Environmental Burden of Childhood Disease in Europe
Abstract
1. Introduction
2. Methods
2.1. Selection of Environmental Risks and Health Outcomes
2.2. Data Collection
2.3. Environmental Burden of Disease
- (1)
- PAF = f × (RR − 1)/f × (RR − 1) + 1,
- (2)
- AP = PAF × P,
- (3)
- DALY = AI × DW × L,
3. Results
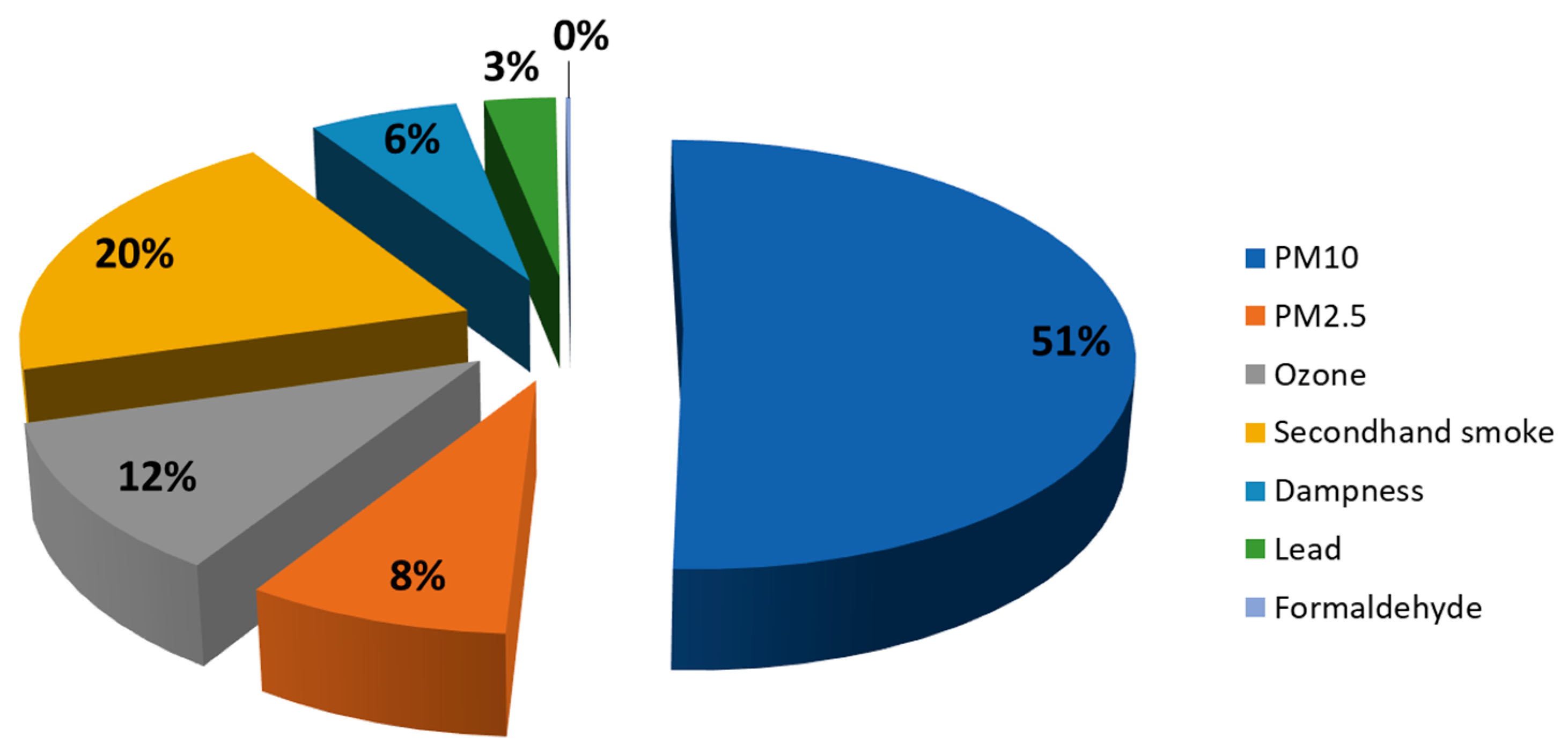
3.1. Environmental Burden of Disease in the EU of 28 Countries
3.1.1. Particulate Matter less than 10 Micrometer of Diameter (PM10) and less than 2.5 Micrometer of Diameter (PM2.5)
3.1.2. Secondhand Smoke
3.1.3. Ozone
3.1.4. Dampness
3.1.5. Lead
3.1.6. Formaldehyde
3.1.7. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Landrigan, P.J.; Schechter, C.B.; Lipton, J.M.; Fahs, M.C.; Schwartz, J. Children’s Health Environmental Pollutants and Disease in American Children: Estimates of. Environ. Health Perspect. 2002, 110, 721–728. [Google Scholar] [CrossRef] [PubMed]
- Forouzanfar, M.H.; Afshin, A.; Alexander, L.T.; Anderson, H.R.; Bhutta, Z.A.; Biryukov, S.; Brauer, M.; Burnett, R.; Cercy, K.; Charlson, F.J.; et al. GBD 2015 Risk Factors Collaborators. Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks in 188 countries, 1990–2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 2016, 380, 2224–2260. [Google Scholar] [CrossRef]
- Hänninen, O.; Knol, A.B.; Jantunen, M.; Lim, T.A.; Conrad, A.; Rappolder, M.; Carrer, P.; Fanetti, A.C.; Kim, R.; Buekers, J.; et al. Environmental Burden of Disease in Europe: Assessing Nine Risk Factors in Six Countries. Environ. Health Perspect. 2014, 122, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Bartlett, E.S.; Trasande, L. Economic impacts of environmentally attributable childhood health outcomes in the European Union. Eur. J. Public Health 2014, 24, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Landrigan, P.J.; Bellinger, D.C. Neurobehavioural effects of developmental toxicity. Lancet. Neurol. 2014, 13, 330–338. [Google Scholar] [CrossRef]
