Prevalence of Seasonal Influenza Vaccination in Chronic Obstructive Pulmonary Disease (COPD) Patients in the Balearic Islands (Spain) and Its Effect on COPD Exacerbations: A Population-Based Retrospective Cohort Study
Abstract
1. Introduction
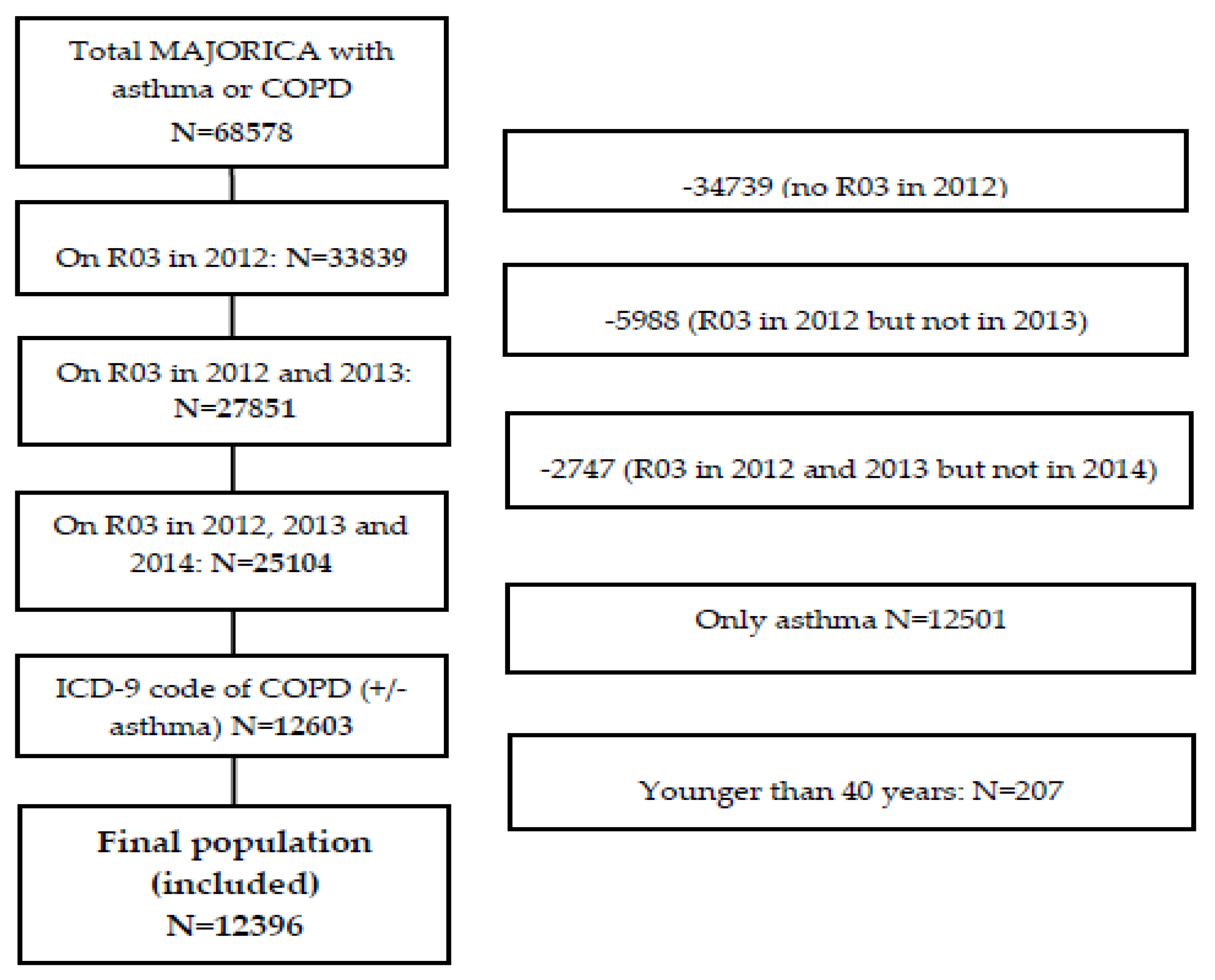
2. Materials and Methods
3. Results
3.1. Population Characteristics
3.2. Prevalence and Characteristics of Influenza Vaccination
3.3. Effects of Influenza Vaccination on COPD Exacerbations
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- From the Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD). 2019. Available online: http://goldcopd.org (accessed on 22 May 2019).
