Antibiotic Resistance in Microbes from Street Fruit Drinks and Hygiene Behavior of the Vendors in Delhi, India
Abstract
1. Introduction
2. Materials and Methods
2.1. Recruitment, Questionnaire Survey, and Sample Collection
2.2. Microbiological Characterization
2.2.1. Enumeration of Bacterial Population (Total Bacterial Count)
2.2.2. Enumeration of Escherichia coli, Salmonella and Vibrio
2.2.3. Isolation and Preservation of Microorganisms
2.3. Antibiotic Susceptibility Test (AST)
2.4. Statistical Analysis
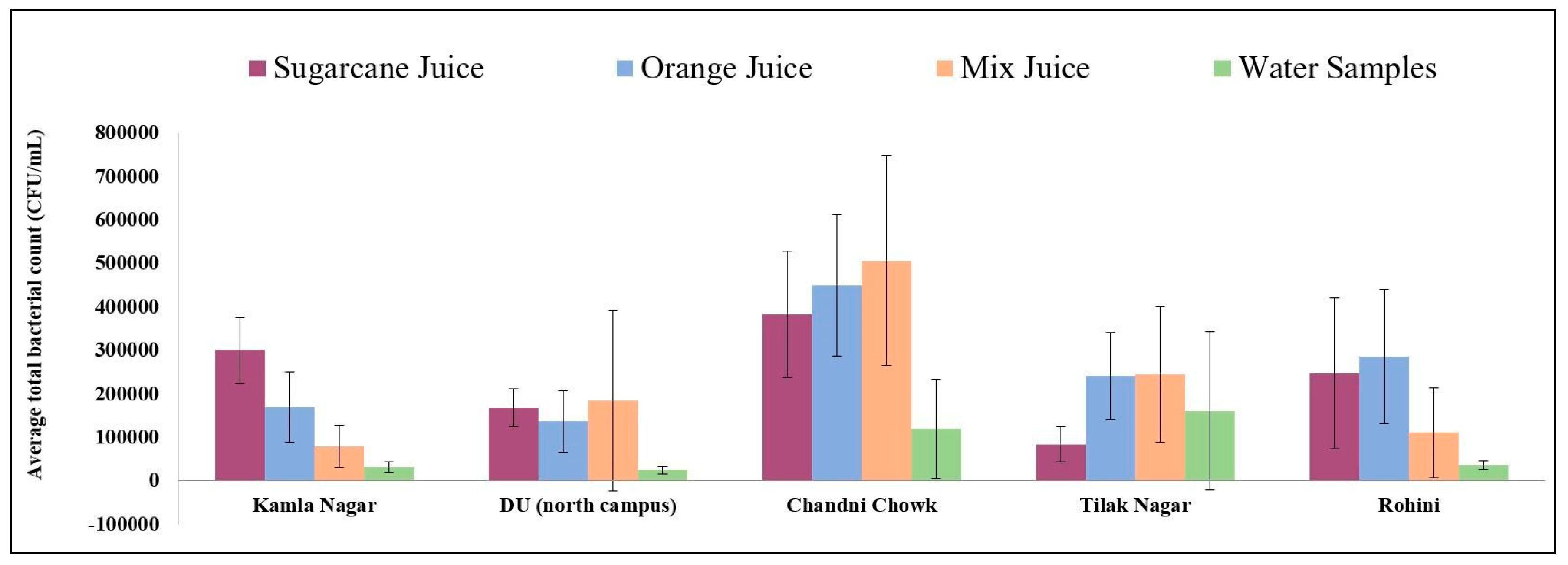
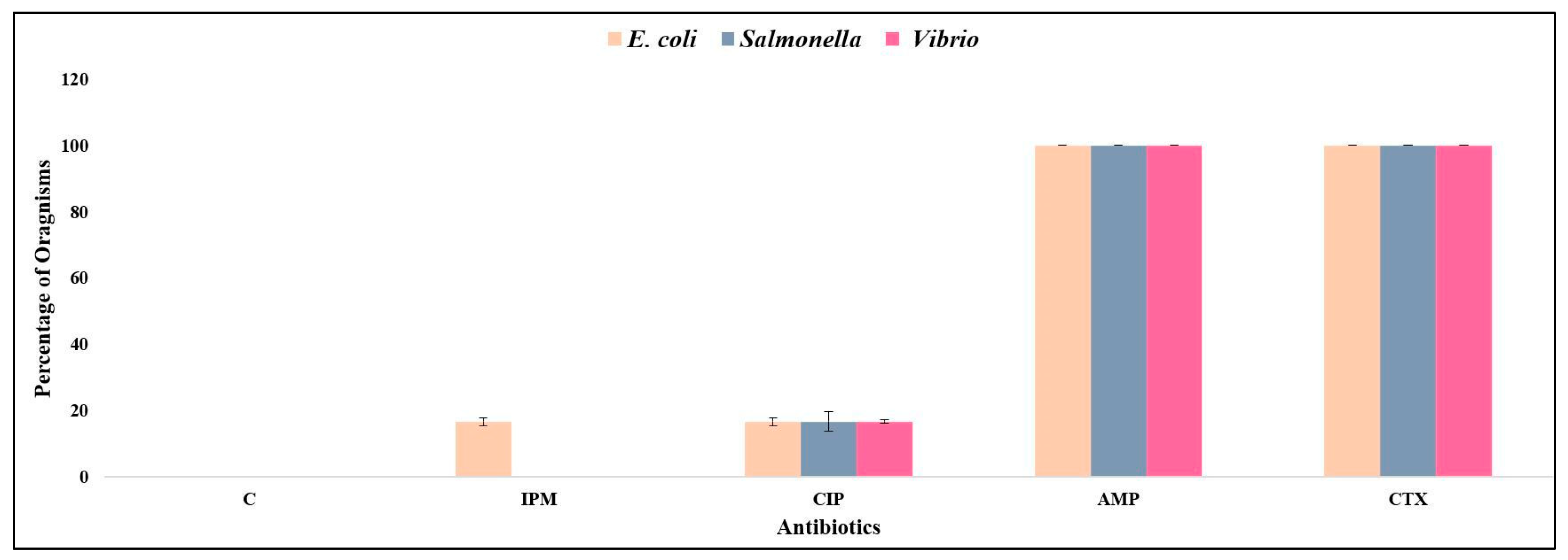
3. Results
3.1. Hygiene Behavior of the Street Juice Vendors
3.2. Bacterial Isolates and AMR Characteristics in Juice Samples
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fellows, P.; Hilmi, M. Selling Street and Snack Foods: FAO Diversification Booklet; Rural Infrastructure and Agro-Industries Division Food and Agriculture Organization of the United Nations: Rome, Italy, 2011. [Google Scholar]
- Lima Tribst, A.A.; de Souza Sant’Ana, A.; de Massaguer, P.R. Microbiological quality and safety of fruit juices—Past, present and future perspectives. Crit. Rev. Microbiol. 2009, 35, 310–339. [Google Scholar] [CrossRef] [PubMed]
- Aneja, K. Microbes associated with fruit juices sold in the retail market and their inactivation with plant antimicrobials. Shodhganga 2016. Available online: http://hdl.handle.net/10603/117765 (accessed on 3 July 2020).
- Callejón, R.M.; Rodriguez-Naranjo, M.I.; Ubeda, C.; Hornedo-Ortega, R.; Garcia-Parrilla, M.C.; Troncoso, A.M. Reported foodborne outbreaks due to fresh produce in the United States and European Union: Trends and causes. Foodborne Pathog. Dis. 2015, 12, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Salomão, B.d.C.M. Pathogens and Spoilage Microorganisms in Fruit Juice: An Overview. In Fruit Juices; Elsevier: San Diego, CA, USA, 2018; pp. 291–308. [Google Scholar]
- Kechero, F.K.; Baye, K.; Tefera, A.T.; Tessema, T.S. Bacteriological quality of commonly consumed fruit juices and vegetable salads sold in some fruit juice houses in Addis Ababa, Ethiopia. J. Food Saf. 2019, 39, e12563. [Google Scholar] [CrossRef]
- Havelaar, A.H.; Kirk, M.D.; Torgerson, P.R.; Gibb, H.J.; Hald, T.; Lake, R.J.; Praet, N.; Bellinger, D.C.; De Silva, N.R.; Gargouri, N. World Health Organization global estimates and regional comparisons of the burden of foodborne disease in 2010. PLoS Med. 2015, 12, e1001923. [Google Scholar] [CrossRef]
- Rane, S. Street vended food in developing world: Hazard analyses. Indian J. Microbiol. 2011, 51, 100–106. [Google Scholar] [CrossRef]
- Sundaram, S.S. National policy for urban street vendors and its impact. Econ. Political Wkly. 2008, 43, 22–25. [Google Scholar]
