Traditional Korean Medicine-Based Forest Therapy Programs Providing Electrophysiological Benefits for Elderly Individuals
Abstract
1. Introduction
2. Materials and Methods
2.1. FTPs
2.2. Sasang Constitutional Medicine
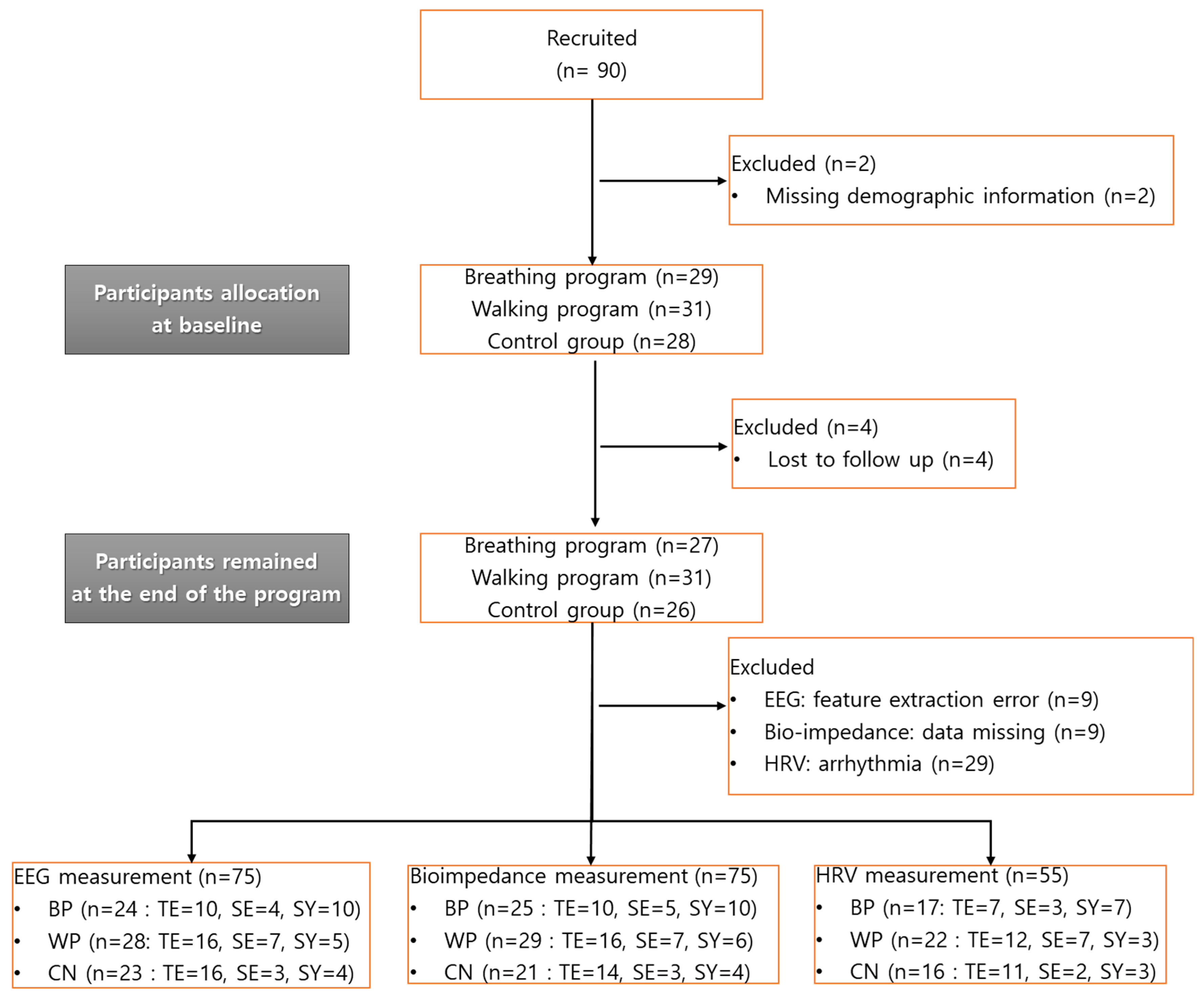
2.3. Subjects and Study Protocol
- not diagnosed with dementia
- without any restrictions on outdoor activity, including walking for more than three hours
- able to communicate and complete the self-reporting questionnaires
- understand the purpose of the study and having voluntarily submitted a consent form.
2.4. Electrophysiological Measurements
2.5. Statistical Analysis
3. Results
3.1. Demographics
3.2. Changes in Electrophysiology According to EEG, Bioimpedance, and HRV
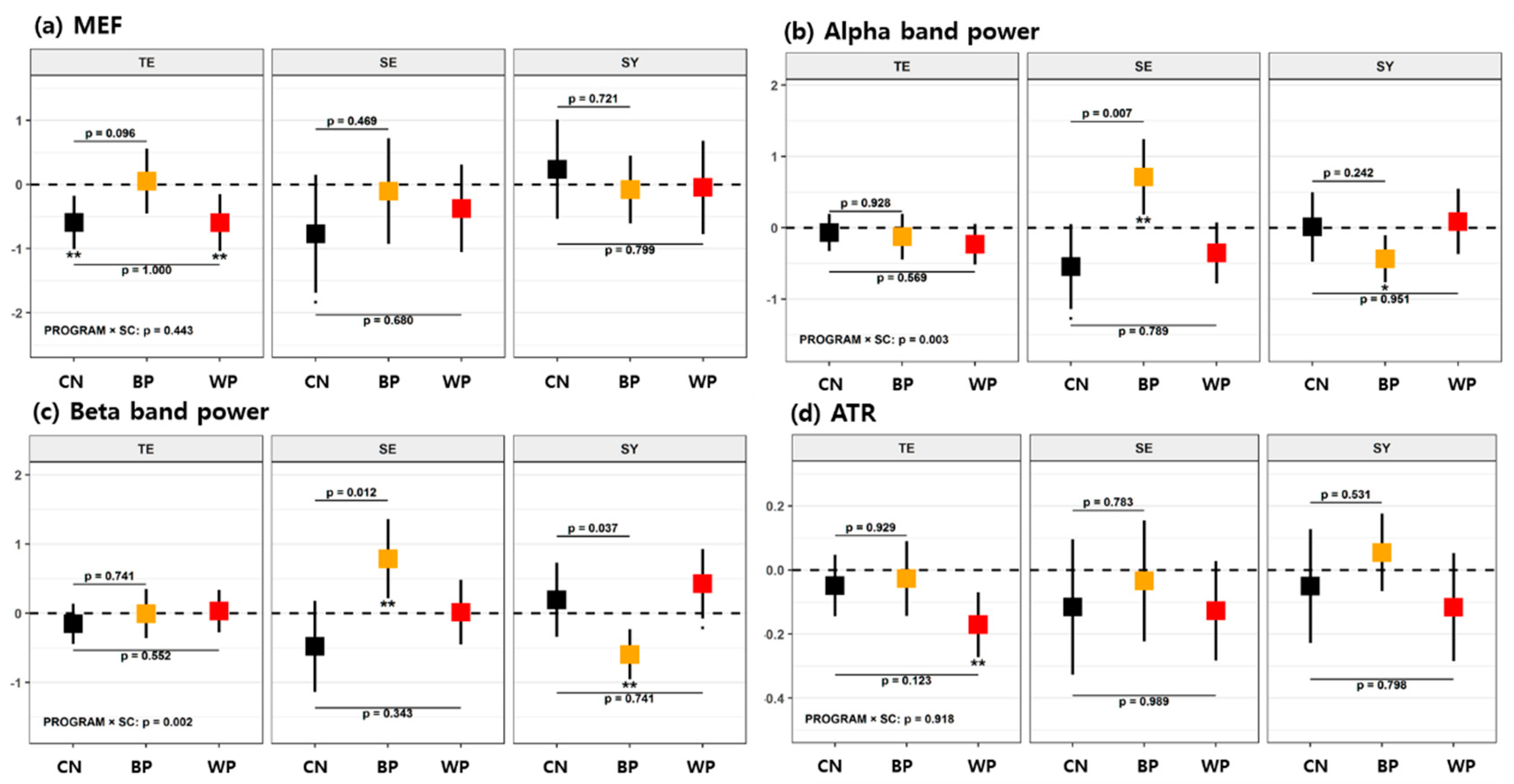
3.2.1. Resting-State EEG
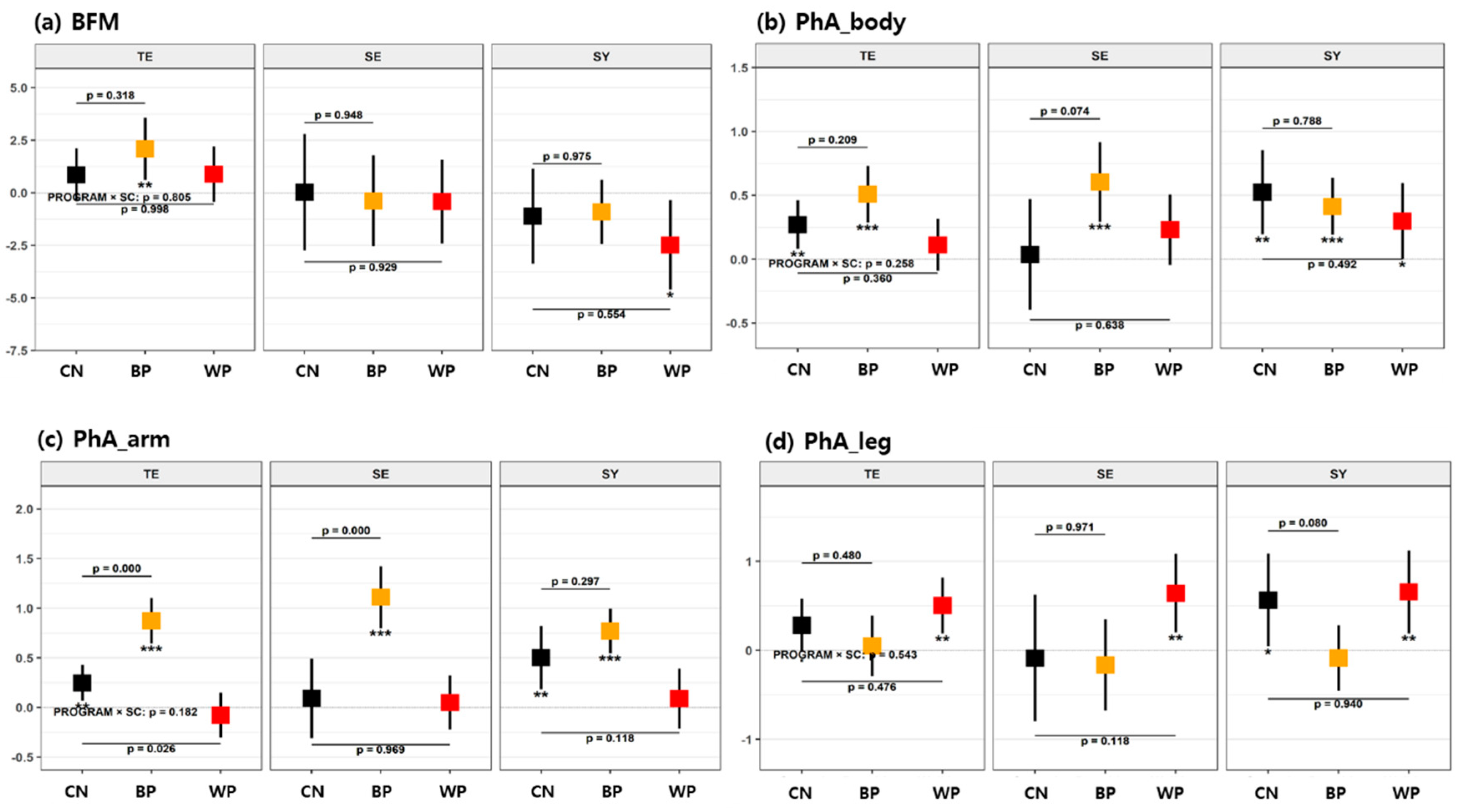
3.2.2. Bioimpedance
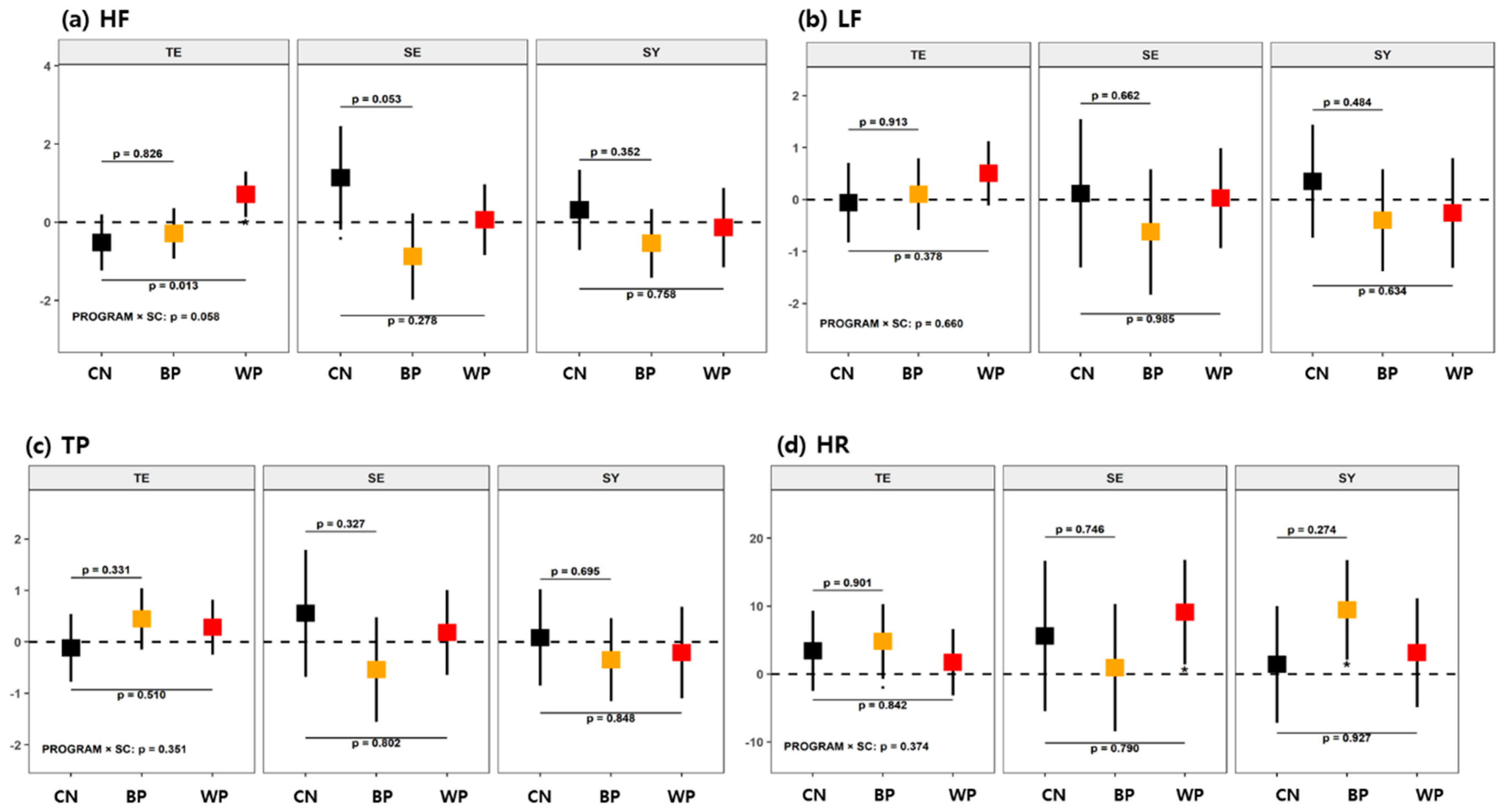
3.2.3. Heart Rate Variability
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| EEG Variable | Control | BP | WP | BP–CN | WP–CN | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SC Type | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | |||||||||
| MEF [Hz] | TE | 8.49 | −0.59 ** (−1.01, −0.18) | 0.74 | 8.62 | 0.05 (−0.46, 0.56) | 0.06 | 8.59 | −0.60 ** (−1.04, −0.15) | 0.67 | 0.65 (−0.10, 1.39) | 0.53 | −0.00 (−0.65, 0.64) | 0.00 |
| SE | 8.36 | −0.77 (−1.69, 0.15) | 0.97 | 8.61 | −0.10 (−0.93, 0.72) | 0.13 | 8.06 | −0.37 (−1.06, 0.31) | 0.41 | 0.67 (−0.76, 2.09) | 0.28 | 0.40 (−0.84, 1.63) | 0.21 | |
| SY | 8.48 | 0.24 (−0.53, 1.01) | 0.31 | 8.08 | −0.08 (−0.61, 0.45) | 0.10 | 8.30 | −0.05 (−0.77, 0.68) | 0.06 | −0.32 (−1.40, 0.76) | 0.19 | −0.29 (−1.48, 0.91) | 0.14 | |
| TE | 2.85 | −0.07 (−0.33, 0.19) | 0.13 | 2.94 | −0.13 (−0.45, 0.19) | 0.25 | 2.62 | −0.23 (−0.52, 0.05) | 0.41 | −0.06 (−0.53, 0.41) | 0.08 | −0.16 (−0.57, 0.25) | 0.24 | |
| SE | 2.10 | −0.54 (−1.14, 0.05) | 1.06 | 3.36 | 0.71 ** (0.18, 1.24) | 1.35 | 2.71 | −0.35 (−0.78, 0.07) | 0.63 | 1.26 ** (0.32, 2.20) | 0.80 | 0.19 (−0.59, 0.97) | 0.16 | |
| SY | 2.62 | 0.01 (−0.47, 0.50) | 0.02 | 2.91 | −0.44 * (−0.77, −0.11) | 0.84 | 2.64 | 0.09 (−0.37, 0.54) | 0.17 | −0.45 (−1.13, 0.23) | 0.43 | 0.08 (−0.68, 0.83) | 0.06 | |
| TE | 1.96 | −0.15 (−0.44, 0.14) | 0.27 | 2.38 | −0.01 (−0.36, 0.35) | 0.01 | 2.02 | 0.03 (−0.28, 0.34) | 0.05 | 0.15 (−0.38, 0.67) | 0.17 | 0.18 (−0.26, 0.63) | 0.24 | |
| SE | 1.15 | −0.48 (−1.14, 0.18) | 0.84 | 2.49 | 0.79 ** (0.22, 1.36) | 1.38 | 2.22 | 0.01 (−0.45, 0.48) | 0.02 | 1.26 * (0.25, 2.28) | 0.74 | 0.49 (−0.38, 1.37) | 0.37 | |
| SY | 2.01 | 0.19 (−0.34, 0.73) | 0.36 | 2.16 | −0.59 ** (−0.96, −0.23) | 1.04 | 2.12 | 0.43 (−0.08, 0.93) | 0.76 | −0.79 * (−1.53, −0.04) | 0.69 | 0.23 (−0.60, 1.06) | 0.17 | |
| ATR | TE | 1.28 | −0.05 (−0.14, 0.05) | 0.26 | 1.20 | −0.03 (−0.14, 0.09) | 0.15 | 1.25 | −0.17 ** (−0.27, −0.07) | 0.84 | 0.02 (−0.15, 0.19) | 0.08 | −0.12 (−0.27, 0.03) | 0.49 |
| SE | 1.24 | −0.12 (−0.33, 0.10) | 0.63 | 1.17 | −0.03 (−0.22, 0.16) | 0.18 | 1.18 | −0.13 (−0.28, 0.03) | 0.62 | 0.08 (−0.25, 0.41) | 0.15 | −0.01 (−0.29, 0.27) | 0.03 | |
| SY | 1.14 | −0.05 (−0.23, 0.13) | 0.28 | 1.17 | 0.05 (−0.07, 0.18) | 0.29 | 1.14 | −0.12 (−0.28, 0.05) | 0.62 | 0.11 (−0.14, 0.35) | 0.28 | −0.07 (−0.34, 0.21) | 0.14 | |
| Bioimpedance | Control | BP | WP | BP−CN | WP−CN | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | SC Type | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | ||||||||
| BFM | TE | 23.53 | 0.86 (−0.39, 2.10) | 0.37 | 25.31 | 2.09 ** (0.61, 3.56) | 0.90 | 22.67 | 0.89 (−0.44, 2.21) | 0.33 | 1.23 (−0.87, 3.33) | 0.35 | 0.03 (−1.90, 1.95) | 0.01 |
| SE | 14.26 | 0.03 (−2.74, 2.79) | 0.01 | 16.65 | −0.39 (−2.55, 1.78) | 0.16 | 12.88 | −0.42 (−2.42, 1.57) | 0.16 | −0.41 (−4.32, 3.49) | 0.06 | −0.45 (−4.00, 3.10) | 0.08 | |
| SY | 14.47 | −1.11 (−3.37, 1.15) | 0.49 | 16.89 | −0.91 (−2.43, 0.62) | 0.38 | 15.02 | −2.48 * (−4.61, −0.35) | 0.95 | 0.20 (−2.80, 3.21) | 0.04 | −1.37 (−4.73, 1.99) | 0.24 | |
| PhA_body | TE | 5.55 | 0.27 ** (0.08, 0.46) | 0.76 | 4.78 | 0.51 *** (0.29, 0.73) | 1.45 | 5.53 | 0.11 (−0.09, 0.32) | 0.28 | 0.24 (−0.10, 0.58) | 0.42 | −0.16 (−0.45, 0.13) | 0.32 |
| SE | 4.63 | 0.04 (−0.40, 0.47) | 0.10 | 5.04 | 0.60 *** (0.29, 0.92) | 1.73 | 5.13 | 0.23 (−0.05, 0.51) | 0.63 | 0.57 (−0.05, 1.18) | 0.56 | 0.19 (−0.36, 0.74) | 0.22 | |