- Miravitlles, M.; Soler-Cataluña, J.J.; Calle, M.; Molina, J.; Almagro, P.; Quintano, J.A.; Trigueros, J.A.; Cosío, B.G.; Casanova, C.; Riesco, J.A.; et al. Spanish COPD guidelines (GesEPOC) 2017. Pharmacological treatment of stable chronic obstructive pulmonary disease. Arch. Bronconeumol. 2017, 53, 324–335. [Google Scholar] [CrossRef] [PubMed]
- Anzueto, A. Impact of exacerbations on COPD. Eur. Respir. Rev. 2010, 19, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Wedzicha, J.A.; Seemungal, T.A. COPD exacerbations: Defining their cause and prevention. Lancet 2007, 370, 786–796. [Google Scholar] [CrossRef]
- Hoogendoorn, M.; Feenstra, T.L.; Hoogenveen, R.T.; Al, M.; Mölken, M.R. Association between lung function and exacerbation frequency in patients with COPD. Int. J. Chron. Obstruct. Pulmon. Dis. 2010, 5, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Sanei, F.; Wilkinson, T. Influenza vaccination for patients with chronic obstructive pulmonary disease: Understanding immunogenicity, efficacy and effectiveness. Ther. Adv. Respir. Dis. 2016, 10, 349–367. [Google Scholar] [CrossRef] [PubMed]
- Lall, D.; Cason, E.; Pasquel, F.J.; Ali, M.K.; Narayan, K.V. Effectiveness of influenza vaccination for individuals with chronic obstructive pulmonary disease (COPD) in low-and middle-income countries. COPD J. Chronic Obstr. Pulm. Dis. 2016, 13, 93–99. [Google Scholar] [CrossRef]
- Kopsaftis, Z.; Wood-Baker, R.; Poole, P. Influenza vaccine for chronic obstructive pulmonary disease (COPD). Cochrane Database Syst. Rev. 2018, CD002733. [Google Scholar] [CrossRef]
- Bekkat-Berkani, R.; Wilkinson, T.; Buchy, P.; Dos Santos, G.; Stefanidis, D.; Devaster, J.M.; Meyer, N. Seasonal influenza vaccination in patients with COPD: A systematic literature review. BMC Pulm. Med. 2017, 17, 79. [Google Scholar] [CrossRef]
- World Health Organization. Vaccines against influenza WHO position paper—November 2012. Wkly Epidemiol. Rec. 2012, 87, 461–476. [Google Scholar]
- European Centre for Disease Prevention and Control (ECDC). Seasonal influenza vaccination in Europe. In Overview of Vaccination Recommendations and Coverage Rates in the EU Member States for the 2012–13 Influenza Season, Technical Report; European centre for disease prevention and control: Stockholm, Sweden, 2015. [Google Scholar]
- Harper, S.A.; Fukuda, K.; Uyeki, T.M.; Cox, N.J.; Bridges, C.B. Advisory Committee onImmunization Practices (ACIP) Centers for Disease Control and Prevention. Pre-vention and control of influenza: Recommendations of the Advisory Committeeon Immunization Practices (ACIP). MMWR Recomm. Rep. 2005, 54, 1–40. [Google Scholar]
- Plans-Rubió, P. Prevention and control of influenza in persons with chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2007, 2, 41–53. [Google Scholar] [CrossRef]
