In Vitro Consequences of Electronic-Cigarette Flavoring Exposure on the Immature Lung
Abstract
1. Introduction
2. Materials and Methods
2.1. Flavored E-Cigarette Refill Solutions
2.2. Cell Culture
2.3. Cell Death
2.4. Isolated Airway and Vessel Studies
2.5. Statistical Analysis
3. Results
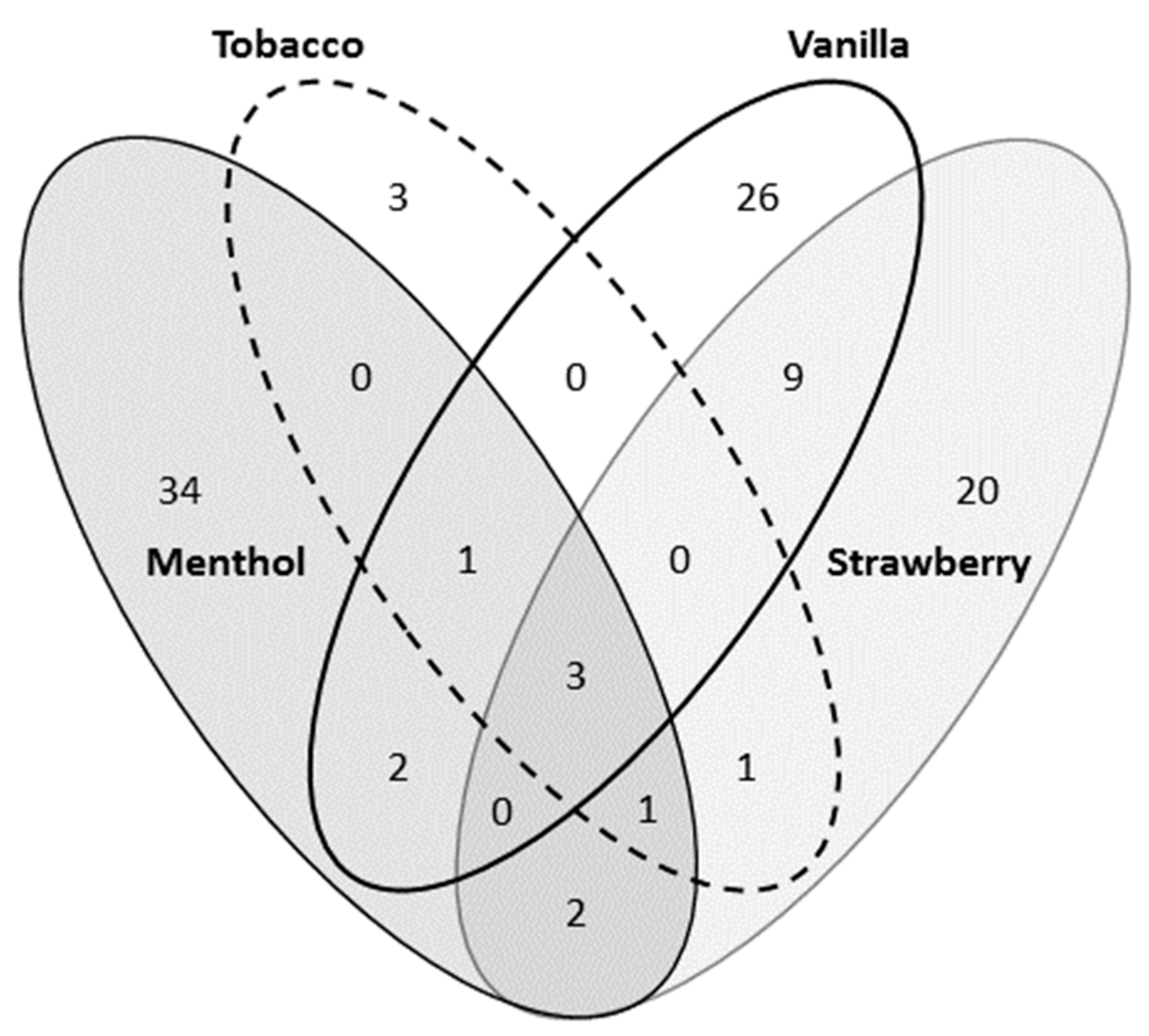