- Trasande, L.; Schechter, C.; Haynes, K.; Landrigan, P.J. Applying cost analyses to drive policy that protects children: mercury as a case study. Ann. N. Y. Acad. Sci. 2006, 1076, 911–923. [Google Scholar] [CrossRef]
- Trasande, L.; Zoeller, R.T.; Hass, U.; Kortenkamp, A.; Grandjean, P.; Myers, J.P.; DiGangi, J.; Bellanger, M.; Hauser, R.; Legler, J.; et al. Estimating burden and disease costs of exposure to endocrine-disrupting chemicals in the European union. J. Clin. Endocrinol. Metab. 2015, 100, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- WHO Europe. Health Risks of Air Pollution in Europe—HRAPIE Project; WHO: Geneva, Switzerland, 2013. [Google Scholar]
- Burnett, R.T.; Pope, C.A., 3rd; Ezzati, M.; Olives, C.; Lim, S.S.; Mehta, S.; Shin, H.H.; Singh, G.; Hubbell, B.; Brauer, M.; et al. An integrated risk function for estimating the global burden of disease attributable to ambient fine particulate matter exposure. Environ. Health Perspect. 2014, 122, 397–403. [Google Scholar] [CrossRef]
- Hurley, F.; Hunt, A.; Cowie, H.; Holland, M.; Miller, B.; Pye, S.; Watkiss, P. Service Contract for Carrying out Cost-Benefit Analysis of Air Quality Related Issues, in particular in the Clean Air for Europe (CAFE) Programme. In Methodology for the Cost-Benefit analysis for CAFE: Volume 2: Health Impact Assessment; AEA Technology Environment: Oxon, UK, 2005; Volume 2, pp. 1–149. [Google Scholar]
- Rumchev, K.B.; Spickett, J.T.; Bulsara, M.K.; Phillips, M.R.; Stick, S.M. Domestic exposure to formaldehyde significantly increases the risk of asthma in young children. Eur. Respir. J. 2002, 20, 403–408. [Google Scholar] [CrossRef]
- Quansah, R.; Jaakkola, M.S.; Hugg, T.T.; Heikkinen, M.; Jaakkola, J.J.K. Residential dampness and molds and the risk of developing asthma: a systematic review and meta-analysis. PLoS ONE 2012, 7, e47526. [Google Scholar] [CrossRef]
- Cal-EPA. Proposed Identification of Environmental Tobacco Smoke As a Toxic Air Contaminant; California Air Resources Board: Sacramento, CA, USA, 2005.
