Perticone, F.; et al. Medication prescription and adherence disparities in non valvular atrial fibrillation patients: An Italian portrait from the ARAPACIS study. Intern. Emerg. Med. 2014, 9, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Pandya, E.Y.; Bajorek, B. Factors affecting patients’ perception on, and adherence to, anticoagulant therapy: Anticipating the role of direct oral anticoagulants. Patient 2017, 10, 163–185. [Google Scholar] [CrossRef] [PubMed]
- Dantas, G.C.; Thompson, B.V.; Manson, J.A.; Tracy, C.S.; Upshur, R.E.G. Patients’ perspectives on taking warfarin: Qualitative study in family practice. BMC Fam. Pract. 2004, 5, 1–9. [Google Scholar] [CrossRef]
- Platt, A.B.; Localio, A.R.; Brensinger, C.M.; Cruess, D.G.; Christie, J.D.; Gross, R.; Parker, C.S.; Price, M.; Metlay, J.P.; Cohen, A.; et al. Risk factors for nonadherence to warfarin: Results from the IN-RANGE study. Pharmacoepidemiol. Drug Saf. 2008, 17, 853–860. [Google Scholar] [CrossRef]
- Orensky, I.A.; Holdford, D.A. Predictors of noncompliance with warfarin therapy in an outpatient anticoagulation clinic. Pharmacotherapy 2005, 25, 1801–1808. [Google Scholar] [CrossRef]
- Choi, J.C.; DiBonaventura, M.D.; Kopenhafer, L.; Nelson, W.W. Survey of the use of warfarin and the newer anticoagulant dabigatran in patients with atrial fibrillation. Patient Prefer. Adherence 2014, 8, 167–177. [Google Scholar] [CrossRef]
- Lee, H.; Park, J.H.; Floyd, J.S.; Park, S.; Kim, H.C. Combined effect of income and medication adherence on mortality in newly treated hypertension: Nationwide study of 16 million person-years. J. Am. Heart Assoc. 2019, 8, e013148. [Google Scholar] [CrossRef]
