Cardiovascular Disease and the Female Disadvantage
Abstract
1. Introduction
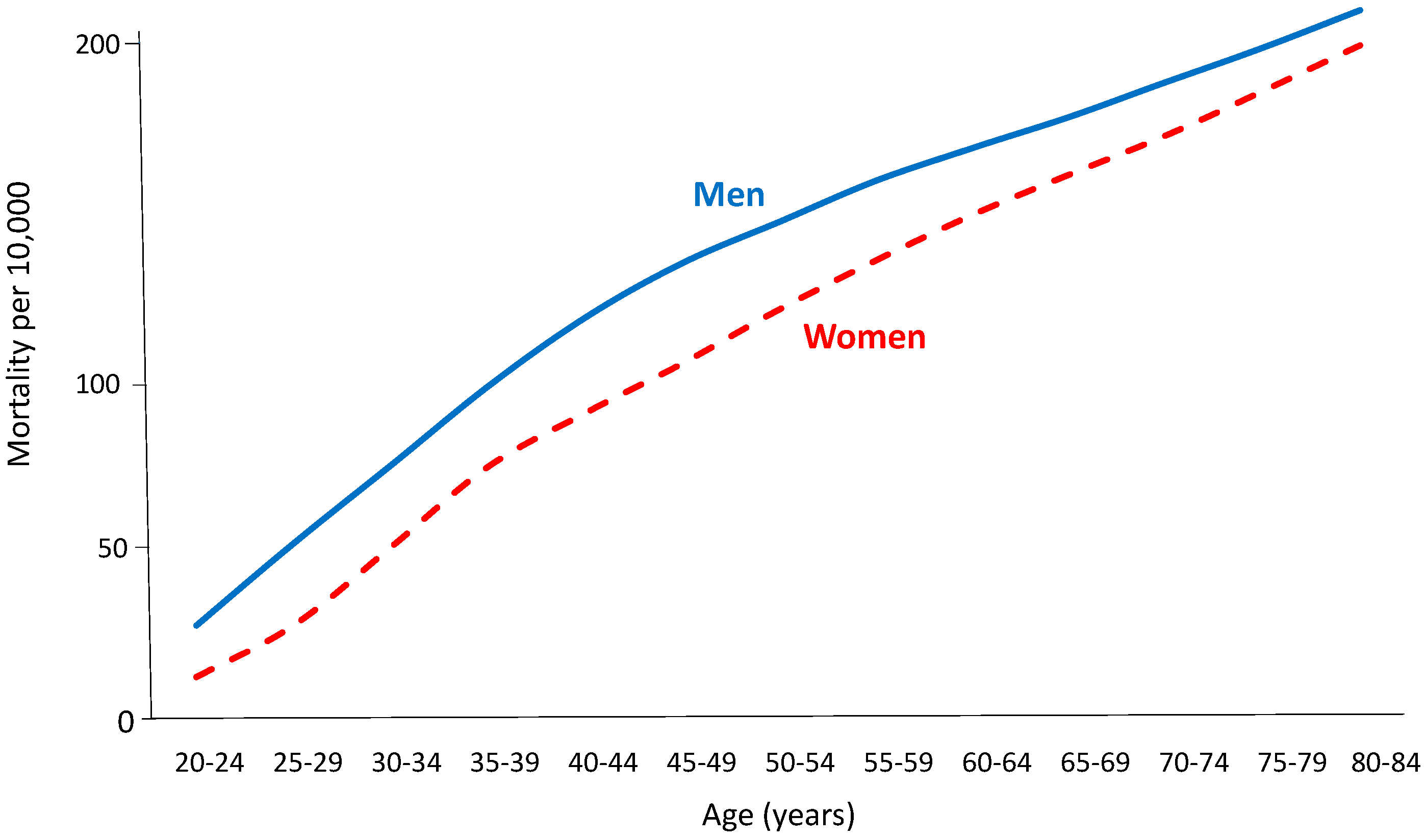
2. The Male Disadvantage
3. Lifetime Risk
4. Manifestations of Female Disadvantage
4.1. Personal Health Care
- <1/2 knew that smoking was a risk factor
- <1/4 named high blood pressure (BP) or cholesterol as risk factors
- <1/3 identified four common symptoms of a heart attack in women
- Of those at high risk (based on medical history and risk factors), 62% rated their risk as low to moderate
- 65% claimed they had most influence on their family’s health.