- Jiménez-García, R.; Ariñez-Fernandez, M.C.; Garcia-Carballo, M.; Hernández-Barrera, V.; de Miguel, A.G.; Carrasco-Garrido, P. Influenza vaccination coverage and related factors among Spanish patients with chronic obstructive pulmonary disease. Vaccine 2005, 23, 3679–3686. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-García, R.; Ariñez-Fernandez, M.C.; Hernández-Barrera, V.; Garcia-Carballo, M.M.; de Miguel, A.G.; Carrasco-Garrido, P. Compliance with influenza and pneumococcal vaccination among patients with chronic obstructive pulmonary disease consulting their medical practitioners in Catalonia, Spain. J. Infect. 2007, 54, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Montserrat-Capdevila, J.; Godoy, P.; Marsal, J.R.; Cruz, I.; Solanes, M. Effectiveness of influenza vaccination in preventing hospital admission due to exacerbations of chronic obstructive pulmonary disease. Enferm. Infecc. Microbiol. Clin. 2014, 32, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Garrastazu, R.; García-Rivero, J.L.; Ruiz, M.; Helguera, J.M.; Arenal, S.; Bonnardeux, C.; León, C.; Llorca, J.; Santibañez, M. Prevalence of Influenza Vaccination in Chronic Obstructive Pulmonary Disease Patients and Impact on the Risk of Severe Exacerbations. Arch. Bronconeumol. 2016, 52, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Hsu, D.J.; North, C.M.; Brode, S.K.; Celli, B.R. Identification of Barriers to Influenza Vaccination in Patients with Chronic Obstructive Pulmonary Disease: Analysis of the 2012 Behavioral Risk Factors Surveillance System. Chronic Obstr. Pulm. Dis. 2016, 3, 620–627. [Google Scholar] [CrossRef]
- Vandenbroucke, J.P.; E Von Elm, D.G.; Altman, P.C.; Gotzsche, C.D.; Mulrow, S.J.; Pocock, C.; Poole, J.J. Schlesselman, and M.; Egger, Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): Explanation and elaboration. Ann. Intern. Med. 2007, 147, 163–194. [Google Scholar] [CrossRef]
- van Boven, J.F.; Roman-Rodriguez, M.; Kocks, J.W.; Soriano, J.B.; Postma, M.J.; van der Molen, T. Predictors of cost-effectiveness of selected COPD treatments in primary care: UNLOCK study protocol. NPJ Prim. Care Respir. Med. 2015, 25, 15051. [Google Scholar] [CrossRef]
- van Boven, J.F.; Roman-Rodriguez, M.; Palmer, J.F.; Toledo-Pons, N.; Cosio, B.G.; Soriano, J.B. Comorbidome, pattern, and impact of asthma-COPD overlap syndrome in real life. Chest 2016, 149, 1011–1020. [Google Scholar] [CrossRef]
- Toledo-Pons, N.; van Boven, J.F.M.; Román-Rodríguez, M.; Pérez, N.; Valera Felices, J.L.; Soriano, J.B.; Cosío, B.G. ACO: Time to move from the description of different phenotypes to the treatable traits. PLoS ONE 2019, 14, e0210915. [Google Scholar]
- Hurst, J.R.; Vestbo, J.; Anzueto, A.; Locantore, N.; Müllerova, H.; Tal-Singer, R.; Miller, B.; Lomas, D.A.; Agusti, A.; MacNee, W.; et al. Susceptibility to exacerbation in chronic obstructive pulmonary disease. N. Engl. J. Med. 2010, 363, 1128–1138. [Google Scholar] [CrossRef] [PubMed]
- Wedzicha, J.A.; Brill, S.E.; Allinson, J.P.; Donaldson, G.C. Mechanisms and impact of the frequent exacerbator phenotype in chronic obstructive pulmonary disease. BMC Med. 2013, 11, 181. [Google Scholar] [CrossRef] [PubMed]
- Instituto de Salud Carlos, I.I.I. Informe de Vigilancia de la Gripe en España. Temporada 2012–2013 (Desde la Semana 40/2012 Hasta la Semana 20/2013). Sistema de Vigilancia de la Gripe en España. Available online: http://vgripe.isciii.es/gripe/documentos/20122013/InformesAnuales/Informe_Vigilancia_GRIPE_2012-13_18sep2013.pdf (accessed on 22 May 2019).
