The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0
Abstract
1. Introduction
2. Seaweeds Biodiversity and Potential to Exploitation
2.1. Green Seaweeds
2.2. Brown Seaweeds
2.3. Red Seaweeds
3. Seaweed Aquaculture: Global Overview
3.1. Environmental Requirements for Seaweed Aquaculture
3.2. Different Seaweed-Aquaculture Techniques
3.2.1. Onshore Cultivation
3.2.2. Offshore Cultivation
3.2.3. Nearshore Cultivation
3.2.4. IMTA Cultivation
3.2.5. Saline Aquaculture
3.3. Seaweeds Aquaculture in Major Cultivated Species
3.3.1. Neopyropia/Pyropia
3.3.2. Gelidium spp. and Pterocladia spp.
3.3.3. Gracilaria/Gracilariopsis
3.3.4. Kappaphycus spp. and Eucheuma spp.
3.3.5. Undaria spp. and Saccharina spp.
3.3.6. Sargassum
4. Seaweed Aquaculture: The Aquaculture 4.0
4.1. Seaweed Productivity and Quality: The Influence of the Abiotic Factors
4.2. New Multidisciplinary Analysis for Optimization of Seaweed Aquaculture
4.2.1. Computational Fluid Dynamics (CFD)
4.2.2. Mechanical and Chemical Engineering
4.2.3. Informatics and Electrotechnics Engineering
4.2.4. Biological Sciences and Engineering
4.3. Aquaculture 4.0: A New Era of Seaweed Cultivation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ferdouse, F.; Holdt, S.L.; Smith, R.; Murúa, P.; Yang, Z. The global status of seaweed production, trade and utilization. FAO Globefish Res. Programme 2018, 124, 120. [Google Scholar]
- Leandro, A.; Pereira, L.; Gonçalves, A.M.M. Diverse Applications of Marine Macroalgae. Mar. Drugs 2019, 18, 17. [Google Scholar] [CrossRef] [PubMed]
- Pereira, L. Edible Seaweeds of the World; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Sánchez-Machado, D.I.; López-Cervantes, J.; López-Hernández, J.; Paseiro-Losada, P. Fatty acids, total lipid, protein and ash contents of processed edible seaweeds. Food Chem. 2004, 85, 439–444. [Google Scholar] [CrossRef]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Macartain, P.; Gill, C.I.R.; Brooks, M.; Campbell, R.; Rowland, I.R. Special Article Nutritional Value of Edible Seaweeds. Nutr. Rev. 2007, 65, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Fleurence, J. Seaweed proteins. In Proteins in Food Processing; Woodhead Publishing Limited: Cambridge, UK, 2004; pp. 197–213. [Google Scholar]
- Mæhre, H.K.; Malde, M.K.; Eilertsen, K.; Elvevoll, E.O. Characterization of protein, lipid and mineral contents in common Norwegian seaweeds and evaluation of their potential as food and feed. J. Sci. Food Agric. 2014, 94, 3281–3290. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Jónsdóttir, R.; Liu, H.; Gu, L.; Kristinsson, H.G.; Raghavan, S.; Ólafsdóttir, G. Antioxidant capacities of phlorotannins extracted from the brown algae fucus vesiculosus. J. Agric. Food Chem. 2012, 60, 5874–5883. [Google Scholar] [CrossRef]
- Fung, A.; Hamid, N.; Lu, J. Fucoxanthin content and antioxidant properties of Undaria pinnatifida. Food Chem. 2013, 136, 1055–1062. [Google Scholar] [CrossRef]
- Mabeau, S.; Fleurence, J. Seaweed in food products: Biochemical and nutritional aspects. Trends Food Sci. Technol. 1993, 4, 103–107. [Google Scholar] [CrossRef]
- Heo, S.J.; Park, E.J.; Lee, K.W.; Jeon, Y.J. Antioxidant activities of enzymatic extracts from brown seaweeds. Bioresour. Technol. 2005, 96, 1613–1623. [Google Scholar] [CrossRef]
- Stengel, D.B.; Connan, S.; Popper, Z.A. Algal chemodiversity and bioactivity: Sources of natural variability and implications for commercial application. Biotechnol. Adv. 2011, 29, 483–501. [Google Scholar] [CrossRef] [PubMed]
- Barbier, M.; Charrier, B.; Araujo, R.; Holdt, S.L.; Jacquemin, B.; Rebours, C. PEGASUS-PHYCOMORPH European Guidelines for a Sustainable Aquaculture of Seaweeds; COST Action FA1406: Roscoff, France, 2019. [Google Scholar]
- Mišurcová, L.; Ambrožová, J.; Samek, D. Seaweed lipids as nutraceuticals. Adv. Food Nutr. Res. 2011, 64, 339–355. [Google Scholar] [PubMed]
- Pereira, H.; Barreira, L.; Figueiredo, F.; Custódio, L.; Vizetto-Duarte, C.; Polo, C.; Rešek, E.; Aschwin, E.; Varela, J. Polyunsaturated fatty acids of marine macroalgae: Potential for nutritional and pharmaceutical applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef] [PubMed]
- Francisco, M.E.Y.; Erickson, K.L. Ma’iliohydrin, a cytotoxic chamigrene dibromohydrin from a Philippine Laurencia species. J. Nat. Prod. 2001, 64, 790–791. [Google Scholar] [CrossRef]
- Smit, A.J. Medicinal and pharmaceutical uses of seaweed natural products: A review. J. Appl. Phycol. 2004, 16, 245–262. [Google Scholar] [CrossRef]
- Dhargalkar, V.K.; Verlecar, X.N. Southern Ocean seaweeds: A resource for exploration in food and drugs. Aquaculture 2009, 287, 229–242. [Google Scholar] [CrossRef]
- Mayer, A.M.S.; Rodríguez, A.D.; Taglialatela-Scafati, O.; Fusetani, N. Marine Pharmacology in 2009–2011: Marine compounds with antibacterial, antidiabetic, antifungal, anti-inflammatory, antiprotozoal, antituberculosis, and antiviral activities; affecting the immune and nervous systems, and other miscellaneous mechanisms. Mar. Drugs 2013, 11, 2510–2573. [Google Scholar] [CrossRef]
- Yuan, Y.; Athukorala, Y. Red Algal Mycosporine-Like Amino Acids (MAAs) as Potential Cosmeceuticals. In Marine Cosmeceuticals; CRC Press: Boca Raton, FL, USA, 2011; pp. 143–168. [Google Scholar]
- Pereira, L. Therapeutic and Nutritional Uses of Algae; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Ruan, B. A Review of the components of seaweeds as potential candidates in cancer therapy. Anticancer. Agents Med. Chem. 2018, 18, 354–366. [Google Scholar] [CrossRef]
- Pangestuti, R.; Kim, S.K. Neuroprotective effects of marine algae. Mar. Drugs 2011, 9, 803–818. [Google Scholar] [CrossRef]
- Barbosa, M.; Valentão, P.; Andrade, P.B. Bioactive compounds from macroalgae in the new millennium: Implications for neurodegenerative diseases. Mar. Drugs 2014, 12, 4934–4972. [Google Scholar] [CrossRef]
- Besednova, N.N.; Zaporozhets, T.S.; Somova, L.M.; Kuznetsova, T.A. Review: Prospects for the use of extracts and polysaccharides from marine algae to prevent and treat the diseases caused by Helicobacter pylori. Helicobacter 2015, 20, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, F.; Alves, R.; Rodrigues, F.; Oliveira, M.B.P.P. Macroalgae-Derived Ingredients for Cosmetic Industry—An Update. Cosmetics 2017, 5, 2. [Google Scholar] [CrossRef]
- Guillerme, J.; Couteau, C.; Coiffard, L. Applications for marine resources in cosmetics. Cosmetics 2017, 4, 35. [Google Scholar] [CrossRef]
- Duarte, C.M.; Wu, J.; Xiao, X.; Bruhn, A.; Krause-Jensen, D. Can seaweed farming play a role in climate change mitigation and adaptation? Front. Mar. Sci. 2017, 4, 1–8. [Google Scholar] [CrossRef]
- Daroch, M.; Geng, S.; Wang, G. Recent advances in liquid biofuel production from algal feedstocks. Appl. Energy 2013, 102, 1371–1381. [Google Scholar] [CrossRef]
- Enquist-Newman, M.; Faust, A.M.E.; Bravo, D.D.; Santos, C.N.S.; Raisner, R.M.; Hanel, A.; Sarvabhowman, P.; Le, C.; Regitsky, D.D.; Cooper, S.R.; et al. Efficient ethanol production from brown macroalgae sugars by a synthetic yeast platform. Nature 2014, 505, 239–243. [Google Scholar] [CrossRef]
- Kraan, S. Mass-cultivation of carbohydrate rich macroalgae, a possible solution for sustainable biofuel production. Mitig. Adapt. Strateg. Glob. Chang. 2013, 18, 27–46. [Google Scholar] [CrossRef]
- Potts, T.; Du, J.; Paul, M.; May, P.; Beitle, R.; Hestekin, J. The production of butanol from Jamaica Bay Macro Algae. Environ. Prog. Sustain. Energy 2012, 31, 29–36. [Google Scholar] [CrossRef]
- Wei, N.; Quarterman, J.; Jin, Y.S. Marine macroalgae: An untapped resource for producing fuels and chemicals. Trends Biotechnol. 2013, 31, 70–77. [Google Scholar] [CrossRef]
- Talep, M. Desintegra.Me. Available online: https://margaritatalep.com/ (accessed on 9 March 2020).
- Reisewitz, S.E.; Estes, J.A.; Simenstad, C.A. Indirect food web interactions: Sea otters and kelp forest fishes in the Aleutian Archipelago. Oecologia 2006, 146, 623–631. [Google Scholar] [CrossRef]
- Leclerc, J.-C.; Riera, P.; Leroux, C.; Lévêque, L.; Laurans, M.; Schaal, G.; Davoult, D. Trophic significance of kelps in kelp communities in Brittany (France) inferred from isotopic comparisons. Mar. Biol. 2013, 160, 3249–3258. [Google Scholar] [CrossRef]
- Smale, D.A.; Burrows, M.T.; Moore, P.; O’Connor, N.; Hawkins, S.J. Threats and knowledge gaps for ecosystem services provided by kelp forests: A northeast Atlantic perspective. Ecol. Evol. 2013, 3, 4016–4038. [Google Scholar] [CrossRef] [PubMed]
- Bertocci, I.; Araújo, R.; Oliveira, P.; Sousa-Pinto, I. Potential effects of kelp species on local fisheries. J. Appl. Ecol. 2015, 52, 1216–1226. [Google Scholar] [CrossRef]
- Almanza, V.; Buschmann, A.H. The ecological importance of Macrocystis pyrifera (Phaeophyta) forests towards a sustainable management and exploitation of chilean coastal benthic co-management areas. Int. J. Environ. Sustain. Dev. 2013, 12, 341–360. [Google Scholar] [CrossRef]
- Vásquez, J.A.; Zuñiga, S.; Tala, F.; Piaget, N.; Rodríguez, D.C.; Vega, J.M.A. Economic valuation of kelp forests in northern Chile: Values of goods and services of the ecosystem. J. Appl. Phycol. 2013, 26, 1081–1088. [Google Scholar] [CrossRef]
- Skjermo, J.; Aasen, I.M.; Arff, J.; Broch, O.J.; Carvajal, A.; Forbord, S.; Olsen, Y.; Reitan, K.I.; Rustad, T.; Sandquist, J.; et al. A new Norwegian Bioeconomy Based on Cultivation and Processing of Seaweeds: Opportunities and R & D Needs; SINTEF Fisheries and Aquaculture: Trondheim, Norway, 2014. [Google Scholar]
- Christianen, M.J.A.; Van Belzen, J.; Herman, P.M.J.; Van Katwijk, M.M.; Lamers, L.P.M.; Van Leent, P.J.M.; Bouma, T.J. Low-canopy seagrass beds still provide important coastal protection services. PLoS ONE 2013, 8, e62413. [Google Scholar] [CrossRef]
- Ondiviela, B.; Losada, I.J.; Lara, J.L.; Maza, M.; Galván, C.; Bouma, T.J.; van Belzen, J. The role of seagrasses in coastal protection in a changing climate. Coast. Eng. 2014, 87, 158–168. [Google Scholar] [CrossRef]
- Ashkenazi, D.Y.; Israel, A.; Abelson, A. A novel two-stage seaweed integrated multi-trophic aquaculture. Rev. Aquac. 2019, 11, 246–262. [Google Scholar] [CrossRef]
- Charrier, B.; Abreu, M.H.; Araujo, R.; Bruhn, A.; Coates, J.C.; De Clerck, O.; Katsaros, C.; Robaina, R.R.; Wichard, T. Furthering knowledge of seaweed growth and development to facilitate sustainable aquaculture. New Phytol. 2017, 216, 967–975. [Google Scholar] [CrossRef]
- Fernand, F.; Israel, A.; Skjermo, J.; Wichard, T.; Timmermans, K.R.; Golberg, A. Offshore macroalgae biomass for bioenergy production: Environmental aspects, technological achievements and challenges. Renew. Sustain. Energy Rev. 2017, 75, 35–45. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture-Contributing to Food Security and Nutrition for All; FAO: Rome, Italy, 2016. [Google Scholar]
- FAO. The State of the World Fisheries and Aquaculture-Meeting the Sustainable Development Goals; FAO: Rome, Italy, 2018; Volume 3. [Google Scholar]
- Cottier-Cook, E.J.; Nagabhatla, N.; Badis, Y.; Campbell, M.; Chopin, T.; Dai, W.; Fang, J.; He, P.; Hewitt, C.; Kim, G.H.; et al. Safeguarding the future of the global seaweed aquaculture industry. In United Nations University and Scottish Association for Marine Science Policy Brief; United Nations University Institue of Water, Environment and Health: Hamilton, ON, Canada, 2016; p. 12. [Google Scholar]
- Rindi, F.; Soler-Vila, A.; Guiry, M.D. Taxonomy of Marine Macroalgae Used as Sources of Bioactive Compounds; Springer: Berlin/Heidelberg, Germany, 2011; Volume 9781461412. [Google Scholar]
- Shannon, E.; Abu-Ghannam, N. Seaweeds as nutraceuticals for health and nutrition. Phycologia 2019, 58, 563–577. [Google Scholar] [CrossRef]
- Rhein-Knudsen, N.; Ale, M.T.; Meyer, A.S. Seaweed hydrocolloid production: An update on enzyme assisted extraction and modification technologies. Mar. Drugs 2015, 13, 3340–3359. [Google Scholar] [CrossRef] [PubMed]
- Zollmann, M.; Robin, A.; Prabhu, M.; Polikovsky, M.; Gillis, A.; Greiserman, S.; Golberg, A. Green technology in green macroalgal biorefineries. Phycologia 2019, 58, 516–534. [Google Scholar] [CrossRef]
- Davis, G.D.J.; Vasanthi, A.H.R. Seaweed metabolite database (SWMD): A database of natural compounds from marine algae. Bioinformation 2011, 5, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Mazarrasa, I.; Olsen, Y.S.; Mayol, E.; Marbà, N.; Duarte, C.M. Global unbalance in seaweed production, research effort and biotechnology markets. Biotechnol. Adv. 2014, 32, 1028–1036. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Camus, C.; Infante, J.; Neori, A.; Israel, Á.; Hernández-González, M.C.; Pereda, S.V.; Gomez-Pinchetti, J.L.; Golberg, A.; Tadmor-Shalev, N.; et al. Seaweed production: Overview of the global state of exploitation, farming and emerging research activity. Eur. J. Phycol. 2017, 52, 391–406. [Google Scholar] [CrossRef]