- Lanphear, B.P.; Hornung, R.; Khoury, J.; Yolton, K.; Baghurst, P.; Bellinger, D.C.; Canfield, R.L.; Dietrich, K.N.; Bornschein, R.; Greene, T.; et al. Low-Level Environmental Lead Exposure and Children’s Intellectual Function: An International Pooled Analysis. Environ. Health Perspect. 2005, 113, 894–899. [Google Scholar] [CrossRef]
- European Commission. Eurostat Database. Internet. 2015. Available online: http://ec.europa.eu/eurostat/web/population-demography-migration-projections/population-data/database (accessed on 26 October 2016).
- Institute for Health Metrics and Evaluation. Epi Visualization. 2016. Available online: http://vizhub.healthdata.org/epi/ (accessed on 1 August 2018).
- Institute for Health Metrics and Evaluation. Global Burden of Disease Study 2015. 2016. Available online: http://ghdx.healthdata.org/gbd-results-tool (accessed on 1 August 2018).
- WHO. Department of Health Statistics and Information Systems. Estim. 2000–2012. 2014. Available online: http://www.who.int/healthinfo/global_burden_disease/estimates/en/index2.html (accessed on 1 August 2018).
- WHO. European Health for All Database (HFA-DB); WHO: Geneva, Switzerland, 2015. [Google Scholar]
- WHO. Health risks of ozone from long-range transboundary air pollution. J. Chem. Inf. Model. 2008, 53, 1689–1699. [Google Scholar] [CrossRef]
- WHO/Europe. Environment and Health Information System (ENHIS). 2012. Available online: http://www.euro.who.int/en/data-and-evidence/environment-and-health-information-system-enhis (accessed on 1 August 2018).
- Ezzati, M.; Lopez, A.D.; Rodgers, A.; Murray, C.J. Comparative Quantification of Health Risks Global and Regional Burden of Disease. Comp. Quantif. Health Risks Glob. Reg. Burd. Dis. 2004. [Google Scholar] [CrossRef]
- Salomon, J.A.; Vos, T.; Hogan, D.R.; Gagnon, M.; Naghavi, M.; Mokdad, A.; Begum, N.; Shah, R.; Karyana, M.; Kosen, S.; et al. Common values in assessing health outcomes from disease and injury: disability weights measurement study for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2129–2143. [Google Scholar] [CrossRef]
- Khreis, H.; Kelly, C.; Tate, J.; Parslow, R.; Lucas, K.; Nieuwenhuijsen, M. Exposure to traf fi c-related air pollution and risk of development of childhood asthma: A systematic review and meta-analysis. Environ. Int. 2017, 100, 1–31. [Google Scholar] [CrossRef]
- Kampa, M.; Castanas, E. Human health effects of air pollution. Environ. Pollut. 2008, 151, 362–367. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fussell, J.C. Air pollution and public health: emerging hazards and improved understanding of risk. Environ. Geochem. Health 2015, 37, 631–649. [Google Scholar] [CrossRef] [PubMed]