- Choudhury, M.; Mahanta, L.; Goswami, J.; Mazumder, M.; Pegoo, B. Socio-economic profile and food safety knowledge and practice of street food vendors in the city of Guwahati, Assam, India. Food Control 2011, 22, 196–203. [Google Scholar] [CrossRef]
- Kristkova, Z.S.; Grace, D.; Kuiper, M. The Economics of Food Safety in India: A Rapid Assessment; Wageningen University & Research: Wageningen, Netherlands, 2017. [Google Scholar]
- Aneja, K.R.; Dhiman, R.; Aggarwal, N.K.; Kumar, V.; Kaur, M. Microbes associated with freshly prepared juices of citrus and carrots. Int. J. Food Sci. 2014, 2014, 408085. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Gaur, S. Microbiological Analysis of Street Vended Sugarcane Juice in Noida City, India. Int. J. Pharma Bio Sci. 2017, 8, 496–499. [Google Scholar] [CrossRef]
- Aruna, N.; Rajan, V. Microbial analysis of street foods of different locations at Chennai city, India. Innov. Int. J. Med. Pharm. Sci. 2017, 2, 21–23. [Google Scholar]
- Cook, K.A.; Dobbs, T.E.; Hlady, W.G.; Wells, J.G.; Barrett, T.J.; Puhr, N.D.; Lancette, G.A.; Bodager, D.W.; Toth, B.L.; Genese, C.A. Outbreak of Salmonella serotype Hartford infections associated with unpasteurized orange juice. JAMA 1998, 280, 1504–1509. [Google Scholar] [CrossRef] [PubMed]
- Castillo, A.; Villarruel-López, A.; Navarro-Hidalgo, V.; Martínez-González, N.; Torres-Vitela, M. Salmonella and Shigella in freshly squeezed orange juice, fresh oranges, and wiping cloths collected from public markets and street booths in Guadalajara, Mexico: Incidence and comparison of analytical routes. J. Food Prot. 2006, 69, 2595–2599. [Google Scholar] [CrossRef]
- Ubong, A.; Tunung, R.; Noorlis, A.; Elexson, N.; Tuan Zainazor, T.; Ghazali, F.; Nakaguchi, Y.; Nishibuchi, M.; Son, R. Prevalence and detection of Vibrio spp. and Vibrio cholerae in fruit juices and flavored drinks. Int. Food Res. J. 2011, 18, 1163–1169. [Google Scholar]
- Uddin, M.E.; Akter, T.; Parvez, M.A.K.; Nahar, S.; Pervin, S.; Debnath, B.; Datta, S. Microbial safety of street vended fruit juices in Dhaka City of Bangladesh. J. Adv. Microbiol. 2017, 3, 1–7. [Google Scholar] [CrossRef]
- Saksena, R.; Malik, M.; Gaind, R. Bacterial contamination and prevalence of antimicrobial resistance phenotypes in raw fruits and vegetables sold in Delhi, India. J. Food Saf. 2020, 40, e12739. [Google Scholar] [CrossRef]
- Malik, Y.; Omar, B.J.; Singh, A. Bacteriological analysis of street-vended fruit juices available in Rishikesh, Uttarakhand. J. Fam. Med. Prim. Care 2020, 9, 938–942. [Google Scholar]
- Sharafati Chaleshtori, R.; Sharafati Chaleshtori, F.; Karimi, A. Antibiotic resistance pattern of staphylococcus strains isolated from orange and apple juices in Shahre-kord, Iran. Pak. J. Med. Sci. 2010, 26, 615–618. [Google Scholar]