- Zerrifi, S.E.A.; Khalloufi, F.E.; Oudra, B.; Vasconcelos, V. Seaweed bioactive compounds against pathogens and microalgae: Potential uses on pharmacology and harmful algae bloom control. Mar. Drugs 2018, 16, 55. [Google Scholar] [CrossRef]
- D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the polyphenols: Status and controversies. Int. J. Mol. Sci. 2010, 11, 1321–1342. [Google Scholar] [CrossRef]
- Zubia, M.; Freile-Pelegrín, Y.; Robledo, D. Photosynthesis, pigment composition and antioxidant defences in the red alga Gracilariopsis tenuifrons (Gracilariales, Rhodophyta) under environmental stress. J. Appl. Phycol. 2014, 26, 2001–2010. [Google Scholar] [CrossRef]
- Cotas, J.; Leandro, A.; Pacheco, D.; Gonçalves, A.M.M.; Pereira, L. A comprehensive review of the nutraceutical and therapeutic applications of red seaweeds (Rhodophyta). Life 2020, 10, 19. [Google Scholar] [CrossRef]
- Mekinić, I.G.; Skroza, D.; Šimat, V.; Hamed, I.; Čagalj, M.; Perković, Z.P. Phenolic content of brown algae (Pheophyceae) species: Extraction, identification, and quantification. Biomolecules 2019, 9, 244. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.; Kim, S.K. Nutritional and Digestive Health Benefits of Seaweed, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2011; Volume 64. [Google Scholar]
- Cherry, P.; O’Hara, C.; Magee, P.J.; McSorley, E.M.; Allsopp, P.J. Risks and benefits of consuming edible seaweeds. Nutr. Rev. 2019, 77, 307–329. [Google Scholar] [CrossRef]
- Pereira, L. A review of the nutrient composition of selected edible seaweed. In Nutrition and Food Science; Nova Science Publishers: Hauppauge, NY, USA, 2011; pp. 15–47. [Google Scholar]
- Pereira, L.; Gheda, S.F.; Ribeiro-claro, P.J.A. Analysis by vibrational spectroscopy of seaweed polysaccharides with potential use in food, pharmaceutical, and cosmetic industries. Int. J. Carbohydr. Chem. 2013, 2013, 7. [Google Scholar] [CrossRef]
- Arunkumar, K. Extraction, Isolation, and Characterization of Alginate. In Industrial Applications of Marine Biopolymers; Taylor & Francis Group: Abingdon, UK; CRC Press: Boca Raton, FL, USA, 2017; pp. 19–35. [Google Scholar]
- Pereira, L. Biological and therapeutic properties of the seaweed polysaccharides. Int. Biol. Rev. 2018, 2, 1–50. [Google Scholar] [CrossRef]
- Holdt, S.L.; Kraan, S. Bioactive compounds in seaweed: Functional food applications and legislation. J. Appl. Phycol. 2011, 23, 543–597. [Google Scholar] [CrossRef]
- Valado, A.; Pereira, M.; Caseiro, A.; Figueiredo, J.P.; Loureiro, H.; Almeida, C.; Cotas, J.; Pereira, L. Effect of carrageenans on vegetable jelly in humans with hypercholesterolemia. Mar. Drugs 2020, 18, 19. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An increase in the Omega-6/Omega-3 fatty acid ratio increases the risk for obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef]
- Commitee on Diet and Health and National Research Council. Diet. and Health: Implications for Reducing Chronic Disease Risk; Motulsky, A.G., Ed.; National Academies Press: Washington, DC, USA, 1989. [Google Scholar]
- Bouga, M.; Combet, E. Emergence of seaweed and seaweed-containing foods in the UK: Focus on labeling, iodine content, toxicity and nutrition. Foods 2015, 4, 240–253. [Google Scholar] [CrossRef]
- Wang, H.M.D.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresour. Technol. 2015, 184, 355–362. [Google Scholar] [CrossRef]
- Barsanti, L.; Gualtieri, P. Algae: Anatomy, Biochemistry, and Biotechnology; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Application of novel extraction technologies for bioactives from marine algae. J. Agric. Food Chem. 2013, 61, 4667–4675. [Google Scholar] [CrossRef]
- Khotimchenko, S.V. Fatty acids of green macrophytic algae from the sea of Japan. Phytochemistry 1993, 32, 1203–1207. [Google Scholar] [CrossRef]
- Thompson, G.A. Lipids and membrane function in green algae. Biochim. Biophys. Acta 1996, 1302, 17–45. [Google Scholar] [CrossRef]
- Khotimchenko, S.V.; Vaskovsky, V.E.; Titlyanova, T.V. Fatty acids of marine algae from the pacific coast of North California. Bot. Mar. 2002, 45, 17–22. [Google Scholar] [CrossRef]
- Li, X.; Fan, X.; Han, L.; Lou, Q. Fatty acids of some algae from the Bohai Sea. Phytochemistry 2002, 59, 157–161. [Google Scholar] [CrossRef]
- Kumari, P.; Kumar, M.; Gupta, V.; Reddy, C.R.K.; Jha, B. Tropical marine macroalgae as potential sources of nutritionally important PUFAs. Food Chem. 2010, 120, 749–757. [Google Scholar] [CrossRef]
- Lahaye, M.; Robic, A. Structure and function properties of Ulvan, a polysaccharide from green seaweeds. Biomacromolecules 2007, 8, 1765–1774. [Google Scholar] [CrossRef]
- Alves, A.; Sousa, R.A.; Reis, R.L. A practical perspective on ulvan extracted from green algae. J. Appl. Phycol. 2013, 25, 407–424. [Google Scholar] [CrossRef]
- Kaeffer, B.; Bénard, C.; Lahaye, M.; Blottière, H.M.; Cherbut, C. Biological Properties of Ulvan, a New Source of Green Seaweed Sulfated Polysaccharides, on Cultured Normal and Cancerous Colonic Epithelial Cells. Planta Med. 1999, 65, 527–531. [Google Scholar] [CrossRef]
- Kidgell, J.T.; Magnusson, M.; de Nys, R.; Glasson, C.R.K. Ulvan: A systematic review of extraction, composition and function. Algal Res. 2019, 39, 101422. [Google Scholar] [CrossRef]
- Wijesekara, I.; Pangestuti, R.; Kim, S.-K. Biological activities and potential health benefits of sulfated polysaccharides derived from marine algae. Carbohydr. Polym. 2011, 84, 14–21. [Google Scholar] [CrossRef]
- Venkatesan, J.; Lowe, B.; Anil, S.; Manivasagan, P.; Kheraif, A.A.A.; Kang, K.H.; Kim, S.K. Seaweed polysaccharides and their potential biomedical applications. Starch/Staerke 2015, 67, 381–390. [Google Scholar] [CrossRef]
- Cunha, L.; Grenha, A. Sulfated Seaweed Polysaccharides as Multifunctional Materials in Drug Delivery Applications. Mar. Drugs 2016, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Lahaye, M.; Ray, B.; Baumberger, S.; Quemener, B.; Axelos, M.A.V. Chemical characterisation and gelling properties of cell wall polysaccharides from species of Ulva (Ulvales, Chlorophyta). Hydrobiologia 1996, 326–327, 473–480. [Google Scholar] [CrossRef]
- Webster, E.A.; Gadd, G.M. Cadmium replaces calcium in the cell wall of Ulva lactuca. BioMetals 1996, 9, 241–244. [Google Scholar] [CrossRef]
- Bocanegra, A.; Bastida, S.; Benedí, J.; Ródenas, S.; Sánchez-Muniz, F.J. Characteristics and nutritional and cardiovascular-health properties of seaweeds. J. Med. Food 2009, 12, 236–258. [Google Scholar] [CrossRef]
- Schijf, J.; Ebling, A.M. Investigation of the ionic strength dependence of Ulva lactuca acid functional group pKas by manual alkalimetric titrations. Environ. Sci. Technol. 2010, 44, 1644–1649. [Google Scholar] [CrossRef]
- Castine, S.A.; McKinnon, A.D.; Paul, N.A.; Trott, L.A.; de Nys, R. Wastewater treatment for land-based Aquaculture: Improvements and value-adding alternatives in model systems from Australia. Aquac. Environ. Interact. 2013, 4, 285–300. [Google Scholar] [CrossRef]
- Lawton, R.J.; Mata, L.; de Nys, R.; Paul, N.A. Algal Bioremediation of Waste Waters from Land-Based Aquaculture Using Ulva: Selecting Target Species and Strains. PLoS ONE 2013, 8, e77344. [Google Scholar] [CrossRef]
- Glasson, C.R.K.; Sims, I.M.; Carnachan, S.M.; de Nys, R.; Magnusson, M. A cascading biorefinery process targeting sulfated polysaccharides (ulvan) from Ulva ohnoi. Algal Res. 2017, 27, 383–391. [Google Scholar] [CrossRef]
- Ortiz-Calderon, C.; Silva, H.C.; Vásquez, D.B. Metal Removal by Seaweed Biomass. In Biomass Volume Estimation and Valorization for Energy; InTech: London, UK, 2017. [Google Scholar]
- Vardhan, K.H.; Kumar, P.S.; Panda, R.C. A review on heavy metal pollution, toxicity and remedial measures: Current trends and future perspectives. J. Mol. Liq. 2019, 290, 111197. [Google Scholar] [CrossRef]
- Dumas, B.; Jaulneau, V.; Lafitte, C.; Jacquet, C.; Fournier, S.; Salamagne, S.; Briand, X.; Esquerré-Tugayé, M.T. Ulvan, a sulfated polysaccharide from green algae, activates plant immunity through the jasmonic acid signaling pathway. J. Biomed. Biotechnol. 2010, 2010, 525291. [Google Scholar]
- Kapetanović, R.; Sladić, D.; Popov, S.; Zlatović, M.; Kljajić, Z.; Gašić, M.J. Sterol composition of the adriatic sea algae Ulva lactuca, codium dichotomum, cystoseira adriatica and fucus virsoides. J. Serbian Chem. Soc. 2005, 70, 1395–1400. [Google Scholar] [CrossRef]
- Green Confertii Extract-NS—The Garden of Naturalsolution. Available online: https://cosmetics.specialchem.com/product/i-natural-solution-green-confertii-extract-ns (accessed on 10 April 2020).
- Dembitsky, V.M.; Maoka, T. Allenic and cumulenic lipids. Prog. Lipid Res. 2007, 46, 328–375. [Google Scholar] [CrossRef]
- Chandini, S.K.; Ganesan, P.; Bhaskar, N. In vitro antioxidant activities of three selected brown seaweeds of India. Food Chem. 2008, 107, 707–713. [Google Scholar] [CrossRef]
- Knowler, D.; Chopin, T.; Martínez-Espiñeira, R.; Neori, A.; Nobre, A.; Noce, A.; Reid, G. The economics of Integrated Multi-Trophic Aquaculture: Where are we now and where do we need to go? Rev. Aquac. 2020, 8. [Google Scholar] [CrossRef]
- Yan, X.; Chuda, Y.; Suzuki, M.; Nagata, T. Fucoxanthin as the major antioxidant in hijikia fusiformis, a common edible seaweed. Biosci. Biotechnol. Biochem. 1999, 63, 605–607. [Google Scholar] [CrossRef]
- Maeda, H.; Hosokawa, M.; Sashima, T.; Funayama, K.; Miyashita, K. Fucoxanthin from edible seaweed, Undaria pinnatifida, shows antiobesity effect through UCP1 expression in white adipose tissues. Biochem. Biophys. Res. Commun. 2005, 332, 392–397. [Google Scholar] [CrossRef]
- Mise, T.; Ueda, M.; Yasumoto, T. Production of fucoxanthin-rich powder from Cladosiphon okamuranus. Adv. J. Food Sci. Technol. 2011, 3, 73–76. [Google Scholar]
- Pigmen, K.; Yip, W.H.; Lim, S.J.; Mustapha, W.A.W.; Maskat, M.Y.; Said, M.; Pigmen, K. Characterisation and Stability of Pigments Extracted from Sargassum binderi Obtained from Semporna, Sabah. Sains Malays. 2014, 43, 1345–1354. [Google Scholar]
- Johns, R.B.; Nichols, P.D.; Perry, G.J. Fatty acid composition of ten marine algae from australian waters. Phytochemistry 1979, 18, 799–802. [Google Scholar] [CrossRef]
- Vaskovsky, V.E.; Khotimchenko, S.V.; Xia, B.; Hefang, L. Polar lipids and fatty acids of some marine macrophytes from the yellow sea. Phytochemistry 1996, 42, 1347–1356. [Google Scholar] [CrossRef]
- Khotimchenko, S.V. Fatty acids of brown algae from the Russian Far East. Phytochemistry 1998, 49, 2363–2369. [Google Scholar] [CrossRef]
- Graeve, M.; Kattner, G.; Wiencke, C.; Karsten, U. Fatty acid composition of Arctic and Antarctic macroalgae: Indicator of phylogenetic and trophic relationships. Mar. Ecol. Prog. Ser. 2002, 231, 67–74. [Google Scholar] [CrossRef]
- Bhaskar, N.; Kazuo, M.; Masashi, H. Comparative Evaluation of Fatty Acid Composition of Different Sargassum (Fucales, Phaeophyta) Species Harvested from Temperate and Tropical Waters. J. Aquat. Food Prod. Technol. 2004, 13, 41–52. [Google Scholar]
- Kamenarska, Z.G.; Dimitrova-Konaklieva, S.D.; Stefanov, K.L.; Popov, S.S. A comparative study on the sterol composition of some brown algae from the Black Sea. J. Serb. Chem. Soc. 2003, 68, 269–275. [Google Scholar] [CrossRef]
- Lopes, G.; Sousa, C.; Bernardo, J.; Andrade, P.B.; Valentão, P.; Ferreres, F.; Mouga, T. Sterol profiles in 18 macroalgae of the portuguese coast. J. Phycol. 2011, 47, 1210–1218. [Google Scholar] [CrossRef]
- Patterson, G.W. The distribution of sterols in algae. Lipids 1971, 6, 120–127. [Google Scholar] [CrossRef]
- Fattorusso, E.; Magno, S.; Mayol, L. Sterols of mediterranean chlorophyceae. Experientia 1980, 36, 1137–1138. [Google Scholar] [CrossRef]
- Lopes, G.; Sousa, C.; Valentão, P.; Andrade, P.B. Sterols in Algae and Health. Bioactive Compounds from Marine Foods; Hernández-Ledesma, B., Herrero, M., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; pp. 173–191. [Google Scholar]
- Reddy, P.; Urban, S. Meroditerpenoids from the southern Australian marine brown alga Sargassum fallax. Phytochemistry 2009, 70, 250–255. [Google Scholar] [CrossRef]
- Kellogg, J.; Grace, M.H.; Lila, M.A. Phlorotannins from alaskan seaweed inhibit carbolytic enzyme activity. Mar. Drugs 2014, 12, 5277–5294. [Google Scholar] [CrossRef] [PubMed]