- Héroux, M.E.; Anderson, H.R.; Atkinson, R.; Brunekreef, B.; Cohen, A.; Forastiere, F.; Hurley, F.; Katsouyanni, K.; Krewski, D.; Krzyzanowski, M.; et al. Quantifying the health impacts of ambient air pollutants: Recommendations of a WHO/Europe project. Int. J. Public Health 2015, 60, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Jassal, M.S. Pediatric asthma and ambient pollutant levels in industrializing nations. Int. Health 2014, 7, 7–15. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Air Quality Guidelines for Particulate Matter, Ozone, Nitrogen Dioxide and Sulfur Dioxide: Global Update 2005: Summary of Risk Assessment; World Health Organization: Geneva, Switzerland, 2006; pp. 1–22. [Google Scholar] [CrossRef]
- Cohen, A.J.; Brauer, M.; Burnett, R.; Anderson, H.R.; Frostad, J.; Estep, K.; Balakrishnan, K.; Brunekreef, B.; Dandona, L.; Dandona, R.; et al. Estimates and 25-year trends of the global burden of disease attributable to ambient air pollution: an analysis of data from the Global Burden of Diseases Study 2015. Lancet 2017, 389, 1907–1918. [Google Scholar] [CrossRef]
- Turner, M.C.; Jerrett, M.; Pope, C.A., III; Krewski, D.; Gapstur, S.M.; Diver, R.W.; Beckerman, B.S.; Marshall, J.D.; Su, J.; Crouse, D.L.; et al. Long-Term Ozone Exposure and Mortality in a Large Prospective Study. Am. J. Respir. Crit. Care Med. 2016, 64, 897–905. [Google Scholar] [CrossRef]
- Lelieveld, J.; Evans, J.S.; Fnais, M.; Giannadaki, D.; Pozzer, A. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 2015, 525, 367–371. [Google Scholar] [CrossRef]
- Karagulian, F.; Dora, C.F.C.; Pruess-Ustuen, A.M.; Bonjour, S.; Adair-Rohani, H.; Amann, M. Contributions to cities’ ambient particulate matter (PM): A systematic review of local source contributions at global level. Atmos. Environ. 2015, 120, 475–483. [Google Scholar] [CrossRef]
- Holman, C.; Harrison, R.; Querol, X. Review of the efficacy of low emission zones to improve urban air quality in European cities. Atmos. Environ. 2015, 111, 161–169. [Google Scholar] [CrossRef]
- Rojas-Rueda, D.; Nazelle, A.; De Teixidó, O.; Nieuwenhuijsen, M.J. Replacing car trips by increasing bike and public transport in the greater Barcelona metropolitan area: a health impact assessment study. Prev. Med. 2013, 57, 573–579. [Google Scholar] [CrossRef]
- Stevenson, M.; Thompson, J.; de Sá, T.H.; Ewing, R.; Mohan, D.; McClure, R.; Roberts, I.; Tiwari, G.; Giles-Corti, B.; Sun, X.; et al. Land use, transport, and population health: estimating the health benefits of compact cities. Lancet 2016, 6736, 1–11. [Google Scholar] [CrossRef]
- Erisman, J.W.; Bleeker, A.; Hensen, A.; Vermeulen, A. Agricultural air quality in Europe and the future perspectives. Atmos. Environ. 2008, 42, 3209–3217. [Google Scholar] [CrossRef]
- Jha, P.; Ramasundarahettige, C.; Landsman, V.; Rostron, B.; Thun, M.; Anderson, R.N.; Tim McAfee, M.D.; Richard Peto, F.R.S. 21st-Century Hazards of Smoking and Benefits of Cessation in the United States. N. Engl. J. Med. 2013, 368, 341–350. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. The Health Consequences of Involuntary Exposure to Tobacco Smoke: A Report of the Surgeon General; U.S. Department of Health and Human Services: Washington, DC, USA, 2006.
