Antimicrobial Efficacy of Fruit Peels Eco-Enzyme against Enterococcus faecalis: An In Vitro Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Eco-Enzyme Extracts
2.2. Bacterial Strain
2.3. Determination of Minimal Inhibitory Concentration (MIC)
2.4. Determination of Minimal Bactericidal Concentration (MBC)
2.5. Statistical Analysis
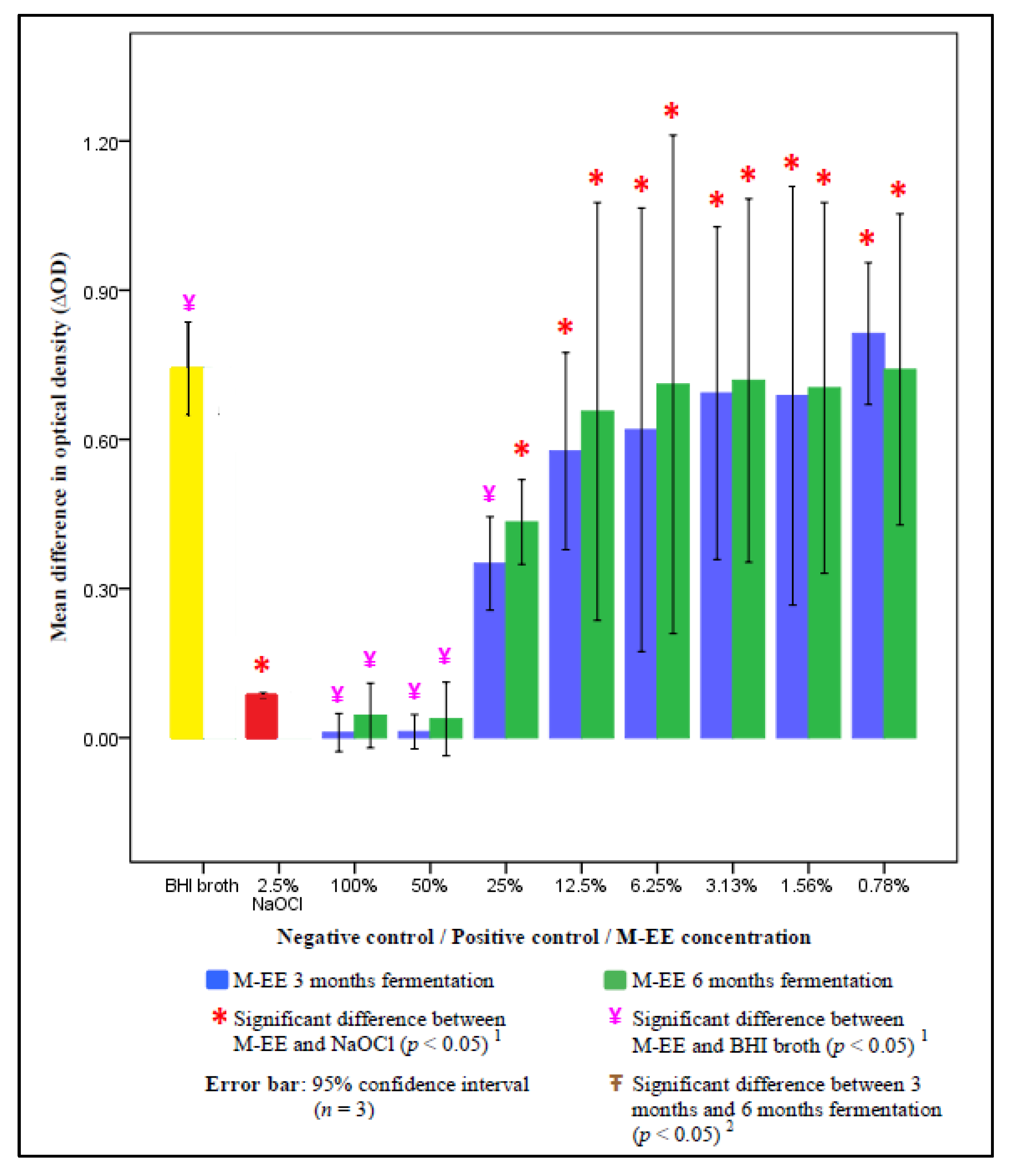
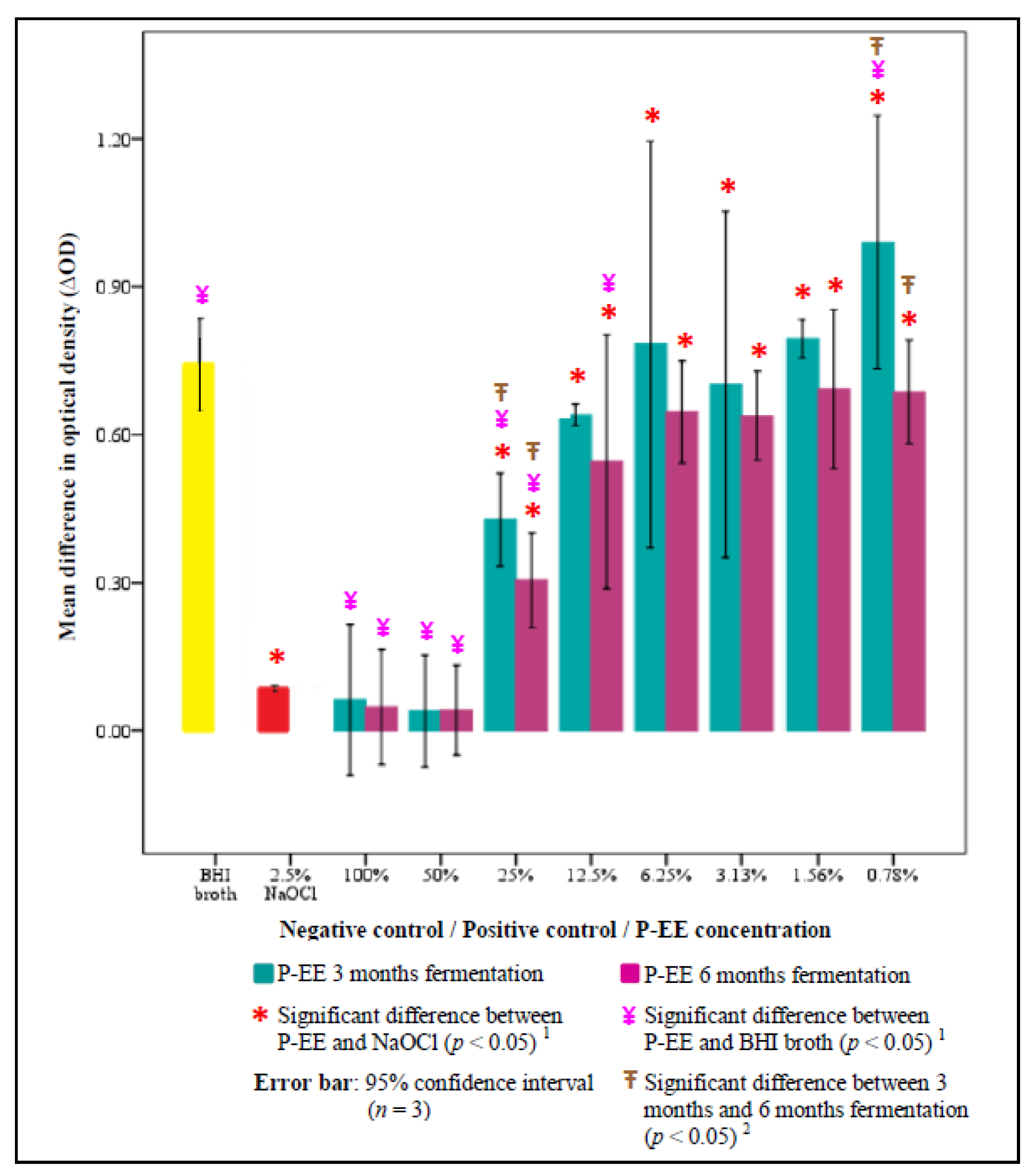
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Swimberghe, R.C.D.; Coenye, T.; De Moor, R.J.G.; Meire, M.A. Biofilm model systems for root canal disinfection: A literature review. Int. Endod. J. 2019, 52, 604–628. [Google Scholar] [CrossRef]
- Alghamdi, F.; Shakir, M. The influence of Enterococcus faecalis as a dental root canal pathogen on endodontic treatment: A systematic review. Cureus 2020, 12, e7257. [Google Scholar] [CrossRef] [PubMed]
- Nair, V.S.; Nayak, M.; Ramya, M.K.; Sivadas, G.; Ganesh, C.; Devi, S.L.; Vedam, V. Detection of adherence of Enterococcus faecalis in infected dentin of extracted human teeth using confocal laser scanning microscope: An in vitro study. J. Pharm. Bioall. Sci. 2017, 9, 41–44. [Google Scholar]
- Zand, V.; Lotfi, M.; Soroush, M.H.; Abdollahi, A.A.; Sadeghi, M.; Mojadadi, A. Antibacterial efficacy of different concentrations of sodium hypochlorite gel and solution on Enterococcus faecalis biofilm. Iran Endod. J. 2016, 11, 315–319. [Google Scholar] [PubMed]
- Estrela, C.; Estrela, C.R.A.; Barbin, E.L.; Spano, J.C.E.; Marchesan, M.A.; Pécora, J.D. Mechanism of action of sodium hypochlorite. Braz. Dent. J. 2002, 13, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Guivarc’h, M.; Ordioni, U.; Ahmed, H.M.A.; Cohen, S.; Catherine, J.-H.; Bukiet, F. Sodium hypochlorite accident: A systematic review. J. Endod. 2017, 43, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, E.; Indushekar, K.R.; Saraf, B.G.; Sheoran, N.; Sardana, D.; Shekhar, A. Exploring the role of Morinda citrifolia and Triphala juice in root canal irrigation: An ex vivo study. J. Conserv. Dent. 2018, 21, 443–449. [Google Scholar]
- Prabhakar, J.; Senthilkumar, M.; Priya, M.S.; Mahalakshmi, K.; Sehgal, P.K.; Sukumaran, V.G. Evaluation of antimicrobial efficacy of herbal alternatives (Triphala and Green Tea Polyphenols), MTAD, and 5% sodium hypochlorite against Enterococcus faecalis biofilm formed on tooth substrate: An in vitro study. J. Endod. 2010, 36, 83–86. [Google Scholar] [CrossRef]