- Lucky, N.A.; Nur, I.T.; Ahmed, T. Microbiological quality assessment for drug resistant pathogenic microorganisms from the fresh vended fruit juices. Stamford J. Microbiol. 2016, 6, 7–10. [Google Scholar] [CrossRef]
- Tadesse, H.A.; Gidey, N.B.; Workelule, K.; Hailu, H.; Gidey, S.; Bsrat, A.; Taddele, H. Antimicrobial Resistance Profile of E. coli Isolated from Raw Cow Milk and Fresh Fruit Juice in Mekelle, Tigray, Ethiopia. Vet. Med. Int. 2018, 2018, 3142. [Google Scholar] [CrossRef]
- Sarker, M.A.R.; Haque, M.M.; Rifa, R.A.; Ema, F.A.; Islam, M.A.; Khatun, M.M. Isolation and identification of bacteria from fresh guava (Psidium guajava) sold at local markets in Mymensingh and their antibiogram profile. Vet. World 2018, 11, 1145–1149. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Mandal, S. Multiple Antibiotic Resistance Indices of Potential Pathogenic Bacteria Isolated from Street Vended Fruit and Sugarcane Juices, Malda Town, India. Acta Sci. Pharm. Sci. 2018, 2, 89–94. [Google Scholar]
- Kebede, H.; Hadush, H.; Gebrecherkos, T.; Chaithanya, K.K. Public health risks and bacterial safety of fruit juices prepared in Axum town, north Ethiopia. J. Pharm. Res. 2018, 12, 509. [Google Scholar]
- Deb, V.K.; Verma, U.; Kumar, A. Isolation and Antibiotics Pattern of Salmonella Typhi From Fruit Juice. Eur. J. Pharm. Med. Res. 2018, 5, 570–575. [Google Scholar]
- Jain, A.K.; Yadav, R. Antibiotic resistance of different bacterial strains isolated from orange juices. Am. J. Phytomed Clin. Ther. 2015, 3, 88–96. [Google Scholar]
- Padamadan, C.J.; Raju, S.; Shenpagam, N.H. Isolation and identification of microorganism from street vented fruit juices and packed juice and its antibiogram. IJAR 2016, 2, 1099–1105. [Google Scholar]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A.G. Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Chauhan, A.; Goyal, P.; Varma, A.; Jindal, T. Microbiological evaluation of drinking water sold by roadside vendors of Delhi, India. Appl. Water Sci. 2017, 7, 1635–1644. [Google Scholar] [CrossRef]
- Duttagupta, I. How FSSAI is ensuring that food offered by street vendors and online aggregators meet safety norms. The Economic Times, 7 October 2018. [Google Scholar]
- World Health Organization. Essential Safety Requirements for Street-Vended Foods; World Health Organization: Geneva, Switzerland, 1996. [Google Scholar]
- Tambekar, D.; Jaiswal, V.; Dhanorkar, D.; Gulhane, P.; Dudhane, M. Microbial quality and safety of street vended fruit juices: A case study of Amravati city. Internet J. Food Saf. 2009, 10, 72–76. [Google Scholar]
- Khan, M.M.; Islam, M.T.; Chowdhury, M.M.H.; Alim, S.R. Assessment of microbiological quality of some drinks sold in the streets of Dhaka University Campus in Bangladesh. Int. J. Food Contam. 2015, 2, 421. [Google Scholar] [CrossRef]