- Ferdinand, K.C.; Senatore, F.F.; Clayton-Jeter, H.; Cryer, D.R.; Lewin, J.C.; Nasser, S.A.; Fiuzat, M.; Califf, R.M. Improving medication adherence in cardiometabolic disease: Practical and regulatory implications. J. Am. Coll. Cardiol. 2017, 69, 437–451. [Google Scholar] [CrossRef]
- Simpson, S.H.; Eurich, D.T.; Majumdar, S.R.; Padwal, R.S.; Tsuyuki, R.T.; Varney, J.; Johnson, J.A. A meta-analysis of the association between adherence to drug therapy and mortality. BMJ 2006, 333, 15. [Google Scholar] [CrossRef] [PubMed]
- Doran, T.; Fullwood, C.; Kontopantelis, E.; Reeves, D. Effect of financial incentives on inequalities in the delivery of primary clinical care in England: Analysis of clinical activity indicators for the quality and outcomes framework. Lancet 2008, 372, 728–736. [Google Scholar] [CrossRef]
- Morgenstern, H. Ecologic studies in epidemiology: Concepts, principles, and methods. Annu. Rev. Public Health 1995, 16, 61–81. [Google Scholar] [CrossRef] [PubMed]
- Radevic, S.; Kocic, S.; Jakovljevic, M. Self-assessed health and socioeconomic inequalities in Serbia: Data from 2013 national health survey. Front. Pharmacol. 2016, 7, 140. [Google Scholar] [CrossRef] [PubMed]
- Sándor, J.; Nagy, A.; Földvári, A.; Szabó, E.; Csenteri, O.; Vincze, F.; Sipos, V.; Kovács, N.; Pálinkás, A.; Papp, M.; et al. Delivery of cardio-metabolic preventive services to Hungarian Roma of different socio-economic strata. Fam. Pract. 2017, 34, 83–89. [Google Scholar] [CrossRef]