- Radwan, I.N.; Randa, B.; Hend, A.N.; Camilia, G. Evaluation of antimicrobial efficacy of four medicinal plants extracts used as root canal irrigant on Enterococcus faecalis: An in vitro study. Int. Dent. Med. J. Adv. Res. 2015, 1, 1–8. [Google Scholar] [CrossRef]
- Costa, E.M.; Evangelista, A.P.; Medeiros, A.C.; Dametto, F.R.; Carvalho, R.A. In vitro evaluation of the root canal cleaning ability of plant extracts and their antimicrobial action. Braz. Oral Res. 2012, 26, 215–221. [Google Scholar] [CrossRef][Green Version]
- Saleem, M.; Saeed, M.T. Potential application of waste fruit peels (orange, yellow lemon and banana) as wide range natural antimicrobial agent. J. King Saud. Univ. Sci. 2020, 32, 805–810. [Google Scholar] [CrossRef]
- Roy, S.; Lingampeta, P. Solid wastes of fruits peels as source of low cost broad spectrum natural antimicrobial compounds—Furanone, furfural and benezenetriol. Int. J. Res. Eng. Technol. 2014, 3, 273–279. [Google Scholar]
- Martins, S.; Mussatto, S.I.; Martínez-Avila, G.; Montañez-Saenz, J.; Aguilar, C.N.; Teixeira, J.A. Bioactive phenolic compounds: Production and extraction by solid-state fermentation. A review. Biotechnol. Adv. 2011, 29, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Sadh, P.K.; Kumar, S.; Chawla, P.; Duhan, J.S. Fermentation: A boon for production of bioactive compounds by processing of food industries wastes (by-products). Molecules 2018, 23, 2560. [Google Scholar] [CrossRef] [PubMed]
- Sagar, N.A.; Pareek, S.; Sharma, S.; Yahia, E.M.; Lobo, M.G. Fruit and vegetable waste: Bioactive compounds, their extraction, and possible utilization. Compr. Rev. Food Sci. Food Saf. 2018, 17, 512–531. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Ballal, S.; Velmurugan, N. Comparative evaluation of the antimicrobial activity of natural extracts of Morinda citrifolia, papain and aloe vera (all in gel formulation), 2% chlorhexidine gel and calcium hydroxide, against Enterococcus faecalis: An in vitro study. J. Conserv. Dent. 2012, 15, 293–297. [Google Scholar] [CrossRef]
- Duarte, M.A.H.; Yamashita, J.C.; Lanza, P.; Fraga, S.C.; Kuga, M.C. The influence of papain gel as endodontic irrigant in the apical leakage. Salusvita 2001, 20, 35–41. [Google Scholar]
- Amri, E.; Mamboya, F. Papain, a plant enzyme of biological importance: A review. Am. J. Biochem. Biotechnol. 2012, 8, 99–104. [Google Scholar]