| SY | 5.33 | 0.52 ** (0.19, 0.85) | 1.59 | 4.98 | 0.41 *** (0.19, 0.64) | 1.17 | 5.47 | 0.30 * (0.00, 0.59) | 0.82 | −0.11 (−0.56, 0.34) | 0.16 | −0.23 (−0.73, 0.28) | 0.27 | |
| PhA_arm | TE | 5.40 | 0.25 ** (0.07, 0.43) | 0.73 | 4.58 | 0.87 *** (0.64, 1.10) | 2.41 | 5.75 | −0.08 (−0.30, 0.15) | 0.17 | 0.63 *** (0.28, 0.97) | 1.08 | −0.32 * (−0.62, −0.03) | 0.66 |
| SE | 4.83 | 0.09 (−0.31, 0.49) | 0.26 | 4.68 | 1.11 *** (0.80, 1.42) | 3.19 | 5.36 | 0.05 (−0.22, 0.32) | 0.14 | 1.02 *** (0.45, 1.59) | 1.09 | −0.04 (−0.58, 0.49) | 0.05 | |
| SY | 5.24 | 0.50 ** (0.18, 0.82) | 1.57 | 4.73 | 0.77 *** (0.55, 0.99) | 2.17 | 5.63 | 0.09 (−0.21, 0.39) | 0.24 | 0.27 (−0.17, 0.71) | 0.39 | −0.41 (−0.91, 0.09) | 0.50 | |
| PhA_leg | TE | 5.68 | 0.28 (−0.02, 0.58) | 0.50 | 4.98 | 0.05 (−0.29, 0.39) | 0.09 | 5.06 | 0.50 ** (0.19, 0.82) | 0.80 | −0.23 (−0.74, 0.27) | 0.27 | 0.22 (−0.26, 0.70) | 0.27 |
| SE | 3.89 | −0.09 (−0.80, 0.62) | 0.14 | 5.45 | −0.16 (−0.68, 0.35) | 0.29 | 4.86 | 0.64 ** (0.20, 1.08) | 1.10 | −0.08 (−1.13, 0.97) | 0.04 | 0.73 (−0.15, 1.61) | 0.53 | |
| SY | 5.32 | 0.56 * (0.04, 1.09) | 1.08 | 5.20 | −0.09 (−0.45, 0.28) | 0.15 | 4.87 | 0.66 ** (0.19, 1.12) | 1.15 | −0.65 (−1.37, 0.06) | 0.59 | 0.09 (−0.71, 0.89) | 0.07 | |
| HRV Variable | Control | BP | WP | BP–CN | WP–CN | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SC Type | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | |||||||||
| HF [ms2] | TE | 3.78 | −0.52 (−1.23, 0.20) | 0.56 | 4.18 | −0.29 (−0.93, 0.36) | 0.32 | 4.23 | 0.71 * (0.13, 1.30) | 0.69 | 0.23 (−0.85, 1.32) | 0.17 | 1.23 * (0.24, 2.22) | 1.02 |
| SE | 3.93 | 1.14 (−0.19, 2.46) | 1.23 | 4.13 | −0.88 (−1.98, 0.23) | 0.93 | 4.80 | 0.06 (−0.84, 0.97) | 0.06 | −2.01 (−4.05, 0.03) | 0.79 | −1.07 (−2.81, 0.66) | 0.54 | |
| SY | 4.09 | 0.31 (−0.71, 1.34) | 0.36 | 4.51 | −0.54 (−1.42, 0.34) | 0.56 | 5.58 | −0.13 (−1.15, 0.88) | 0.13 | −0.85 (−2.40, 0.69) | 0.45 | −0.45 (−2.14, 1.25) | 0.21 | |
| LF [ms2] | TE | 3.21 | −0.06 (−0.83, 0.71) | 0.06 | 3.58 | 0.10 (−0.59, 0.79) | 0.11 | 3.76 | 0.51 (−0.11, 1.12) | 0.46 | 0.16 (−0.99, 1.32) | 0.11 | 0.56 (−0.50, 1.63) | 0.44 |
| SE | 3.48 | 0.12 (−1.31, 1.54) | 0.12 | 4.53 | −0.62 (−1.83, 0.58) | 0.61 | 3.90 | 0.03 (−0.94, 0.99) | 0.02 | −0.74 (−2.99, 1.51) | 0.26 | −0.09 (−1.93, 1.75) | 0.04 | |
| SY | 4.22 | 0.35 (−0.74, 1.44) | 0.38 | 4.45 | −0.40 (−1.38, 0.58) | 0.37 | 4.86 | −0.26 (−1.31, 0.80) | 0.25 | −0.75 (−2.41, 0.91) | 0.37 | −0.61 (−2.35, 1.14) | 0.28 | |
| TP [ms2] | TE | 5.35 | −0.12 (−0.77, 0.54) | 0.14 | 5.61 | 0.45 (−0.15, 1.04) | 0.54 | 6.02 | 0.28 (−0.25, 0.82) | 0.30 | 0.56 (−0.42, 1.55) | 0.45 | 0.40 (−0.52, 1.33) | 0.36 |
| SE | 5.11 | 0.55 (−0.68, 1.79) | 0.65 | 6.14 | −0.54 (−1.55, 0.48) | 0.62 | 6.04 | 0.18 (−0.64, 1.01) | 0.20 | −1.09 (−3.00, 0.81) | 0.46 | −0.37 (−1.97, 1.23) | 0.20 | |
| SY | 5.97 | 0.09 (−0.85, 1.02) | 0.11 | 6.16 | −0.35 (−1.15, 0.46) | 0.39 | 6.69 | −0.21 (−1.10, 0.68) | 0.24 | −0.43 (−1.84, 0.98) | 0.25 | −0.30 (−1.79, 1.20) | 0.16 | |
| HR [bpm] | TE | 68.63 | 3.42 (−2.49, 9.32) | 0.44 | 68.02 | 4.79 (−0.71, 10.29) | 0.62 | 64.79 | 1.73 (−3.17, 6.63) | 0.20 | 1.37 (−7.67, 10.41) | 0.12 | −1.69 (−10.04, 6.66) | 0.17 |
| SE | 70.54 | 5.59 (−5.48, 16.67) | 0.72 | 70.60 | 0.93 (−8.46, 10.33) | 0.12 | 63.63 | 9.11 * (1.40, 16.82) | 1.07 | −4.66 (−21.76, 12.44) | 0.22 | 3.51 (−11.07, 18.10) | 0.21 | |
| SY | 68.57 | 1.41 (−7.21, 10.02) | 0.19 | 65.27 | 9.45 * (2.13, 16.77) | 1.17 | 62.81 | 3.14 (−4.87, 11.15) | 0.40 | 8.04 (−4.87, 20.95) | 0.50 | 1.73 (−11.99, 15.46) | 0.10 | |
References
- Statistics Korea. 2017 Population and Housing Census; Statistics Korea: Daejeon, Korea, 2018.
- Livingston, G.; Sommerlad, A.; Orgeta, V.; Costafreda, S.G.; Huntley, J.; Ames, D.; Ballard, C.; Banerjee, S.; Burns, A.; Cohen-Mansfield, J.; et al. The Lancet International Commission on Dementia Prevention and Care. Lancet 2017, 390, 2673–2734. [Google Scholar] [CrossRef]