- Muinde, O.; Kuria, E. Hygienic and sanitary practices of vendors of street foods in Nairobi, Kenya. Afr. J. Food Agric. Dev. 2005, 1, 1–5. [Google Scholar]
- Cortese, R.D.M.; Veiros, M.B.; Feldman, C.; Cavalli, S.B. Food safety and hygiene practices of vendors during the chain of street food production in Florianopolis, Brazil: A cross-sectional study. Food Control 2016, 62, 178–186. [Google Scholar] [CrossRef]
- Reddi, S.L.; Kumar, R.N.; Balakrishna, N.; Rao, V.S. Microbiological quality of street vended fruit juices in Hyderabad, India and their association between food safety knowledge and practices of fruit juice vendors. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 970–982. [Google Scholar]
- Kotwal, V.; Satya, S.; Naik, S.N. Food Safety & Hygiene Practices Among Urban And Rural Street Food Vendors: A Comparative Study In And Around Delhi, India. Int. J. Curr. Res. 2018, 10, 65500–65506. [Google Scholar]
- Heaton, J.C.; Jones, K. Microbial contamination of fruit and vegetables and the behaviour of enteropathogens in the phyllosphere: A review. J. Appl. Microbiol. 2008, 104, 613–626. [Google Scholar] [CrossRef] [PubMed]
- Enache, E.; Chen, Y.; Elliott, P.H. Inactivation of Escherichia coli O157: H7 in single-strength lemon and lime juices. J. Food Prot. 2009, 72, 235–240. [Google Scholar] [CrossRef]
- Tomotake, H.; Koga, T.; Yamato, M.; KASSU, A.; OTA, F. Antibacterial activity of citrus fruit juices against Vibrio species. J. Nutr. Sci. Vitaminol. 2006, 52, 157–160. [Google Scholar] [CrossRef]
- Reddy, K.S.; Reddy, B.S.; Doshi, D.; Reddy, P.; Kulkarni, S. Identification of specific microorganisms in fresh squeezed street vended fruit juices. J. Indian Assoc. Public Health Dent. 2016, 14, 84. [Google Scholar] [CrossRef]
- Sharma, P.U. Bacteriological analysis of street vended fruit juices available in Vidarbha. Int. J. Curr. Microbiol. App. Sci. 2013, 2, 178–183. [Google Scholar]
- Rashed, N.; Aftab, U.M.; Azizul, H.M.; Saurab, K.; Mrityunjoy, A.; Majibur, R. Microbiological study of vendor and packed fruit juices locally available in Dhaka city, Bangladesh. Int. Food Res. J. 2013, 20, 1011–1015. [Google Scholar]
- Sultana, F.; Marzan, L.; Mina, S. Microbiological quality assessment of locally vended and commercially packed fruit juices in Chittagong city of Bangladesh. J. Bio Sci. 2019, 27, 43–58. [Google Scholar] [CrossRef]