- Pandey, S.; Cabot, P.J.; Shaw, P.N.; Hewavitharana, A.K. Anti-inflammatory and immunomodulatory properties of Carica papaya. J. Immunotoxicol. 2016, 13, 590–602. [Google Scholar] [CrossRef]
- Arun, C.; Sivashanmugam, P. Study on optimization of process parameters for enhancing the multi-hydrolytic enzyme activity in garbage enzyme produced from preconsumer organic waste. Bioresour. Technol. 2017, 226, 200–210. [Google Scholar] [CrossRef]
- Gunwantrao, B.B.; Bhausaheb, S.K.; Ramrao, B.S.; Subhash, K.S. Antimicrobial activity and phytochemical analysis of orange (Citrus aurantium L.) and pineapple (Ananas comosus (L.) Merr.) peel extract. Ann. Phytomed. 2016, 5, 156–160. [Google Scholar] [CrossRef]
- Li, T.; Shen, P.; Liu, W.; Liu, C.; Liang, R.; Yan, N.; Chen, J. Major polyphenolics in pineapple peels and their antioxidant interactions. Int. J. Food Prop. 2014, 17, 1805–1817. [Google Scholar] [CrossRef]
- Ana, C.-C.; Jesus, P.-V.; Hugo, E.-A.; Teresa, A.-T.; Ulises, G.-C.; Neith, P. Antioxidant capacity and UPLC–PDA ESI–MS polyphenolic profile of Citrus aurantium extracts obtained by ultrasound assisted extraction. J. Food Sci. Technol. 2018, 55, 5106–5114. [Google Scholar] [CrossRef] [PubMed]
- Liliany, D.; Widyarman, A.S.; Erfan, E.; Sudiono, J.; Djamil, M.S. Enzymatic activity of bromelain isolated pineapple (Ananas comosus) hump and its antibacterial effect on Enterococcus faecalis. Sci. Dent. J. 2018, 2, 41–52. [Google Scholar]
- Al Qarni, F.M.; Elfasakhany, F.M.; Kenawi, L.M.; Moustafa, A.M. Antimicrobial activity of Azadirachta indica (neem) and Salvadora persica (miswak) extracts as endodontic irrigants. Endo EPT 2019, 13, 237–245. [Google Scholar]
- Basrani, B.; Tjaderhane, L.; Santos, J.M.; Pascon, E.; Grad, H.; Lawrence, H.P.; Friedman, S. Efficacy of chlorhexidine- and calcium hydroxide- containing medicaments against Enterococcus faecalis in vitro. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2003, 96, 618–624. [Google Scholar] [CrossRef]
- Diogo, P.; Mota, M.; Fernandes, C.; Sequeira, D.; Palma, P.; Caramelo, F.; Neves, M.G.P.M.S.; Faustino, M.A.F.; Goncalves, T.; Santos, J.M. Is the chlorophyll derivative Zn(II)e6Me a good photosensitizer to be used in root canal disinfection? Photodiagnosis Photodyn. Ther. 2018, 22, 205–211. [Google Scholar] [CrossRef]