- World Health Organization. Meeting on the Implementation of the Global Action Plan on the Public Health Response to Dementia; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Intzandt, B.; Black, S.E.; Lanctot, K.L.; Herrmann, N.; Oh, P.; Middleton, L.E. Is Cardiac Rehabilitation Exercise Feasible for People with Mild Cognitive Impairment? Can. Geriatr. J. 2015, 18, 65–72. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bratman, G.N.; Daily, G.C.; Levy, B.J.; Gross, J.J. The benefits of nature experience: Improved affect and cognition. Landsc. Urban Plan. 2015, 138, 41–50. [Google Scholar] [CrossRef]
- Berman, M.G.; Kross, E.; Krpan, K.M.; Askren, M.K.; Burson, A.; Deldin, P.J.; Kaplan, S.; Sherdell, L.; Gotlib, I.H.; Jonides, J. Interacting with nature improves cognition and affect for individuals with depression. J. Affect. Disord. 2012, 140, 300–305. [Google Scholar] [CrossRef]
- Shin, W.S.; Shin, C.S.; Yeoun, P.S. The influence of forest therapy camp on depression in alcoholics. Environ. Health Prev. Med. 2012, 17, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Shin, W.S.; Shin, C.S.; Yeoun, P.S.; Kim, J.J. The Influence of interaction with forest on cognitive function. Scand. J. For. Res. 2011, 26, 595–598. [Google Scholar] [CrossRef]
- Hartig, T.; Mitchell, R.; Vries, S.D.; Frumkin4, H. Nature and Health. Annu. Rev. Public Health 2014, 35, 207–228. [Google Scholar] [CrossRef]
- Gallis, C. Green Care: For Human Therapy, Social Innovation, Rural Economy, and Education; Nova Science Publishers: Hauppauge, NY, USA, 2013. [Google Scholar]
- Oh, B.; Lee, K.J.; Zaslawski, C.; Yeung, A.; Rosenthal, D.; Larkey, L.; Back, M. Health and well-being benefits of spending time in forests: Systematic review. Environ. Health Prev. Med. 2017, 22, 71. [Google Scholar] [CrossRef]
- Song, C.; Ikei, H.; Miyazaki, Y. Physiological Effects of Nature Therapy: A Review of the Research in Japan. Int. J. Environ. Res. Public Health 2016, 13, 781. [Google Scholar] [CrossRef]
- Li, Q. Forest Medicine; Nova Science Publishers: Hauppauge, NY, USA, 2012. [Google Scholar]
- Li, Q.; Morimotoi, K.; Nakadai, A.; Inagaki, H.; Katsumata, M.; Shimizu, T.; Hirata, Y.; Hirata, K.; Suzuki, H.; Miyazaki, Y.; et al. Forest bathing enhances human natural killer activity and expression of anti-cancer proteins. Int. J. Immunopathol. Pharmacol. 2007, 20, 3–8. [Google Scholar] [CrossRef]
- Park, B.-J.; Tsunetsugu, Y.; Kasetani, T.; Hirano, H.; Kagawa, T.; Sato, M.; Miyazaki, Y. Physiological Effects of Shinrin-yoku (Taking in the Atmosphere of the Forest)—Using Salivary Cortisol and Cerebral Activity as Indicators. J. Physiol. Anthropol. 2007, 26, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Tsunetsugu, Y.; Kasetani, T.; Kagawa, T.; Miyazaki, Y. The physiological effects of Shinrin-yoku (taking in the forest atmosphere or forest bathing): Evidence from field experiments in 24 forests across Japan. Environ. Health Prev. Med. 2010, 15, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.; Choi, H.; Bang, K.S.; Kim, S.; Song, M.; Lee, B. Effects of Forest Therapy on Depressive Symptoms among Adults: A Systematic Review. Int. J. Environ. Res. Public Health 2017, 14, 321. [Google Scholar] [CrossRef] [PubMed]
- Han, J.W.; Choi, H.; Jeon, Y.H.; Yoon, C.H.; Woo, J.M.; Kim, W. The Effects of Forest Therapy on Coping with Chronic Widespread Pain: Physiological and Psychological Differences between Participants in a Forest Therapy Program and a Control Group. Int. J. Environ. Res. Public Health 2016, 13, 255. [Google Scholar] [CrossRef]
- Chun, M.H.; Chang, M.C.; Lee, S.J. The effects of forest therapy on depression and anxiety in patients with chronic stroke. Int. J. Neurosci. 2017, 127, 199–203. [Google Scholar] [CrossRef]
- Deckers, K.; van Boxtel, M.P.; Schiepers, O.J.; de Vugt, M.; Munoz Sanchez, J.L.; Anstey, K.J.; Brayne, C.; Dartigues, J.F.; Engedal, K.; Kivipelto, M.; et al. Target risk factors for dementia prevention: A systematic review and Delphi consensus study on the evidence from observational studies. Int. J. Geriatr. Psychiatry 2015, 30, 234–246. [Google Scholar] [CrossRef]
- Jung, J. A Systematic Review of Forest Therapy Program on Older Adults. J. Humanit. Soc. Sci. 2019, 21, 1017–1032. [Google Scholar]
- Lee, H.J.; Son, S.A. Psychological and Physical Effects of 10 Weeks Urban Forest Therapy Program on Dementia Prevention in Low-Income Elderly Living Alone. J. People Plants Environ. 2018, 21, 557–564. [Google Scholar] [CrossRef]