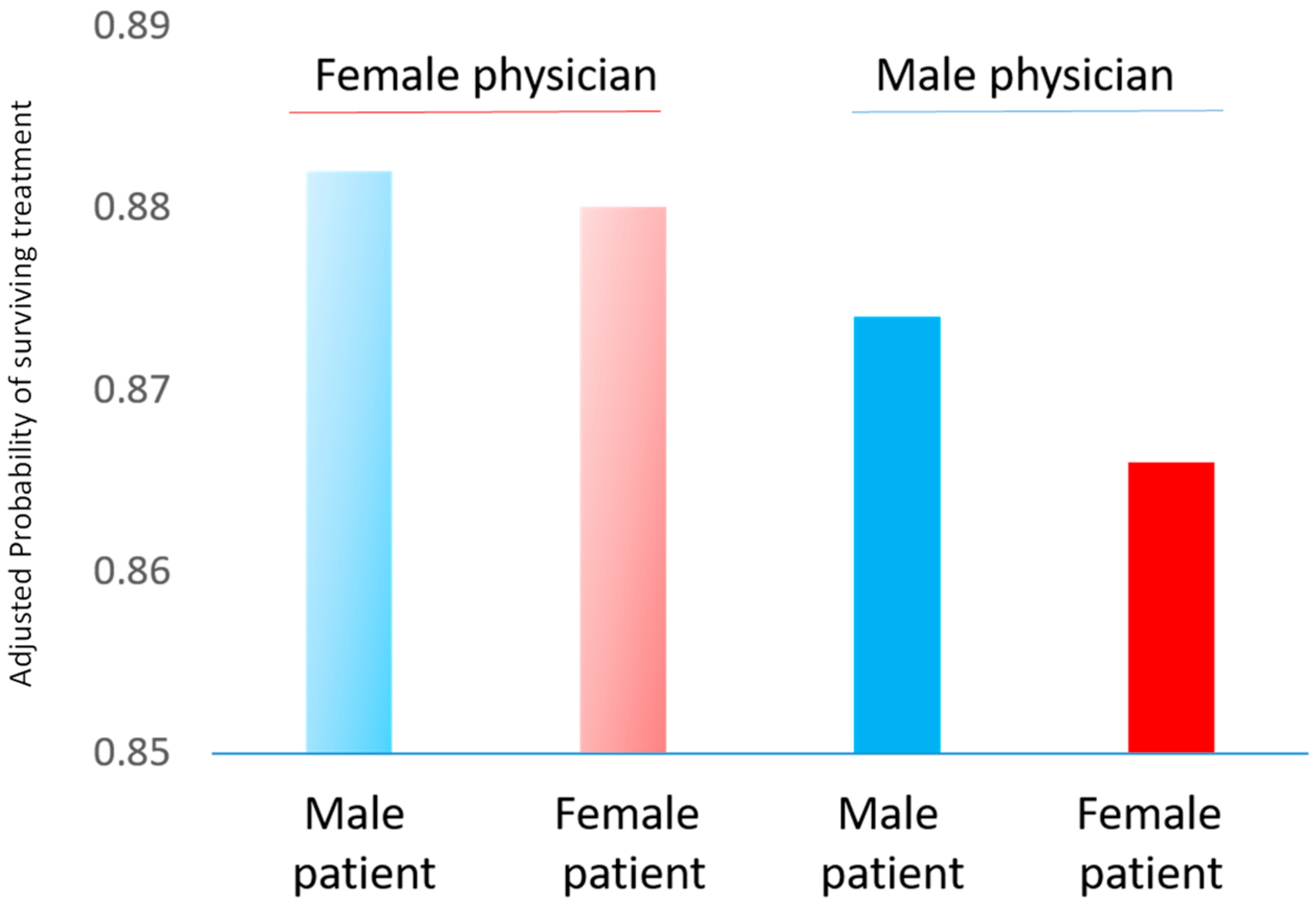
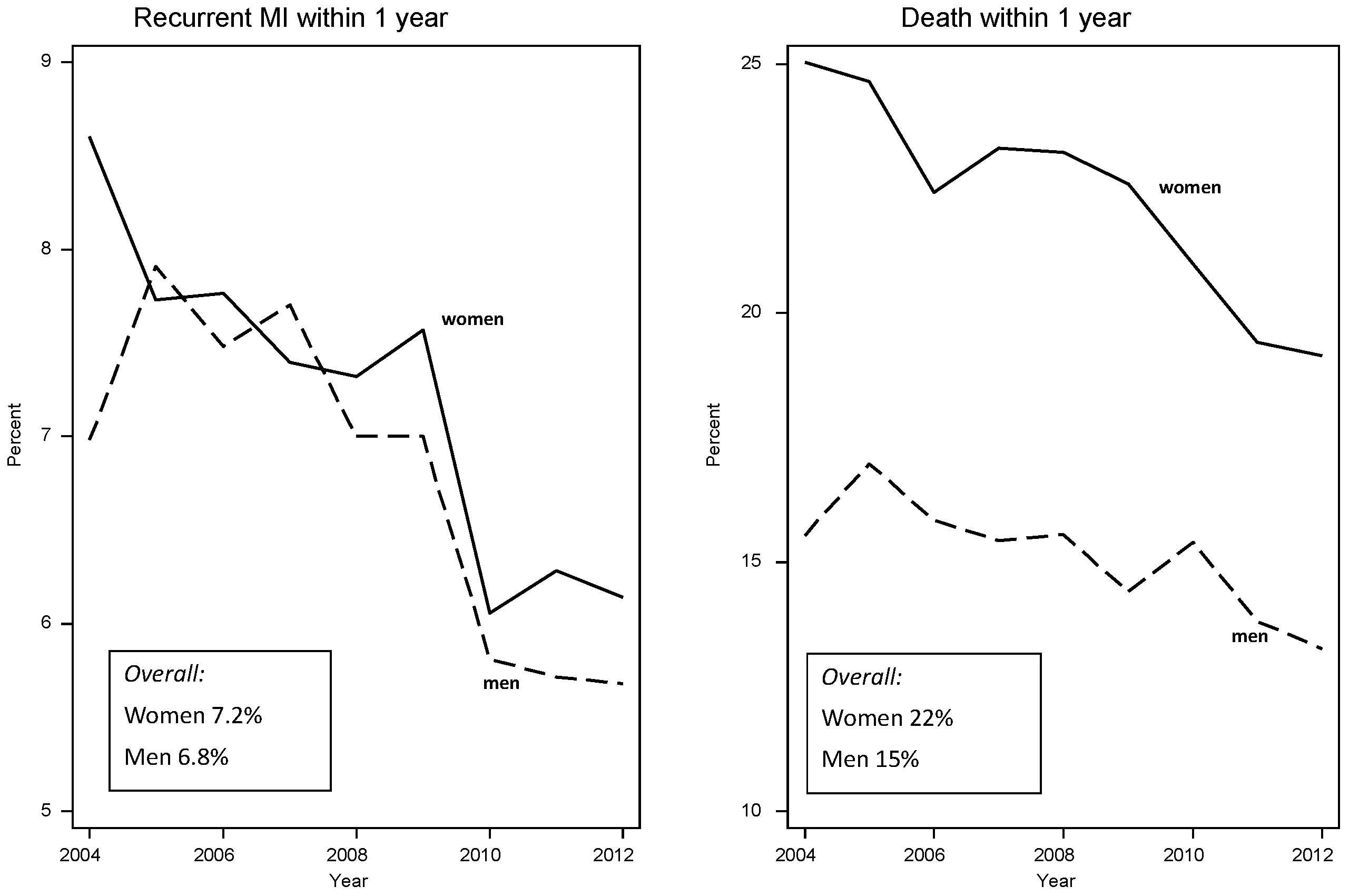
4.2. Professional Health Care
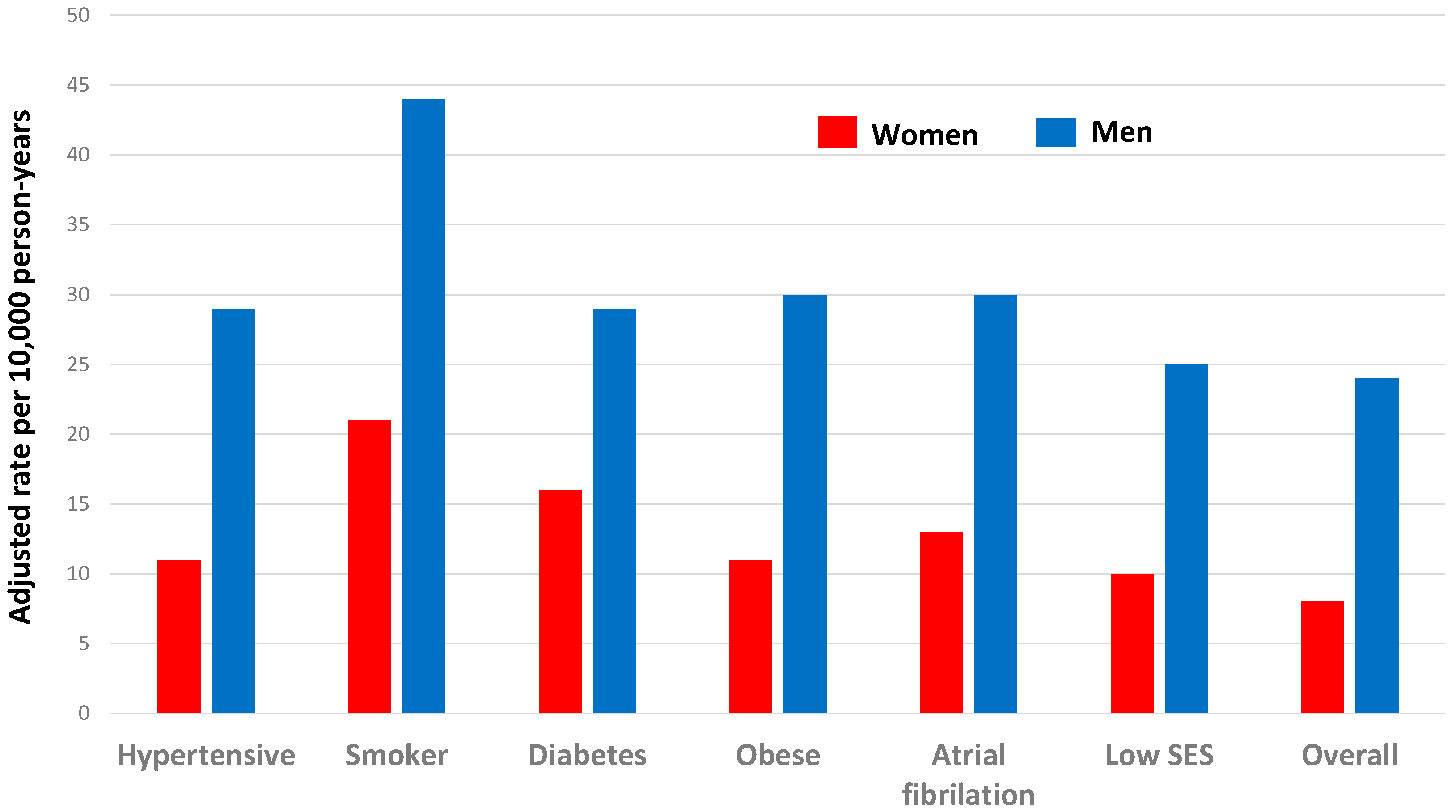
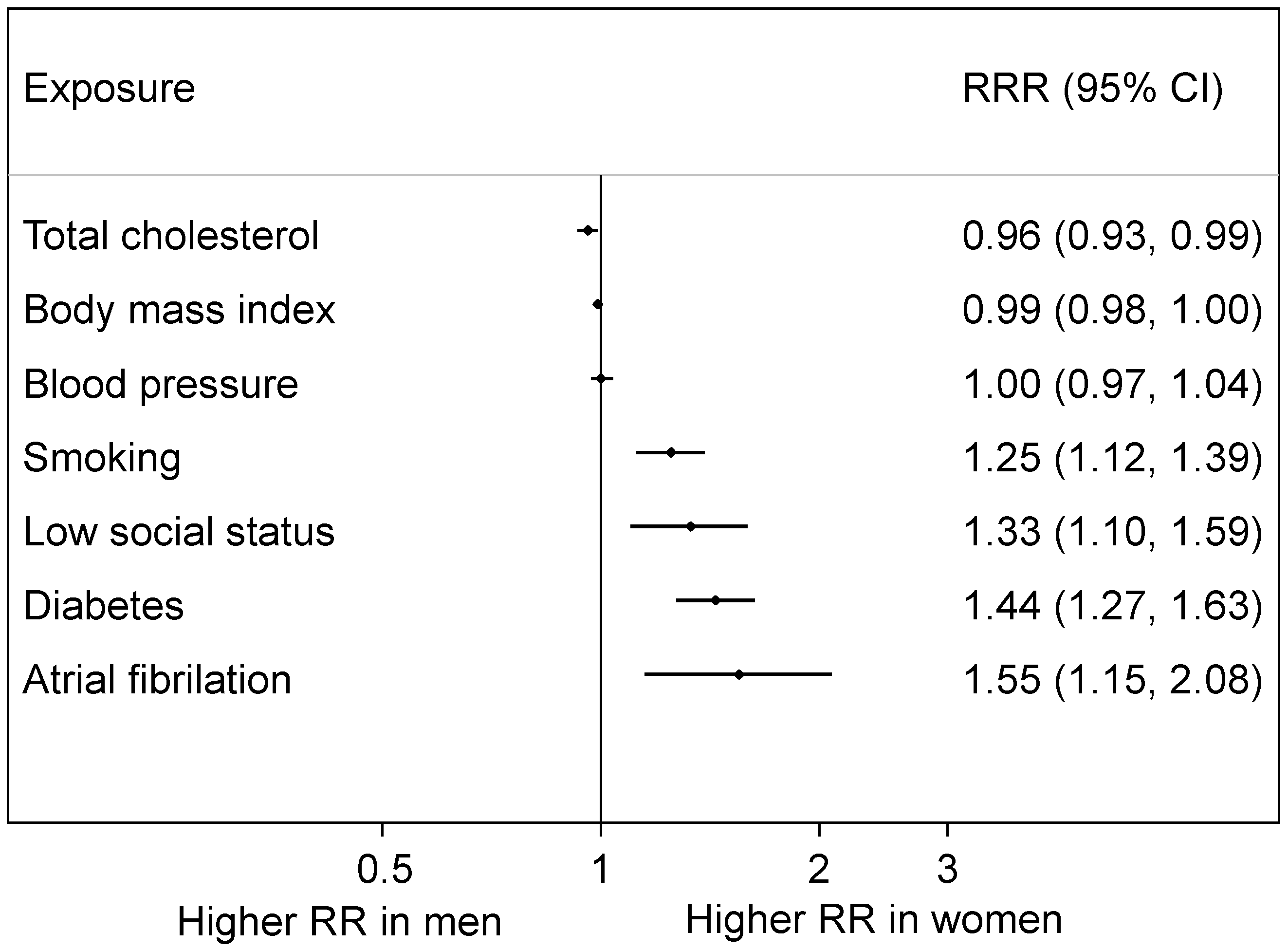
4.3. Risk Factors for both Sexes
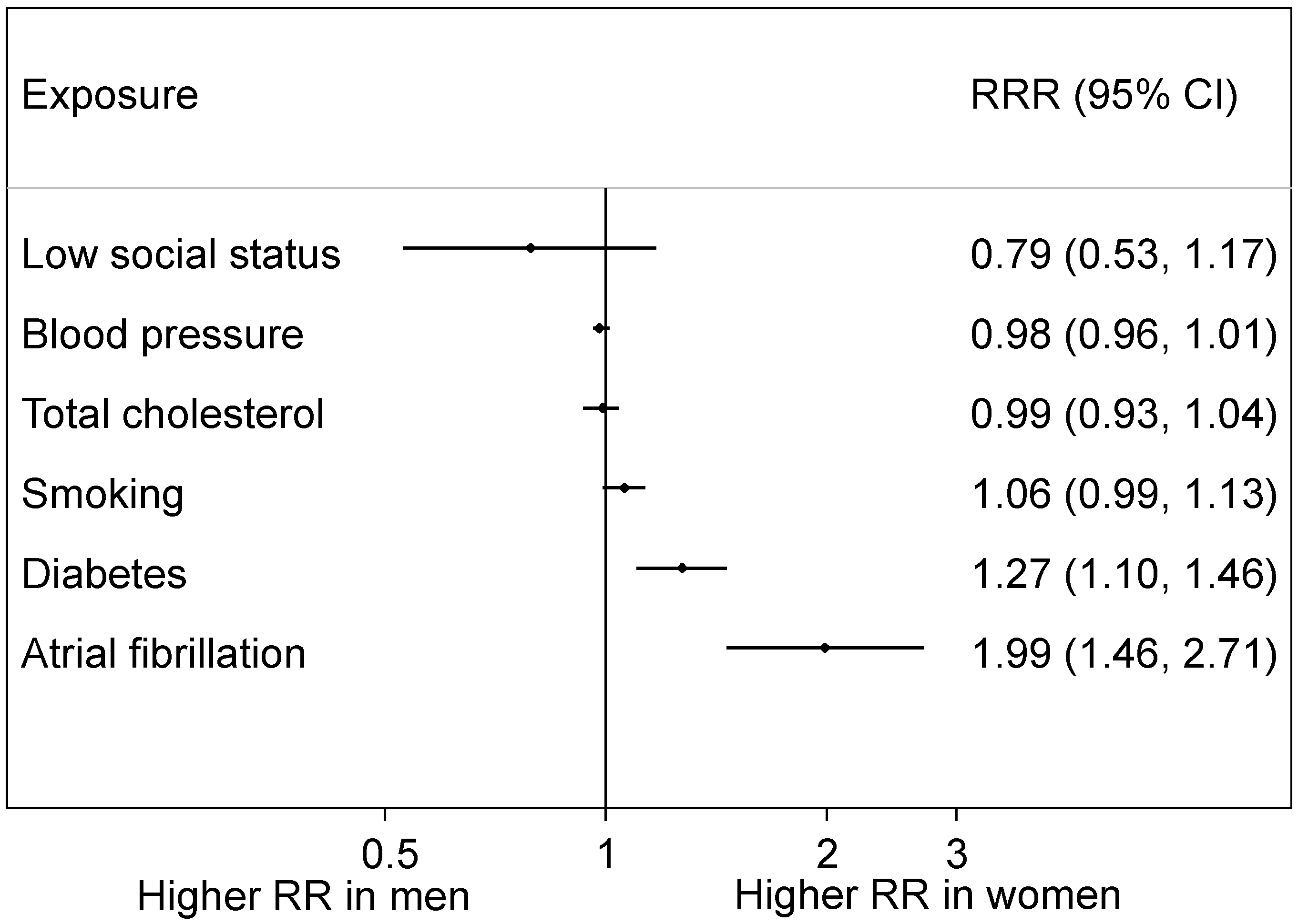
4.4. Female Risk Factors
- Early menarche
- Early menopause
- History of hysterectomy
- Early age at first birth
- History of miscarriage
- History of stillbirth.
5. Discussion
6. Conclusions
- A broader women’s health agenda is needed, integrating sexual and reproductive health with CVD and other non-communicable diseases—taking a lifecourse approach to women’s health
- Sufficient numbers of women should be included in studies of CVD (and more generally)
- A sex-disaggregated approach should be taken to collecting, analysing and reporting CVD (and more generally); given that roughly 50% of the population is female, it seems reasonable that responsible official bodies and journals should mandate that all research (except in extenuating circumstances) should report results by sex
- Where appropriate, male controls should be included in studies of women’s health.
Funding
Acknowledgments
Conflicts of Interest
References
- Available online: https://vizhub.healthdata.org/gbd-compare/ (accessed on 17 March 2019).