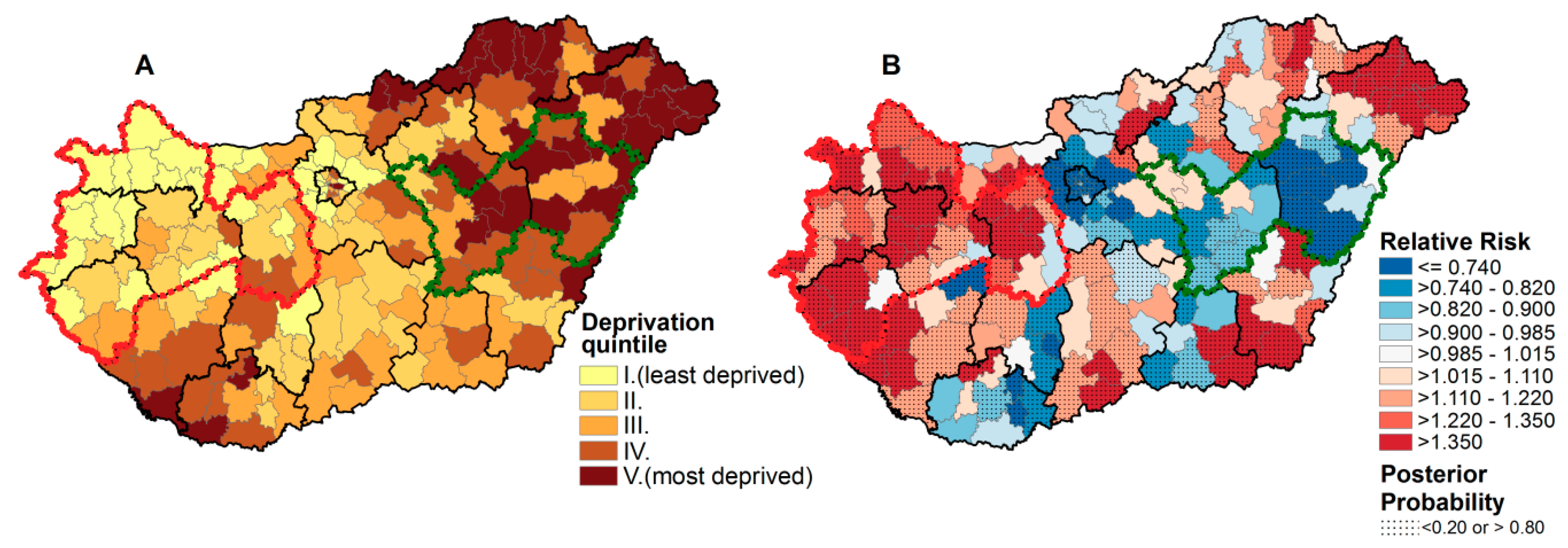
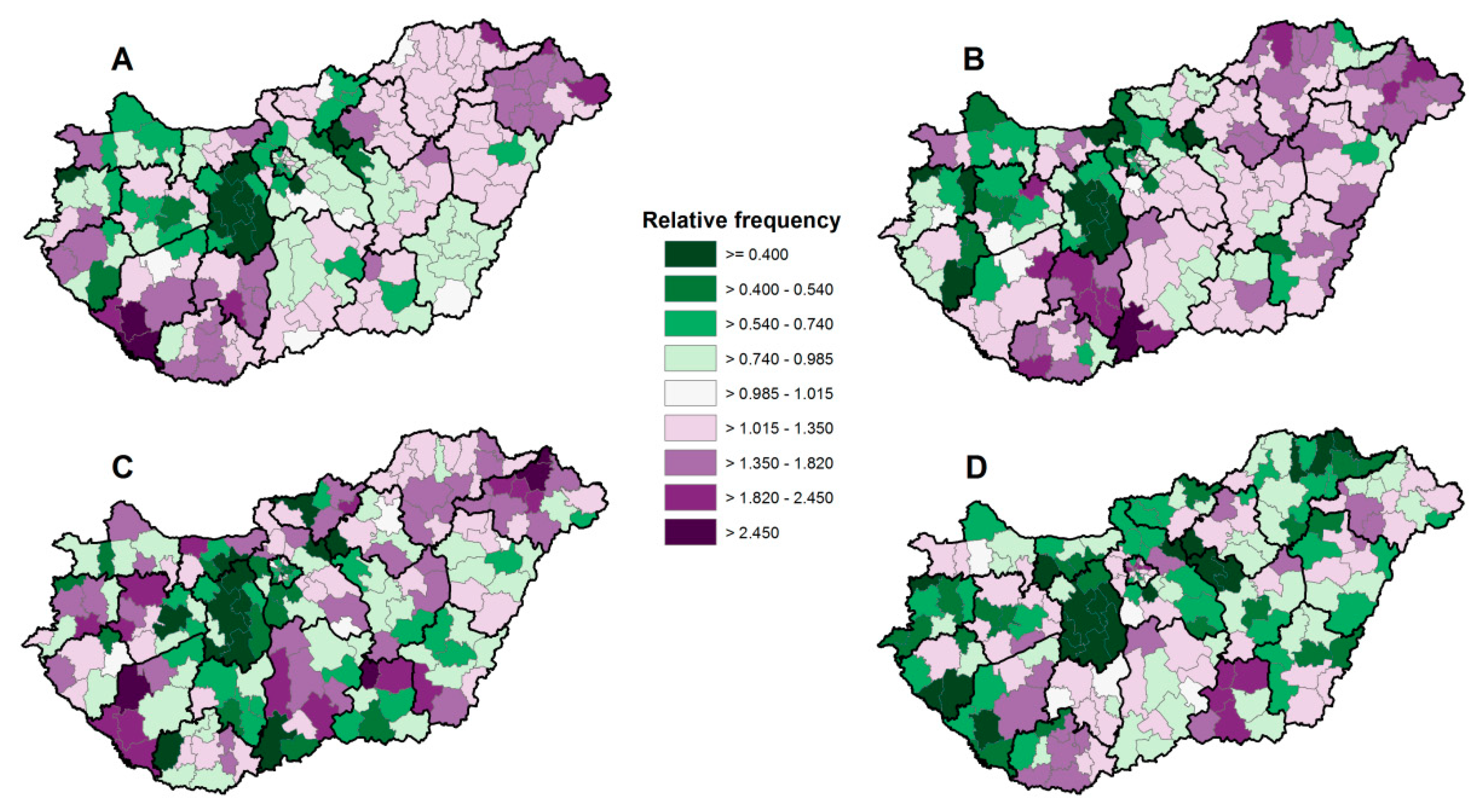
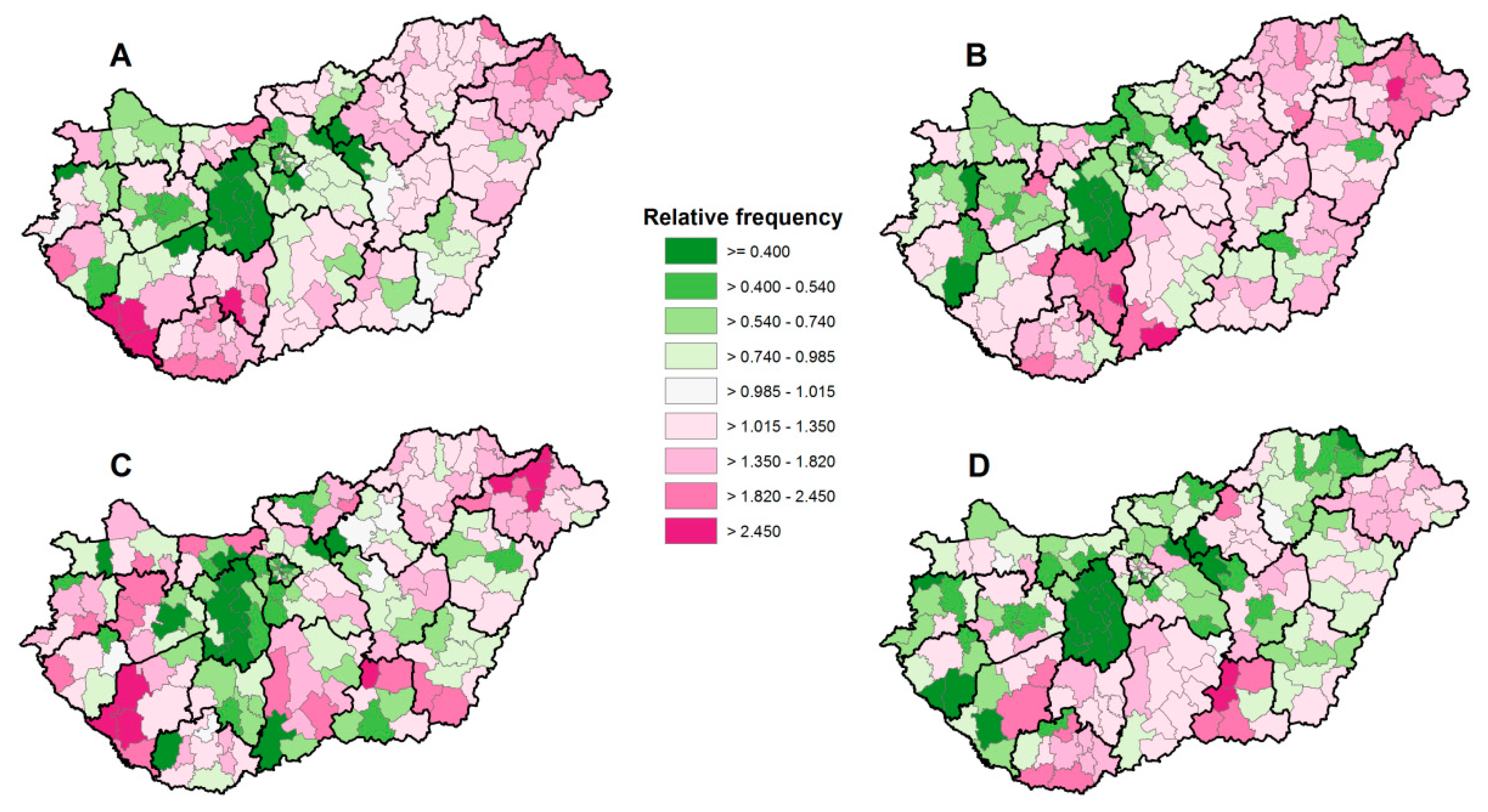
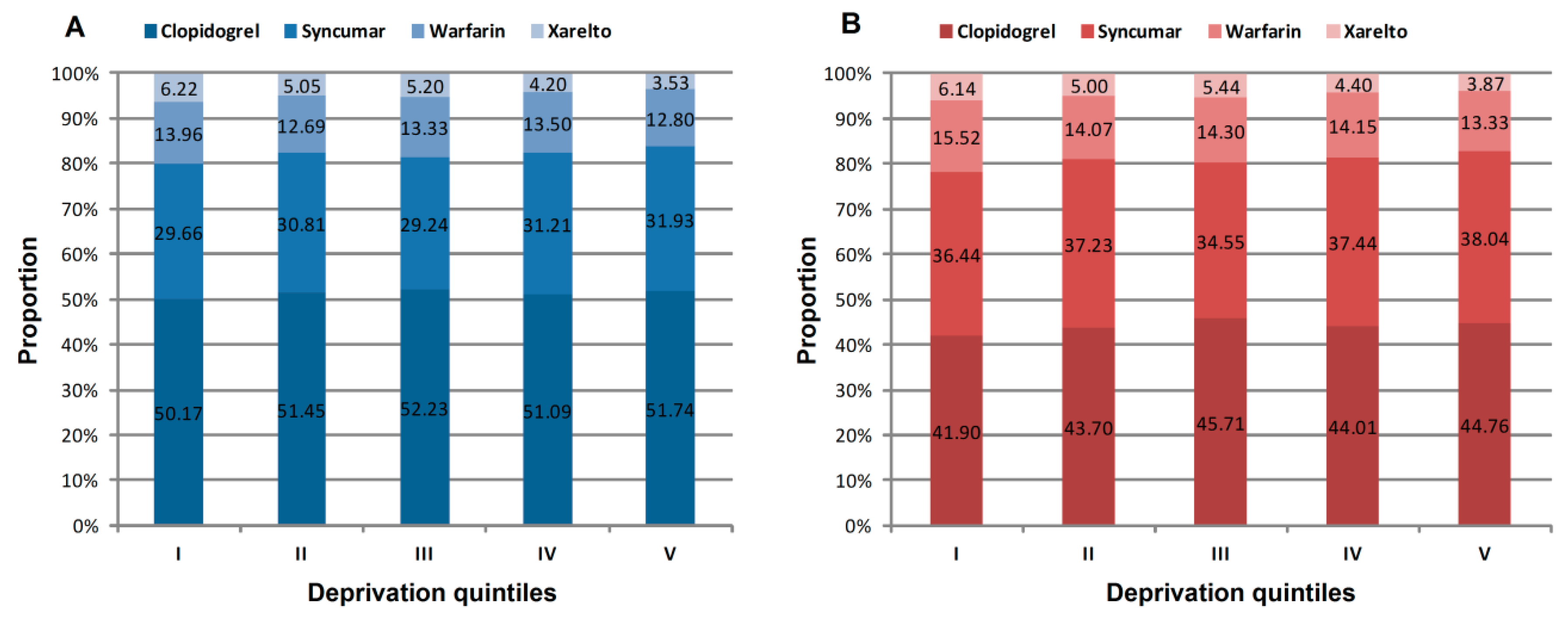
| DI Quintiles | Observed Cases | Incidence Rate Ratio (95%CI) |
|---|---|---|
| I. (least deprived) | 1093 | 1 |
| II. | 1306 | 1.229 (1.025–1.473) |
| III. | 1378 | 1.284 (1.070–1.541) |
| IV. | 1070 | 1.387 (1.154–1.666) |
| V. (most deprived) | 735 | 1.304 (1.081–1.574) |
| p-trend | 0.0067 |
| Drug Types | Total Number of Prescriptions | Total Number of Redemptions | Frequency of Prescription | Frequency of Redemption | Redemption Rate (%) |
|---|---|---|---|---|---|
| (Per 100 Persons Aged 40+ Years) | |||||
| Clopidogrel | 1,729,058 | 941,921 | 32.322 (32.290–32.355) | 17.608 (17.578–17.638) | 54.476 (54.402–54.550) |
| Syncumar | 1,024,233 | 780,918 | 19.147 (19.118–19.176) | 14.598 (14.571–14.625) | 76.244 (76.162–76.326) |
| Warfarin | 447,050 | 305,034 | 8.357 (8.334–8.380) | 5.702 (5.0–5.721) | 68.232 (68.096–68.369) |
| Xarelto | 165,816 | 107,069 | 3.099 (3.085–3.114) | 2.002 (1.990–2.013) | 64.571 (64.34–64.801) |
| Drug Types | DI Quintiles | p-Trend | ||||
|---|---|---|---|---|---|---|
| I. (Least Deprived) | II. | III. | IV. | V. (Most Deprived) | ||
| Prescription | ||||||
| Frequency (Per 100 Persons Aged 40+ Years) (95% CI) | ||||||
| Clopidogrel | 26.373 (26.308–26.438) | 29.378 (29.308–29.448) | 34.007 (33.941–34.072) | 35.900 (35.827–35.986) | 40.654 (40.562–40.746) | <0.0001 |