- Lee, H.J.; Son, S.A. Qualitative Assessment of Experience on Urban Forest Therapy Program for Preventing Dementia of the Elderly Living Alone in Low-Income Class. J. People Plants Environ. 2018, 21, 565–574. [Google Scholar] [CrossRef]
- Han, I.D.; Koo, C.-D. The Effect of Forest Healing Program on the Resilience of Elderly People in Urban Forest. J. People Plants Environ. 2018, 21, 293–303. [Google Scholar] [CrossRef]
- Kim, J.Y.; Shin, C.S.; Lee, J.K. The Effects of Forest Healing Program on Mental Health and Melatonin of the Elderly in the Urban Forest. J. Korea Soc. Plants People Environ. 2017, 20, 95–106. [Google Scholar] [CrossRef]
- Choi, J.-H.; Shin, C.-S.; Yeoun, P.-S. Effects of Forest-Walking Exercise on Functional Fitness and Gait Pattern in the Elderly. J. Korean For. Soc. 2014, 103, 503–509. [Google Scholar] [CrossRef][Green Version]
- Mao, G.; Cao, Y.; Wang, B.; Wang, S.; Chen, Z.; Wang, J.; Xing, W.; Ren, X.; Lv, X.; Dong, J.; et al. The Salutary Influence of Forest Bathing on Elderly Patients with Chronic Heart Failure. Int. J. Environ. Res. Public Health 2017, 14, 368. [Google Scholar] [CrossRef] [PubMed]
- Mao, G.X.; Cao, Y.B.; Lan, X.G.; He, Z.H.; Chen, Z.M.; Wang, Y.Z.; Hu, X.L.; Lv, Y.D.; Wang, G.F.; Yan, J. Therapeutic effect of forest bathing on human hypertension in the elderly. J. Cardiol. 2012, 60, 495–502. [Google Scholar] [CrossRef]
- Kim, J.Y.; Pham, D.D. Sasang constitutional medicine as a holistic tailored medicine. Evid. Based Complementary Altern. Med. 2009, 6 (Suppl. 1), 9–11. [Google Scholar] [CrossRef]
- Kim, J.U.; Ku, B.; Kim, Y.M.; Do, J.H.; Jang, J.S.; Jang, E.; Jeon, Y.J.; Kim, K.H.; Kim, J.Y. The concept of sasang health index and constitution-based health assessment: An integrative model with computerized four diagnosis methods. Evid. Based Complementary Altern. Med. 2013, 2013, 879420. [Google Scholar] [CrossRef]
- Gates, N.; Fiatarone Singh, M.A.; Sachdev, P.S.; Valenzuela, M. The effect of exercise training on cognitive function in older adults with mild cognitive impairment: A meta-analysis of randomized controlled trials. Am. J. Geriatr. Psychiatry 2013, 21, 1086–1097. [Google Scholar] [CrossRef]
- Hong, S.G.; Kim, J.H.; Jun, T.W. Effects of 12-Week Resistance Exercise on Electroencephalogram Patterns and Cognitive Function in the Elderly with Mild Cognitive Impairment: A Randomized Controlled Trial. Clin. J. Sport Med. 2018, 28, 500–508. [Google Scholar] [CrossRef]
- Sung, S.-H.; Park, J.-H.; Lee, Y.-J.; Han, C.-H. The Analysis of Research Trends on Forest Therapy in the Korean Journal. J. Korean Med. Rehabil. 2015, 25, 63–70. [Google Scholar] [CrossRef]
- Chaochao, Y.; Le, W.; Lihong, K.; Feng, S.; Chaoyang, M.; Yanjun, D.; Hua, Z. Acupoint combinations used for treatment of Alzheimer’s disease: A data mining analysis. J. Tradit. Chin. Med. 2018, 38, 943–952. [Google Scholar] [CrossRef]
- Shen, K.; Cho, Y.; Pascoe, E.M.; Hawley, C.M.; Oliver, V.; Hughes, K.M.; Baer, R.; Frazier, J.; Jarvis, E.; Tan, K.S.; et al. The SIESTA Trial: A Randomized Study Investigating the Efficacy, Safety, and Tolerability of Acupressure versus Sham Therapy for Improving Sleep Quality in Patients with End-Stage Kidney Disease on Hemodialysis. Evid. Based Complementary Altern. Med. 2017, 2017, 7570352. [Google Scholar] [CrossRef] [PubMed]
- Shang, C. Emerging paradigms in mind-body medicine. J. Altern. Complementary Med. 2001, 7, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Visocchi, M. Spinal cord stimulation and cerebral haemodynamics. Hydrocephalus 2006, 99, 111–116. [Google Scholar]
- Wolter, T.; Kieselbach, K. Cervical Spinal Cord Stimulation: An Analysis of 23 Patients with Long-term Follow-up. Pain Physician 2012, 15, 203–212. [Google Scholar] [PubMed]
- Kim, J.-H.; Cho, M.-R.; Park, G.-C.; Lee, J.-S. Effects of different acupuncture treatment methods on mild cognitive impairment: A study protocol for a randomized controlled trial. Trials 2019, 20, 551. [Google Scholar] [CrossRef]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Song, I.-B. An Introduction to Sasang Constitutional Medicine; Jimoondang: Seoul, Korea, 2005. [Google Scholar]
- Han, Y.R.; Lee, H.B.; Han, S.Y.; Kim, B.J.; Lee, S.J.; Chae, H. Systematic review of type-specific pathophysiological symptoms of Sasang typology. Integr. Med. Res. 2016, 5, 83–98. [Google Scholar] [CrossRef]