- Murray, M.; Dordevic, A.L.; Ryan, L.; Bonham, M.P. The impact of a single dose of a polyphenol-rich seaweed extract on postprandial glycaemic control in healthy adults: A randomised cross-over trial. Nutrients 2018, 10, 270. [Google Scholar] [CrossRef] [PubMed]
- Artan, M.; Li, Y.; Karadeniz, F.; Lee, S.H.; Kim, M.M.; Kim, S.K. Anti-HIV-1 activity of phloroglucinol derivative, 6,6′-bieckol, from Ecklonia cava. Bioorganic Med. Chem. 2008, 16, 7921–7926. [Google Scholar] [CrossRef] [PubMed]
- Yoon, N.Y.; Chung, H.Y.; Kim, H.R.; Choi, J.S. Acetyl- and butyrylcholinesterase inhibitory activities of sterols and phlorotannins from Ecklonia stolonifera. Fish. Sci. 2008, 74, 200–207. [Google Scholar] [CrossRef]
- Shim, S.Y.; Choi, J.S.; Byun, D.S. Inhibitory effects of phloroglucinol derivatives isolated from Ecklonia stolonifera on FcεRI expression. Bioorganic Med. Chem. 2009, 17, 4734–4739. [Google Scholar] [CrossRef]
- Ah, H.; Ho, S.; Sue, J. Bioorganic & Medicinal Chemistry Letters Molecular docking studies of phlorotannins from Eisenia bicyclis with BACE1 inhibitory activity. Bioorganic Med. Chem. Lett. 2010, 20, 3211–3215. [Google Scholar]
- Kim, S.K.; Kong, C.S. Anti-adipogenic effect of dioxinodehydroeckol via AMPK activation in 3T3-L1 adipocytes. Chem. Biol. Interact. 2010, 186, 24–29. [Google Scholar] [CrossRef]
- Whitfield, F.B.; Helidoniotis, F.; Shaw, K.J.; Svoronos, D. Distribution of bromophenols in species of marine algae from eastern Australia. J. Agric. Food Chem. 1999, 47, 2367–2373. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Milley, J.E.; Lall, S.P. Nutritional quality of some wild and cultivated seaweeds: Nutrient composition, total phenolic content and in vitro digestibility. J. Appl. Phycol. 2016, 28, 3575–3585. [Google Scholar] [CrossRef]
- Roy, M.C.; Anguenot, R.; Fillion, C.; Beaulieu, M.; Bérubé, J.; Richard, D. Effect of a commercially-available algal phlorotannins extract on digestive enzymes and carbohydrate absorption in vivo. Food Res. Int. 2011, 44, 3026–3029. [Google Scholar] [CrossRef]
- Douglas, T.E.L.; Dokupil, A.; Reczyńska, K.; Brackman, G.; Krok-Borkowicz, M.; Keppler, J.K.; Božič, M.; Van Der Voort, P.; Pietryga, K.; Samal, S.K.; et al. Enrichment of enzymatically mineralized gellan gum hydrogels with phlorotannin-rich Ecklonia cava extract Seanol® to endow antibacterial properties and promote mineralization. Biomed. Mater. 2016, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Andrade, L.R.; Salgado, L.T.; Farina, M.; Pereira, M.S.; Mourão, P.A.S.; Amado Filho, G.M. Ultrastructure of acidic polysaccharides from the cell walls of brown algae. J. Struct. Biol. 2004, 145, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Wiltshire, K.H.; Tanner, J.E.; Gurgel, C.F.D.; Deveney, M.R. Feasibility study for integrated multitrophic aquaculture in southern Australia. In Report to the Fisheries Research & Development Corporation; SARDI: Adelaide, Australia, 2015. [Google Scholar]
- White, W.L.; Wilson, P. Chapter 2: World Seaweed Utilization; Elsevier Inc.: Amsterdam, The Netherlands, 2015. [Google Scholar]
- McHugh, D.J. A Guide to the Seaweed Industry; FAO Fisher: Rome, Italy, 2003. [Google Scholar]
- Imeson, A. Food Stabilisers, Thickeners and Gelling Agents; Imeson, A., Ed.; Wiley-Blackwell: Oxford, UK, 2009. [Google Scholar]
- Fleurence, J. The enzymatic degradation of algal cell walls: A useful approach for improving protein accessibility? J. Appl. Phycol. 1999, 11, 313–314. [Google Scholar] [CrossRef]
- Sandberg, A.S.; Andersson, H.; Bosaeus, I.; Carlsson, N.G.; Hasselblad, K.; Harrod, M. Alginate, small bowel sterol excretion, and absorption of nutrients in ileostomy subjects. Am. J. Clin. Nutr. 1994, 60, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Wolf, B.W.; Lai, C.S.; Kipnes, M.S.; Ataya, D.G.; Wheeler, K.B.; Zinker, B.A.; Garleb, K.A.; Firkins, J.L. Glycemic and insulinemic responses of nondiabetic healthy adult subjects to an experimental acid-induced viscosity complex incorporated into a glucose beverage. Nutrition 2002, 18, 621–626. [Google Scholar] [CrossRef]
- Black, W.A.P.; Cornhill, W.J.; Dewar, E.T.; Woodward, F.N. Manufactureof algal chemicals. III. Laboratory-scale isolation oflaminarin from brown marine algae. J. Appl. Chem. 1951, 1, 505–517. [Google Scholar] [CrossRef]
- Annan, W.D.; Hirst, E.; Manners, D.J. The constitution of laminarin. Part V. The location of 1,6-glucosidic linkages. J. Chem. Soc. 1965, 1, 885–891. [Google Scholar]
- Kadam, S.U.; Tiwari, B.K.; O’Donnell, C.P. Extraction, structure and biofunctional activities of laminarin from brown algae. Int. J. Food Sci. Technol. 2015, 50, 24–31. [Google Scholar] [CrossRef]
- Rioux, L.E.; Turgeon, S.L.; Beaulieu, M. Structural characterization of laminaran and galactofucan extracted from the brown seaweed Saccharina longicruris. Phytochemistry 2010, 71, 1586–1595. [Google Scholar] [CrossRef]
- Devillé, C.; Damas, J.; Forget, P.; Dandrifosse, G.; Peulen, O. Laminarin in the dietary fibre concept. J. Sci. Food Agric. 2004, 84, 1030–1038. [Google Scholar] [CrossRef]
- Déville, C.; Gharbi, M.; Dandrifosse, G.; Peulen, O. Study on the effects of laminarin, a polysaccharide from seaweed, on gut characteristics. J. Sci. Food Agric. 2007, 1725, 1717–1725. [Google Scholar] [CrossRef]
- Imbs, T.I.; Skriptsova, A.V.; Zvyagintseva, T.N. Antioxidant activity of fucose-containing sulfated polysaccharides obtained from Fucus evanescens by different extraction methods. J. Appl. Phycol. 2014, 27, 545–553. [Google Scholar] [CrossRef]
- Ponce, N.M.A.; Pujol, C.A.; Damonte, E.B.; Flores, M.L.; Stortz, C.A. Fucoidans from the brown seaweed Adenocystis utricularis: Extraction methods, antiviral activity and structural studies. Carbohydr. Res. 2003, 338, 153–165. [Google Scholar] [CrossRef]
- Li, B.; Lu, F.; Wei, X.; Zhao, R. Fucoidan: Structure and bioactivity. Molecules 2008, 13, 1671–1695. [Google Scholar] [CrossRef] [PubMed]
- Mandal, P.; Mateu, C.G.; Chattopadhyay, K.; Pujol, C.A.; Damonte, E.B.; Ray, B. Structural features and antiviral activity of sulphated fucans from the brown seaweed Cystoseira indica. Antivir. Chem. Chemother. 2007, 18, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Chandía, N.P.; Matsuhiro, B. Characterization of a fucoidan from Lessonia vadosa (Phaeophyta) and its anticoagulant and elicitor properties. Int. J. Biol. Macromol. 2008, 42, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Chizhov, A.O.; Dell, A.; Morris, H.R.; Haslam, S.M.; McDowell, R.A.; Shashkov, A.S.; Nifant’ev, N.E.; Khatuntseva, E.A.; Usov, A.I. A study of fucoidan from the brown seaweed Chorda filum. Carbohydr. Res. 1999, 320, 108–119. [Google Scholar] [CrossRef]
- Wijesinghe, W.A.J.P.; Jeon, Y. Biological activities and potential industrial applications of fucose rich sulfated polysaccharides and fucoidans isolated from brown seaweeds: A review. Carbohydr. Polym. 2012, 88, 13–20. [Google Scholar] [CrossRef]
- Peng, Y.; Xie, E.; Zheng, K.; Fredimoses, M.; Yang, X.; Zhou, X.; Wang, Y.; Yang, B.; Lin, X.; Liu, J.; et al. Nutritional and chemical composition and antiviral activity of cultivated seaweed sargassum naozhouense Tseng et Lu. Mar. Drugs 2013, 11, 20–32. [Google Scholar] [CrossRef]
- Osea Malibu. Available online: https://oseamalibu.com/ (accessed on 14 December 2018).
- Cabarry, C. SpecialChem-Connect, Innovate, Accelerate. Available online: https://cosmetics.specialchem.com/ (accessed on 25 February 2020).
- Bommers, M. La-Mer. Available online: https://www.la-mer.com/en/ (accessed on 26 February 2020).
- Cian, R.E.; Caballero, M.S.; Sabbag, N.; González, R.J.; Drago, S.R. Bio-accessibility of bioactive compounds (ACE inhibitors and antioxidants) from extruded maize products added with a red seaweed Porphyra columbina. LWT Food Sci. Technol. 2014, 55, 51–58. [Google Scholar] [CrossRef]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef]
- Sekar, S.; Chandramohan, M. Phycobiliproteins as a commodity: Trends in applied research, patents and commercialization. J. Appl. Phycol. 2008, 20, 113–136. [Google Scholar] [CrossRef]
- Kendel, M.; Wielgosz-Collin, G.; Bertrand, S.; Roussakis, C.; Bourgougnon, N.; Bedoux, G. Lipid Composition, Fatty Acids and Sterols in the Seaweeds Ulva armoricana, and Solieria chordalis from Brittany (France): An Analysis from Nutritional, Chemotaxonomic, and Antiproliferative Activity Perspectives. Mar. Drugs 2015, 13, 5606–5628. [Google Scholar] [CrossRef] [PubMed]
- Kumari, P.; Bijo, A.J.; Mantri, V.A.; Reddy, C.R.K.; Jha, B. Fatty acid profiling of tropical marine macroalgae: An analysis from chemotaxonomic and nutritional perspectives. Phytochemistry 2013, 86, 44–56. [Google Scholar] [CrossRef]
- Namvar, F.; Mohamed, S.; Fard, S.G.; Behravan, J.; Mustapha, N.M.; Alitheen, N.B.M.; Othman, F. Polyphenol-rich seaweed (Eucheuma cottonii) extract suppresses breast tumour via hormone modulation and apoptosis induction. Food Chem. 2012, 130, 376–382. [Google Scholar] [CrossRef]
- Guihéneuf, F.; Gietl, A.; Stengel, D.B. Temporal and spatial variability of mycosporine-like amino acids and pigments in three edible red seaweeds from western Ireland. J. Appl. Phycol. 2018, 30, 2573–2586. [Google Scholar] [CrossRef]
- Torres, P.; Santos, J.P.; Chow, F.; Pena Ferreira, M.J.; dos Santos, D.Y.A.C. Comparative analysis of in vitro antioxidant capacities of mycosporine-like amino acids (MAAs). Algal Res. 2018, 34, 57–67. [Google Scholar] [CrossRef]
- Usov, A.I. Polysaccharides of the red algae. In Advances in Carbohydrate Chemistry and Biochemistry; Elsevier Inc.: Amsterdam, The Netherlands, 2011; Volume 65, pp. 115–217. [Google Scholar]
- Armisen, R. World-wide use and importance of Gracilaria. J. Appl. Phycol. 1995, 7, 231–243. [Google Scholar] [CrossRef]
- Lee, W.K.; Lim, Y.Y.; Leow, A.T.C.; Namasivayam, P.; Abdullah, J.O.; Ho, C.L. Factors affecting yield and gelling properties of agar. J. Appl. Phycol. 2017, 29, 1527–1540. [Google Scholar] [CrossRef]
- Yarnpakdee, S.; Benjakul, S.; Kingwascharapong, P. Physico-chemical and gel properties of agar from Gracilaria tenuistipitata from the lake of Songkhla, Thailand. Food Hydrocoll. 2015, 51, 217–226. [Google Scholar] [CrossRef]
- Daugherty, B.K.; Bird, K.T. Salinity and temperature effects on agar production from Gracilaria verrucosa Strain G-16. Aquaculture 1988, 75, 105–113. [Google Scholar] [CrossRef]
- Cardozo, K.H.M.M.; Guaratini, T.; Barros, M.P.; Falcão, V.R.; Tonon, A.P.; Lopes, N.P.; Campos, S.; Torres, M.A.; Souza, A.O.; Colepicolo, P.; et al. Metabolites from algae with economical impact. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2007, 146, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Arbit, N.I.S.; Omar, S.B.A.; Soekendarsi, E.; Yasir, I.; Tresnati, J.; Tuwo, A. Morphological and genetic analysis of Gracilaria sp. cultured in ponds and coastal waters. In IOP Conference Series: Earth and Environmental Science; Institute of Physics Publishing: Bristol, UK, 2019; Volume 370. [Google Scholar]
- Silva, D.M.; Valente, L.M.P.; Sousa-Pinto, I.; Pereira, R.; Pires, M.A.; Seixas, F.; Rema, P. Evaluation of IMTA-produced seaweeds (Gracilaria, Porphyra, and Ulva) as dietary ingredients in Nile tilapia, Oreochromis niloticus L., juveniles. Effects on growth performance and gut histology. J. Appl. Phycol. 2015, 27, 1671–1680. [Google Scholar] [CrossRef]
- Abreu, M.H.; Pereira, R.; Yarish, C.; Buschmann, A.H.; Sousa-Pinto, I. IMTA with Gracilaria vermiculophylla: Productivity and nutrient removal performance of the seaweed in a land-based pilot scale system. Aquaculture 2011, 312, 77–87. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Mora, O.A.; Gómez, P.; Böttger, M.; Buitano, S.; Retamales, C.; Vergara, P.A.; Gutierrez, A. Gracilaria chilensis outdoor tank cultivation in Chile: Use of land-based salmon culture effluents. Aquac. Eng. 1994, 13, 283–300. [Google Scholar] [CrossRef]
- Troell, M.; Halling, C.; Nilsson, A.; Buschmann, A.H.; Kautsky, N.; Kautsky, L. Integrated marine cultivation of Gracilaria chilensis (Gracilariales, Rhodophyta) and salmon cages for reduced environmental impact and increased economic output. Aquaculture 1997, 156, 45–61. [Google Scholar] [CrossRef]
- Seedevi, P.; Moovendhan, M.; Viramani, S.; Shanmugam, A. Bioactive potential and structural chracterization of sulfated polysaccharide from seaweed (Gracilaria corticata). Carbohydr. Polym. 2017, 155, 516–524. [Google Scholar] [CrossRef]
- Chen, H.M.; Zheng, L.; Yan, X.J. The preparation and bioactivity research of agaro-oligosaccharides. Food Technol. Biotechnol. 2005, 43, 29–36. [Google Scholar]
- Jin, M.; Liu, H.; Hou, Y.; Chan, Z.; Di, W.; Li, L.; Zeng, R. Preparation, characterization and alcoholic liver injury protective effects of algal oligosaccharides from Gracilaria lemaneiformis. Food Res. Int. 2017, 100, 186–195. [Google Scholar] [CrossRef]