| Syncumar | 15.51 (15.454–15.565) | 17.546 (17.485–17.606) | 19.061 (19.003–19.120) | 22 (21.928–22.073) | 25.342 (25.254–25.431) | <0.0001 |
| Warfarin | 7.328 (7.285–7.372) | 7.246 (7.201–7.292) | 8.691 (8.644–8.738) | 9.5 (9.443–9.558) | 10.051 (9.980–10.122) | <0.0001 |
| Xarelto | 3.255 (3.225–3.285) | 2.876 (2.847–2.906) | 3.387 (3.357–3.418) | 2.96 (2.926–2.994) | 2.795 (2.756–2.835) | <0.0001 |
| Drug Types | Redemption | p-Trend | ||||
| Frequency (per 100 Persons Aged 40+ Years) (95% CI) | ||||||
| Clopidogrel | 12.774 (12.719–12.828) | 15.750 (15.688–15.812) | 18.897 (18.835–18.959) | 20.118 (20.042–20.193) | 24.166 (24.072–24.261) | <0.0001 |
| Syncumar | 11.054 (11.004–11.104) | 13.378 (13.322–13.434) | 14.294 (14.240–14.349) | 17.169 (17.100–17.238) | 20.791 (20.705–20.878) | <0.0001 |
| Warfarin | 4.724 (4.688–4.760) | 5.066 (5.028–5.105) | 5.918 (5.879–5.958) | 6.477 (6.429–6.527) | 7.219 (7.158–7.281) | <0.0001 |
| Xarelto | 1.866 1.843–1.889] | 1.796 (1.772–1.820) | 2.249 (2.224–2.274) | 2.017 (1.989–2.046) | 2.111 (2.076–2.146) | <0.0001 |
| Drug Types | Redemption Rate (%) (95% CI) | p-Trend | ||||
| Clopidogrel | 48.599 (48.43–48.769) | 53.622 (53.457–53.788) | 55.572 (55.427–55.718] | 55.952 (55.783–56.121) | 59.18 (58.992–59.368) | <0.0001 |
| Syncumar | 71.258 (71.059–71.458) | 76.224 (76.041–76.407) | 75.013 (74.844–75.183] | 78.033 (77.853–78.214) | 82.008 (81.821–82.196) | <0.0001 |
| Warfarin | 64.402 (64.095–64.71) | 69.851 (69.543–70.159) | 68.093 (67.823–68.363] | 68.268 (67.961–68.577) | 72.025 (71.68–72.372) | <0.0001 |