- Jin, H.J.; Baek, Y.; Kim, H.S.; Ryu, J.; Lee, S. Constitutional multicenter bank linked to Sasang constitutional phenotypic data. BMC Complement. Altern. Med. 2015, 15, 46. [Google Scholar] [CrossRef]
- Baek, Y.-H.; Jang, E.-S.; Park, K.-H.; Yoo, J.-H.; Jin, H.-J.; Lee, S.-W. Development and Validation of Brief KS-15 (Korea Sasang Constitutional Diagnostic Questionnaire) Based on Body Shape, Temperament and Symptoms. J. Sasang Const. Med. 2015, 27, 211–221. [Google Scholar] [CrossRef]
- Choi, S.-H. Longevity and Life Preservation in Eastern Medicine; Kyung Hee University Press: Seoul, Korea, 2009. [Google Scholar]
- Chin, J.; Park, J.; Yang, S.J.; Yeom, J.; Ahn, Y.; Baek, M.J.; Ryu, H.J.; Lee, B.H.; Han, N.E.; Ryu, K.H.; et al. Re-standardization of the Korean-Instrumental Activities of Daily Living (K-IADL): Clinical Usefulness for Various Neurodegenerative Diseases. Dement. Neurocognitive Disord. 2018, 17, 11–22. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1982, 17, 37–49. [Google Scholar] [CrossRef]
- Cho, M.J.; Bae, J.N.; Suh, G.H.; Hahm, B.J.; Kim, J.K.; Lee, D.W.; Kang, M.H. Validation of Geriatric Depression Scale, Korean Version (GDS) in the Assessment of DSM-III-R Major Depression. J. Korean Neuropsychiatr. Assoc. 1999, 38, 48–63. [Google Scholar]
- Han, J.W.; Kim, T.H.; Jhoo, J.H.; Park, J.H.; Kim, J.L.; Ryu, S.H.; Moon, S.W.; Choo, I.H.; Lee, D.W.; Yoon, J.C.; et al. A Normative Study of the Mini-Mental State Examination for Dementia Screening (MMSE-DS) and Its Short form (SMMSE-DS) in the Korean Elderly. J. Korean Geriatr. Psychiatry 2010, 14, 27–37. [Google Scholar]
- Choi, J.; Ku, B.; You, Y.G.; Jo, M.; Kwon, M.; Choi, Y.; Jung, S.; Ryu, S.; Park, E.; Go, H.; et al. Resting-state prefrontal EEG biomarkers in correlation with MMSE scores in elderly individuals. Sci. Rep. 2019, 9, 10468. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing, Vienna, Austria, 2011. R Foundation for Statistical Computing: Vienna, Austria, 2011. [Google Scholar]
- Van Buuren, S.; Groothuis-Oudshoorn, K. Mice: Multivariate Imputation by Chained Equations in R. J. Stat. Softw. 2010, 45, 67. [Google Scholar] [CrossRef]
- Rosnow, R.L.; Rosenthal, R.; Rubin, D.B. Contrasts and Correlations in Effect-Size Estimation. Psychol. Sci. 2016, 11, 446–453. [Google Scholar] [CrossRef]
- Task Force of The European Society of Cardiology; The North American Society of Pacing and Electrophysiology. Heart rate variability; Standards of measurement, physiological interpretation, and clinical use. Eur. Heart J. 1996, 17, 354–381. [Google Scholar] [CrossRef]
- Song, C.; Ikei, H.; Kobayashi, M.; Miura, T.; Taue, M.; Kagawa, T.; Li, Q.; Kumeda, S.; Imai, M.; Miyazaki, Y. Effect of forest walking on autonomic nervous system activity in middle-aged hypertensive individuals: A pilot study. Int. J. Environ. Res. Public Health 2015, 12, 2687–2699. [Google Scholar] [CrossRef]
- Lee, J.; Tsunetsugu, Y.; Takayama, N.; Park, B.J.; Li, Q.; Song, C.; Komatsu, M.; Ikei, H.; Tyrvainen, L.; Kagawa, T.; et al. Influence of forest therapy on cardiovascular relaxation in young adults. Evid. Based Complementary Altern. Med. 2014, 2014, 834360. [Google Scholar] [CrossRef]
- Aspinall, P.; Mavros, P.; Coyne, R.; Roe, J. The urban brain: Analysing outdoor physical activity with mobile EEG. Br. J. Sports Med. 2015, 49, 272–276. [Google Scholar] [CrossRef]
- Neale, C.; Aspinall, P.; Roe, J.; Tilley, S.; Mavros, P.; Cinderby, S.; Coyne, R.; Thin, N.; Bennett, G.; Thompson, C.W. The Aging Urban Brain: Analyzing Outdoor Physical Activity Using the Emotiv Affectiv Suite in Older People. J. Urban Health 2017, 94, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Shafer, K.J.; Siders, W.A.; Johnson, L.K.; Lukaski, H.C. Validity of segmental multiple-frequency bioelectrical impedance analysis to estimate body composition of adults across a range of body mass indexes. Nutrition 2009, 25, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Raja, M.K.; Raymer, G.H.; Moran, G.R.; Marsh, G.; Thompson, R.T. Changes in tissue water content measured with multiple-frequency bioimpedance and metabolism measured with 31P-MRS during progressive forearm exercise. J. Appl. Physiol. 2006, 101, 1070–1075. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.E.; Snyder, P.J. Electroencephalography and event-related potentials as biomarkers of mild cognitive impairment and mild Alzheimer’s disease. Alzheimer’s Dement 2008, 4 (Suppl. 1), 137–143. [Google Scholar] [CrossRef]
