Long-Term Combined Training in Idiopathic Pulmonary Fibrosis: A Case Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Laboratory Tests
2.2. Training Program
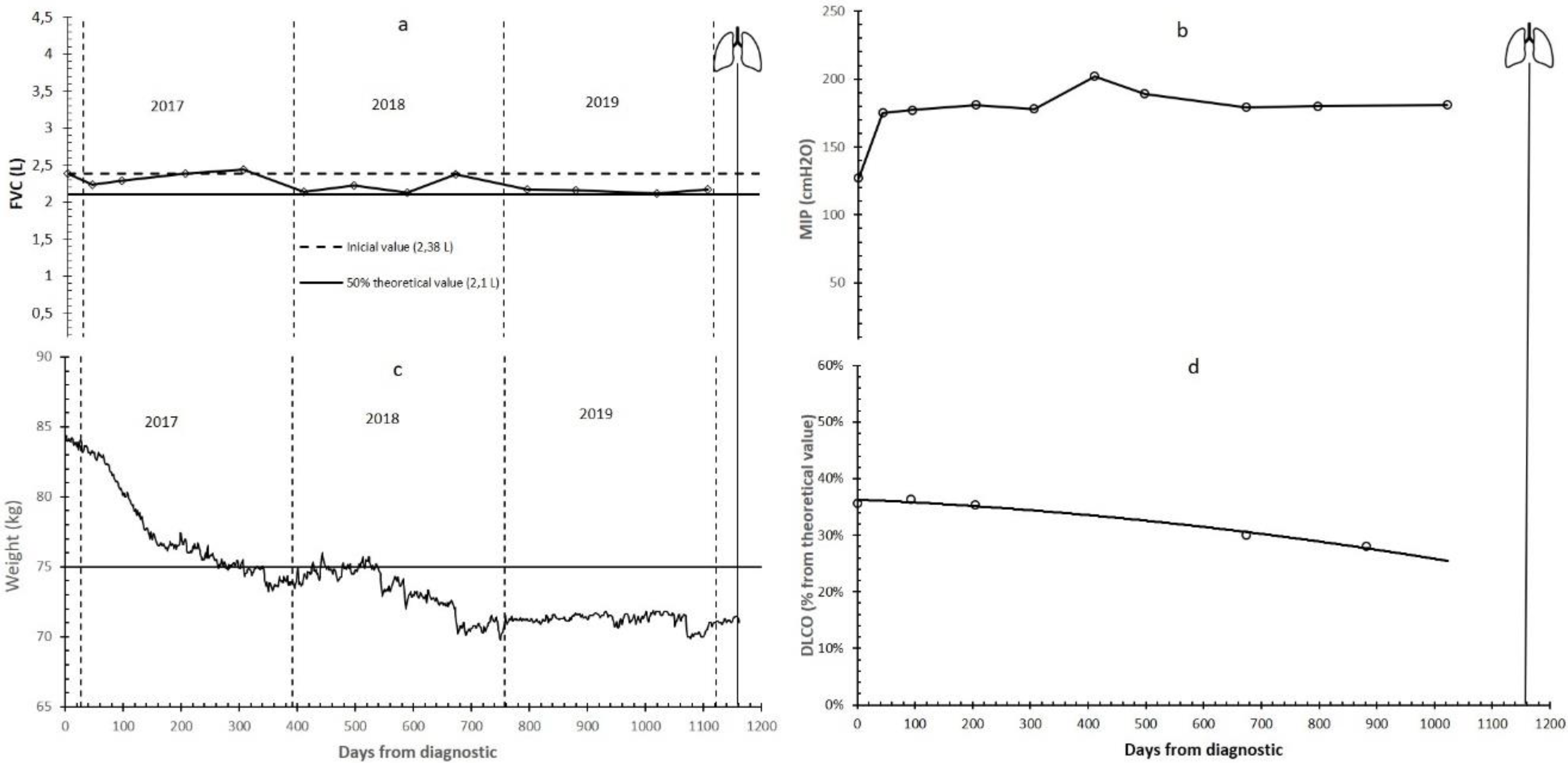
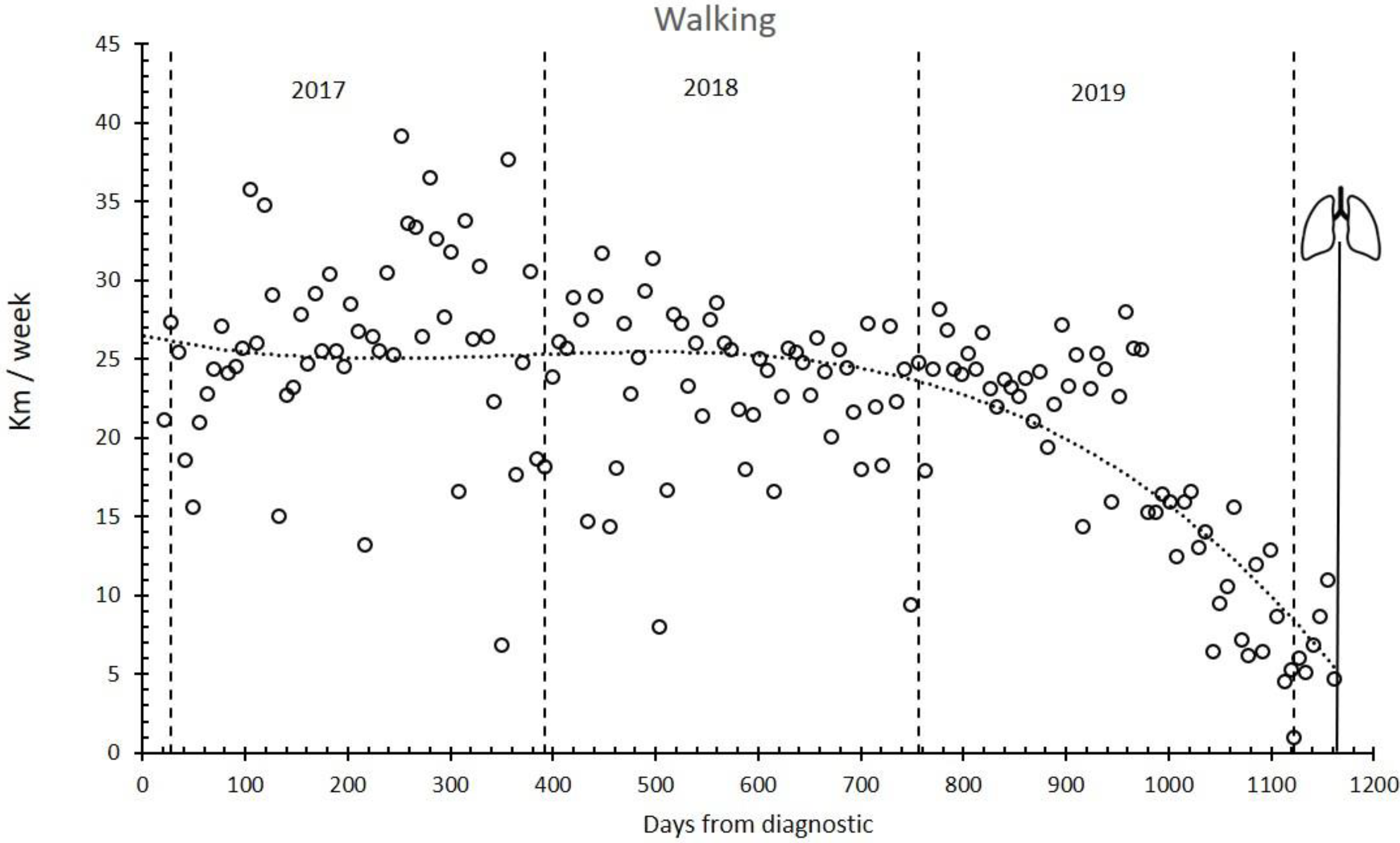