- World Health Organization. World Health Organization Model List of Essential Medicines: 21st List 2019; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- Peechakara, B.V.; Basit, H.; Gupta, M. Ampicillin; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Arumugham, V.B.; Cascella, M. Third Generation Cephalosporins. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Kumar, Y.; Sharma, A.; Mani, K.R. Antibiogram profile of Salmonella enterica serovar typhi in India–A two year study. Trop. Life Sci. Res. 2013, 24, 45. [Google Scholar] [PubMed]
- Jayaraman, Y.; Veeraraghavan, B.; Purushothaman, G.K.C.; Sukumar, B.; Kangusamy, B.; Kapoor, A.N.; Gupta, N.; Mehendale, S.M.; Hospital Based Sentinel Surveillance of Bacterial Meningitis (HBSSBM) Network Team. Burden of bacterial meningitis in India: Preliminary data from a hospital based sentinel surveillance network. PLoS ONE 2018, 13, e0197198. [Google Scholar] [CrossRef] [PubMed]
| Hygiene Behavior | Locations Showing the Highest % of Behavior | Highest Frequency of Response at a Location/Total Response | Combined Response of all Five Locations (n = 50) n (%) | Comparison between all Locations p-Values |
|---|---|---|---|---|
| Positive Behavior | ||||
| Hand washing with water | Kamla Nagar and Chandni Chowk | 3/10 | 10 (20) | 0.44 |
| Hand washing with soap and water | University of Delhi (DU) (North Campus) | 10/10 | 40 (80) | 0.44 |
| Utensil washing with water | Chandni Chowk | 7/10 | 19 (38) | 0.13 |
| Utensil washing with soap and water | Kamla Nagar and DU (North Campus) | 8/10 | 31 (62) | 0.12 |
| Storage of peeled fruits in closed container | Kamla Nagar, Tilak Nagar and Rohini | 2/10 | 10 (20) | 0.99 |
| Peeling of fruit just before juice extraction | DU (North Campus) | 7/10 | 21 (42) | 0.26 |
| Keep grinded sugar in closed container | Kamla Nagar and DU (North Campus) | 8/10 | 29 (58) | 0.01 * |
| Uses packaged drinking water | DU (North Campus) | 1/10 | 1 (2) | NA |
| Uses mosquito spray | DU (North Campus) and Tilak Nagar | 6/10 | 22 (44) | 0.13 |
| Uses waste bin with lid | DU (North Campus) and Tilak Nagar | 6/10 | 22 (44) | 0.14 |
| Negative Behavior | ||||
| Uses already peeled fruits | Kamla Nagar | 8/10 | 29 (58) | 0.26 |
| Keep peeled fruits in open container | Kamla Nagar | 7/10 | 24 (48) | 0.36 |
| Keep grinded sugar in open container | Chandni Chowk | 9/10 | 21 (42) | 0.01 * |
| Uses municipal water | Kamla Nagar, Tilak Nagar, Rohini and Chandni Chowk | 10/10 | 49 (98) | 0.39 |
| Uses ice bought from local ice vendor | Chandni Chowk, Tilak Nagar and Rohini | 9/10 | 40 (80) | 0.11 |
| Uses ice from own source of water | Kamla Nagar | 5/10 | 10 (20) | 0.12 |
| Do not use mosquito spray | Chandni Chowk | 9/10 | 28 (56) | 0.14 |
| Do not use bin | Chandni Chowk | 7/10 | 17 (34) | 0.02 * |
| Uses bin without lid | Kamla Nagar | 4/10 | 11 (22) | 0.41 |
| Tobacco chewing | Chandni Chowk | 10/10 | 32 (64) | 0.03 * |
| Scalp scratching | Chandni Chowk | 3/10 | 11 (22) | 0.97 |