- Rabe, K.F.; Hurd, S.; Anzueto, A.; Barnes, P.J.; Buist, S.A.; Calverley, P.; Fukuchi, Y.; Jenkins, C.; Rodriguez-Roisin, R.; Van Weel, C.; et al. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am. J. Respir. Crit. Care Med. 2013, 187, 347–365. [Google Scholar] [CrossRef]
- Poole, P.; Chacko, E.; Wood-Baker, R.; Cates, C. Influenza vaccine for patients with chronic obstructive pulmonary disease. Cochrane Database Syst. Rev. 2006, CD002733. [Google Scholar] [CrossRef] [PubMed]
- Instituto de Salud Carlos, I.I.I. Informe de Vigilancia de la Gripe en España. Temporada 2011–2012 (Desde la semana 40/2011 hasta la semana 20/2012). Sistema de Vigilancia de la Gripe en España. Available online: http://vgripe.isciii.es/gripe/documentos/20112012/InformesAnuales/Informe GRIPETemporada2011-12 v.3septiembre2012.pdf (accessed on 22 May 2019).
- Center for Biologics Evaluation and Research (CBER). Guidance for Industry: Clinical Data Needed to Support the Licensure of Seasonal Inactivated Influenza Vaccines; Food and drug administration (FDA): Silver Spring, MD, USA, 2007.
- Committee for Proprietary Medicinal Products (CPMP). Note for Guidance on Harmonisation of Requirements for Influenza Vaccines; The European agency for the evaluation of medicinal products: London, UK, 1997.
- Hovden, A.O.; Cox, R.J.; Haaheim, L.R. Influenza: The virus and prophylaxis with inactivated influenza vaccine in ‘at risk’ groups, including COPD patients. Int. J. Chron. Obstruct. Pulmon. Dis. 2007, 2, 229–240. [Google Scholar] [PubMed]
- Yip, N.H.; Yuen, G.; Lazar, E.J.; Regan, B.K.; Brinson, M.D.; Taylor, B.; George, L.; Karbowitz, S.R.; Stumacher, R.; Schluger, N.W.; et al. Analysis of hospitalizations for COPD exacerbation: Opportunities for improving care. COPD 2010, 7, 85–92. [Google Scholar] [CrossRef]
- Nicholson, K.G.; Kent, J.; Hammersley, V. Influenzae A among community-dwelling elderly persons in Leicestershine during Winter 1993–4; cigarrette smoking as a risk factor and the efficacy of influenzae vaccination. Epidemiol. Infect. 1999, 123, 103–108. [Google Scholar] [CrossRef]
- Santos-Sancho, J.M.; Jimenez-Trujillo, I.; Hernández-Barrera, V.; López-de Andrés, A.; Carrasco-Garrido, P.; Ortega-Molina, P.; Jimenez-Garcia, R. Influenza vaccination coverage uptake predictors among Spanish adults suffering, COPD. Hum. Vaccin. Immunother. 2012, 8, 938–945. [Google Scholar] [CrossRef]
- Ting, S.C.; Crooks, S.W.; South, G. The effect of influenza vaccination on the inci-dence of chronic obstructive pulmonary disease exacerbations in the immediatepostvaccination period. J. Epidemiol. Commun. Health 2011, 65, 157–159. [Google Scholar] [CrossRef]