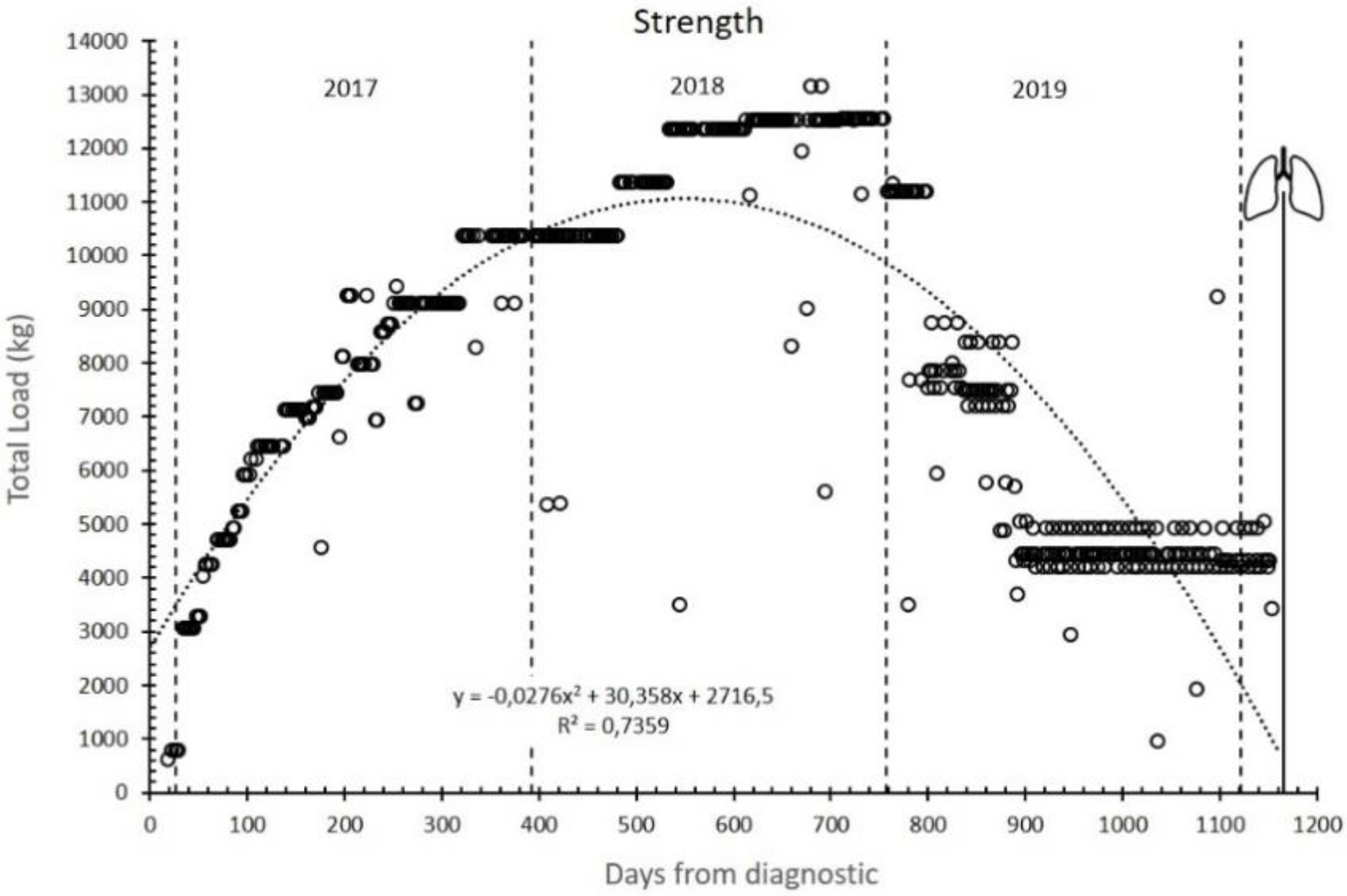
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kim, D.S.; Collard Collard, H.R.; King, T.E. Classification and natural history of the idiopathic interstitial pneumonias. Proc. Am. Thorac. Soc. 2006, 3, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Sociedad Española de Neumología y Cirugía Torácica. Resultados del Registro de Fibrosis Pulmonar Idiopática. Available online: https://www.separ.es/sites/default/files/SEPAR_afectados_fpi.pdf (accessed on 11 June 2020).