- Souza, B.W.S.; Cerqueira, M.A.; Bourbon, A.I.; Pinheiro, A.C.; Martins, J.T.; Teixeira, J.A.; Coimbra, M.A.; Vicente, A.A. Chemical characterization and antioxidant activity of sulfated polysaccharide from the red seaweed Gracilaria birdiae. Food Hydrocoll. 2012, 27, 287–292. [Google Scholar] [CrossRef]
- Mazumder, S.; Ghosal, P.K.; Pujol, C.A.; Carlucci, M.J.; Damonte, E.B.; Ray, B. Isolation, chemical investigation and antiviral activity of polysaccharides from Gracilaria corticata (Gracilariaceae, Rhodophyta). Int. J. Biol. Macromol. 2002, 31, 87–95. [Google Scholar] [CrossRef]
- Bhattarai, Y.; Kashyap, P.C. Agaro-oligosaccharides: A new frontier in the fight against colon cancer? Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 310, G335–G336. [Google Scholar] [CrossRef] [PubMed]
- Coura, C.O.; Souza, R.B.; Rodrigues, J.A.G.; Vanderlei, E.D.S.O.; De Araújo, I.W.F.; Ribeiro, N.A.; Frota, A.F.; Ribeiro, K.A.; Chaves, H.V.; Pereira, K.M.A.; et al. Mechanisms involved in the anti-inflammatory action of a polysulfated fraction from Gracilaria cornea in rats. PLoS ONE 2015, 10, e0119319. [Google Scholar] [CrossRef]
- Hehemann, J.H.; Correc, G.; Thomas, F.; Bernard, T.; Barbeyron, T.; Jam, M.; Helbert, W.; Michel, G.; Czjzek, M. Biochemical and structural characterization of the complex agarolytic enzyme system from the marine bacterium Zobellia galactanivorans. J. Biol. Chem. 2012, 287, 30571–30584. [Google Scholar] [CrossRef] [PubMed]
- Higashimura, Y.; Naito, Y.; Takagi, T.; Mizushima, K.; Hirai, Y.; Harusato, A.; Ohnogi, H.; Yamaji, R.; Inui, H.; Nakano, Y.; et al. Oligosaccharides from agar inhibit murine intestinal inflammation through the induction of heme oxygenase-1 expression. J. Gastroenterol. 2013, 48, 897–909. [Google Scholar] [CrossRef]
- Jang, M.K.; Lee, D.G.; Kim, N.Y.; Yu, K.H.; Jang, H.J.; Lee, S.W.; Jang, H.J.; Lee, Y.J.; Lee, S.H. Purification and characterization of neoagarotetraose from hydrolyzed agar. J. Microbiol. Biotechnol. 2009, 19, 1197–1200. [Google Scholar] [PubMed]
- Liu, Q.M.; Xu, S.S.; Li, L.; Pan, T.M.; Shi, C.L.; Liu, H.; Cao, M.J.; Su, W.J.; Liu, G.M. In vitro and in vivo immunomodulatory activity of sulfated polysaccharide from Porphyra haitanensis. Carbohydr. Polym. 2017, 165, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Yun, E.J.; Yu, S.; Kim, K.H. Current knowledge on agarolytic enzymes and the industrial potential of agar-derived sugars. Appl. Microbiol. Biotechnol. 2017, 101, 5581–5589. [Google Scholar] [CrossRef]
- Kazłowski, B.; Pan, C.L.; Ko, Y.T. Monitoring and preparation of neoagaro- and agaro-oligosaccharide products by high performance anion exchange chromatography systems. Carbohydr. Polym. 2015, 122, 351–358. [Google Scholar] [CrossRef]
- Freile-Pelegrín, Y.; Robledo, D. Influence of alkali treatment on agar from Gracilaria cornea from Yucatan, Mexico. J. Appl. Phycol. 1997, 9, 533–539. [Google Scholar]
- Freile-Pelegrín, Y.; Murano, E. Agars from three species of Gracilaria (Rhodophyta) from Yucatán Peninsula. Bioresour. Technol. 2005, 96, 295–302. [Google Scholar] [CrossRef]
- Marinho-Soriano, E.; Bourret, E. Polysaccharides from the red seaweed Gracilaria dura (Gracilariales, Rhodophyta). Bioresour. Technol. 2005, 96, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.M.M.; Alves, V.D.; Morais, S.; Delerue-Matos, C.; Gonçalves, M.P. Agar extraction from integrated multitrophic aquacultured Gracilaria vermiculophylla: Evaluation of a microwave-assisted process using response surface methodology. Bioresour. Technol. 2010, 101, 3258–3267. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, R.D.; Sousa, A.M.M.; Gonçalves, M.P.; Nilsson, M.; Hilliou, L. Production and properties of agar from the invasive marine alga, Gracilaria vermiculophylla (Gracilariales, Rhodophyta). J. Appl. Phycol. 2010, 22, 211–220. [Google Scholar] [CrossRef]
- Meireles, F. Carrageenan Yield and Quality of Chondrus Crispus Stackhouse (Rhodophyta) Cultivated in an Integrated Multi-Trophic Aquaculture (IMTA) System. Master’s Thesis, University of Coimbra, Coimbra, Portugal, 2013. [Google Scholar]
- Torres, M.D.; Flórez-Fernández, N.; Domínguez, H. Integral Utilization of Red Seaweed for Bioactive Production. Mar. Drugs 2019, 17, 314. [Google Scholar] [CrossRef]
- Gurgel, C.F.D.; Lopez-Bautista, J. Red Algae. Encycl. Life Sci. 2007, 1–5. [Google Scholar] [CrossRef]
- Pereira, L.; Mesquita, J.F. Carrageenophytes of occidental Portuguese coast: 1-spectroscopic analysis in eight carrageenophytes from Buarcos bay. Biomol. Eng. 2003, 20, 217–222. [Google Scholar] [CrossRef]
- Schultz-Johansen, M.; Bech, P.K.; Hennessy, R.C.; Glaring, M.A.; Barbeyron, T.; Czjzek, M.; Stougaard, P. A Novel Enzyme Portfolio for Red Algal Polysaccharide Degradation in the Marine Bacterium Paraglaciecola hydrolytica S66T Encoded in a Sizeable Polysaccharide Utilization Locus. Front. Microbiol. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Ghanbarzadeh, M.; Golmoradizadeh, A.; Homaei, A. Carrageenans and carrageenases: Versatile polysaccharides and promising marine enzymes. Phytochem. Rev. 2018, 17, 535–571. [Google Scholar] [CrossRef]
- Collén, J.; Cornish, M.L.; Craigie, J.; Ficko-Blean, E.; Hervé, C.; Krueger-Hadfield, S.A.; Leblanc, C.; Michel, G.; Potin, P.; Tonon, T.; et al. Chondrus Crispus-A Present and Historical Model. Organism for Red Seaweeds; Elsevier: Amsterdam, The Netherlands, 2014; Volume 71. [Google Scholar]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar] [CrossRef]
- De Sousa Oliveira Vanderlei, E.; De Araújo, I.W.F.; Quinderé, A.L.G.; Fontes, B.P.; Eloy, Y.R.G.; Rodrigues, J.A.G.; Silva, A.A.R.E.; Chaves, H.V.; Jorge, R.J.B.; De Menezes, D.B.; et al. The involvement of the HO-1 pathway in the anti-inflammatory action of a sulfated polysaccharide isolated from the red seaweed Gracilaria birdiae. Inflamm. Res. 2011, 60, 1121–1130. [Google Scholar] [CrossRef]
- Cáceres, P.J.; Carlucci, M.J.; Damonte, E.B.; Matsuhiro, B.; Zúñiga, E.A. Carrageenans from chilean samples of Stenogramme interrupta (Phyllophoraceae): Structural analysis and biological activity. Phytochemistry 2000, 53, 81–86. [Google Scholar] [CrossRef]
- Chattopadhyay, K.; Ghosh, T.; Pujol, C.A.; Carlucci, M.J.; Damonte, E.B.; Ray, B. Polysaccharides from Gracilaria corticata: Sulfation, chemical characterization and anti-HSV activities. Int. J. Biol. Macromol. 2008, 43, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Inic-Kanada, A.; Stein, E.; Stojanovic, M.; Schuerer, N.; Ghasemian, E.; Filipovic, A.; Marinkovic, E.; Kosanovic, D.; Barisani-Asenbauer, T. Effects of iota-carrageenan on ocular Chlamydia trachomatis infection in vitro and in vivo. J. Appl. Phycol. 2018, 30, 2601–2610. [Google Scholar] [CrossRef] [PubMed]
- Talarico, L.B.; Zibetti, R.G.M.; Faria, P.C.S.; Scolaro, L.A.; Duarte, M.E.R.; Noseda, M.D.; Pujol, C.A.; Damonte, E.B. Anti-herpes simplex virus activity of sulfated galactans from the red seaweeds Gymnogongrus griffithsiae and Cryptonemia crenulata. Int. J. Biol. Macromol. 2004, 34, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Talarico, L.B.; Pujol, C.A.; Zibetti, R.G.M.; Faría, P.C.S.; Noseda, M.D.; Duarte, M.E.R.; Damonte, E.B. The antiviral activity of sulfated polysaccharides against dengue virus is dependent on virus serotype and host cell. Antivir. Res. 2005, 66, 103–110. [Google Scholar] [CrossRef]
- Shi, Q.; Wang, A.; Lu, Z.; Qin, C.; Hu, J.; Yin, J. Overview on the antiviral activities and mechanisms of marine polysaccharides from seaweeds. Carbohydr. Res. 2017, 453–454, 1–9. [Google Scholar] [CrossRef]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Bioactivity of sulfated polysaccharides from the edible red seaweed Mastocarpus stellatus. Bioact. Carbohydr. Diet. Fibre 2014, 3, 29–40. [Google Scholar] [CrossRef]
- Yuan, H.; Song, J.; Li, X.; Li, N.; Dai, J. Immunomodulation and antitumor activity of κ-carrageenan oligosaccharides. Cancer Lett. 2006, 243, 228–234. [Google Scholar] [CrossRef]
- Liu, J.; Hafting, J.; Critchley, A.T.; Banskota, A.H.; Prithiviraj, B. Components of the cultivated red seaweed Chondrus crispus enhance the immune response of Caenorhabditis elegans to Pseudomonas aeruginosa through the pmk-1, daf-2/daf-16, and skn-1 pathways. Appl. Environ. Microbiol. 2013, 79, 7343–7350. [Google Scholar] [CrossRef]
- Souza, M.P.; Vaz, A.F.M.; Costa, T.B.; Cerqueira, M.A.; De Castro, C.M.M.B.; Vicente, A.A.; Carneiro-da-Cunha, M.G. Construction of a Biocompatible and Antioxidant Multilayer Coating by Layer-by-Layer Assembly of κ-Carrageenan and Quercetin Nanoparticles. Food Bioprocess. Technol. 2018, 11, 1050–1060. [Google Scholar] [CrossRef]
- Sun, L.; Wang, S.; Gong, X.; Zhao, M.; Fu, X.; Wang, L. Isolation, purification and characteristics of R-phycoerythrin from a marine macroalga Heterosiphonia japonica. Protein Expr. Purif. 2009, 64, 146–154. [Google Scholar] [CrossRef] [PubMed]
- Poupard, N.; Badarou, P.; Fasani, F.; Groult, H.; Bridiau, N.; Sannier, F.; Bordenave-Juchereau, S.; Kieda, C.; Piot, J.M.; Grillon, C.; et al. Assessment of heparanase-mediated angiogenesis using microvascular endothelial cells: Identification of λ-Carrageenan derivative as a potent anti angiogenic agent. Mar. Drugs 2017, 15, 134. [Google Scholar] [CrossRef] [PubMed]
- Ghannam, A.; Abbas, A.; Alek, H.; Al-Waari, Z.; Al-Ktaifani, M. Enhancement of local plant immunity against tobacco mosaic virus infection after treatment with sulphated-carrageenan from red alga (Hypnea musciformis). Physiol. Mol. Plant. Pathol. 2013, 84, 19–27. [Google Scholar] [CrossRef]
- Mercier, L.; Lafitte, C.; Borderies, G.; Briand, X.; Esquerré-Tugayé, M.T.; Fournier, J. The algal polysaccharide carrageenans can act as an elicitor of plant defence. New Phytol. 2001, 149, 43–51. [Google Scholar] [CrossRef]
- Nagorskaya, V.P.; Reunov, A.V.; Lapshina, L.A.; Ermak, I.M.; Barabanova, A.O. Inhibitory effect of κ/β-carrageenan from red alga Tichocarpus crinitus on the development of a potato virus X infection in leaves of Datura stramonium L. Biol. Bull. 2010, 37, 653–658. [Google Scholar] [CrossRef]
- Sangha, J.S.; Ravichandran, S.; Prithiviraj, K.; Critchley, A.T.; Prithiviraj, B. Sulfated macroalgal polysaccharides λ-carrageenan and ι-carrageenan differentially alter Arabidopsis thaliana resistance to Sclerotinia sclerotiorum. Physiol. Mol. Plant. Pathol. 2010, 75, 38–45. [Google Scholar] [CrossRef]
- Sangha, J.S.; Khan, W.; Ji, X.; Zhang, J.; Mills, A.A.S.; Critchley, A.T.; Prithiviraj, B. Carrageenans, sulphated polysaccharides of red seaweeds, differentially affect Arabidopsis thaliana resistance to Trichoplusia ni (Cabbage Looper). PLoS ONE 2011, 6, e26834. [Google Scholar] [CrossRef]
- Sangha, J.S.; Kandasamy, S.; Khan, W.; Bahia, N.S.; Singh, R.P.; Critchley, A.T.; Prithiviraj, B. λ-carrageenan suppresses tomato chlorotic dwarf viroid (TCDVd) replication and symptom expression in tomatoes. Mar. Drugs 2015, 13, 2875–2889. [Google Scholar] [CrossRef]
- Shukla, P.S.; Borza, T.; Critchley, A.T.; Prithiviraj, B. Carrageenans from Red Seaweeds As Promoters of Growth and Elicitors of Defense Response in Plants. Front. Mar. Sci. 2016, 3, 1–9. [Google Scholar] [CrossRef]
- Vera, J.; Castro, J.; Contreras, R.A.; González, A.; Moenne, A. Oligo-carrageenans induce a long-term and broad-range protection against pathogens in tobacco plants (var. Xanthi). Physiol. Mol. Plant. Pathol. 2012, 79, 31–39. [Google Scholar] [CrossRef]
- Bi, Y.; Hu, Y.; Zhou, Z. Genetic variation of Laminaria japonica (Phaeophyta) populations in China as revealed by RAPD markers. Acta Oceanol. Sin. 2011, 30, 103–112. [Google Scholar] [CrossRef]
- Zou, P.; Lu, X.; Jing, C.; Yuan, Y.; Lu, Y.; Zhang, C.; Meng, L.; Zhao, H.; Li, Y. Low-molecular-weightt polysaccharides from Pyropia yezoensis enhance tolerance of wheat seedlings (Triticum aestivum L.) to salt stress. Front. Plant. Sci. 2018, 9, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Bixler, H.J.; Porse, H. A decade of change in the seaweed hydrocolloids industry. J. Appl. Phycol. 2011, 23, 321–335. [Google Scholar] [CrossRef]
- Ioannou, E.; Roussis, V. Natural Products from Seaweeds. In Plant.-Derived Natural Products; Osbourn, A., Lanzotti, V., Eds.; Springer: New York, NY, USA, 2009. [Google Scholar]
- Jiao, G.; Yu, G.; Zhang, J.; Ewart, H.S. Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar. Drugs 2011, 9, 196–233. [Google Scholar] [CrossRef]
- Morrice, L.M.; McLean, M.W.; Long, W.F.; Williamson, F.B. Porphyran primary structure. Eur. J. Biochem. 1983, 133, 673–684. [Google Scholar] [CrossRef]