| Itching on body parts | Kamla Nagar and Rohini | 1/10 | 2 (4) | NA |
| Wiping hands with clothing | Chandni Chowk | 8/10 | 21 (42) | 0.05 |
| Sugarcane Juice | Orange Juice | Mix Juice | |||||||
|---|---|---|---|---|---|---|---|---|---|
| E. coli (CFU/mL) | Salmonella (CFU/mL) | Vibrio (CFU/mL) | E. coli (CFU/mL) | Salmonella (CFU/mL) | Vibrio (CFU/mL) | E. coli (CFU/mL) | Salmonella (CFU/mL) | Vibrio (CFU/mL) | |
| Kamla Nagar | 3.5 × 104 (±39,393.5) | 5.5 × 104 (±33,842.7) | 6.3 × 102 (±664.2) | 1.3 × 104 (±12,834.7) | 0 | 4.4 × 103 (±3413.6) | 5.7 × 104 (±61,675.7) | 4.8 × 104 (±66,468.0) | 0 |
| DU (North campus) | 9.7 × 104 (±58,488.6) | 6.3 × 104 (±8838.8) | 8.2 × 102 (±56.5) | 1.6 × 104 (±2886.7) | 1.1 × 104 (±12,288.2) | 1 × 103 (±1979.8) | 1.3 × 104 (±1044.03) | 7.3 × 103 (±318.19) | 4.3 × 103 (±296.9) |
| Chandni Chowk | 2 × 105 (±62,010.7) | 9.3 × 104 (±49,369.3) | 4.8 × 103 (±2709.8) | 1.7 × 105 (±127,719) | 1.8 × 105 (±111,519) | 1.5 × 103 (±494.9) | 1.7 × 105 (±158,104) | 2.2 × 105 (±127,899) | 1.4 × 103 (±253.8) |
| Tilak Nagar | 4 × 104 (±2309.4) | 1.6 × 105 (±133,180) | 1.1 × 104 (±15,044.3) | 3.7 × 104 (±1778.5) | 7.5 × 104 (±84,902.3) | 7.7 × 104 (±87,726.6) | 1.7 × 105 (±150,414) | 1.1 × 105 (±75,438.7) | 4.5 × 104 (±36,693.7) |
| Rohini | 1.8 × 105 (±216,569) | 8.4 × 104 (±118,392) | 3.2 × 103 (±3364.5) | 6.1 × 104 (±32,896.1) | 9.6 × 104 (±47,619.2) | 3 × 104 (±2474.8) | 2.5 × 104 (±20,297.7) | 2.6 × 104 (±4596.1) | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharma, N.; Singh, K.; Toor, D.; Pai, S.S.; Chakraborty, R.; Khan, K.M. Antibiotic Resistance in Microbes from Street Fruit Drinks and Hygiene Behavior of the Vendors in Delhi, India. Int. J. Environ. Res. Public Health 2020, 17, 4829. https://doi.org/10.3390/ijerph17134829
Sharma N, Singh K, Toor D, Pai SS, Chakraborty R, Khan KM. Antibiotic Resistance in Microbes from Street Fruit Drinks and Hygiene Behavior of the Vendors in Delhi, India. International Journal of Environmental Research and Public Health. 2020; 17(13):4829. https://doi.org/10.3390/ijerph17134829
Chicago/Turabian StyleSharma, Neha, Kamalpreet Singh, Devinder Toor, Somnath S. Pai, Rishika Chakraborty, and Khalid M. Khan. 2020. "Antibiotic Resistance in Microbes from Street Fruit Drinks and Hygiene Behavior of the Vendors in Delhi, India" International Journal of Environmental Research and Public Health 17, no. 13: 4829. https://doi.org/10.3390/ijerph17134829
APA StyleSharma, N., Singh, K., Toor, D., Pai, S. S., Chakraborty, R., & Khan, K. M. (2020). Antibiotic Resistance in Microbes from Street Fruit Drinks and Hygiene Behavior of the Vendors in Delhi, India. International Journal of Environmental Research and Public Health, 17(13), 4829. https://doi.org/10.3390/ijerph17134829