- Plantier, L.; Cazes, A.; Dinh-Xuan, A.T.; Bancal, C.; Marchand-Adam, S.; Crestani, B. Physiology of the lung in idiopathic pulmonary fibrosis. Eur. Respir. Rev. 2018, 27, 174. [Google Scholar] [CrossRef] [PubMed]
- Molgat-Seon, Y.; Schaeffer, M.R.; Ryerson, C.J.; Guenette, J.A. Exercise pathophysiology in interstitial lung disease. Clin. Chest Med. 2019, 40, 405–420. [Google Scholar] [CrossRef] [PubMed]
- Puhan, M.A.; Lareau, S. Evidence-based outcomes from pulmonary rehabilitation in the chronic obstructive pulmonary disease patient. Clin. Chest Med. 2014, 35, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Rochester, C.L.; Fairburn, C.; Crouch, R.H. Pulmonary Rehabilitation for respiratory disorders other than chronic obstructive pulmonary disease. Clin. Chest Med. 2014, 35, 369–389. [Google Scholar] [CrossRef] [PubMed]
- Rochester, C.L.; Vogiatzis, I.; Holland, A.E.; Lareau, S.C.; Marciniuk, D.D.; Puhan, M.A.; Spruit, M.A.; Masefield, S.; Casaburi, R.; Clini, E.; et al. An official american thoracic society/european respiratory society policy statement: Enhancing implementation, use, and delivery of pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 2015, 192, 1373–1386. [Google Scholar] [CrossRef]
- Kenn, K.; Gloeckl, R.; Behr, J. Pulmonary rehabilitation in patients with idiopathic pulmonary fibrosis: A review. Respiration 2013, 86, 89–99. [Google Scholar] [CrossRef]
- Yu, X.; Li, X.; Wang, L.; Liu, R.; Xie, Y.; Li, S.; Li, J.S. Pulmonary rehabilitation for exercise tolerance and quality of life in ipf patients: A systematic review and meta-analysis. BioMed Res. Int. 2019, 2019, 8498603. [Google Scholar] [CrossRef]
- Vainshelboim, B.; Oliveira, J.; Fox, B.D.; Soreck, Y.; Fruchter, O.; Kramer, M.R. Long-term effects of a 12-week exercise training program on clinical outcomes in idiopathic pulmonary fibrosis. Lung 2015, 193, 345–354. [Google Scholar] [CrossRef]
- Vainshelboim, B.; Oliveira, J.; Yehoshua, L.; Weiss, I.; Fox, B.D.; Fruchter, O.; Kramer, M.R. Exercise training-based pulmonary rehabilitation program is clinically beneficial for idiopathic pulmonary fibrosis. Respiration 2014, 88, 378–388. [Google Scholar] [CrossRef]
- Holland, A.E.; Hill, C. Physical training for interstitial lung disease. Cochrane Database Syst. Rev. 2008, 4. [Google Scholar] [CrossRef]
- Vainshelboim, B.; Myers, J.; Oliveira, J.; Izhakian, S.; Unterman, A.; Kramer, M.R. Physiological responses and prognostic value of common exercise testing modalities in idiopathic pulmonary fibrosis. J. Cardiopulm. Rehabil. Prev. 2019, 39, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Vainshelboim, B.; Oliveira, J.; Fox, B.D.; Kramer, M.R. The prognostic role of ventilatory inefficiency and exercise capacity in idiopathic pulmonary Fibrosis. Respir. Care 2016, 61, 1100–1109. [Google Scholar] [CrossRef] [PubMed]
- Farina, S.; Correale, M.; Bruno, N.; Paolillo, S.; Salvioni, E.; Badagliacca, R.; Agostoni, P. The role of cardiopulmonary exercise tests in pulmonary arterial hypertension. Eur. Respir. Rev. 2018, 27, 148. [Google Scholar] [CrossRef]