- Lemmens, V.; Oenema, A.; Knut, I.K.; Brug, J. Effectiveness of smoking cessation interventions among adults: a systematic review of reviews. Eur. J. Cancer Prev. 2008, 17, 535–544. [Google Scholar] [CrossRef] [PubMed]
- Frazer, K.; Callinan, J.E.; McHugh, J.; van Baarsel, S.; Clarke, A.; Doherty, K.; Kelleher, C. Legislative smoking bans for reducing harms from secondhand smoke exposure, smoking prevalence and tobacco consumption (Review) Summary of Findings for the Main Comparison. Cochrane Database Syst. Rev. 2016, 2. [Google Scholar] [CrossRef]
- McAfee, T.; Davis, K.C.; Alexander, R.L.; Pechacek, T.F.; Bunnell, R. Effect of the first federally funded US antismoking national media campaign. Lancet 2013, 382, 2003–2011. [Google Scholar] [CrossRef]
- Institute of Medicine of the National Academies. Damp Indoor Spaces and Health; National Academies Press: Washington, DC, USA, 2004. [Google Scholar]
- Krieger, J.; Jacobs, D.E.; Ashley, P.J.; Baeder, A.; Chew, G.L.; Dearborn, D.; Hynes, H.P.; Miller, J.D.; Morley, R.; Rabito, F.; et al. Housing Interventions and Control of Asthma-Related Indoor Biologic Agents: A Review of the Evidence. J. Public Health Manag. Pract. 2010, 16, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Fewtrell, L.; Prüss-üstün, A.; Campbell-lendrum, D.; Corvalán, C.; Woodward, A. Introduction and Methods: Assessing the Environmental Burden of Disease at National and Local Levels; Environmental Burden of Disease Series No. 1; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- Tong, S.; von Schirnding, Y.E.; Prapamontol, T. Environmental lead exposure: a public health problem of global dimensions. Bull. World Health Organ. 2000, 78, 1068–1077. [Google Scholar] [CrossRef] [PubMed]
- WHO/Europe. WHO Guidelines for Indoor Air Quality: Selected Pollutants; Bonn, Ger. puncto druck+ Medien GmbH; World Health Organization: Geneva, Switzerland, 2010. [Google Scholar] [CrossRef]
- Hänninen, O. European Perspectives on Environmental Burden of Disease Estimates for Nine Stressors in Six European Countries; National Institute for Health and Welfare: Helsinki, Finland, 2011. [Google Scholar]
- Murray, C.J.; Ezzati, M.; Lopez, A.D.; Rodgers, A.; Vander Hoorn, S. Comparative quantification of health risks: Conceptual framework and methodological issues. Popul. Health Metr. 2003, 1, 1. [Google Scholar] [CrossRef] [PubMed]
- Vrijheid, M.; Slama, R.; Robinson, O.; Chatzi, L.; Coen, M.; van den Hazel, P.; Thomsen, C.; Wright, J.; Athersuch, T.J.; Avellana, N.; et al. The human early-life exposome (HELIX): Project rationale and design. Environ. Health Perspect. 2014, 122, 535–544. [Google Scholar] [CrossRef]
- Vineis, P.; Chadeau-Hyam, M.; Gmuender, H.; Gulliver, J.; Herceg, Z.; Kleinjans, J.; Kogevinas, M.; Kyrtopoulos, S.; Nieuwenhuijsen, M.; Phillips, D.H.; et al. The exposome in practice: Design of the EXPOsOMICS project. Int. J. Hyg. Environ. Health 2016, 220 Pt A, 142–151. [Google Scholar] [CrossRef]
- HEALS. Health and Environment-Wide Associations Based on Large Population Surveys (HEALS). 2017. Available online: http://www.heals-eu.eu/index.php/project/ (accessed on 1 August 2018).
| Risk Factor | Health Outcome | Population | Exposure Estimate | Unit of Exposure | DRF | Reference |
|---|---|---|---|---|---|---|
| PM10 | Asthma | 5–18 years | Ambient levels | μg/m3 | 1.028 (1.0006–1.051) | [8] |
| PM10 | Infant mortality | <1 year | Ambient levels | μg/m3 | 1.04 (1.02–1.07) | [8] |
| PM2.5 | Low respiratory infections | <18 years | Ambient levels | μg/m3 | Function * | [9] |
| Ozone | Cough days | 5–14 years | Ambient levels | μg/m3 | 0.093 (0.019–0.22) | [10] |
| Ozone | Low respiratory symptoms | 5–14 years | Ambient levels | μg/m3 | 0.016 (−0.043–0.08) | [10] |