- Tawakoli, P.N.; Ragnarsson, K.T.; Rechenberg, D.K.; Mohn, D.; Zehnder, M. Effect of endodontic irrigants on biofilm matrix polysaccharides. Int. Endod. J. 2017, 50, 153–160. [Google Scholar] [CrossRef]
- CLSI. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard: Ninth Edition; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012; p. M07-A9. [Google Scholar]
- Cieplik, F.; Kara, E.; Muehler, D.; Enax, J.; Hiller, K.-A.; Maisch, T.; Buchalla, W. Antimicrobial efficacy of alternative compounds for use in oral care toward biofilms from caries-associated bacteria in vitro. Microbiologyopen 2019, 8, e695. [Google Scholar] [CrossRef]
- Kush, A.; Thakur, R.; Patil, S.D.S.; Paul, S.T.; Kakanur, M. Evaluation of antimicrobial action of Carie CareTM and Papacarie DuoTM on Aggregatibacter actinomycetemcomitans a major periodontal pathogen using polymerase chain reaction. Contemp. Clin. Dent. 2015, 6, 534–538. [Google Scholar]
- Lakhdar, L.; Farah, A.; Tahar, B.; Rida, S.; Bouziane, A.; Ennibi, O. In vitro antibacterial activity of essentials oils from Mentha pulegium, Citrus aurantium and Cymbopogon citratus on virulent strains of Aggregatibacter actinomycetemcomitans. Int. J. Pharmacog. Phytochem. Res. 2014, 6, 1035–1042. [Google Scholar]
- Reddy, V.K.; Nagar, P.; Reddy, S.; Ragulakollu, R.; Tirupathi, S.P.; Ravi, R.; Purumadla, U. Bromelain vs. papain gel for caries removal in primary teeth. J. Contemp. Dent. Pract. 2019, 20, 1345–1349. [Google Scholar] [PubMed]
- Praveen, N.C.; Rajesh, A.; Madan, M.; Chaurasia, V.R.; Hiremath, N.V.; Sharma, M.A. In vitro evaluation of antibacterial efficacy of pineapple extract (bromelain) on periodontal pathogens. J. Int. Oral Health 2014, 6, 96–98. [Google Scholar] [PubMed]
- EUCAST. Determination of minimum inhibitory concentrations (MICs) of antibacterial agents by broth dilution: E.Dis 5.1. European Committee for Antimicrobial Susceptibility Testing (EUCAST) of the European Society of Clinical Microbiology and Infectious Diseases (ESCMID). Clin. Microbiol. Infect. 2003, 9, 1–7. [Google Scholar]
- Neupane, K.; Khadka, R. Production of garbage enzyme from different fruit and vegetable wastes and evaluation of its enzymatic and antimicrobial efficacy. Tribhuvan Univ. J. Microbiol. 2019, 6, 113–118. [Google Scholar] [CrossRef]
- Saramanda, G.; Kaparapu, J. Antimicrobial activity of fermented citrus fruit peel extract. Int. J. Eng. Res. Appl. 2017, 7, 25–28. [Google Scholar]