- Adolfo, J.R.; Dhein, W.; Sbruzzi, G. Intensity of physical exercise and its effect on functional capacity in COPD: Systematic review and meta-analysis. J. Bras. Pneumol. 2019, 45, 1. [Google Scholar] [CrossRef]
- Gaunaurd, I.A.; Gómez-Marín, O.W.; Ramos, C.F.; Sol, C.M.I.; Cohen, M.; Cahalin, L.P.; Cardenas, D.D.; Jackson, R.M. Physical activity and quality of life improvements of patients with idiopathic pulmonary fibrosis completing a pulmonary rehabilitation program. Respir. Care 2014, 59, 1872–1879. [Google Scholar] [CrossRef] [PubMed]
- Vainshelboim, B. Exercise training in idiopathic pulmonary fibrosis: Is it of benefit? Breathe 2016, 12, 130–138. [Google Scholar] [CrossRef]
- Langer, D. Rehabilitation in patients before and after lung transplantation. Respiration 2015, 89, 353–362. [Google Scholar] [CrossRef]
- Osho, A.; Mulvihill, M.; Lamba, N.; Hirji, S.; Yerokun, B.; Bishawi, M.; Spencer, P.; Panda, N.; Villavicencio, M.; Hartwig, M. Is functional independence associated with improved long-term survival after lung transplantation? Ann. Thorac. Surg. 2018, 106, 79–84. [Google Scholar] [CrossRef]
- Maury, G.; Langer, D.; Verleden, G.; Dupont, L.; Gosselink, R.; Decramer, M.; Troosters, T. Skeletal muscle force and functional exercise tolerance before and after lung transplantation: A cohort study. Arab. Archaeol. Epigr. 2008, 8, 1275–1281. [Google Scholar] [CrossRef]
- Walsh, J.R.; Chambers, D.C.; Davis, R.J.; Morris, N.R.; Seale, H.E.; Yerkovich, S.T.; Hopkins, P.M. Impaired exercise capacity after lung transplantation is related to delayed recovery of muscle strength. Clin. Transplant. 2013, 27, E504–E511. [Google Scholar] [CrossRef] [PubMed]
- Wickerson, L.; Mathur, S.; Singer, L.G.; Brooks, D. Physical activity levels early after lung transplantation. Phys. Ther. 2015, 95, 517–525. [Google Scholar] [CrossRef] [PubMed]
- LaPorta, R.; Carrasco, M.T.L.; De Ugarte, A.V.; Gil, P.U. Seguimiento a largo plazo del paciente trasplantado pulmonar. Arch. Bronconeumol. 2014, 50, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Jastrzebski, D.; Kozielski, J.; Żebrowska, A. Pulmonary rehabilitation in patients with idiopathic pulmonary fibrosis with inspiratory muscle training. Pneumonol. I Alergol. Polska 2008, 76, 131–141. [Google Scholar]
- Beckerman, M.; Magadle, R.; Weiner, P.; Weiner, P. The effects of 1 year of specific inspiratory muscle training in patients with COPD. Chest 2005, 128, 3177–3182. [Google Scholar] [CrossRef]
- Santana-Sosa, E.; González-Saiz, L.; Groeneveld, I.F.; Villa-Asensi, J.R.; Aguero, M.I.B.G.D.; Fleck, S.J.; López-Mojares, L.M.; Pérez, M.; Lucia, A. Benefits of combining inspiratory muscle with ‘whole muscle’ training in children with cystic fibrosis: A randomised controlled trial. Br. J. Sports Med. 2013, 48, 1513–1517. [Google Scholar] [CrossRef]
- González-Saiz, L.; Fiuza-Luces, C.; Sanchis-Gomar, F.; Santos-Lozano, A.; Quezada-Loaiza, C.A.; Flox-Camacho, A.; Munguía-Izquierdo, D.; Ara, I.; Santalla, A.; Morán, M.; et al. Benefits of skeletal-muscle exercise training in pulmonary arterial hypertension: The WHOLEi+12 trial. Int. J. Cardiol. 2017, 231, 277–283. [Google Scholar] [CrossRef]