- Tata, L.J.; West, J.; Harrison, T.; Farrington, P.; Smith, C.; Hubbard, R. Doesinfluenza vaccination increase consultations, corticosteroid prescriptions, orexacerbations in subjects with asthma or COPD. Thorax 2003, 58, 835–839. [Google Scholar] [CrossRef]
- Glasgow, R.E.; Magid, D.J.; Beck, A.; Ritzwoller, D.; Estabrooks, P.A. Practical clinical trials for translating research to practice: Design and measurement recommendations. Med. Care 2005, 43, 551–557. [Google Scholar] [CrossRef] [PubMed]
- Julian, D.G.; Pocock, S.J. Interpreting a Trial Report; WB Sanders: London, UK, 1997. [Google Scholar]
- Persaud, N.; Mamdani, M.M. External validity: The neglected dimension in evidence ranking. J. Eval. Clin. Pract. 2006, 12, 450–453. [Google Scholar] [CrossRef] [PubMed]
| Unvaccinated | Vaccinated | Total | |||||
|---|---|---|---|---|---|---|---|
| N = 5003 | % Row | N = 7393 | % Row | N = 12,396 | % column | p Value | |
| Sex | |||||||
| Women | 1884 | 46.4% | 2174 | 53.6% | 4058 | 32.7% | <0.001 |
| Men | 3119 | 37.4% | 5219 | 62.6% | 8338 | 67.3% | |
| Age | |||||||
| Mean [SD] | 66.56 | 11.52 | 71.884 | 10.08 | 69.735 | 11 | <0.001 |
| BMI | |||||||
| Mean [SD] | 29.93 | 6.38 | 30.17 | 7.94 | 30.09 | 7.42 | 0.003 |
| Normal weight (18.5–24.9) | 594 | 41.1% | 850 | 58.9% | 1444 | 16.90% * | <0.001 |
| Overweight (25–29.9) | 1045 | 34.1% | 2019 | 65.9% | 3064 | 35.80% | |
| Obesity (≥30) | 1381 | 34.8% | 2583 | 65.2% | 3964 | 46.30% | |
| Underweight (<18.5) | 43 | 48.9% | 45 | 51.1% | 88 | 1.00% | |
| Missing values | 1940 | 50.6% | 1896 | 49.4% | 3836 | 30.9% | |
| BMI > 21 | 2903 | 35.3% | 5311 | 64.7% | 8214 | 96.00% | <0.001 |
| BMI ≤21 | 160 | 46.2% | 186 | 53.8% | 346 | 4.00% | |
| Missing values | 1940 | 50.6% | 1896 | 49.4% | 3836 | 30.9% | |
| Smoking habit | |||||||
| Never | 1233 | 34.2% | 2368 | 65.8% | 3601 | 32.30% * | <0.001 |
| Former | 1439 | 32.9% | 2930 | 67.1% | 4369 | 39.20% | |
| Current | 1735 | 54.6% | 1443 | 45.4% | 3178 | 28.50% | |
| Missing values | 596 | 47.8% | 652 | 52.2% | 1248 | 10.1% | |
| FEV1 | |||||||
| Mean [SD] | 64.19% | 19.56 | 61.74% | 19.79 | 62.66% | 19.74 | <0.001 |
| Obstruction severity (FEV1) | |||||||
| GOLD 1 (≥80%) | 467 | 42.5% | 633 | 57.53% | 1100 | 19.60% * | <0.001 |
| GOLD 2 (≥50–80%) | 1133 | 37.8% | 1862 | 62.2% | 2995 | 53.40% | |
| GOLD 3 (≥30–49.9%) | 449 | 33.7% | 884 | 66.3% | 1333 | 23.80% | |
| GOLD 4 (<30%) | 64 | 35.0% | 119 | 65.0% | 183 | 3.30% | |
| Missing values | 2890 | 42.6% | 3895 | 57.4% | 6785 | 54.7% | |
| History of moderate exacerbations 2012 | |||||||
| Number of exacerbations, mean [SD] | 0.7 | 1 | 0.84 | 1.09 | 0.78 | 1.06 | <0.001 |