- Rahman, S.; Haque, I.; Goswami, R.C.D.; Barooah, P.; Sood, K. Characterization and FPLC analysis of garbage enzyme: Biocatalytic and antimicrobial activity. Waste Biomass Valor. 2020. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef]
- Basudan, S.O. Sodium hypochlorite use, storage, and delivery methods: A survey. Saudi. Endod. J. 2019, 9, 27–33. [Google Scholar]
- Dutner, J.; Mines, P.; Anderson, A. Irrigation trends among American Association of Endodontists members: A web-based survey. J. Endod. 2012, 38, 27–40. [Google Scholar] [CrossRef]
- Gopikrishna, V.; Pare, S.; Pradeep Kumar, A.R.; Narayanan, L.L. Irrigation protocol among endodontic faculty and post-graduate students in dental colleges of India: A survey. J. Conserv. Dent. 2013, 16, 394–398. [Google Scholar] [CrossRef]
- Basrani, B.; Haapasalo, M. Update on endodontic irrigating solutions. Endod Topics 2012, 27, 74–102. [Google Scholar] [CrossRef]
- Iandolo, A.; Dagna, A.; Poggio, C.; Capar, I.; Amato, A.; Abdellatif, D. Evaluation of the actual chlorine concentration and the required time for pulp dissolution using different sodium hypochlorite irrigating solutions. J. Conserv. Dent. 2019, 22, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, Y.T.; Shehab, N.F.; Zakaria, N.A. Efficiency of sodium hypochlorite as root canal disinfectant against Enterococcus faecalis: An in vitro study. EC Microbiol. 2019, 15, 288–294. [Google Scholar]
- Murad, C.F.; Sassone, L.M.; Souza, M.C.; Fidel, R.A.S.; Fidel, S.R.; Junior, R.H. Antimicrobial activity of sodium hypochlorite, chlorhexidine and MTAD® against Enterococcus faecalis biofilm on human dentin matrix in vitro. Rev. Bras. Odontol. 2012, 9, 143–150. [Google Scholar]
- Ravinanthanan, M.; Hegde, M.N.; Shetty, V.; Kumari, S. Cytotoxicity effects of endodontic irrigants on permanent and primary cell lines. Biomed. Biotechnol. Res. J. 2018, 2, 59–62. [Google Scholar] [CrossRef]
- Marion, J.J.C.; Manhães, F.C.; Bajo, H.; Duque, T.M. Efficiency of different concentrations of sodium hypochlorite during endodontic treatment. Literature review. Dent. Press Endod. 2012, 2, 32–37. [Google Scholar]
- Verma, N.; Sangwan, P.; Tewari, S.; Duhan, J. Effect of different concentrations of sodium hypochlorite on outcome of primary root canal treatment: A randomized controlled trial. J. Endod. 2019, 45, 357–363. [Google Scholar] [CrossRef]