- Sanchis-Gomar, F.; Gonzalez-Saiz, L.; Ayán, P.S.; Fiuza-Luces, C.; Quezada-Loaiza, C.A.; Flox-Camacho, Á.; Santalla, A.; Munguía-Izquierdo, D.; Santos-Lozano, A.; Pareja-Galeano, H.; et al. Rationale and design of a randomized controlled trial evaluating whole muscle exercise training effects in outpatients with pulmonary arterial hypertension (WHOLEi+12). Cardiovasc. Drugs Ther. 2015, 29, 543–550. [Google Scholar] [CrossRef]
- Lopes, A.; Menezes, S.; Dias, C.; Oliveira, J.; Mainenti, M.; Guimarães, F. Cardiopulmonary exercise testing variables as predictors of long-term outcome in thoracic sarcoidosis. Braz. J. Med. Biol. Res. 2012, 45, 256–263. [Google Scholar] [CrossRef]
- Sun, X.G.; Hansen, J.E.; Garatachea, N.; Storer, T.W.; Wasserman, K. Ventilatory efficiency during exercise in healthy subjects. Am. J. Respir. Crit. Care Med. 2002, 166, 1443–1448. [Google Scholar] [CrossRef]
- Brown, S.J.; Raman, A.; Schlader, Z.; Stannard, S.R. Ventilatory efficiency in juvenile elite cyclists. J. Sci. Med. Sport 2013, 16, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Ross, L.M.; Porter, R.R.; Durstine, J.L. High-intensity interval training (HIIT) for patients with chronic diseases. J. Sport Health Sci. 2016, 5, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Black, L.F.; Hyatt, R.E. Maximal respiratory pressures: Normal values and relationship to age and sex. Am. Rev. Respir. Dis. 1969, 99, 696–702. [Google Scholar] [PubMed]
- Shei, R.J.; Paris, H.L.R.; Wilhite, D.P.; Chapman, R.; Mickleborough, T.D. The role of inspiratory muscle training in the management of asthma and exercise-induced bronchoconstriction. Physician Sportsmed. 2016, 44, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Dempsey, J.A.; Romer, L.; Rodman, J.; Miller, J.; Smith, C. Consequences of exercise-induced respiratory muscle work. Respir. Physiol. Neurobiol. 2006, 151, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Hilton, N.; Solis-Moya, A. Respiratory muscle training for cystic fibrosis. Cochrane Database Syst. Rev. 2018, 5, CD006112. [Google Scholar] [CrossRef]
- Rodriguez, D.A.; Del Rio, F.; Fuentes, M.E.; Naranjo, S.; Moradiellos, J.; Gomez, D.; Rubio, J.; Calvo, E.; Varela, A. Trasplante de pulmón con donantes no controlados a corazón parado. Factores pronósticos dependientes del donante y evolución inmediata postrasplante. Arch. Bronconeumol. 2011, 47, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Florian, J.; Watte, G.; Teixeira, P.J.Z.; Altmayer, S.; Schio, S.M.; Sanchez, L.B.; Nascimento, D.Z.; Camargo, S.M.; Perin, F.A.; Camargo, J.D.J.; et al. Pulmonary rehabilitation improves survival in patients with idiopathic pulmonary fibrosis undergoing lung transplantation. Sci. Rep. 2019, 9, 1–6. [Google Scholar] [CrossRef]


| PeakVO2 | VE/VO2 | VE/VCO2 | DeltaCO2 | VEmax | VT/Ti max | Ti/Ttot | |
|---|---|---|---|---|---|---|---|
| (mL/kg/min) | (at VT1) | (at VT1) | (L/min) | (L/s) | |||
| 28/11/2016 | 16.6 | 40 | 37 | 33.6 | 57.9 | 2.8 | 0.3 |
| 09/01/2017 | 14.7 | 31 | 38 | 31.3 | 54.1 | 2.5 | 0.3 |
| 01/03/2017 | 16.2 | 33 | 36 | 31.8 | 61.9 | 2.8 | 0.3 |
| 20/06/2017 | 20.2 | 42 | 35 | 32 | 75 | 3.2 | 0.3 |
| 29/09/2017 | 22.3 | 37 | 35 | 30.1 | 80 | 3.2 | 0.3 |
| 12/01/2018 | 19.8 | 40 | 34 | 29.4 | 57.7 | 2.7 | 0.3 |