| Xarelto | 57.279 (56.805–57.756) | 62.418 (61.905–62.935) | 66.357 (65.919–66.796] | 68.246 (67.696–68.8) | 75.63 (75.004–76.26) | <0.0001 |
| Less Deprived Counties with High Mortality | Highly Deprived Counties with Low Mortality Rate | Hungarian National Average | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Standardized * Death Rate (Per 100,000 Persons Aged 40+ Years) (95% CI) | 92.097 (87.140–97.282) | 49.728 [44.749–55.121] | 66.493 [64.645–68.394] | |||||||||
| Clopidogrel | Syncumar | Warfarin | Xarelto | Clopidogrel | Syncumar | Warfarin | Xarelto | Clopidogrel | Syncumar | Warfarin | Xarelto | |
| Prescription frequency (per 100 persons aged 40+ years) [95% CI] | 23.769 (23.684–23.832) | 13.102 (13.035–13.161) | 7.537 (7.485–7.582) | 2.146 (2.119–2.171) | 30.867 (30.737–30.966) | 20.234 (20.122–20.335) | 7.577 (7.503–7.639) | 2.664 (2.619–2.703) | 32.322 (32.290–32.355) | 19.147 (19.118–19.176) | 8.357 (8.334–8.380) | 3.099 (3.085–3.114) |
| Redemption frequency (per 100 persons aged 40+ years) [95% CI] | 12.857 (12.790–12.925) | 9.965 (9.905–10.017) | 5.320 (5.275–5.349) | 1.369 (1.346–1.369) | 18.026 (17.918–18.14) | 16.025 (15.922–16.096) | 5.125 (5.063–5.17) | 1.863 (1.825–1.864) | 17.608 (17.578–17.638) | 14.598 (14.571–14.625) | 5.702 (5.683–5.721) | 2.002 (1.990–2.013) |