- Yamashita, J.C.; Filho, M.T.; Leonardo, M.R.; Rossi, M.A.; Silva, L.A.B. Scanning electron microscopic study of the cleaning ability of chlorhexidine as a root-canal irrigant. Int. Endod. J. 2003, 36, 391–394. [Google Scholar] [CrossRef]
- Muñoz, A.F.; Nolf, M.R.; Campusano, C.B.; Cea, D.C.; Cumsille, P.A.; Schnake, V.C.; Lopez, I.Y. Ethylenediaminetetraacetic acid as an irrigant between 5% sodium hypochlorite and 2% chlorhexidine in the formation of para-chloroaniline related precipitate. EC Dent. Sci. 2017, 10, 158–164. [Google Scholar]
- Ketnawa, S.; Chaiwut, P.; Rawdkuen, S. Extraction of bromelain from pineapple peels. Food Sci. Technol. Int. 2011, 17, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Chaiwut, P.; Nitsawang, S.; Shank, L.; Kanasawud, P. A comparative study on properties and proteolytic components of papaya peel and latex proteases. Chiang Mai J. Sci. 2007, 34, 109–118. [Google Scholar]
- Jayahari, N.K.; Niranjan, N.T.; Kanaparthy, A. The efficacy of passion fruit juice as an endodontic irrigant compared with sodium hypochlorite solution: An in vitro study. J. Investig. Clin. Dent. 2014, 5, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Seenivasan, R.; Roopa, L.; Geetha, S. Investigations on purification, characterization and antimicrobial activity of enzyme papain from Carica papaya Linn. J. Pharm. Res. 2010, 3, 1092–1095. [Google Scholar]
- Molobela, I.P.; Cloete, T.E.; Beukes, M. Protease and amylase enzymes for biofilm removal and degradation of extracellular polymeric substances (EPS) produced by Pseudomonas fluorescens bacteria. Afr. J. Microbiol. Res. 2010, 4, 1515–1524. [Google Scholar]
- Arun, C.; Sivashanmugam, P. Identification and optimization of parameters for the semi-continuous production of garbage enzyme from pre-consumer organic waste by green RP-HPLC method. J. Waste Manag. 2015, 44, 28–33. [Google Scholar] [CrossRef]
- Lund, P.; Tramonti, A.; Biase, D.D. Coping with low pH: Molecular strategies in neutralophilic bacteria. FEMS Microbiol. Rev. 2014, 38, 1091–1125. [Google Scholar] [CrossRef]
- Halstead, F.D.; Rauf, M.; Moiemen, N.S.; Bamford, A.; Wearn, C.M.; Fraise, A.P.; Lund, P.A.; Oppenheim, B.A.; Webber, M.A. The antibacterial activity of acetic acid against biofilm-producing pathogens of relevance to burns patients. PLoS ONE 2015, 10, e0136190. [Google Scholar] [CrossRef]
- Arun, C.; Sivashanmugam, P. Solubilization of waste activated sludge using a garbage enzyme produced from different pre-consumer organic waste. RCS Adv. 2015, 5, 51421–51427. [Google Scholar] [CrossRef]