- Stanley, P.; Turvey, J.R.; Rees, D.A. Carbohydrates of the red alga, porphyra umbilicalis. J. Chem. Soc. 1961, 1590–1595. [Google Scholar]
- Ishihara, K.; Oyamada, C.; Matsushima, R.; Murata, M.; Muraoka, T. Inhibitory effect of porphyran, prepared from dried “Nori”, on contact hypersensitivity in mice. Biosci. Biotechnol. Biochem. 2005, 69, 1824–1830. [Google Scholar] [CrossRef]
- Liu, Z.; Gao, T.; Yang, Y.; Meng, F.; Zhan, F.; Jiang, Q.; Sun, X. Anti-Cancer Activity of Porphyran and Carrageenan from Red Seaweeds. Molecules 2019, 24, 4286. [Google Scholar] [CrossRef]
- Noda, H. Health benefits and nutritional properties of nori. J. Appl. Phycol. 1993, 5, 255–258. [Google Scholar] [CrossRef]
- Zhang, Q.; Qi, H.; Zhao, T.; Deslandes, E.; Ismaeli, N.M.; Molloy, F.; Critchley, A.T. Chemical characteristics of a polysaccharide from Porphyra capensis (Rhodophyta). Carbohydr. Res. 2005, 340, 2447–2450. [Google Scholar] [CrossRef]
- He, D.; Wu, S.; Yan, L.; Zuo, J.; Cheng, Y.; Wang, H.; Liu, J.; Zhang, X.; Wu, M.; Choi, J.-I.; et al. Antitumor bioactivity of porphyran extracted from Pyropia yezoensis Chonsoo2 on human cancer cell lines. J. Sci. Food Agric. 2019, 99, 6722–6730. [Google Scholar] [CrossRef] [PubMed]
- Kwon, M.J.; Nam, T.J. Porphyran induces apoptosis related signal pathway in AGS gastric cancer cell lines. Life Sci. 2006, 79, 1956–1962. [Google Scholar] [CrossRef]
- Yu, X.; Zhou, C.; Yang, H.; Huang, X.; Ma, H.; Qin, X.; Hu, J. Effect of ultrasonic treatment on the degradation and inhibition cancer cell lines of polysaccharides from Porphyra yezoensis. Carbohydr. Polym. 2015, 117, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Inoue, N.; Yamano, N.; Sakata, K.; Nagao, K.; Hama, Y.; Yanagita, T. The sulfated polysaccharide porphyran reduces apolipoprotein B100 secretion and lipid synthesis in HepG2 cells. Biosci. Biotechnol. Biochem. 2009, 73, 447–449. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Noda, H.; Amano, H.; Nishino, T.; Nishizawa, K. Study on Antihypertensive and Antihyperlipidemic Effects of Marine Algae. Fish. Sci. 1994, 60, 83–88. [Google Scholar] [CrossRef]
- Tsuge, K.; Okabe, M.; Yoshimura, T.; Sumi, T.; Tachibana, H.; Yamada, K. Dietary effects of porphyran from Porphyra yezoensis on growth and lipid metabolism of Sprague-Dawley rats. Food Sci. Technol. Res. 2004, 10, 147–151. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Q.; Wang, J.; Zhang, H.; Niu, X.; Li, P. Preparation of the different derivatives of the low-molecular-weight porphyran from Porphyra haitanensis and their antioxidant activities in vitro. Int. J. Biol. Macromol. 2009, 45, 22–26. [Google Scholar] [CrossRef]
- Zhao, T.; Zhang, Q.; Qi, H.; Zhang, H.; Niu, X.; Xu, Z.; Li, Z. Degradation of porphyran from Porphyra haitanensis and the antioxidant activities of the degraded porphyrans with different molecular weight. Int. J. Biol. Macromol. 2006, 38, 45–50. [Google Scholar] [CrossRef]
- Lee, H.A.; Kim, I.H.; Nam, T.J. Bioactive peptide from Pyropia yezoensis and its anti-inflammatory activities. Int. J. Mol. Med. 2015, 36, 1701–1706. [Google Scholar] [CrossRef]
- Wang, Y.; Hwang, J.Y.; Park, H.-B.; Yadav, D.; Oda, T.; Jin, J.-O. Porphyran isolated from Pyropia yezoensis inhibits lipopolysaccharide-induced activation of dendritic cells in mice. Carbohydr. Polym. 2020, 229, 115457. [Google Scholar] [CrossRef]
- Yanagido, A.; Ueno, M.; Jiang, Z.; Cho, K.; Yamaguchi, K.; Kim, D.; Oda, T. Increase in anti-inflammatory activities of radical-degraded porphyrans isolated from discolored nori (Pyropia yezoensis). Int. J. Biol. Macromol. 2018, 117, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, S.; Rathee, P.; Sharma, K.; Chaugule, B.B.; Kar, N.; Bera, T. Immuno-modulation effect of sulphated polysaccharide (porphyran) from Porphyra vietnamensis. Int. J. Biol. Macromol. 2013, 57, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Escrig, A.; Gómez-Ordóñez, E.; Rupérez, P. Brown and red seaweeds as potential sources of antioxidant nutraceuticals. J. Appl. Phycol. 2012, 24, 1123–1132. [Google Scholar] [CrossRef]
- Liu, Y.J.; Deng, Z.; Geng, L.; Wang, J.; Zhang, Q. In vitro evaluation of the neuroprotective effect of oligo-porphyran from Porphyra yezoensis in PC12 cells. J. Appl. Phycol. 2019, 31, 2559–2571. [Google Scholar] [CrossRef]
- Ueno, M.; Cho, K.; Isaka, S.; Nishiguchi, T.; Yamaguchi, K.; Kim, D.; Oda, T. Inhibitory effect of sulphated polysaccharide porphyran (isolated from Porphyra yezoensis) on RANKL-induced differentiation of RAW264.7 cells into osteoclasts. Phyther. Res. 2018, 32, 452–458. [Google Scholar] [CrossRef]
- Bito, T.; Teng, F.; Watanabe, F. Bioactive Compounds of Edible Purple Laver Porphyra sp. (Nori). J. Agric. Food Chem. 2017, 65, 10685–10692. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Wang, J.; Wang, S.; Xu, X. Porphyra species: A mini-review of its pharmacological and nutritional properties. J. Med. Food 2016, 19, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Araki, T. Novel β-Agarase, Process for Producing the Same and Use Thereof. U.S. Patent JP386,580,1B2, 27 April 1995. [Google Scholar]
- Sugihara, Y.; Taniguchi, A.; Masatsugu, K.; Kazuhiro, N.; Miho, T.; Takashi, A. Polysaccharide containing composition and tear film stabilizing ophthalmic solution. U.S. Patent KR200,601,211,51A, 10 October 2004. [Google Scholar]
- Bhatia, S.; Sharma, A.; Sharma, K.; Kavale, M.; Chaugule, B.B.; Dhalwal, K.; Namdeo, A.G.; Mahadik, K.R. Novel Algal Polysaccharides from Marine Source: Porphyran. Phcog. Rev. 2008, 2, 271–276. [Google Scholar]
- Kim, J.K.; Yarish, C.; Hwang, E.K.; Park, M.; Kim, Y. Seaweed aquaculture: Cultivation technologies, challenges and its ecosystem services. Algae 2017, 32, 1–13. [Google Scholar] [CrossRef]
- Cheng, T.-H. Production of kelp—A major aspect of China’s exploitation of the sea. Econ. Bot. 1969, 23, 215–236. [Google Scholar] [CrossRef]
- FAO. The State of World Fisheries and Aquaculture—Opportunities and Challenges; FAO: Rome, Italy, 2014. [Google Scholar]
- Bostock, J.; Lane, A.; Hough, C.; Yamamoto, K. An assessment of the economic contribution of EU aquaculture production and the influence of policies for its sustainable development. Aquac. Int. 2016, 24, 699–733. [Google Scholar] [CrossRef]
- Goecke, F.; Klemetsdal, G.; Ergon, Å. Cultivar development of kelps for commercial cultivation—Past lessons and future prospects. Front. Mar. Sci. 2020, 8, 1–17. [Google Scholar] [CrossRef]
- Lehahn, Y.; Nivrutti, K.; Golberg, A. Global potential of offshore and shallow waters macroalgal biorefineries to provide for food, chemicals and energy: Feasibility and sustainability. Algal Res. 2016, 17, 150–160. [Google Scholar] [CrossRef]
- Callaway, R.; Shinn, A.P.; Grenfell, S.E.; Bron, J.E.; Burnell, G.; Cook, E.J.; Crumlish, M.; Culloty, S.; Davidson, K.; Ellis, R.P.; et al. Review of climate change impacts on marine aquaculture in the UK and Ireland. Aquat. Conserv. Mar. Freshw. Ecosyst. 2012, 22, 389–421. [Google Scholar] [CrossRef]
- Hughes, A.D.; Kelly, M.S.; Black, K.D.; Stanley, M.S. Biogas from macroalgae: Is it time to revisit the idea? Biotechnol. Biofuels 2012, 5, 1–7. [Google Scholar] [CrossRef]
- Peteiro, C.; Sánchez, N.; Martínez, B. Mariculture of the Asian kelp Undaria pinnatifida and the native kelp Saccharina latissima along the Atlantic coast of Southern Europe: An overview. Algal Res. 2016, 15, 9–23. [Google Scholar] [CrossRef]
- Stévant, P.; Rebours, C.; Chapman, A. Seaweed aquaculture in Norway: Recent industrial developments and future perspectives. Aquac. Int. 2017, 25, 1373–1390. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Prescott, S.; Potin, P.; Faugeron, S.; Vásquez, J.A.; Camus, C.; Infante, J.; Hernández-gonzález, M.C.; Gutíerrez, A.; Varela, D.A. The status of kelp exploitation and marine agronomy, with emphasis on Macrocystis pyrifera, in Chile. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2014; Volume 71, pp. 161–188. [Google Scholar]
- Camus, C.; Infante, J.; Buschmann, A.H. Overview of 3 year precommercial seafarming of Macrocystis pyrifera along the Chilean coast. Rev. Aquac. 2016, 3, 543–559. [Google Scholar] [CrossRef]
- Pellizzari, F.; Reis, R.P. Seaweed cultivation on the Southern and Southeastern Brazilian Coast. Braz. J. Pharmacogn. 2011, 21, 305–312. [Google Scholar] [CrossRef]
- Augyte, S.; Yarish, C.; Redmond, S.; Kim, J.K. Cultivation of a morphologically distinct strain of the sugar kelp, Saccharina latissima forma angustissima, from coastal Maine, USA, with implications for ecosystem services. J. Appl. Phycol. 2017, 29, 1967–1976. [Google Scholar] [CrossRef]
- Msuya, F.E. The impact of seaweed farming on the socioeconomic status of coastal communities in Zanzibar, Tanzania. World Aquac. 2011, 42, 45–48. [Google Scholar]
- Hafting, J.T.; Craigie, J.S.; Stengel, D.B.; Loureiro, R.R.; Buschmann, A.H.; Yarish, C.; Edwards, M.D.; Critchley, A.T. Prospects and challenges for industrial production of seaweed bioactives. J. Phycol. 2015, 51, 821–837. [Google Scholar] [CrossRef] [PubMed]
- Dawes, C.J.; Orduña-Rojas, J.; Robledo, D. Response of the tropical red seaweed Gracilaria cornea to temperature, salinity and irradiance. J. Appl. Phycol. 1998, 10, 419–425. [Google Scholar] [CrossRef]
- Choi, T.S.; Kang, E.J.; Kim, J.; Kim, K.Y. Effect of salinity on growth and nutrient uptake of Ulva pertusa (Chlorophyta) from an eelgrass bed. Algae 2010, 25, 17–26. [Google Scholar] [CrossRef]
- Guo, H.; Yao, J.; Sun, Z.; Duan, D. Effect of temperature, irradiance on the growth of the green alga Caulerpa lentillifera (Bryopsidophyceae, Chlorophyta). J. Appl. Phycol. 2015, 27, 879–885. [Google Scholar] [CrossRef]
- Campbell, I.; Macleod, A.; Sahlmann, C.; Neves, L.; Funderud, J.; Øverland, M.; Hughes, A.D.; Stanley, M. The environmental risks associated with the development of seaweed farming in Europe-prioritizing key knowledge gaps. Front. Mar. Sci. 2019, 6, 107. [Google Scholar] [CrossRef]
- Cumming, E.E.; Matthews, T.G.; Sanderson, C.J.; Ingram, B.A.; Bellgrove, A. Optimal spawning conditions of Phyllospora comosa (Phaeophyceae, Fucales) for mariculture. J. Appl. Phycol. 2019, 31, 3041–3050. [Google Scholar] [CrossRef]
- Radulovich, R.; Neori, A.; Valderrama, D.; Reddy, C.R.K.; Cronin, H.; Forster, J. Farming of Seaweeds; Elsevier Inc.: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Sudhakar, K.; Mamat, R.; Samykano, M.; Azmi, W.H.; Ishak, W.F.W.; Yusaf, T. An overview of marine macroalgae as bioresource. Renew. Sustain. Energy Rev. 2018, 91, 165–179. [Google Scholar] [CrossRef]
- Craigie, J.S.; Shacklock, P.F. Culture of Irish Moss, 2nd ed.; The Canadian Institute for Research on Regional Development: Moncton, NB, Canada, 1995. [Google Scholar]
- Hafting, J.T.; Critchley, A.T.; Cornish, M.L.; Hubley, S.A.; Archibald, A.F. On-land cultivation of functional seaweed products for human usage. J. Appl. Phycol. 2012, 24, 385–392. [Google Scholar] [CrossRef]
- Currie, M.E. The Growing Sustainable Seaweed Industry: A Comparison of Australian State Governance Directing Current and Future Seaweed Cultivation. Independent Study Project (ISP) Collection. 2018. Available online: http://digitalcollections.sit.edu/isp_collection/2956 (accessed on 3 March 2020).
- Reid, G.K.; Lefebvre, S.; Filgueira, R.; Robinson, S.M.C.; Broch, O.J.; Dumas, A.; Chopin, T.B.R. Performance measures and models for open-water integrated multi-trophic aquaculture. Rev. Aquac. 2020, 12, 47–75. [Google Scholar] [CrossRef]
- Kim, J.K.; Kraemer, G.P.; Yarish, C. Field scale evaluation of seaweed aquaculture as a nutrient bioextraction strategy in Long Island Sound and the Bronx River Estuary. Aquaculture 2014, 433, 148–156. [Google Scholar] [CrossRef]
- Di Trapani, A.M.; Sgroi, F.; Testa, R.; Tudisca, S. Economic comparison between offshore and inshore aquaculture production systems of European sea bass in Italy. Aquaculture 2014, 434, 334–339. [Google Scholar] [CrossRef]
- Buck, B.H.; Troell, M.F.; Krause, G.; Angel, D.L.; Grote, B.; Chopin, T. State of the art and challenges for offshore integrated multi-trophic aquaculture (IMTA). Front. Mar. Sci. 2018, 5, 1–21. [Google Scholar] [CrossRef]
- Rosenthal, H.; Costa-Pierce, B.A.; Krause, G.; Buck, B. Bremerhaven Declaration on the Future of Global Open Ocean. Aquaculture, Part. I: Preamble and Recommendations; Workshop: Open Ocean Aquaculture Development, Aquaculture Forum; The Bremerhaven Economic Development Company: Bremerhaven, Germany, 2012; Available online: http://www.aquaculture-forum.de/fileadmin/pdfs/BremerhavenDeclaration-Part1_07-2012_final.pdf (accessed on 3 March 2020).