- Moretti, D. Individual analysis of EEG frequency and band power in mild Alzheimer’s disease. Clin. Neurophysiol. 2004, 115, 299–308. [Google Scholar] [CrossRef]
- Hogan, M.J.; Swanwick, G.R.J.; Kaiser, J.; Rowan, M.; Lawlor, B. Memory-related EEG power and coherence reductions in mild Alzheimer’s disease. Int. J. Psychophysiol. 2003, 49, 147–163. [Google Scholar] [CrossRef]
- Sardinha, L.B. Physiology of exercise and phase angle: Another look at BIA. Eur. J. Clin. Nutr. 2018, 72, 1323–1327. [Google Scholar] [CrossRef]
- Souza, M.F.; Tomeleri, C.M.; Ribeiro, A.S.; Schoenfeld, B.J.; Silva, A.M.; Sardinha, L.B.; Cyrino, E.S. Effect of resistance training on phase angle in older women: A randomized controlled trial. Scand. J. Med. Sci. Sports 2017, 27, 1308–1316. [Google Scholar] [CrossRef]
- Jun, M.H.; Kim, S.; Ku, B.; Cho, J.; Kim, K.; Yoo, H.R.; Kim, J.U. Glucose-independent segmental phase angles from multi-frequency bioimpedance analysis to discriminate diabetes mellitus. Sci. Rep. 2018, 8, 648. [Google Scholar] [CrossRef]
- Kyle, U.G.; Genton, L.; Pichard, C. Low phase angle determined by bioelectrical impedance analysis is associated with malnutrition and nutritional risk at hospital admission. Clin. Nutr. 2013, 32, 294–299. [Google Scholar] [CrossRef]
- Stapel, S.N.; Looijaard, W.G.P.M.; Dekker, I.M.; Girbes, A.R.J.; Weijs, P.J.M.; Straaten, H.M.O.V. Bioelectrical impedance analysis-derived phase angle at admission as a predictor of 90-day mortality in intensive care patients. Eur. J. Clin. Nutr. 2018, 72, 1019–1025. [Google Scholar] [CrossRef] [PubMed]
- Maddocks, M.; Kon, S.S.; Jones, S.E.; Canavan, J.L.; Nolan, C.M.; Higginson, I.J.; Gao, W.; Polkey, M.I.; Man, W.D. Bioelectrical impedance phase angle relates to function, disease severity and prognosis in stable chronic obstructive pulmonary disease. Clin. Nutr. 2015, 34, 1245–1250. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, O.; Yoon, S.L.; Williams, J.J. The value of bioelectrical impedance analysis and phase angle in the evaluation of malnutrition and quality of life in cancer patients—A comprehensive review. Eur. J. Clin. Nutr. 2015, 69, 1290–1297. [Google Scholar] [CrossRef] [PubMed]
- Shim, E.B.; Leem, C.H.; Kim, J.J.; Kim, J.Y. Lower cellular metabolic power can be an explanation for obesity trend in Tae-Eum type: Hypothesis and clinical observation. Integr. Med. Res. 2017, 6, 254–259. [Google Scholar] [CrossRef]
- Shim, E.B.; Lee, S.W.; Kim, J.Y.; Leem, C.H.; Earm, Y.E. Taeeum-type people in Sasang constitutional medicine have a reduced mitochondrial metabolism. Integr. Med. Res. 2012, 1, 41–45. [Google Scholar] [CrossRef]
- Baek, Y.; Park, K.; Lee, S.; Jang, E. The prevalence of general and abdominal obesity according to sasang constitution in Korea. BMC Complement. Altern. Med. 2014, 14, 298. [Google Scholar] [CrossRef]
- Cho, N.H.; Kim, J.Y.; Kim, S.S.; Lee, S.K.; Shin, C. Predicting type 2 diabetes using Sasang constitutional medicine. J. Diabetes Investig. 2014, 5, 525–532. [Google Scholar] [CrossRef]
- Song, K.H.; Yu, S.G.; Kim, J.Y. Prevalence of Metabolic Syndrome according to Sasang Constitutional Medicine in Korean Subjects. Evid. Based Complementary Altern. Med. 2012, 2012, 646794. [Google Scholar] [CrossRef]
- Lee, S.K.; Yoon, D.W.; Yi, H.; Lee, S.W.; Kim, J.Y.; Kim, J.K.; Hong, J.H.; Shin, C. So-eum type as an independent risk factor for irritable bowel syndrome: A population-based study in Korea. J. Altern. Complement. Med. 2014, 20, 846–852. [Google Scholar] [CrossRef]

| Dataset | Variable | Explanation |
|---|---|---|
| EEG | MEF [Hz] | Median frequency: the median frequency in the dominant intrinsic oscillatory frequency band of 4–13 Hz of the power spectrum |
| Pα [μV2] | Alpha band power: The spectral power integrated over the frequency range between 8 and 13 Hz (natural logarithmic scale) | |
| Pβ [μV2] | Beta band power: The spectral power integrated over the frequency range between 13 and 30 Hz (the natural logarithmic scale) | |