- Verma, D.; Singh, A.N.; Shukla, A.K. Use of garbage enzyme for treatment of waste water. Int. J. Sci. Res. Rev. 2019, 7, 201–205. [Google Scholar]
- Mótyán, J.A.; Tóth, F.; Tőzsér, J. Research applications of proteolytic enzymes in molecular biology. Biomolecules 2013, 3, 923–942. [Google Scholar] [CrossRef] [PubMed]
- Milošević, J.; Janković, B.; Prodanović, R.; Polović, N. Comparative stability of ficin and papain in acidic conditions and the presence of ethanol. Amino Acids 2019, 51, 829–838. [Google Scholar] [CrossRef] [PubMed]
- dos Anjos, M.M.; da Silva, A.A.; de Pascoli, I.C.; Mikcha, J.M.G.; Machinski Jr, M.; Peralta, R.M.; de Abreu Filho, B.A. Antibacterial activity of papain and bromelain on Alicyclobacillus spp. Int. J. Food Microbiol. 2016, 216, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Man, A.; Gaz, A.S.; Mare, A.D.; Berta, L. Effects of low-molecular weight alcohols on bacterial viability. Rev. Rom. Med. Lab. 2017, 25, 335–343. [Google Scholar] [CrossRef]
- Jayaprakashvel, M.; Akila, S.; Venkatramani, M.; Vinothini1, S.; Bhagat, S.J.; Hussain, A.J. Production of bioethanol from papaya and pineapple wastes using marine associated microorganisms. Biosci. Biotechnol. Res. Asia 2014, 11, 193–199. [Google Scholar] [CrossRef]
- Girish, V.; Kumar, K.R.; Girisha, S.T. Estimation of sugar and bio ethanol from different decaying fruits extract. Adv. Appl. Sci. Res. 2014, 5, 106–110. [Google Scholar]
- Khandaker, M.M.; Qiamuddin, K.; Majrashi, A.; Dalorima, T.; Sajili, M.H.; Hossain, A.S. Bio-ethanol production from fruit and vegetable waste by using Saccharomyces cerevisiae. Biosci. Res. 2018, 15, 1703–1711. [Google Scholar]
- Rasit, N.; Mohammad, F.S. Production and characterization of bio catalytic enzyme produced from fermentation of fruit and vegetable wastes and its influence on aquaculture sludge. Int. J. Sci. Technol. 2018, 4, 12–26. [Google Scholar] [CrossRef][Green Version]
- Tang, F.E.; Tong, C.W. A study of the garbage enzyme’s effects in domestic wastewater. Int. J. Environ. Ecol. Eng. 2011, 5, 887–892. [Google Scholar]
- Morandi, S.; Brasca, M.; Alfieri, P.; Lodi, R.; Tamburini, A. Influence of pH and temperature on the growth of Enterococcus faecium and Enterococcus faecalis. Le Lait 2005, 85, 181–192. [Google Scholar] [CrossRef]