| Formaldehyde | Asthma | <3 years | Indoor levels | μg/m3 | 1.017 (1.004–1.025) | [11] |
| Dampness | Asthma | <14 years | Percent exposed | House Yes/no | 1.33 (1.12–1.56) | [12] |
| Mold | Asthma | <14 years | Percent exposed | House Yes/no | 1.29 (1.04–1.6) | [12] |
| SHS | Asthma | <14 years | Percent exposed | Parental Yes/no | 1.32 (1.24–1.41) | [13] |
| SHS | Low respiratory infections | <5 years | Percent exposed | Parental Yes/no | 1.55 (1.42–1.69) | [2] |
| SHS | Otitis media | <5 years | Percent exposed | Parental Yes/no | 1.37 (1.24–1.50) | [2] |
| Lead | IQ loss | <5 years | Blood levels | mg/L | 0.051 (0.032–0.07) | [14] |
| Lead | Mild mental retardation | <5 years | Blood levels | mg/L | Function * | [14] |
| Risk Factor | Health Outcome | Population | Cases | DALYs | LCI | UCI | DALYs/100,000 Population | Total DALYs by Exposure |
|---|---|---|---|---|---|---|---|---|
| Lead | Mild mental retardation | <5 years | 138,646 | 6216 | 2699 | 11,414 | 15.09 | 6216 |
| PM10 | Asthma | 5–18 years | 43,402 | 13,904 | 462 | 15,181 | 17.80 | |
| Infant mortality | <1 year | 1078 | 93,147 | 45,106 | 166,668 | 16,324 | 107,051 | |
| PM2.5 | Low respiratory infections | <18 years | 134,032 | 17,453 | 8042 | 29,659 | 21.14 | 17,453 |
| Secondhand smoke | Asthma | <14 years | 106,085 | 20,880 | 15,645 | 25,065 | 28.03 | |
| Low respiratory infections | <5 years | 142,530 | 9728 | 5942 | 14,040 | 37.79 | ||
| Otitis media | <5 years | 821,499 | 2062 | 1132 | 3396 | 8.01 | 42,501 | |
| Ozone | Cough days | 5–14 years | 52,436,762 * | 10,057 | 902 | 10,177 | 19.78 | |
| Low respiratory symptoms days | 5–14 years | 52,059,353 * | 14,122 | 760 | 14,207 | 27.78 | 24,179 | |
| Dampness | Asthma | <14 years | 65,815 | 12,954 | 3022 | 31,646 | 17.39 | 12,954 |
| Formaldehyde | Asthma | <3 years | 423 | 33 | 4 | 83 | 0.60 | 423 |
| Total | 210,777 |
| Stakeholder | Recommendations |
|---|---|
| Authorities | Develop evidence-based policies to reduce child exposure to environmental risk factors across Europe, with special attention to major risk factors as secondhand smoke and air pollution |
| Public health practitioners | Create and develop European health databases with harmonized exposure data for (old and new) environmental risk factors for children across all European countries |
| Researchers | Develop epidemiological studies on multiple environmental risk factors, with special attention to providing dose-response functions with harmonized exposure and outcome definitions |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rojas-Rueda, D.; Vrijheid, M.; Robinson, O.; Gunn Marit, A.; Gražulevičienė, R.; Slama, R.; Nieuwenhuijsen, M. Environmental Burden of Childhood Disease in Europe. Int. J. Environ. Res. Public Health 2019, 16, 1084. https://doi.org/10.3390/ijerph16061084
Rojas-Rueda D, Vrijheid M, Robinson O, Gunn Marit A, Gražulevičienė R, Slama R, Nieuwenhuijsen M. Environmental Burden of Childhood Disease in Europe. International Journal of Environmental Research and Public Health. 2019; 16(6):1084. https://doi.org/10.3390/ijerph16061084
Chicago/Turabian StyleRojas-Rueda, David, Martine Vrijheid, Oliver Robinson, Aasvang Gunn Marit, Regina Gražulevičienė, Remy Slama, and Mark Nieuwenhuijsen. 2019. "Environmental Burden of Childhood Disease in Europe" International Journal of Environmental Research and Public Health 16, no. 6: 1084. https://doi.org/10.3390/ijerph16061084
APA StyleRojas-Rueda, D., Vrijheid, M., Robinson, O., Gunn Marit, A., Gražulevičienė, R., Slama, R., & Nieuwenhuijsen, M. (2019). Environmental Burden of Childhood Disease in Europe. International Journal of Environmental Research and Public Health, 16(6), 1084. https://doi.org/10.3390/ijerph16061084