| None (0 exacerbations) | 2800 | 43.0% | 3714 | 57.0% | 6514 | 52.5% | |
| 1 exacerbation | 1332 | 38.9% | 2096 | 61.1% | 3428 | 27.7% | |
| ≥2 exacerbations | 871 | 35.5% | 1583 | 64.5% | 2454 | 19.8% | |
| Admitted for COPD exacerbation 2012 | |||||||
| Number of COPD admissions, mean [SD] | 0.08 | 0.34 | 0.1 | 0.39 | 0.09 | 0.37 | 0.003 |
| None (0 COPD admissions) | 4648 | 40.7% | 6763 | 59.3% | 11411 | 92.1% | 0.01 |
| 1 COPD admission | 292 | 36.5% | 509 | 63.5% | 801 | 6.5% | |
| ≥2 severe exacerbations | 63 | 34.2% | 121 | 65.8% | 184 | 1.5% | |
| History of overall exacerbations 2012 | |||||||
| Number of exacerbations, mean [SD] | 0.79 | 1.15 | 0.94 | 1.26 | 0.88 | 1.22 | <0.001 |
| None (0 exacerbations) | 2737 | 43.1% | 3614 | 56.9% | 6351 | 51.2% | <0.001 |
| 1 exacerbation | 1254 | 38.6% | 1993 | 61.4% | 3247 | 26.2% | |
| ≥2 exacerbations | 1012 | 36.2% | 1786 | 63.8% | 2798 | 22.6% | |
| Exacerbator phenotype 2012 | |||||||
| No | 3931 | 41.6% | 5514 | 58.4% | 9445 | 76.2% | <0.001 |
| Yes | 1072 | 36.3% | 1879 | 63.7% | 2951 | 23.8% | |
| Unvaccinated | Vaccinated | Total | |||||
|---|---|---|---|---|---|---|---|
| N = 5003 | % | N = 7393 | % | N = 12,396 | % | p Value | |
| Moderate COPD exacerbations 2013 | |||||||
| Number of exacerbations. | |||||||
| None (0 exacerbations) | 2750 | 55.0% | 3640 | 49.2% | 6390 | 51.5% | <0.001 |
| 1 exacerbation | 1279 | 25.6% | 1943 | 26.3% | 3222 | 26.0% | |
| ≥2 exacerbations | 974 | 19.5% | 1810 | 24.5% | 2784 | 22.5% | |
| Severe COPD exacerbations 2013 | |||||||
| Number of COPD admissions. | |||||||
| None (0 COPD admissions) | 4618 | 92.3% | 6722 | 90.9% | 11340 | 91.5% | 0.003 |
| 1 COPD admission | 320 | 6.4% | 522 | 7.1% | 842 | 6.8% | |
| ≥2 COPD admissions | 65 | 1.3% | 149 | 2.0% | 214 | 1.7% | |
| Overall COPD exacerbations 2013 | |||||||
| Number of exacerbations. | |||||||
| None (0 exacerbations) | 2701 | 54.0% | 3564 | 48.2% | 6265 | 50.5% | <0.001 |
| 1 exacerbation | 1187 | 23.7% | 1815 | 24.6% | 3002 | 24.2% | |
| ≥2 exacerbations | 1115 | 22.3% | 2014 | 27.2% | 3129 | 25.2% | |
| Exacerbator phenotype 2013 | |||||||
| No | 3842 | 76.8% | 5311 | 71.8% | 9153 | 73.8% | <0.001 |
| Yes | 1161 | 23.2% | 2082 | 28.2% | 3243 | 26.2% | |
| Severe Exacerbations (Admitted for COPD Exacerbation) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2013 Complete Year | 2013 Epidemic Period Only | |||||||||||
| None | At Least One | None | At Least One | |||||||||
| N= 11,340 | N = 1056 | OR | (95% CI) | ORa | (95% CI) | N = 11,997 | N = 399 | OR | (95% CI) | ORa | (95% CI) | |
| All COPD | ||||||||||||
| (N = 12,396) | ||||||||||||
| Unvaccinated | 4618 | 385 | 1 | -- | 1 | -- | 4846 | 157 | 1 | -- | 1 | -- |