- Bots, S.H.; Peters, S.A.E.; Woodward, M. Sex differences in coronary heart disease and stroke mortality: A global assessment of the effect of ageing between 1980 and 2010. BMJ Glob. Health 2017, 2, e000298. [Google Scholar] [CrossRef]
- Millett, E.R.C.; Peters, S.A.E.; Woodward, M. Sex differences in risk factors for myocardial infarction: Cohort study of UK Biobank participants. BMJ 2018, 363, k4247. [Google Scholar] [CrossRef]
- Leening, M.J.; Ferket, B.S.; Steyerberg, E.W.; Kavousi, M.; Deckers, J.W.; Nieboer, D.; Heeringa, J.; Portegies, M.L.; Hofman, A.; Ikram, M.A. Sex differences in lifetime risk and first manifestation of cardiovascular disease: Prospective population based cohort study. BMJ 2014, 349, g5992. [Google Scholar] [CrossRef] [PubMed]
- Lumsden, M.A.; Sassarini, J. The evolution of the human menopause. Climacteric 2019, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Tunstall-Pedoe, H. Myth and paradox of coronary risk and the menopause. Lancet 1998, 351, 1425–1427. [Google Scholar] [CrossRef]
- Boardman, H.M.; Hartley, L.; Eisinga, A.; Main, C.; Roqué i Figuls, M.; Bonfill Cosp, X.; Gabriel Sanchez, R.; Knight, B. Hormone therapy for preventing cardiovascular disease in post-menopausal women. Cochrane Database Syst. Rev. 2015, 3, CD002229. [Google Scholar] [CrossRef]
- McDonnell, L.A.; Pipe, A.L.; Westcott, C.; Perron, S.; Younger-Lewis, D.; Elias, N.; Nooyen, J.; Reid, R.D. Perceived vs. actual knowledge and risk of heart disease in women: Findings from a Canadian survey on heart health awareness, attitudes, and lifestyle. Can. J. Cardiol. 2014, 30, 827–834. [Google Scholar] [CrossRef]
- Lundberg, G.P.; Mehta, L.S.; Sanghani, R.M.; Patel, H.N.; Aggarwal, N.R.; Aggarwal, N.T.; Braun, L.T.; Lewis, S.J.; Mieres, J.H.; Wood, M.J.; et al. Heart Centers for Women. Circulation 2018, 138, 1155–1165. [Google Scholar] [CrossRef]
- Daugherty, S.L.; Blair, I.V.; Havranek, E.P.; Furniss, A.; Dickinson, L.M.; Karimkhani, E.; Main, D.S.; Masoudi, F.A. Implicit gender bias and the use of cardiovascular tests among cardiologists. J. Am. Heart Assoc. 2017, 6, e006872. [Google Scholar] [CrossRef]
- Hyun, K.; Redfern, J.; Patel, A.; Peiris, D.; Brieger, D.; Sullivan, D.; Harris, M.; Usherwood, T.; MacMahon, S.; Lyford, M.; et al. Gender inequalities in cardiovascular risk factor assessment and management in primary healthcare. Heart 2017, 103, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Vaartjes, I.; Graham, I.; Grobbee, D.; Spiering, W.; Klipstein-Grobusch, K.; Woodward, M.; Peters, S.A. Sex differences in risk factor management of coronary heart disease across three regions. Heart 2017, 103, 1587–1594. [Google Scholar] [CrossRef]
- Peters, S.A.E.; Colantonio, L.D.; Zhao, H.; Bittner, V.; Dai, Y.; Farkouh, M.E.; Monda, K.L.; Safford, M.M.; Muntner, P.; Woodward, M. Sex differences in high-intensity statin use following myocardial infarction in the United States. J. Am. Coll. Cardiol. 2018, 71, 1729–1737. [Google Scholar] [CrossRef]
- Wu, J.; Gale, C.P.; Hall, M.; Dondo, T.B.; Metcalfe, E.; Oliver, G.; Batin, P.D.; Hemingway, H.; Timmis, A.; West, R.M. Impact of initial hospital diagnosis on mortality for acute myocardial infarction: A national cohort study. Eur. Heart J. Acute Cardiovasc. Care 2018, 7, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, B.N.; Carnahanb, S.; Huang, L. Patient–physician gender concordance and increased mortality among female heart attack patients. Proc. Natl. Acad. Sci. USA 2018, 115, 8569–8574. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health and Welfare 2018. Transition between Hospital and Community Care for Patients with Coronary Heart Disease: New South Wales and Victoria, 2012–2015; Cat. No. CDK 9; AIHW: Canberra, Australia, 2018.
- Available online: https://www.fda.gov/ScienceResearch/SpecialTopics/WomensHealthResearch/ucm131182.htm#Timeline (accessed on 17 March 2019).
- Clayton, J.A. Studying both sexes: A guiding principle for biomedicine. FASEB J. 2016, 30, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.E.; Unger, E.F.; Jenkins, M.R.; Southworth, M.R.; McDowell, T.Y.; Geller, R.J.; Elahi, M.; Temple, R.J.; Woodcock, J. Participation of women in clinical trials supporting FDA approval of cardiovascular drugs. J. Am. Coll. Cardiol. 2018, 71, 1960–1969. [Google Scholar] [CrossRef] [PubMed]
- Mehta, L.S.; Beckie, T.M.; DeVon, H.A.; Grines, C.L.; Krumholz, H.M.; Johnson, M.N.; Lindley, K.J.; Vaccarino, V.; Wang, T.Y.; Watson, K.E.; et al. Acute myocardial infarction in women: A Scientific Statement from the American Heart Association. Circulation 2016, 133, 916–947. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Q.D.; Peters, E.; Wassef, A.; Desmarais, P.; Rémillard-Labrosse, D.; Tremblay-Gravel, M. Evolution of age and female representation in the most-cited randomized controlled trials of cardiology of the last 20 years. Circ. Cardiovasc. Qual. Outcomes 2018, 11, e004713. [Google Scholar] [CrossRef]
- Carcel, C.; Woodward, M.; Balicki, G.; Koroneos, G.L.; Sousa, D.A.; Cordonnier, C.; Lukaszyk, C.; Thompson, K.; Wang, X.; Davies, L.; et al. Trends in recruitment of women and reporting of sex differences in large-scale published randomised controlled trials in stroke. Int. J. Stroke. in press.