- Rosenthal, H.; Costa-Pierce, B.A.; Krause, G.; Buck, B. Bremerhaven Declaration on the Future of Global Open Ocean Aquaculture—Part II: Recommendations on Subject Areas and Justifications; Workshop: Open Ocean Aquaculture Development, Aquaculture Forum; The Bremerhaven Economic Development Company: Bremerhaven, Germany, 2012; Available online: http://www.aquaculture-forum.de/fileadmin/pdfs/BremerhavenDeclaration-Part2_07-2012_final.pdf (accessed on 3 March 2020).
- Bird, K.T. Cost analyses of energy from marine biomass. Dev. Aquac. Fish. Sci. 1987, 16, 327–350. [Google Scholar]
- Buck, B.H.; Buchholz, C.M. The offshore-ring: A new system design for the open ocean aquaculture of macroalgae. J. Appl. Phycol. 2004, 16, 355–368. [Google Scholar] [CrossRef]
- Marine Biomass from Offshore Wind Parks. Available online: http://www.submariner-project.eu/index.php?Option=com_content&view=article&id=159:marine-biomass-from-offshore-wind-parks & catid=62:regionalactivitiesdenmark & Itemid=402 (accessed on 3 March 2020).
- Kirkman, H.; Kendrick, G.A. Ecological significance and commercial harvesting of drifting and beach-cast macro-algae and seagrasses in Australia: A review. J. Appl. Phycol. 1997, 9, 311–326. [Google Scholar] [CrossRef]
- Lenzi, M.; Finoia, M.G.; Persia, E.; Comandi, S.; Gargiulo, V.; Solari, D.; Gennaro, P.; Porrello, S. Biogeochemical effects of disturbance in shallow water sediment by macroalgae harvesting boats. Mar. Pollut. Bull. 2004, 50, 512–519. [Google Scholar] [CrossRef]
- De Góes, H.G.; Reis, R.P. An initial comparison of tubular netting versus tie—Tie methods of cultivation for Kappaphycus alvarezii (Rhodophyta, Solieriaceae) on the south coast of Rio de Janeiro State, Brazil. J. Appl. Phycol. 2011, 23, 607–613. [Google Scholar] [CrossRef]
- Peteiro, C.; Freire, Ó. Outplanting time and methodologies related to mariculture of the edible kelp Undaria pinnatifida in the Atlantic coast of Spain. J. Appl. Phycol. 2012, 24, 1361–1372. [Google Scholar] [CrossRef]
- Peteiro, C.; Sánchez, N.; Dueñas-liaño, C.; Martínez, B. Open-sea cultivation by transplanting young fronds of the kelp Saccharina latissima. J. Appl. Phycol. 2014, 26, 519–528. [Google Scholar] [CrossRef]
- Grote, B. Recent developments in aquaculture of Palmaria palmata (Linnaeus) (Weber & Mohr 1805): Cultivation and uses. Rev. Aquac. 2019, 11, 25–41. [Google Scholar]
- Troell, M.; Joyce, A.; Chopin, T.; Neori, A.; Buschmann, A.H.; Fang, J.G. Ecological engineering in aquaculture—Potential for integrated multi-trophic aquaculture (IMTA) in marine offshore systems. Aquaculture 2009, 297, 1–9. [Google Scholar] [CrossRef]
- Troell, M.; Hecht, T.; Beveridge, M.; Stead, S.; Bryceson, I.; Kautsky, N.; Mmochi, A.; Ollevier, F. Mariculture in the WIO Region: Challenges and Prospects; WIOMSA Book Series: Zanzibar, Tanzania, 2009; pp. 8–59. Available online: https://www.wiomsa.org/download/book_series/mariculture_publication_final.pdf (accessed on 10 March 2020).
- Harrison, P.J.; Hurd, C.L. Nutrient physiology of seaweeds: Application of concepts to aquaculture. Cah. Biol. Mar. 2001, 42, 71–82. [Google Scholar]
- Handå, A.; Forbord, S.; Wang, X.; Broch, O.J.; Dahle, S.W.; Størseth, T.R.; Reitan, K.I.; Olsen, Y.; Skjermo, J. Seasonal- and depth-dependent growth of cultivated kelp (Saccharina latissima) in close proximity to salmon (Salmo salar) aquaculture in Norway. Aquaculture 2013, 414–415, 191–201. [Google Scholar] [CrossRef]
- Fletcher, R.L. Epiphytism and fouling in Gracilaria cultivation: An overview. J. Appl. Phycol. 1995, 7, 325–333. [Google Scholar] [CrossRef]
- Vairappan, C.S.; Chung, C.S.; Hurtado, A.Q.; Soya, F.E.; Lhonneur, G.B.; Critchley, A. Distribution and symptoms of epiphyte infection in major carrageenophyte-producing farms. J. Appl. Phycol. 2008, 20, 477–483. [Google Scholar] [CrossRef]
- Troell, M.; Jonell, M.; John, P.; Henriksson, G. Ocean space for seafood. Nat. Ecol. Evol. 2017, 1, 1224–1225. [Google Scholar] [CrossRef]
- Oyinlola, M.A.; Reygondeau, G.; Wabnitz, C.C.C.; Troell, M.; Cheung, W.W.L. Global estimation of areas with suitable environmental conditions for mariculture species. PLoS ONE 2018, 13, 1–19. [Google Scholar] [CrossRef]
- Polk, M. Open Ocean Aquaculture. In Proceedings of the International Conference on Open Ocean Aquaculture, Portland, OR, USA, 8–10 May 1996; p. 640. [Google Scholar]
- Hesley, C. Open Ocean. Aquaculture ’97 Charting the Future of Ocean. Farming: Proceedings of the International Conference, April 23–25, 1997, Maui, Hawaii; University of Hawaii Sea Grant College Program: Maui, HI, USA, 1997; p. 353. [Google Scholar]
- Stickney, R.R. Joining forces with industry. In Proceedings of the Third International Conference on Open Ocean Aquaculture, Corpus Christi, TX, USA, 10–15 May 1999. [Google Scholar]
- Bridger, C.J.; Costa-Pierce, B.A. Open Ocean. Aquaculture: From Research to Commercial Reality; Baton Rouge, L.W.A.S., Ed.; Academic Press: New York, NY, USA, 2003. [Google Scholar]
- Ferreira, J.G.; Sequeira, A.; Hawkins, A.J.S.; Newton, A.; Nickell, T.D.; Pastres, R.; Forte, J.; Bodoy, A.; Bricker, S.B. Analysis of coastal and offshore aquaculture: Application of the FARM model to multiple systems and shellfish species. Aquaculture 2009, 292, 129–138. [Google Scholar] [CrossRef]
- Chopin, T.; Robinson, S.; Reid, G.; Ridler, N. Prospects for Integrated Multi-Trophic Aquaculture (IMTA) in the open ocean. Bull. Aquac. Assoc. 2013, 111, 28–35. [Google Scholar]
- Jansen, H.M.; Van Den Burg, S.; Bolman, B.; Jak, R.G.; Kamermans, P.; Poelman, M.; Stuiver, M. The feasibility of offshore aquaculture and its potential for multi-use in the North Sea. Aquac. Int. 2016, 24, 735–756. [Google Scholar] [CrossRef]
- Buck, B.H.; Langan, R. Aquaculture Perspective of Multi-Use Sites in the Open Ocean.: The Untapped Potential for Marine Resources in the Anthropocene; Springer: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Valderrama, D.; Cai, J.; Hishamunda, N.; Ridler, N. Social and Economic Dimensions of Carrageenan Seaweed Farming; FAO: Rome, Italy, 2013. [Google Scholar]
- Bak, U.G.; Gregersen, Ó.; Infante, J. Technical challenges for offshore cultivation of kelp species: Lessons learned and future directions. Bot. Mar. 2020, 63, 341–353. [Google Scholar] [CrossRef]
- Soto, D.; Wurmann, C. Offshore Aquaculture: A Needed New Frontier for Farmed Fish at Sea. In The Future of Ocean Governance and Capacity Development; Brill|Nijhoff: Leiden, The Netherlands, 2019; pp. 379–384. [Google Scholar]
- Zheng, Y.; Jin, R.; Zhang, X.; Wang, Q.; Wu, J. The considerable environmental benefits of seaweed aquaculture in China. Stoch. Environ. Res. Risk Assess. 2019, 33, 1203–1221. [Google Scholar] [CrossRef]
- Chávez-Crooker, P.; Obreque-Contreras, J. Bioremediation of aquaculture wastes. Curr. Opin. Biotechnol. 2010, 21, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Granada, L.; Sousa, N.; Lopes, S.; Lemos, M.F.L. Is integrated multitrophic aquaculture the solution to the sectors’ major challenges?—A review. Rev. Aquac. 2016, 8, 283–300. [Google Scholar] [CrossRef]
- Neori, A.; Chopin, T.; Troell, M.; Buschmann, A.H.; Kraemer, G.P.; Halling, C.; Shpigel, M.; Yarish, C. Integrated aquaculture: Rationale, evolution and state of the art emphasizing seaweed biofiltration in modern mariculture. Aquaculture 2004, 231, 361–391. [Google Scholar] [CrossRef]
- Samocha, T.M.; Fricker, J.; Ali, A.M.; Shpigel, M.; Neori, A. Growth and Nutrient Uptake of the Macroalga Gracilaria Tikvahiae Cultured with the Shrimp Litopenaeus Vannamei in an Integrated Multi-Trophic Aquaculture (IMTA) System; Elsevier: Amsterdam, The Netherlands, 2015; Volume 446. [Google Scholar]
- Tanaka, Y.; Ashaari, A.; Mohamad, F.S.; Lamit, N. Bioremediation potential of tropical seaweeds in aquaculture: Low-salinity tolerance, phosphorus content, and production of UV-absorbing compounds. Aquaculture 2020, 518, 734853. [Google Scholar] [CrossRef]
- Hernández, I.; Fernández-Engo, M.A.; Pérez-Lloréns, J.L.; Vergara, J.J. Integrated outdoor culture of two estuarine macroalgae as biofilters for dissolved nutrients from Sparus aurata waste waters. J. Appl. Phycol. 2005, 17, 557–567. [Google Scholar] [CrossRef]
- Buschmann, A.; Varela, D.; Hernández-González, M.C.; Huovinen, P. Opportunities and challenges for the development of an integrated seaweed-based aquaculture activity in Chile: Determining the physiological capabilities of Macrocystis and Gracilaria as biofilters. J. Appl. Phycol. 2008, 20, 571–577. [Google Scholar] [CrossRef]
- Sanderson, J.C.; Dring, M.J.; Davidson, K.; Kelly, M.S. Culture, yield and bioremediation potential of Palmaria palmata (Linnaeus) Weber & Mohr and Saccharina latissima (Linnaeus) C.E. Lane, C. Mayes, Druehl & G.W. Saunders adjacent to fish farm cages in northwest Scotland. Aquaculture 2012, 354–355, 128–135. [Google Scholar]
- Hadley, S.; Wild-Allen, K.; Johnson, C.; Macleod, C. Modeling macroalgae growth and nutrient dynamics for integrated multi-trophic aquaculture. J. Appl. Phycol. 2015, 27, 901–916. [Google Scholar] [CrossRef]
- Redmond, S.; Green, L.; Yarish, C.; Kim, J.; Neefus, C. New England Nursery Systems. Connecicut, Sea Grant CTSG-14-01. Available online: https://www.researchgate.net/publication/311561368_New_England_Seaweed_Culture_Handbook (accessed on 19 August 2020).
- Hadley, S.A. Farming Macroalgae to Mitigate Coastal Nutrification from Finfish Aquaculture: A Modelling Study. Ph.D. Thesis, University of Tasmania, Hobart, Australia, 2015; p. 164. [Google Scholar]
- Michalak, I.; Chojnacka, K. Algal extracts: Technology and advances. Eng. Life Sci. 2014, 14, 581–591. [Google Scholar] [CrossRef]
- Pereira, L.; Bahcevandziev, K.; Joshi, N.H. Seaweeds as Plant. Fertilizer, Agricultural Biostimulants and Animal Fodder; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Li, X.; Norman, H.C.; Kinley, R.; Laurence, M.; Wilmot, M.; Bender, H.; de Nys, R.; Tomkins, N. Asparagopsis taxiformis decreases enteric methane production from sheep. Anim. Prod. Sci. 2016, 58. [Google Scholar] [CrossRef]
- Troell, M.; Halling, C.; Neori, A.; Chopin, T.; Buschmann, A.H.; Kautsky, N.; Yarish, C. Integrated mariculture: Asking the right questions. Aquaculture 2003, 226, 69–90. [Google Scholar] [CrossRef]
- Hossain, A.B.M.S.; Salleh, A.; Boyce, A.N.; Chowdhury, P.; Naqiuddin, M. Biodiesel fuel production from algae as renewable energy. Am. J. Biochem. Biotechnol. 2008, 4, 250–254. [Google Scholar] [CrossRef]
- Duarte, C.M.; Losada, I.J.; Hendriks, I.E.; Mazarrasa, I.; Marbà, N. The role of coastal plant communities for climate change mitigation and adaptation. Nat. Clim. Chang. 2013, 3, 961–968. [Google Scholar] [CrossRef]
- Feng, Y.Y.; Hou, L.C.; Ping, N.X.; Ling, T.D.; Kyo, C.I. Development of mariculture and its impacts in Chinese coastal waters. Rev. Fish. Biol. Fish. 2004, 14, 1–10. [Google Scholar] [CrossRef]
- Chopin, T.; Buschmann, A.; Hulling, C.; Troell, M.; Kautsky, N.; Neori, A.; Kraemer, G.; Zertuche-González, J.A.; Yarish, C.; Neefus, C. Integrating seaweeds into marine aquaculture systems: A key toward sustainability. J. Phycol. 2001, 37, 975–986. [Google Scholar] [CrossRef]
- FAO. State of World Aquaculture; FAO: Rome, Italy, 2006. [Google Scholar]
- Buchholz, C.M.; Krause, G.; Buck, B.H. Seaweed and Man. In Seaweed Biology; Wiencke, C., Bischof, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2012; Volume 219, pp. 471–493. [Google Scholar]
- Chopin, T.; Robinson, S.M.C.; Troell, M.; Neori, A.; Buschmann, A.H.; Fang, J. Multitrophic integration for sustainable marine aquaculture. In Encyclopedia of Ecology; Elsevier: Amsterdam, The Netherlands, 2008; pp. 2463–2475. [Google Scholar]
- Allan, G.L.; Fielder, D.S.; Fitzsimmons, K.M.; Applebaum, S.L.; Raizada, S. Inland Saline Aquaculture; Woodhead Publishing Limited: Cambridge, UK, 2009. [Google Scholar]
- Thi, H.; Bui, T. Technical Feasibility of Cultivating Local Seaweed Species in Inland Saline Water of Western Australia. Ph.D. Thesis, Curtin University, Perth, Australia, 2018. [Google Scholar]
- Bracken, M.E.S.; Stachowicz, J.J. Seaweed diversity enhances nitrogen uptake via complementary use of nitrate and ammonium. Ecology 2006, 87, 2397–2403. [Google Scholar] [CrossRef]
- Lüning, K.; Pang, S. Mass cultivation of seaweeds: Current aspects and approaches. J. Appl. Phycol. 2003, 15, 115–119. [Google Scholar] [CrossRef]
- Mumford, T.F.; Miura, A. Porphyra as food: Cultivation and economics. In Algae and Human Affairs; Lembi, C.A., Waaland, J.R., Eds.; Cambridge University Press: London, UK, 1988; pp. 87–117. [Google Scholar]
- Pereira, R.; Yarish, C. Mass Production of Marine Macroalgae. Encycl. Ecol. 2008, 5, 2236–2247. [Google Scholar]
- Guiry, M.D.; Guiry, G.M. AlgaeBase. Available online: http://www.algaebase.org (accessed on 7 March 2020).