| Vaccinated | 6722 | 671 | 1.20 | (1.05–1.37) | 0.93 | (0.74–1.18) | 7151 | 242 | 1.05 | (0.85–1.28) | 0.82 | (0.58–1.16) |
| GOLD 1 (N = 1100) | ||||||||||||
| Unvaccinated | 448 | 19 | 1 | -- | 1 | -- | 461 | 6 | 1 | -- | 1 | -- |
| Vaccinated | 603 | 30 | 1.17 | (0.65–2.11) | 0.87 | (0.45–1.69) | 623 | 10 | 1.23 | (0.45–3.42) | 0.94 | (0.31–2.84) |
| GOLD 2 (N = 2995) | ||||||||||||
| Unvaccinated | 1050 | 83 | 1 | -- | 1 | -- | 1095 | 38 | 1 | -- | 1 | -- |
| Vaccinated | 1741 | 121 | 0.88 | (0.66–1.18) | 0.72 | (0.52–1.00) | 1813 | 49 | 0.78 | 0.51–1.20 | 0.71 | 0.44–1.16) |
| GOLD 3 (N = 1333) | ||||||||||||
| Unvaccinated | 398 | 51 | 1 | -- | 1 | -- | 428 | 21 | 1 | -- | 1 | -- |
| Vaccinated | 757 | 127 | 1.31 | (0.93–1.85) | 1.07 | (0.71–1.61) | 842 | 42 | 1.02 | (0.59–1.74) | 0.78 | (0.42–1.44) |
| GOLD 4 (N = 183) | ||||||||||||
| Unvaccinated | 55 | 9 | 1 | -- | 1 | -- | 60 | 4 | 1 | -- | 1 | -- |
| Vaccinated | 91 | 28 | 1.88 | (0.83–4.28) | 6.09 | (1.77–20.97) | 109 | 10 | 1.38 | (0.41–4.58) | 2.17 | 0.40–11.73 |
| Moderate Exacerbations | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 2013 Complete Year | 2013 Epidemic Period Only | |||||||||||
| None | At Least One | None | At Least One | |||||||||
| N = 6390 | N = 6006 | OR | (95% CI) | ORa | (95% CI) | N = 9345 | N = 3045 | OR | (95% CI) | ORa | (95% CI) | |
| Influenza Vaccination (N = 12,396) | ||||||||||||
| Unvaccinated | 2750 | 2253 | 1 | -- | 1 | -- | 3891 | 1112 | 1 | -- | 1 | -- |
| Vaccinated | 3640 | 3753 | 1.26 | (1.17–1.35) | 1.10 | (0.97–1.24) | 5460 | 1933 | 1.24 | (1.14–1.35) | 1.11 | (0.96–1.28) |
| GOLD 1 (N = 1100) | ||||||||||||
| Unvaccinated | 285 | 182 | 1 | -- | 1 | -- | 383 | 84 | 1 | -- | 1 | -- |
| Vaccinated | 329 | 304 | 1.45 | (1.14–1.84) | 1.21 | (0.91–1.61) | 475 | 158 | 1.52 | (1.13–2.04) | 1.33 | (0.95–1.86) |
| GOLD 2 (N = 2995) | ||||||||||||
| Unvaccinated | 648 | 485 | 1 | -- | 1 | -- | 892 | 241 | 1 | -- | 1 | -- |
| Vaccinated | 964 | 898 | 1.25 | (1.07–1.44) | 1.11 | (0.93–1.31) | 1404 | 458 | 1.21 | (1.01–1.44) | 1.05 | (0.86–1.28) |
| GOLD 3 (N = 1333) | ||||||||||||
| Unvaccinated | 203 | 246 | 1 | -- | 1 | -- | 323 | 126 | 1 | -- | 1 | -- |
| Vaccinated | 388 | 496 | 1.06 | (0.84–1.33) | 0.84 | (0.65–1.09) | 606 | 278 | 1.18 | (0.92–1.51) | 1.00 | (0.75–1.33) |
| GOLD 4 (N = 183) | ||||||||||||
| Unvaccinated | 37 | 27 | 1 | -- | 1 | -- | 47 | 17 | 1 | -- | 1 | -- |
| Vaccinated | 45 | 74 | 2.25 | (1.21–4.19) | 4.54 | (1.77–11.67) | 77 | 42 | 1.51 | (0.77–2.95) | 3.93 | 1.39–11.13 |
| 2013 Frequent Exacerbator Phenotype | ||||||
|---|---|---|---|---|---|---|
| No | Yes | |||||