- Peters, S.A.E.; Muntner, P.; Woodward, M. Sex differences in the prevalence of, and trends in, cardiovascular risk factors, treatment, and control in the United States, 2001 to 2016. Circulation 2019, 139, 1025–1035. [Google Scholar] [CrossRef]
- Stevens, G.A.; Singh, G.M.; Lu, Y.; Danaei, G.; Lin, J.K.; Finucane, M.M.; Bahalim, A.N.; McIntire, R.K.; Gutierrez, H.R.; Cowan, M.; et al. National, regional, and global trends in adult overweight and obesity prevalences. Popul. Health Metr. 2012, 10, 22. [Google Scholar] [CrossRef]
- Huxley, R.R.; Woodward, M. Cigarette smoking as a risk factor for coronary heart disease in women compared with men. Lancet 2011, 378, 1297–1305. [Google Scholar] [CrossRef]
- Peters, S.A.E.; Huxley, R.R.; Woodward, M. Diabetes as a risk factor for incident coronary heart disease in women compared with men. Diabetalogia 2014, 57, 1542–1551. [Google Scholar] [CrossRef]
- Emdin, C.; Wong, C.X.; Hsiao, A.J.; Altman, D.; Peters, S.; Woodward, M.; Odutayo, A. Atrial fibrillation as a risk factor for cardiovascular disease and death in women compared to men. BMJ 2016, 532, h7013. [Google Scholar] [CrossRef]
- Backholer, K.; Peters, S.A.E.; Bots, S.H.; Peeters, A.; Huxley, R.R.; Woodward, M. Sex differences in the relationship between socioeconomic status and cardiovascular disease. J. Epidemiol. Community Health 2017, 71, 550–557. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.A.; Singhateh, Y.; Mackay, D.; Huxley, R.R.; Woodward, M. Total cholesterol as a risk factor for coronary heart disease and stroke in women compared with men. Atherosclerosis 2016, 248, 123–131. [Google Scholar] [CrossRef]
- Peters, S.A.; Huxley, R.; Woodward, M. Comparison of the sex-specific associations between systolic blood pressure and the risk of CVD. Stroke 2013, 44, 2394–2401. [Google Scholar] [CrossRef] [PubMed]
- Mongraw-Chaffin, M.L.; Peters, S.A.; Huxley, R.R.; Woodward, M. The sex-specific association between BMI and coronary heart disease. Lancet Diabetes Endocrinol. 2015, 3, 437–449. [Google Scholar] [CrossRef]
- Peters, S.A.E.; Huxley, R.R.; Woodward, M. Diabetes as a risk factor for stroke in women compared with men. Lancet 2014, 383, 1973–1980. [Google Scholar] [CrossRef]
- Peters, S.A.; Huxley, R.R.; Woodward, M. Smoking as a risk factor for stroke in women compared with men. Stroke 2013, 44, 2821–2828. [Google Scholar] [CrossRef]
- Woodward, M.; Peters, S.A.; Huxley, R.R. Diabetes and the female disadvantage. Womens Health 2015, 11, 833–839. [Google Scholar] [CrossRef]
- Health and Social Care Information Centre. National Diabetes Audit 2012–2013– Report 1: Care Processes and Treatment Targets 2014. Available online: www.hscic.gov.uk/catalogue/PUB14970 (accessed on 1 April 2019).
- Logue, J.; Walker, J.J.; Colhoun, H.M.; Leese, G.P.; Lindsay, R.S.; McKnight, J.A.; Morris, A.D.; Pearson, D.W.; Petrie, J.R.; Philip, S.; et al. Do men develop type 2 diabetes at lower body mass indices than women? Diabetologia 2011, 54, 3003–3006. [Google Scholar] [CrossRef]
- Peters, S.A.E.; Huxley, R.R.; Woodward, M. Sex differences in body anthropometry and composition in individuals with and without diabetes in the UK Biobank. BMJ Open 2016, 6, e010007. [Google Scholar] [CrossRef]
- Peters, S.A.; Woodward, M. Women’s reproductive factors and incident cardiovascular disease in the UK Biobank. Heart 2018, 104, 1069–1075. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.A.E.; Yang, L.; Guo, Y.; Chen, Y.; Bian, Z.; Millwood, I.Y.; Wang, S.; Yang, L.; Hu, Y.; Liu, J.; et al. Parenthood and the risk of cardiovascular diseases among 0.5 million men and women: Findings from the China Kadoorie Biobank. Int. J. Epidemiol. 2017, 46, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Tobias, D.K.; Stuart, J.J.; Li, S.; Chavarro, J.; Rimm, E.B.; Rich-Edwards, J.; Hu, F.B.; Manson, J.; Zhang, C. Association of history of gestational diabetes with long-term cardiovascular disease risk in a large prospective cohort of US women. JAMA Intern. Med. 2017, 177, 1735–1742. [Google Scholar] [CrossRef] [PubMed]