- Mubarak, Z.; Soraya, C. The acid tolerance response and pH adaptation of Enterococcus faecalis in extract of lime Citrus aurantiifolia from Aceh Indonesia. F1000Research 2018, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
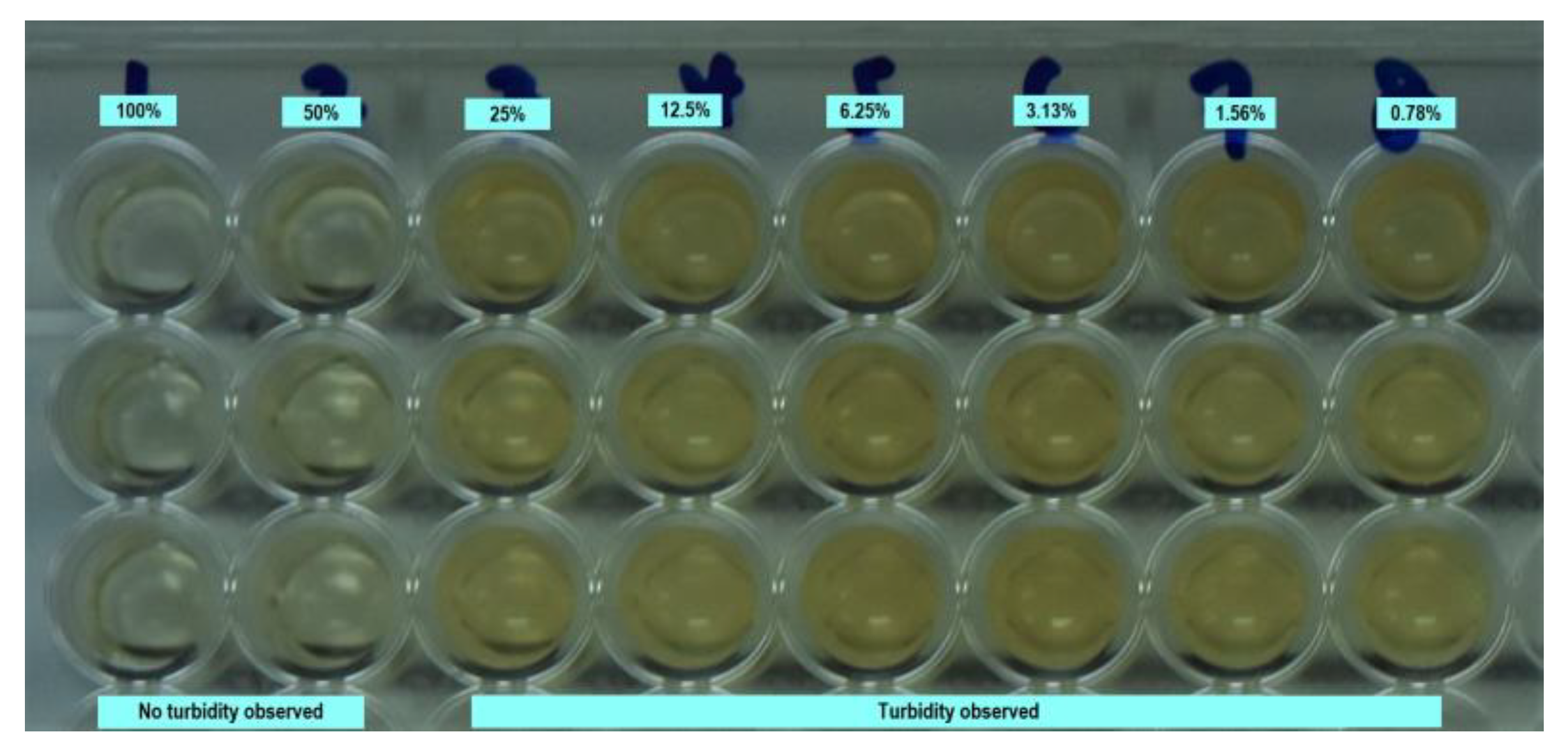

| Concentration (%) | 100 | 50 | 25 | 12.5 | 6.25 | 3.13 | 1.56 | 0.78 |
|---|---|---|---|---|---|---|---|---|
| 3 months fermentation | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| 6 months fermentation | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| Concentration (%) | 100 | 50 | 25 | 12.5 | 6.25 | 3.13 | 1.56 | 0.78 |
|---|---|---|---|---|---|---|---|---|
| 3 months fermentation | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| 6 months fermentation | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 |
| Type of Endodontic Irrigant | Fermentation Period (months) | Concentration (%) | Sensitivity against Enterococcus faecalis |
|---|---|---|---|
| M-EE | 3 | 100 | 1 |
| 50 | 2 | ||
| 6 | 100 | 1 | |
| 50 | 2 | ||
| P-EE | 3 | 100 | 1 |
| 50 | 2 | ||
| 6 | 100 | 1 | |
| 50 | 2 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mavani, H.A.K.; Tew, I.M.; Wong, L.; Yew, H.Z.; Mahyuddin, A.; Ahmad Ghazali, R.; Pow, E.H.N. Antimicrobial Efficacy of Fruit Peels Eco-Enzyme against Enterococcus faecalis: An In Vitro Study. Int. J. Environ. Res. Public Health 2020, 17, 5107. https://doi.org/10.3390/ijerph17145107
Mavani HAK, Tew IM, Wong L, Yew HZ, Mahyuddin A, Ahmad Ghazali R, Pow EHN. Antimicrobial Efficacy of Fruit Peels Eco-Enzyme against Enterococcus faecalis: An In Vitro Study. International Journal of Environmental Research and Public Health. 2020; 17(14):5107. https://doi.org/10.3390/ijerph17145107
Chicago/Turabian StyleMavani, Hetal Ashvin Kumar, In Meei Tew, Lishen Wong, Hsu Zenn Yew, Alida Mahyuddin, Rohi Ahmad Ghazali, and Edmond Ho Nang Pow. 2020. "Antimicrobial Efficacy of Fruit Peels Eco-Enzyme against Enterococcus faecalis: An In Vitro Study" International Journal of Environmental Research and Public Health 17, no. 14: 5107. https://doi.org/10.3390/ijerph17145107
APA StyleMavani, H. A. K., Tew, I. M., Wong, L., Yew, H. Z., Mahyuddin, A., Ahmad Ghazali, R., & Pow, E. H. N. (2020). Antimicrobial Efficacy of Fruit Peels Eco-Enzyme against Enterococcus faecalis: An In Vitro Study. International Journal of Environmental Research and Public Health, 17(14), 5107. https://doi.org/10.3390/ijerph17145107