| N = 9153 | N = 3243 | OR | (95% CI) | ORa | (95% CI) | |
| Influenza Vaccination (2012–2013 Campaign) | ||||||
| Total (N= 12,396) | ||||||
| Unvaccinated | 3842 | 1161 | 1 | -- | 1 | -- |
| Vaccinated | 5311 | 2082 | 1.30 | (1.19–1.41) | 1.02 | (0.88–1.19) |
| GOLD 1 (N = 1100) | ||||||
| Unvaccinated | 384 | 83 | 1 | -- | 1 | -- |
| Vaccinated | 488 | 145 | 1.38 | (1.02–1.86) | 1.15 | (0.80–1.63) |
| GOLD 2 (N = 2995) | ||||||
| Unvaccinated | 876 | 257 | 1 | -- | 1 | -- |
| Vaccinated | 1409 | 453 | 1.10 | (0.92–1.31) | 0.91 | (0.74–1.12) |
| GOLD 3 (N = 1333) | ||||||
| Unvaccinated | 315 | 134 | 1 | -- | 1 | -- |
| Vaccinated | 569 | 315 | 1.30 | (1.02–1.66) | 1.03 | (0.77–1.39) |
| GOLD 4 (N = 183) | ||||||
| Unvaccinated | 41 | 23 | 1 | -- | 1 | -- |
| Vaccinated | 65 | 54 | 1.48 | (0.79–2.77) | 3.72 | (1.48–9.33) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz Azcona, L.; Roman-Rodriguez, M.; Llort Bove, M.; van Boven, J.F.; Santibáñez Margüello, M. Prevalence of Seasonal Influenza Vaccination in Chronic Obstructive Pulmonary Disease (COPD) Patients in the Balearic Islands (Spain) and Its Effect on COPD Exacerbations: A Population-Based Retrospective Cohort Study. Int. J. Environ. Res. Public Health 2020, 17, 4027. https://doi.org/10.3390/ijerph17114027
Ruiz Azcona L, Roman-Rodriguez M, Llort Bove M, van Boven JF, Santibáñez Margüello M. Prevalence of Seasonal Influenza Vaccination in Chronic Obstructive Pulmonary Disease (COPD) Patients in the Balearic Islands (Spain) and Its Effect on COPD Exacerbations: A Population-Based Retrospective Cohort Study. International Journal of Environmental Research and Public Health. 2020; 17(11):4027. https://doi.org/10.3390/ijerph17114027
Chicago/Turabian StyleRuiz Azcona, Laura, Miguel Roman-Rodriguez, Montserrat Llort Bove, Job FM van Boven, and Miguel Santibáñez Margüello. 2020. "Prevalence of Seasonal Influenza Vaccination in Chronic Obstructive Pulmonary Disease (COPD) Patients in the Balearic Islands (Spain) and Its Effect on COPD Exacerbations: A Population-Based Retrospective Cohort Study" International Journal of Environmental Research and Public Health 17, no. 11: 4027. https://doi.org/10.3390/ijerph17114027
APA StyleRuiz Azcona, L., Roman-Rodriguez, M., Llort Bove, M., van Boven, J. F., & Santibáñez Margüello, M. (2020). Prevalence of Seasonal Influenza Vaccination in Chronic Obstructive Pulmonary Disease (COPD) Patients in the Balearic Islands (Spain) and Its Effect on COPD Exacerbations: A Population-Based Retrospective Cohort Study. International Journal of Environmental Research and Public Health, 17(11), 4027. https://doi.org/10.3390/ijerph17114027