- He, P.; Yarish, C. The developmental regulation of mass cultures of free-living conchocelis for commercial net seeding of Porphyra leucosticta from Northeast America. Aquaculture 2006, 257, 373–381. [Google Scholar] [CrossRef]
- He, P.; Xu, S.; Zhang, H.; Wen, S.; Dai, Y.; Lin, S.; Yarish, C. Bioremediation efficiency in the removal of dissolved inorganic nutrients by the red seaweed, Porphyra yezoensis, cultivated in the open sea. Water Res. 2008, 42, 1281–1289. [Google Scholar] [CrossRef] [PubMed]
- Peteiro, C. Alginate Production from Marine Macroalgae, with Emphasis on Kelp Farming. In Alginates and Their Biomedical Applications; Springer: Singapore, 2018; pp. 27–66. [Google Scholar]
- Andersen, R.A. Algal Culturing Techniques; Andersen, R.A., Ed.; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Pereira, R.; Yarish, C.; Critchley, A. Seaweed Aquaculture for Human Foods in Land-Based and IMTA Systems. In Sustainable Food Production; Christou, P., Savin, R., Costa-Pierce, B.A., Misztal, I., Whitelaw, C.B.A., Eds.; Springer: New York, NY, USA, 2013; Volume 15. [Google Scholar]
- Kim, J.K.; Lindell, S.; Green-Beach, E.; Peach, M.; Beals, M.; Yarish, C. Multi-cropping seaweed Gracilaria tikvahiae with oysters for nutrient bioextraction and sea vegeta bles in Waquoit Bay, Massachusetts. Phycologia 2013, 52, 53. [Google Scholar]
- Friedlander, M. Advances in cultivation of Gelidiales. J. Appl. Phycol. 2007, 20, 1–6. [Google Scholar]
- Qi, Z.; Liu, H.; Li, B.; Mao, Y.; Jiang, Z.; Zhang, J.; Fang, J. Suitability of two seaweeds, Gracilaria lemaneiformis and Sargassum pallidum, as feed for the abalone Haliotis discus hannai Ino. Aquaculture 2010, 300, 189–193. [Google Scholar] [CrossRef]
- Johnson, R.B.; Kim, J.K.; Armbruster, L.C.; Yarish, C. Nitrogen allocation of Gracilaria tikvahiae grown in urbanized estuaries of Long Island Sound and New York City, USA: A preliminary evaluation of ocean farmed Gracilaria for alternative fish feeds. Algae 2014, 29, 227–235. [Google Scholar] [CrossRef]
- Kim, J.K.; Mao, Y.; Kraemer, G.; Yarish, C. Growth and pigment content of Gracilaria tikvahiae McLachlan under fluorescent and LED lighting. Aquaculture 2015, 436, 52–57. [Google Scholar] [CrossRef]
- Kim, J.K.; Yarish, C.; Pereira, R. Tolerances to hypo-osmotic and temperature stresses in native and invasive species of Gracilaria (Rhodophyta). Phycologia 2016, 55, 257–264. [Google Scholar] [CrossRef]
- Wu, H.; Huo, Y.; Han, F.; Liu, Y.; He, P. Bioremediation using Gracilaria chouae co-cultured with Sparus macrocephalus to manage the nitrogen and phosphorous balance in an IMTA system in Xiangshan Bay, China. Mar. Pollut. Bull. 2015, 91, 272–279. [Google Scholar] [CrossRef]
- Gorman, L.; Kraemer, G.P.; Yarish, C.; Boo, S.M.; Kim, J.K. The effects of temperature on the growth rate and nitrogen content of invasive Gracilaria vermiculophylla and native Gracilaria tikvahiae from Long Island Sound, USA. Algae 2017, 32, 57–66. [Google Scholar] [CrossRef]
- Yokoya, N.S.; Kakita, H.; Obika, H.; Kitamura, T. Effects of environmental factors and plant growth regulators on growth of the red alga Gracilaria vermiculophylla from Shikoku Island, Japan. Hydrobiologia 1999, 398–399, 339–347. [Google Scholar] [CrossRef]
- Weinberger, F.; Buchholz, B.; Karez, R.; Wahl, M. The invasive red alga Gracilaria vermiculophylla in the Baltic Sea: Adaptation to brackish water may compensate for light limitation. Aquat. Biol. 2008, 3, 251–264. [Google Scholar] [CrossRef]
- Raikar, S.V.; Iima, M.; Fujita, Y. Effect of temperature, salinity and light intensity on the growth of Gracilaria spp. (Gracilariales, Rhodophyta) from Japan, Malaysia and India. Indian J. Mar. Sci. 2001, 30, 98–104. [Google Scholar]
- Oliveira, E.C.; Alveal, K.; Anderson, R.J. Mariculture of the agar-producing Gracilarioid red algae. Rev. Fish. Sci. 2000, 8, 345–377. [Google Scholar] [CrossRef]
- Alamsjah, M.A. Producing new variety of Gracilaria sp. through cross breeding. Res. J. Fish. Hydrobiol. 2010, 5, 159–167. [Google Scholar]
- Ask, E.I.; Azanza, R.V. Advances in cultivation technology of commercial eucheumatoid species: A review with suggestions for future research. Aquaculture 2002, 206, 257–277. [Google Scholar] [CrossRef]
- Hayashi, L.; Hurtado, A.Q.; Msuya, F.E.; Bleicher-Lhonneur, G.; Critchley, A.T. A review of Kappaphycus farming: Prospects and constraints. In Seaweed and Their Role in Globally Changing Environment; Israel, A., Einav, R., Seckbach, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Hurtado, A.Q.; Critchley, A.T.; Trespoey, A.; Lhonneur, G.B. Occurrence of Polysiphonia epiphytes in Kappaphycus farms at Calaguas Is., Camarines Norte, Phillippines. J. Appl. Phycol. 2006, 18, 301–306. [Google Scholar] [CrossRef]
- Hwang, E.K.; Gong, Y.G.; Hwang, I.K.; Park, E.J.; Park, C.S. Cultivation of the two perennial brown algae Ecklonia cava and E. stolonifera for abalone feeds in Korea. J. Appl. Phycol. 2013, 25, 825–829. [Google Scholar] [CrossRef]
- Kawashima, Y.; Tokuda, H. Regeneration from callus of Undaria pinnatifida (Harvey) Suringar (Laminariales, Phaeophyta). Hydrobiologia 1993, 260–261, 385–389. [Google Scholar] [CrossRef]
- Li, X.; Cong, Y.; Yang, G.; Shi, Y.; Qu, S.; Li, Z.; Wang, G.; Zhang, Z.; Luo, S.; Dai, H.; et al. Trait evaluation and trial cultivation of Dongfang No. 2, the hybrid of a male gametophyte clone of Laminaria longissima (Laminariales, Phaeophyta) and a female one of L. japonica. J. Appl. Phycol. 2007, 19, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.; Winberg, P.; Kirkendale, L. Genetic improvement of macroalgae: Status to date and needs for the future. J. Appl. Phycol. 2013, 25, 703–716. [Google Scholar] [CrossRef]
- Sohn, C.H. The seaweed resources of Korea. In Seaweed Resources of the World; Critchley, A.T., Ohno, M., Eds.; Japan International Cooperation Agency: Yokosuka, Japan, 1998; pp. 15–33. [Google Scholar]
- Hwang, E.-K.; Ha, D.-S.; Baek, J.-M.; Wee, M.-Y.; Park, C.-S. Effects of pH and Salinity on the Cultivated Brown Alga Sargassumfulvellum and Associated Animals. Algae 2006, 21, 317–321. [Google Scholar] [CrossRef][Green Version]
- Redmond, S.; Kim, J.K.; Yarish, C.; Pietrak, M.; Bricknell, I. Culture of Sargassum in Korea: Techniques and Potential for Culture in the U.S.; Maine Sea Grant: Orono, ME, USA, 2014. [Google Scholar]
- Sulaiman, O.O.; Magee, A.; Nik, W.B.W.; Saharuddin, A.H.; Kader, A.S.A. Design and Model Testing of Offshore Acquaculture Floating Structure for Seaweed Oceanic Plantation. Biosci. Biotechnol. Res. Asia 2012, 9, 477–494. [Google Scholar] [CrossRef]
- Shan, T.F.; Pang, S.J.; Li, J.; Gao, S.Q. Breeding of an elite cultivar Haibao no. 1 of Undaria pinnatifida (phaeophyceae) through gametophyte clone crossing and consecutive selection. J. Appl. Phycol. 2016, 28, 2419–2426. [Google Scholar] [CrossRef]
- Zhao, X.B.; Pang, S.J.; Liu, F.; Shan, T.F.; Li, J.; Gao, S.Q.; Kim, H.G. Intraspecific crossing of Saccharina japonica using distantly related unialgal gametophytes benefits kelp farming by improving blade quality and productivity at Sanggou Bay, China. J. Appl. Phycol. 2016, 28, 449–455. [Google Scholar] [CrossRef]
- Correa, T.; Gutiérrez, A.; Flores, R.; Buschmann, A.H.; Cornejo, P.; Bucarey, C. Production and economic assessment of giant kelp Macrocystis pyrifera cultivation for abalone feed in the south of Chile. Aquac. Res. 2016, 47, 698–707. [Google Scholar] [CrossRef]
- Zuniga-Jara, S.; Marín-Riffo, M.C.; Bulboa-Contador, C. Bioeconomic analysis of giant kelp Macrocystis pyrifera cultivation (Laminariales; Phaeophyceae) in northern Chile. J. Appl. Phycol. 2016, 28, 405–416. [Google Scholar] [CrossRef]
- Beauchamp, E. Effects of UV radiation and salinity on the intertidal macroalgae Palmaria palmata and Ulva lactuca; effects on photosynthetic performance, growth and pigments. Plymouth Stud. Sci. 2012, 5, 3–22. [Google Scholar]
- Cotas, J.; Figueirinha, A.; Pereira, L.; Batista, T. The effect of salinity on Fucus ceranoides (Ochrophyta, Phaeophyceae) in the Mondego River (Portugal). J. Oceanol. Limnol. 2019, 37, 881–891. [Google Scholar] [CrossRef]
- Suthar, P.; Gajaria, T.K.; Reddy, C.R.K. Production of quality seaweed biomass through nutrient optimization for the sustainable land-based cultivation. Algal Res. 2019, 42, 101583. [Google Scholar] [CrossRef]
- Pliego-Cortés, H.; Bedoux, G.; Boulho, R.; Taupin, L.; Freile-Pelegrín, Y.; Bourgougnon, N.; Robledo, D. Stress tolerance and photoadaptation to solar radiation in Rhodymenia pseudopalmata (Rhodophyta) through mycosporine-like amino acids, phenolic compounds, and pigments in an Integrated Multi-Trophic Aquaculture system. Algal Res. 2019, 41, 101542. [Google Scholar] [CrossRef]
- Zepeda, E.; Freile-Pelegrín, Y.; Robledo, D. Nutraceutical assessment of Solieria filiformis and Gracilaria cornea (Rhodophyta) under light quality modulation in culture. J. Appl. Phycol. 2020. [Google Scholar] [CrossRef]
- Aquaculture 4.0: Applying Industry Strategy to Fisheries Management. Available online: https://www.governmenteuropa.eu/aquaculture-4-0/93038/ (accessed on 6 March 2020).
- Santisteban, A.; Piquero, A.; Ortiz, F.; Delgado, F.; Ortiz, A. Thermal Modelling of a Power Transformer Disc Type Winding Immersed in Mineral and Ester-Based Oils Using Network Models and CFD. IEEE Access 2019, 7, 174651–174661. [Google Scholar] [CrossRef]
- Cotas, C.I.P. Modelling of Fiber Suspensions Flow in Pipes. Ph.D. Thesis, University of Coimbra, Coimbra, Portugal, 2015. [Google Scholar]
- Bitog, J.P.; Lee, I.B.; Lee, C.G.; Kim, K.S.; Hwang, H.S.; Hong, S.W.; Seo, I.H.; Kwon, K.S.; Mostafa, E. Application of computational fluid dynamics for modeling and designing photobioreactors for microalgae production: A review. Comput. Electron. Agric. 2011, 76, 131–147. [Google Scholar] [CrossRef]
- Neori, A. Essential role of seaweed cultivation in integrated multi-trophic aquaculture farms for global expansion of mariculture: An analysis. J. Appl. Phycol. 2008, 20, 567–570. [Google Scholar] [CrossRef]
- Behroozi, L.; Couturier, M.F. Prediction of water velocities in circular aquaculture tanks using an axisymmetric CFD model. Aquac. Eng. 2019, 85, 114–128. [Google Scholar] [CrossRef]
- Labatut, R.A.; Ebeling, J.M.; Bhaskaran, R.; Timmons, M.B. Exploring flow discharge strategies of a mixed-cell raceway (MCR) using 2-D computational fluid dynamics (CFD). Aquac. Eng. 2015, 66, 68–77. [Google Scholar] [CrossRef]
- Gorle, J.M.R.; Terjesen, B.F.; Summerfelt, S.T. Hydrodynamics of octagonal culture tanks with Cornell-type dual-drain system. Comput. Electron. Agric. 2018, 151, 354–364. [Google Scholar] [CrossRef]
- Olanrewaju, S.O.; Magee, A.; Kader, A.S.A.; Tee, K.F. Simulation of offshore aquaculture system for macro algae (seaweed) oceanic farming. Ships Offshore Struct. 2017, 12, 553–562. [Google Scholar] [CrossRef]
- Sato, K.; Eksangsri, T.; Egashira, R. Ammonia-Nitrogen Uptake by Seaweed for Water Quality Control in Intensive Mariculture Ponds. J. Chem. Eng. 2006, 39, 247–255. [Google Scholar] [CrossRef]
- Troell, M.; Rönnbäck, P.; Halling, C.; Kautsky, N.; Buschmann, A. Ecological engineering in aquaculture: Use of seaweeds for removing nutrients from intensive mariculture. In Sixteenth International Seaweed Symposium; Springer: Amsterdam, The Netherlands, 1999; pp. 603–611. [Google Scholar]
- Xing, Q.; An, D.; Zheng, X.; Wei, Z.; Wang, X.; Li, L.; Tian, L.; Chen, J. Monitoring seaweed aquaculture in the Yellow Sea with multiple sensors for managing the disaster of macroalgal blooms. Remote Sens. Environ. 2019, 231, 111279. [Google Scholar] [CrossRef]
- Mantri, V.A.; Ashok, K.S.; Saminathan, K.R.; Rajasankar, J.; Harikrishna, P. Concept of triangular raft design: Achieving higher yield in Gracilaria edulis. Aquac. Eng. 2015, 69, 1–6. [Google Scholar] [CrossRef]
- Azevedo, I.C.; Duarte, P.M.; Marinho, G.S.; Neumann, F.; Sousa-Pinto, I. Growth of Saccharina latissima (Laminariales, Phaeophyceae) cultivated offshore under exposed conditions. Phycologia 2019, 58, 504–515. [Google Scholar] [CrossRef]
- Davison, A.V.; Piedrahita, R.H. Temperature modeling of a land-based aquaculture system for the production of Gracilaria pacifica: Possible system modifications to conserve heat and extend the growing season. Aquac. Eng. 2015, 66, 1–10. [Google Scholar] [CrossRef]
- Valero, M.; Guillemin, M.-L.; Destombe, C.; Jacquemin, B.; Gachon, C.M.M.; Badis, Y.; Buschmann, A.H.; Camus, C.; Faugeron, S. Perspectives on domestication research for sustainable seaweed aquaculture. Perspect. Phycol. 2017, 4, 33–46. [Google Scholar] [CrossRef]
- Myles, S.; Boyko, A.R.; Owens, C.L.; Brown, P.J.; Grassi, F.; Aradhya, M.K.; Prins, B.; Reynolds, A.; Chia, J.M.; Ware, D.; et al. Genetic structure and domestication history of the grape. Proc. Natl. Acad. Sci. USA 2011, 108, 3530–3535. [Google Scholar] [CrossRef]
- Chen, T.T.; Lin, C.M.; Chen, M.J.; Lo, J.H.; Chiou, P.P.; Gong, H.Y.; Wu, J.L.; Chen, M.H.C.; Yarish, C. Transgenic technology in marine organisms. In Springer Handbook of Marine Biotechnology; Springer: Berlin/Heidelberg, Germany, 2015; pp. 387–412. [Google Scholar]
- Patwary, M.U.; van der Meer, J.P. Genetics and breeding of cultivated seaweeds. Korean J. Phycol. 1992, 7, 281–318. [Google Scholar]
- Gangl, D.; Zedler, J.A.Z.; Rajakumar, P.D.; Martinez, E.M.R.; Riseley, A.; Włodarczyk, A.; Purton, S.; Sakuragi, Y.; Howe, C.J.; Jensen, P.E.; et al. Biotechnological exploitation of microalgae. J. Exp. Bot. 2015, 66, 6975–6990. [Google Scholar] [CrossRef]
- Henley, W.J.; Litaker, R.W.; Novoveská, L.; Duke, C.S.; Quemada, H.D.; Sayre, R.T. Initial risk assessment of genetically modified (GM) microalgae for commodity-scale biofuel cultivation. Algal Res. 2012, 2, 66–77. [Google Scholar] [CrossRef]
- Nobre, A.M.; Musango, J.K.; de Wit, M.P.; Ferreira, J.G. A dynamic ecological-economic modeling approach for aquaculture management. Ecol. Econ. 2009, 68, 3007–3017. [Google Scholar] [CrossRef]
- Sustainable European Aquaculture 4.0: Nutrition and Breeding. Available online: https://cordis.europa.eu/programme/id/H2020_DT-BG-04-2018-2019 (accessed on 6 March 2020).