| ATR | Alpha/theta ratio: the power ratio of alpha rhythms (8–13 Hz) to theta rhythms (4–8 Hz) | |
| Bioimpedance | FFM [kg] | Fat-free mass |
| BFM [kg] | Body fat mass | |
| %BF [%] | Percent body fat (body fat/whole body mass) | |
| PhA_body | Phase angle of the whole body = (reactance of the whole body)/(impedance of the whole body) | |
| Imp_arm [Ω] | Impedance averaged over both arms | |
| Imp_leg [Ω] | Impedance averaged over both legs | |
| Reactance_arm [Ω] | Reactance averaged over both arms | |
| Reactance_leg [Ω] | Reactance averaged over both legs | |
| PhA_arm | Phase angle of both arms | |
| PhA_leg | Phase angle of both legs | |
| HRV | HF [msec2] | Spectral power in the high frequency (HF) range of HRV (0.15–0.4 Hz) |
| LF [msec2] | Spectral power in the low frequency (LF) range of HRV (0.04–0.15 Hz) | |
| %LF | LF power/(LF+HF power) | |
| HR [bpm] | Heart rate |
| Demographic Variable | Control Group | Breathing Program | Walking Program | p-Value |
|---|---|---|---|---|
| N (%) | 28 (31.8%) | 29 (33.0%) | 31 (35.2%) | |
| Missing cases | ||||
| EEG | 6 (20.7%) | 5 (17.2%) | 3 (9.7%) | 0.486 |
| HRV | 16 (61.5%) | 13 (44.8%) | 9 (29.0%) | 0.413 |
| Bioimpedance | 8 (27.6%) | 4 (13.8%) | 2 (6.5%) | 0.075 |
| SC Type | 0.214 | |||
| TE | 18 (64.3%) | 12 (41.4%) | 16 (51.6%) | |
| SE | 3 (10.7%) | 5 (17.2%) | 8 (25.8%) | |
| SY | 7 (25.0%) | 12 (41.4%) | 7 (22.6%) | |
| Sex: Female | 20 (69.0%) | 22 (75.9%) | 28 (90.3%) | 0.118 |
| Age [yr] | 74.4 ± 4.9 | 78.5 ± 6.9 | 72.9 ± 6.2 | 0.002 |
| Height [cm] | 154.9 ± 6.6 | 153.3 ± 10.2 | 151.9 ± 5.5 | 0.325 |
| Weight [kg] | 60.8 ± 8.1 | 58.4 ± 9.7 | 56.5 ± 8.6 | 0.178 |
| BMI [kg/m2] | 25.3 ± 3.2 | 23.8 ± 2.9 | 24.4 ± 3.3 | 0.168 |
| MMSE | 25.3 ± 3.5 | 23.5 ± 4.0 | 26.3 ± 4.1 | 0.024 |
| Smoking: Yes | 1 (3.6%) | 4 (13.8%) | 1 (3.2%) | 0.191 |
| Alcohol: Yes | 1 (3.6%) | 3 (10.3%) | 2 (6.5%) | 0.595 |
| Religion: Yes | 22 (78.6%) | 15 (51.7%) | 20 (64.5%) | 0.105 |
| Marital status: Married | 14 (50.0%) | 4 (13.8%) | 16 (51.6%) | 0.004 |
| Education level | 0.005 | |||
| None | 3 (10.7%) | 12 (41.4%) | 1 (3.2%) | |
| 1~3 years | 2 (7.1%) | 3 (10.3%) | 7 (22.6%) | |
| 4~6 years | 13 (46.4%) | 10 (34.5%) | 10 (32.3%) | |
| 7~9 years | 3 (10.7%) | 1 (3.4%) | 2 (6.5%) | |
| More than 10 years | 7 (25.0%) | 3 (10.3%) | 11 (35.5%) | |
| Medical history | ||||
| Hypertension | 13 (46.4%) | 17 (58.6%) | 15 (48.4%) | 0.609 |
| Diabetes | 5 (17.9%) | 7 (24.1%) | 5 (16.1%) | 0.714 |
| Dyslipidemia | 13 (46.4%) | 11 (37.9%) | 8 (25.8%) | 0.253 |
| Arthritis | 15 (53.6%) | 17 (58.6%) | 12 (38.7%) | 0.275 |
| Cerebrovascular disease | 1 (3.6%) | 2 (6.9%) | 1 (3.2%) | 0.758 |
| Depression | 1 (3.6%) | 2 (6.9%) | 0 (0.0%) | 0.338 |
| Parkinson’s disease | 0 (0.0%) | 0 (0.0%) | 1 (3.2%) | 0.395 |
| Etc. | 5 (17.9%) | 13 (44.8%) | 8 (25.8%) | 0.071 |
| Visits to forest [per month] | 2.9 ± 5.1 | 1.4 ± 5.6 | 1.8 ± 3.7 | 0.491 |
| Willing to participate in an FTP | 24 (85.7%) | 27 (93.1%) | 31 (100.0%) | 0.094 |
| Daily activity hours [hour/day] | 1.3 ± 1.7 | 0.9 ± 1.0 | 2.1 ± 3.2 | 0.114 |
| EEG | Control | Breathing Program | Walking Program | BP–CN | WP–CN | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | ||||||||
| MEF [Hz] | 8.47 | −0.45 ** (−0.78, −0.11) | 0.57 | 8.40 | −0.02 (−0.38, 0.34) | 0.02 | 8.40 | −0.40 * (−0.77, −0.03) | 0.40 | 0.43 (−0.11, 0.97) | 0.45 | 0.05 (−0.48, 0.57) | 0.05 |
| 2.71 | −0.10 (−0.33, 0.13) | 0.18 | 3.00 | −0.17 (−0.42, 0.08) | 0.28 | 2.65 | −0.15 (−0.42, 0.11) | 0.22 | −0.07 (−0.45, 0.31) | 0.11 | −0.06 (−0.42, 0.31) | 0.09 | |
| 1.86 | −0.10 (−0.37, 0.16) | 0.17 | 2.31 | −0.16 (−0.44, 0.12) | 0.24 | 2.09 | 0.13 (−0.16, 0.42) | 0.17 | −0.06 (−0.49, 0.37) | 0.08 | 0.23 (−0.18, 0.64) | 0.32 | |
| ATR | 1.25 | −0.06 (−0.13, 0.02) | 0.33 | 1.18 | 0.00 (−0.08, 0.08) | 0.02 | 1.22 | −0.15 *** (−0.23, −0.07) | 0.68 | 0.06 (−0.06, 0.18) | 0.29 | −0.09 (−0.21, 0.03) | 0.44 |
| Bioimpedance | Control | Breathing Program | Walking Program | BP–CN | WP–CN | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | ||||||||
| FFM [kg] | 40.70 | 0.72 (−0.30, 1.74) | 0.31 | 37.47 | −0.26 (−1.26, 0.74) | 0.10 | 38.28 | 0.20 (−0.92, 1.31) | 0.07 | −0.98 (−2.52, 0.55) | 0.36 | −0.53 (−2.01, 0.96) | 0.20 |