| Redemption rate [95% CI] | 54.09 (53.887–54.271) | 76.056 (75.819–76.227) | 70.584 (70.252–70.751) | 63.742 (63.085–63.749) | 58.398 (58.148–58.632) | 79.196 (78.941–79.338) | 67.631 (67.152–67.917) | 69.926 (69.131–69.935) | 54.476 (54.402–54.550) | 76.244 (76.162–76.326) | 68.232 (68.096–68.369) | 64.571 (64.34–64.801) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juhász, A.; Nagy, C.; Varga, O.; Boruzs, K.; Csernoch, M.; Szabó, Z.; Ádány, R. Antithrombotic Preventive Medication Prescription Redemption and Socioeconomic Status in Hungary in 2016: A Cross-Sectional Study. Int. J. Environ. Res. Public Health 2020, 17, 6855. https://doi.org/10.3390/ijerph17186855
Juhász A, Nagy C, Varga O, Boruzs K, Csernoch M, Szabó Z, Ádány R. Antithrombotic Preventive Medication Prescription Redemption and Socioeconomic Status in Hungary in 2016: A Cross-Sectional Study. International Journal of Environmental Research and Public Health. 2020; 17(18):6855. https://doi.org/10.3390/ijerph17186855
Chicago/Turabian StyleJuhász, Attila, Csilla Nagy, Orsolya Varga, Klára Boruzs, Mária Csernoch, Zoltán Szabó, and Róza Ádány. 2020. "Antithrombotic Preventive Medication Prescription Redemption and Socioeconomic Status in Hungary in 2016: A Cross-Sectional Study" International Journal of Environmental Research and Public Health 17, no. 18: 6855. https://doi.org/10.3390/ijerph17186855
APA StyleJuhász, A., Nagy, C., Varga, O., Boruzs, K., Csernoch, M., Szabó, Z., & Ádány, R. (2020). Antithrombotic Preventive Medication Prescription Redemption and Socioeconomic Status in Hungary in 2016: A Cross-Sectional Study. International Journal of Environmental Research and Public Health, 17(18), 6855. https://doi.org/10.3390/ijerph17186855