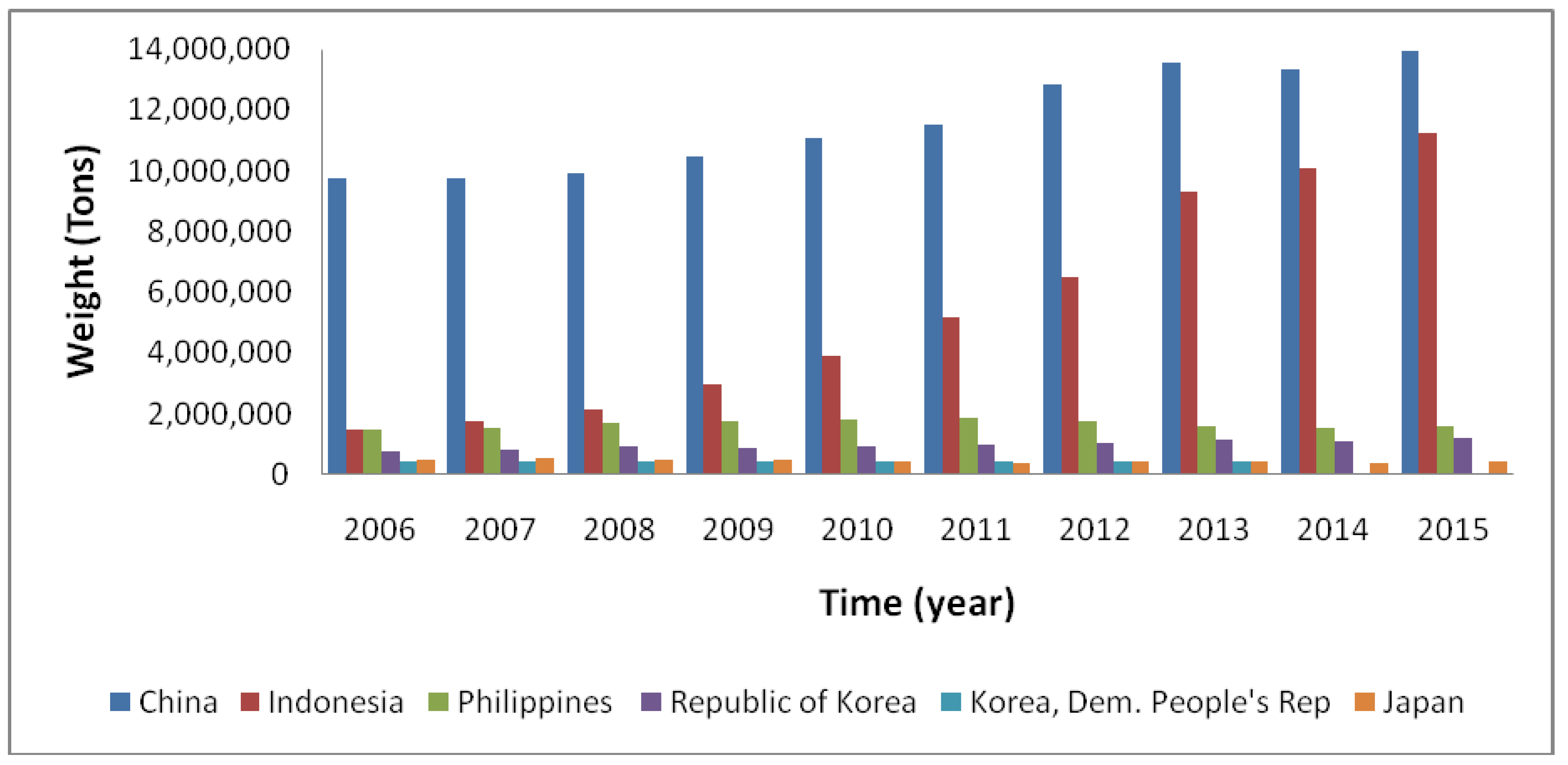
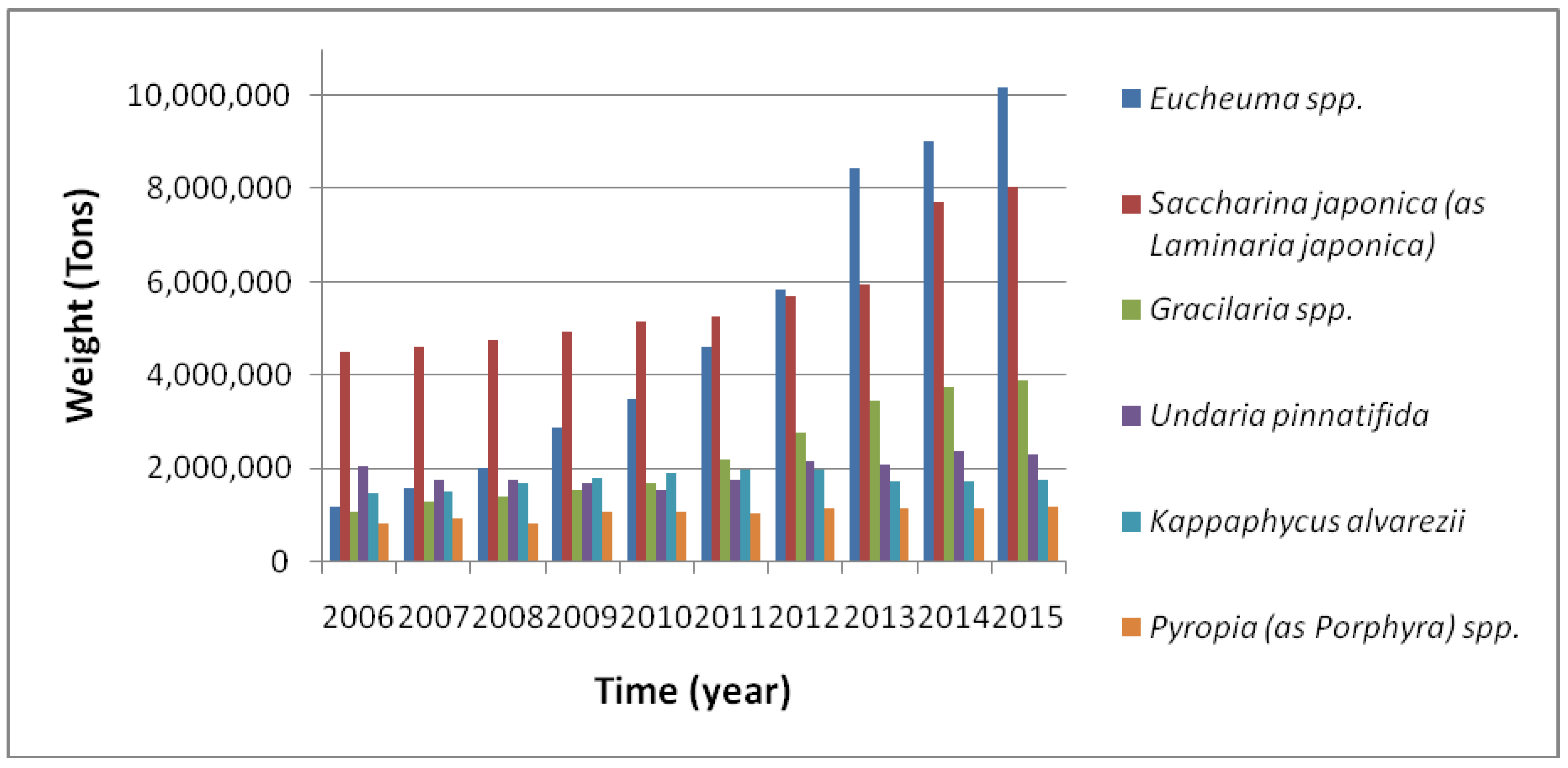
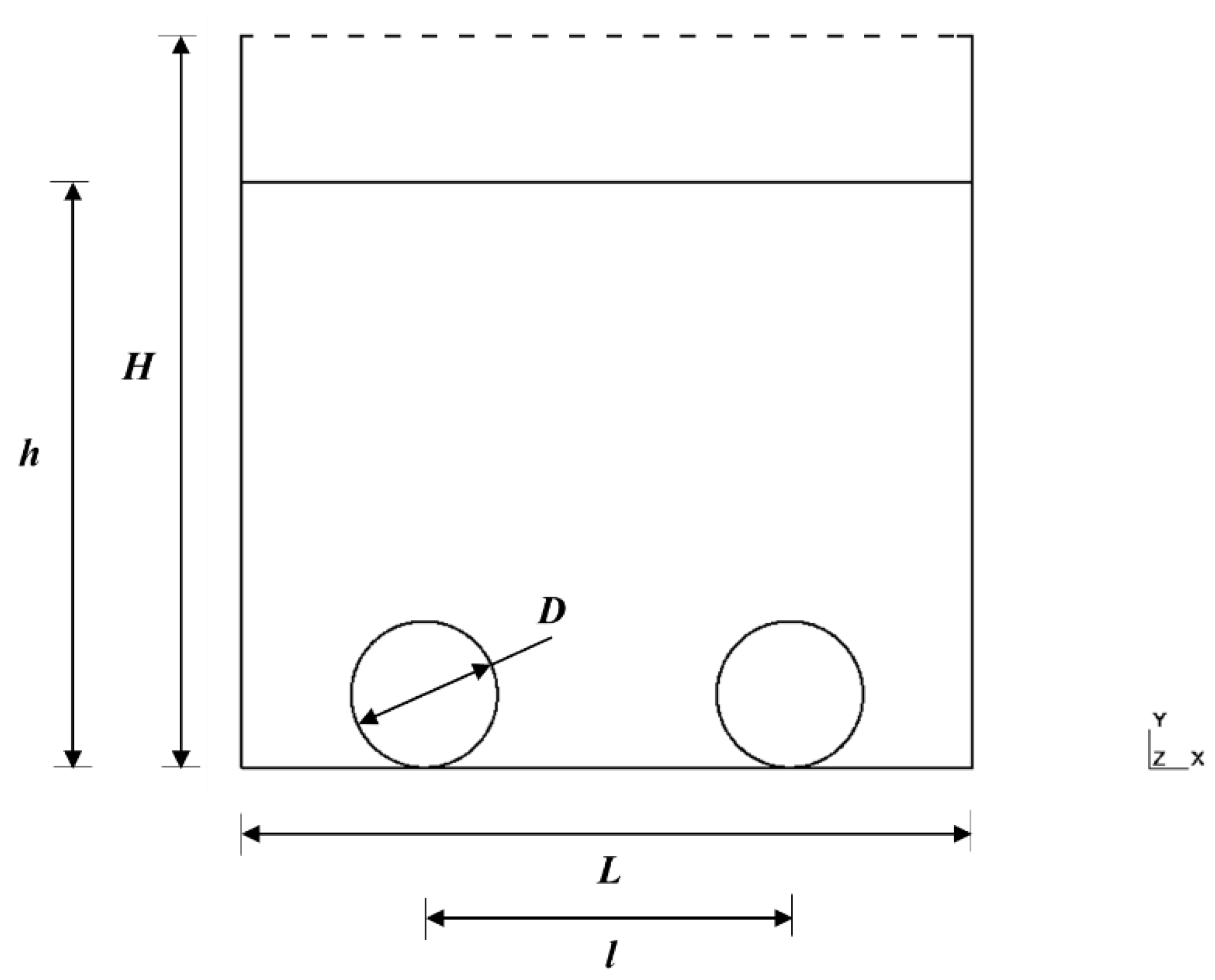
| Onshore Methods | Offshore Methods | |
|---|---|---|
| Line cultivation: -Off-bottom -Submerged hanging line -Floating line (long-line) | X | X |
| Net cultivation (depth, floating at the surface or slightly submerged) | X | X |
| Floating raft cultivation | X | X |
| Tank or pond cultivation | X | |
| Rock-based farming—direct planting on the ocean bottom or attached to artificial substrate | X | |
| Onshore and offshore seaweed cultivation methods are identified by the c‘olor/shadow and a cross (X). | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Poza, S.; Leandro, A.; Cotas, C.; Cotas, J.; Marques, J.C.; Pereira, L.; Gonçalves, A.M.M. The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. Int. J. Environ. Res. Public Health 2020, 17, 6528. https://doi.org/10.3390/ijerph17186528
García-Poza S, Leandro A, Cotas C, Cotas J, Marques JC, Pereira L, Gonçalves AMM. The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. International Journal of Environmental Research and Public Health. 2020; 17(18):6528. https://doi.org/10.3390/ijerph17186528
Chicago/Turabian StyleGarcía-Poza, Sara, Adriana Leandro, Carla Cotas, João Cotas, João C. Marques, Leonel Pereira, and Ana M. M. Gonçalves. 2020. "The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0" International Journal of Environmental Research and Public Health 17, no. 18: 6528. https://doi.org/10.3390/ijerph17186528
APA StyleGarcía-Poza, S., Leandro, A., Cotas, C., Cotas, J., Marques, J. C., Pereira, L., & Gonçalves, A. M. M. (2020). The Evolution Road of Seaweed Aquaculture: Cultivation Technologies and the Industry 4.0. International Journal of Environmental Research and Public Health, 17(18), 6528. https://doi.org/10.3390/ijerph17186528