- McDonald, S.D.; Malinowski, A.; Zhou, Q.; Yusuf, S.; Devereaux, P.J. Cardiovascular sequelae of preeclampsia/eclampsia: A systematic review and meta-analyses. Am. Heart J. 2008, 156, 918–930. [Google Scholar] [CrossRef] [PubMed]
| Rank | Global | United States of America | High-Income Countries | India | Low-Income Countries |
|---|---|---|---|---|---|
| 1 | CHD | CHD | CHD | CHD | Respir Infect |
| 2 | Stroke | Alzheimer’s | Alzheimer’s | COPD | Neonatal disorders |
| 3 | Alzheimer’s | Stroke | Stroke | Diarrhoea | Diarrhoea |
| 4 | COPD | COPD | Lung cancer | Stroke | Malaria |
| 5 | Respir Infect | Lung cancer | COPD | Respir Infect | CHD |
| 6 | Diarrhoea | Breast cancer | Respir Infect | Neonatal disorders | Stroke |
| 7 | Neonatal disorders | Respir Infect | Breast cancer | TB | HIV/AIDS |
| 8 | Diabetes | CKD | Colorectal cancer | Asthma | TB |
| 9 | Breast cancer | Colorectal cancer | CKD | Diabetes | Congenital defects |
| 10 | Lung cancer | Diabetes | Diabetes | Falls | COPD |
| Rank | Global | United States of America | High-Income Countries | India | Low-Income Countries |
|---|---|---|---|---|---|
| 1 | Neonatal disorders | CHD | Low back pain | Neonatal disorders | Neonatal disorders |
| 2 | CHD | Low back pain | CHD | CHD | Respir Infect |
| 3 | Stroke | COPD | Stroke | Diarrhoea | Malaria |
| 4 | Respir Infect | Drug use | Alzheimer’s | Respir Infect | Diarrhoea |
| 5 | Diarrhoea | Stroke | Headaches | COPD | HIV/AIDS |
| 6 | COPD | Headaches | COPD | Dietary iron deficiency | Congenital defects |
| 7 | Low back pain | Diabetes | Diabetes | Stroke | TB |
| 8 | Headaches | Lung cancer | Depressive disorders | Headaches | Maternal disorders |
| 9 | Diabetes | Depressive disorders | Breast cancer | TB | Malnutrition |
| 10 | Congenital defects | Alzheimer’s | Lung cancer | Congenital defects | Stroke |
| Rank | Global | United States of America | High-Income Countries | India | Low–Income Countries |
|---|---|---|---|---|---|
| 1 | CHD | CHD | CHD | CHD | Neonatal disorders |
| 2 | Stroke | Lung | Lung cancer | COPD | Respir infect |
| 3 | COPD | Alzheimer’s | Stroke | Stroke | Diarrhoea |
| 4 | Respir infect | COPD | Alzheimer’s | Diarrhoea | CHD |
| 5 | Lung cancer | Stroke | COPD | TB | Malaria |
| 6 | Neonatal disorders | Respir infect | Respir infect | Respir infect | TB |
| 7 | RTI | CKD | Colorectal cancer | Neonatal disorders | Stroke |
| 8 | Cirrhosis | Drug use | Prostate cancer | RTI | HIV/AIDS |
| 9 | Alzheimer’s | Colorectal cancer | Cirrhosis | Cirrhosis | RTI |
| 10 | TB | Cirrhosis | Self-harm | CKD | Congenital defects |
| Rank | Global | United States of America | High-Income Countries | India | Low–Income Countries |
|---|---|---|---|---|---|
| 1 | CHD | CHD | CHD | Neonatal disorders | Neonatal disorders |
| 2 | Neonatal disorders | Drug use | Low back pain | CHD | Respir infect |
| 3 | Stroke | COPD | Lung cancer | COPD | Diarrhoea |
| 4 | Respir infect | Diabetes | Stroke | Respir infect | Malaria |
| 5 | RTI | Lung cancer | Diabetes | Diarrhoea | HIV/AIDS |
| 6 | COPD | Low back pain | COPD | TB | Congenital defects |
| 7 | Diarrhoea | RTI | Drug use | Stroke | TB |
| 8 | Diabetes | Stroke | Self-harm | RTI | RTI |
| 9 | Congenital defects | Self-harm | RTI | Diabetes | CHD |
| 10 | Low back pain | Cirrhosis | Falls | Self-harm | Meningitis |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woodward, M. Cardiovascular Disease and the Female Disadvantage. Int. J. Environ. Res. Public Health 2019, 16, 1165. https://doi.org/10.3390/ijerph16071165
Woodward M. Cardiovascular Disease and the Female Disadvantage. International Journal of Environmental Research and Public Health. 2019; 16(7):1165. https://doi.org/10.3390/ijerph16071165
Chicago/Turabian StyleWoodward, Mark. 2019. "Cardiovascular Disease and the Female Disadvantage" International Journal of Environmental Research and Public Health 16, no. 7: 1165. https://doi.org/10.3390/ijerph16071165
APA StyleWoodward, M. (2019). Cardiovascular Disease and the Female Disadvantage. International Journal of Environmental Research and Public Health, 16(7), 1165. https://doi.org/10.3390/ijerph16071165