| 09/04/2018 | 18.4 | 48 | 40 | 36.3 | 63.9 | 2.6 | 0.3 |
| 10/07/2018 | 18.0 | 37 | 40 | 34.5 | 50.1 | 2.3 | 0.3 |
| 03/10/2018 | 14.7 | 45 | 46 | 40 | 45.3 | 2.1 | 0.3 |
| 04/02/2019 | 11.4 | 61 | 59 | 54 | 51.9 | 2.4 | 0.3 |
| 29/04/2019 | 12.5 | 48 | 53 | 49.5 | 49.2 | 2.4 | 0.3 |
| 17/09/2019 | 10.9 | 50 | 54 | 37.9 | 38.6 | 1.9 | 0.3 |
| Kms | Min | Steps | Kcal | ||
| 2017 | TOTAL | 1357 | 18,497 | 2,036,787 | 223,807 |
| Daily average ± SD | 3.7 ± 1.4 | 51 ± 23 | 5580 ± 2431 | 613 ± 293 | |
| Weekly average ± SD | 26 ± 5.2 | 355 ± 78 | 39,062 ± 6832 | 4292 ± 822 | |
| 2018 | TOTAL | 1221 | 17,535 | 1,833,589 | 180,219 |
| Daily average ± SD | 4.3 ± 1.8 | 62 ± 25 | 5024 ± 2653 | 494 ± 276 | |
| Weekly average ± SD | 23.4 ± 4.9 | 336 ± 73 | 35,180 ± 7458 | 3457.6 ± 797 | |
| 2019 | TOTAL | 969 | 14,425 | 1,455,616 | 133,821 |
| Daily average ± SD | 2.7 ± 1.1 | 40 ± 15 | 3988 ± 1620 | 367 ± 194 | |
| Weekly average ± SD | 18.6 ± 6.9 | 277 ± 94 | 27,964 ± 10,438 | 2572.2 ± 1159 |
| Exercises | 2016 December | 2017 June | 2017 December | 2018 June | 2018 December | 2019 June | 2019 December |
|---|---|---|---|---|---|---|---|
| Leg Press | 3 × 6 × 30 kg (3–94%) | 3 × 10 × 61 kg (4–94%) | 3 × 10 × 69 kg (4–94%) | 3 × 10 × 69 kg (4–94%) | 3 × 10 × 70 kg (5–92%) | --- | --- |
| Butterfly | --- | --- | --- | --- | --- | 3 × 8 × 30 kg (3–91%) | 3 × 8 × 20 kg (5–91%) |
| Chest press | 2 × 6 × 15 kg (3–95%) | 3 × 10 × 35 kg (4–94%) | 3 × 10 × 35 kg (4–94%) | 3 × 10 × 35 kg (4–94%) | 3 × 10 × 40 kg (4–92%) | 3 × 8 × 30 kg (3–94%) | 3 × 8 × 30 kg (5–95%) |
| Back Row | 2 × 6 × 7 kg (4–95%) | 3 × 10 × 35 kg (4–94%) | 3 × 10 × 35 kg (4–94%) | 3 × 10 × 35 kg (4–94%) | 3 × 10 × 35 kg (3–93%) | 3 × 8 × 30 kg (3–94%) | 3 × 8 × 25 kg (3–95%) |
| Abdominal | 2 × 10 × 10 kg (4–94%) | 8 × 15 × 30 kg (4–93%) | 10 × 20 × 25 kg (4–94%) | 10 × 20 × 35 kg (5–93%) | 10 × 20 × 35 kg (5–92%) | 8 × 15 × 25 kg (6–94%) | 8 × 15 × 25 kg (6–96%) |
| Lat Pull Down | 2 × 6 × 25 kg (4–95%) | 3 × 10 × 35 kg (4–93%) | 3 × 10 × 40 kg (4–93%) | 3 × 10 × 40 kg (4–94%) | 3 × 10 × 40 kg (5–93%) | 3 × 8 × 30 kg (3–92%) | 3 × 8 × 30 kg (5–95%) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naranjo-Orellana, J.; Santalla, A. Long-Term Combined Training in Idiopathic Pulmonary Fibrosis: A Case Study. Int. J. Environ. Res. Public Health 2020, 17, 5091. https://doi.org/10.3390/ijerph17145091
Naranjo-Orellana J, Santalla A. Long-Term Combined Training in Idiopathic Pulmonary Fibrosis: A Case Study. International Journal of Environmental Research and Public Health. 2020; 17(14):5091. https://doi.org/10.3390/ijerph17145091
Chicago/Turabian StyleNaranjo-Orellana, José, and Alfredo Santalla. 2020. "Long-Term Combined Training in Idiopathic Pulmonary Fibrosis: A Case Study" International Journal of Environmental Research and Public Health 17, no. 14: 5091. https://doi.org/10.3390/ijerph17145091
APA StyleNaranjo-Orellana, J., & Santalla, A. (2020). Long-Term Combined Training in Idiopathic Pulmonary Fibrosis: A Case Study. International Journal of Environmental Research and Public Health, 17(14), 5091. https://doi.org/10.3390/ijerph17145091