| BFM [kg] | 20.48 | 0.26 (−0.79, 1.30) | 0.11 | 20.21 | 0.62 (−0.43, 1.67) | 0.24 | 18.72 | 0.02 (−1.15, 1.18) | 0.01 | 0.36 (−1.30, 2.02) | 0.12 | −0.24 (−1.88, 1.40) | 0.08 |
| %BF [%] | 33.00 | −0.36 (−1.85, 1.13) | 0.11 | 34.48 | 0.75 (−0.76, 2.26) | 0.20 | 32.25 | −0.41 (−2.12, 1.31) | 0.09 | 1.11 (−1.26, 3.47) | 0.26 | −0.04 (−2.39, 2.30) | 0.01 |
| PhA_body | 5.38 | 0.29 *** (0.14, 0.44) | 0.85 | 4.91 | 0.48 *** (0.34, 0.63) | 1.30 | 5.42 | 0.20 * (0.03, 0.36) | 0.44 | 0.19 (−0.05, 0.43) | 0.45 | −0.09 (−0.32, 0.13) | 0.24 |
| Imp_arm [Ω] | 343.69 | 3.22 (−4.38, 10.82) | 0.18 | 346.11 | 14.25 *** (6.54, 21.96) | 0.74 | 347.20 | −1.19 (−9.63, 7.25) | 0.05 | 11.03 (−1.01, 23.08) | 0.51 | −4.41 (−16.13, 7.32) | 0.21 |
| Imp_leg [Ω] | 168.44 | 3.41 (−2.91, 9.74) | 0.24 | 180.63 | −3.86 (−10.13, 2.40) | 0.25 | 168.65 | 8.76 * (1.65, 15.87) | 0.46 | −7.28 (−17.27, 2.71) | 0.41 | 5.35 (−4.37, 15.07) | 0.31 |
| Reactance_arm [Ω] | 31.45 | 1.74 * (0.29, 3.19) | 0.52 | 28.07 | 6.32 *** (4.70, 7.95) | 1.55 | 33.92 | −0.56 (−2.18, 1.06) | 0.13 | 4.58 *** (2.14, 7.03) | 1.05 | −2.30 * (−4.57, −0.02) | 0.57 |
| Reactance_leg [Ω] | 15.52 | 1.23 * (0.23, 2.23) | 0.53 | 16.24 | −0.37 (−1.39, 0.65) | 0.14 | 14.58 | 2.25 *** (1.12, 3.39) | 0.74 | −1.60 (−3.21, 0.01) | 0.56 | 1.02 (−0.56, 2.60) | 0.37 |
| PhA_arm | 5.28 | 0.28 *** (0.14, 0.43) | 0.85 | 4.66 | 0.87 *** (0.70, 1.03) | 2.13 | 5.63 | 0.01 (−0.17, 0.20) | 0.03 | 0.58 *** (0.33, 0.84) | 1.29 | −0.27 * (−0.51, −0.03) | 0.64 |
| PhA_leg | 5.35 | 0.29 * (0.06, 0.52) | 0.54 | 5.16 | −0.06 (−0.30, 0.17) | 0.11 | 4.97 | 0.60 *** (0.35, 0.85) | 0.89 | −0.35 (−0.71, 0.02) | 0.54 | 0.31 (−0.04, 0.67) | 0.50 |
| HRV | Control | Breathing Program | Walking Program | BP–CN | WP–CN | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | (95% CI) | (95% CI) | (95% CI) | (95% CI) | (95% CI) | ||||||||
| HF [msec2] | 3.88 | −0.13 (−0.69, 0.44) | 0.13 | 4.27 | −0.46 (−0.98, 0.05) | 0.46 | 4.61 | 0.41 (−0.09, 0.92) | 0.35 | −0.34 (−1.22, 0.55) | 0.28 | 0.54 (−0.31, 1.39) | 0.48 |
| LF [msec2] | 3.50 | 0.00 (−0.55, 0.56) | 0.00 | 4.03 | −0.16 (−0.68, 0.35) | 0.16 | 3.99 | 0.28 (−0.22, 0.78) | 0.24 | −0.17 (−1.05, 0.72) | 0.14 | 0.27 (−0.56, 1.11) | 0.25 |
| %LF | 46.89 | −1.38 (−6.31, 3.55) | 0.16 | 48.25 | 1.48 (−3.19, 6.14) | 0.16 | 46.50 | −1.38 (−5.90, 3.14) | 0.13 | 2.86 (−5.00, 10.71) | 0.26 | 0.00 (−7.40, 7.40) | 0.00 |
| TP [msec2] | 5.47 | −0.04 (−0.53, 0.46) | 0.04 | 5.88 | 0.05 (−0.40, 0.50) | 0.06 | 6.14 | 0.17 (−0.27, 0.62) | 0.17 | 0.09 (−0.69, 0.86) | 0.08 | 0.21 (−0.54, 0.96) | 0.21 |
| HR [bpm] | 68.93 | 2.87 (−1.58, 7.33) | 0.38 | 67.64 | 5.97 ** (1.82, 10.11) | 0.73 | 64.17 | 3.02 (−1.04, 7.09) | 0.32 | 3.09 (−3.85, 10.04) | 0.32 | 0.15 (−6.61, 6.91) | 0.02 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yi, J.; Ku, B.; Kim, S.G.; Khil, T.; Lim, Y.; Shin, M.; Jeon, S.; Kim, J.; Kang, B.; Shin, J.; et al. Traditional Korean Medicine-Based Forest Therapy Programs Providing Electrophysiological Benefits for Elderly Individuals. Int. J. Environ. Res. Public Health 2019, 16, 4325. https://doi.org/10.3390/ijerph16224325
Yi J, Ku B, Kim SG, Khil T, Lim Y, Shin M, Jeon S, Kim J, Kang B, Shin J, et al. Traditional Korean Medicine-Based Forest Therapy Programs Providing Electrophysiological Benefits for Elderly Individuals. International Journal of Environmental Research and Public Health. 2019; 16(22):4325. https://doi.org/10.3390/ijerph16224325
Chicago/Turabian StyleYi, Jiyune, Boncho Ku, Seul Gee Kim, Taegyu Khil, Youngsuwn Lim, Minja Shin, Sookja Jeon, Jingun Kim, Byunghoon Kang, Jongyeon Shin, and et al. 2019. "Traditional Korean Medicine-Based Forest Therapy Programs Providing Electrophysiological Benefits for Elderly Individuals" International Journal of Environmental Research and Public Health 16, no. 22: 4325. https://doi.org/10.3390/ijerph16224325
APA StyleYi, J., Ku, B., Kim, S. G., Khil, T., Lim, Y., Shin, M., Jeon, S., Kim, J., Kang, B., Shin, J., Kim, K., Jeong, A. Y., Park, J. H., Choi, J., Cha, W., Shin, C., Shin, W., & Kim, J. U. (2019). Traditional Korean Medicine-Based Forest Therapy Programs Providing Electrophysiological Benefits for Elderly Individuals. International Journal of Environmental Research and Public Health, 16(22), 4325. https://doi.org/10.3390/ijerph16224325