3.1. Chemical Analysis of Flavored Liquid Solutions
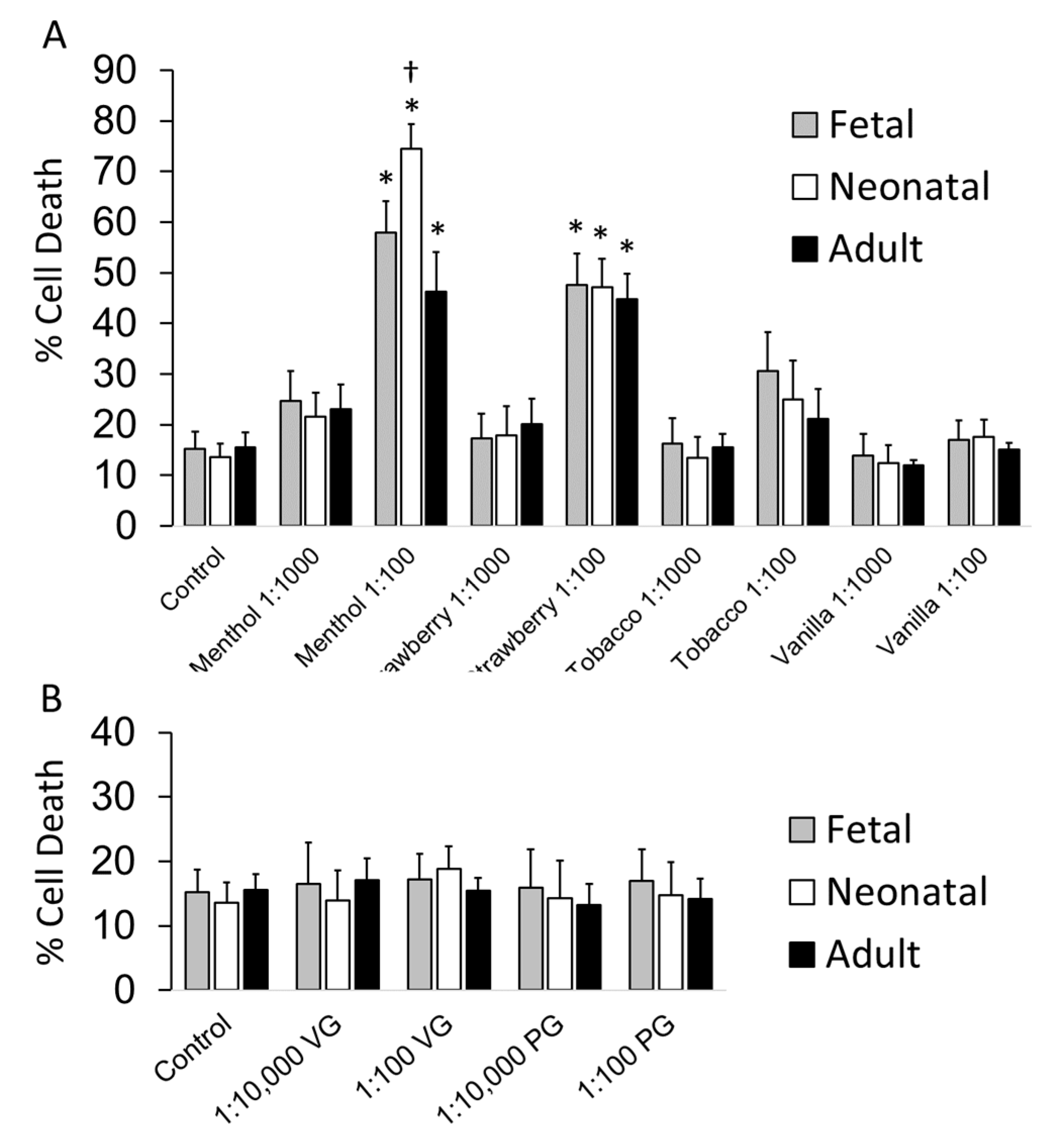
3.2. Cell Toxicity Studies
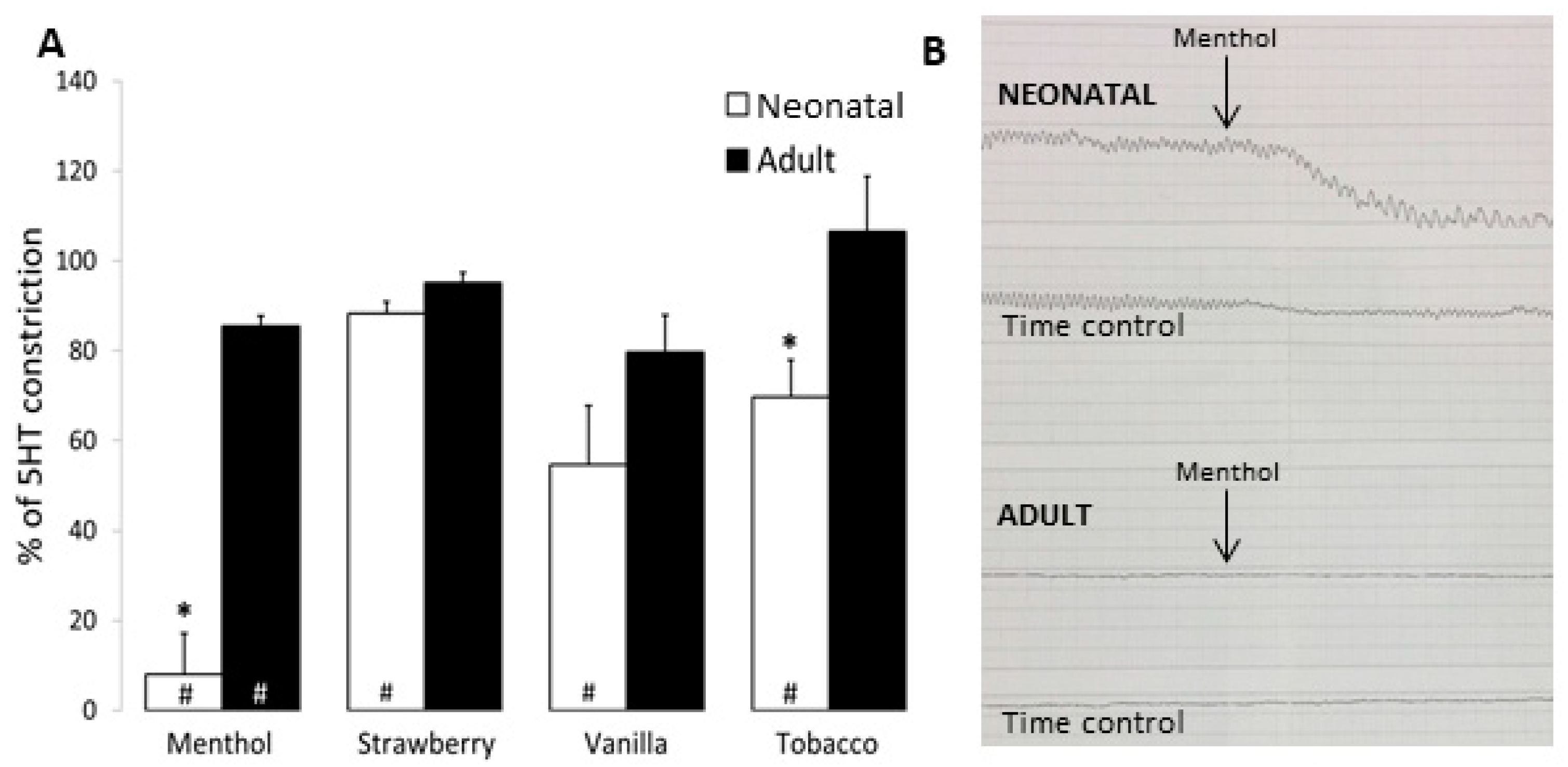
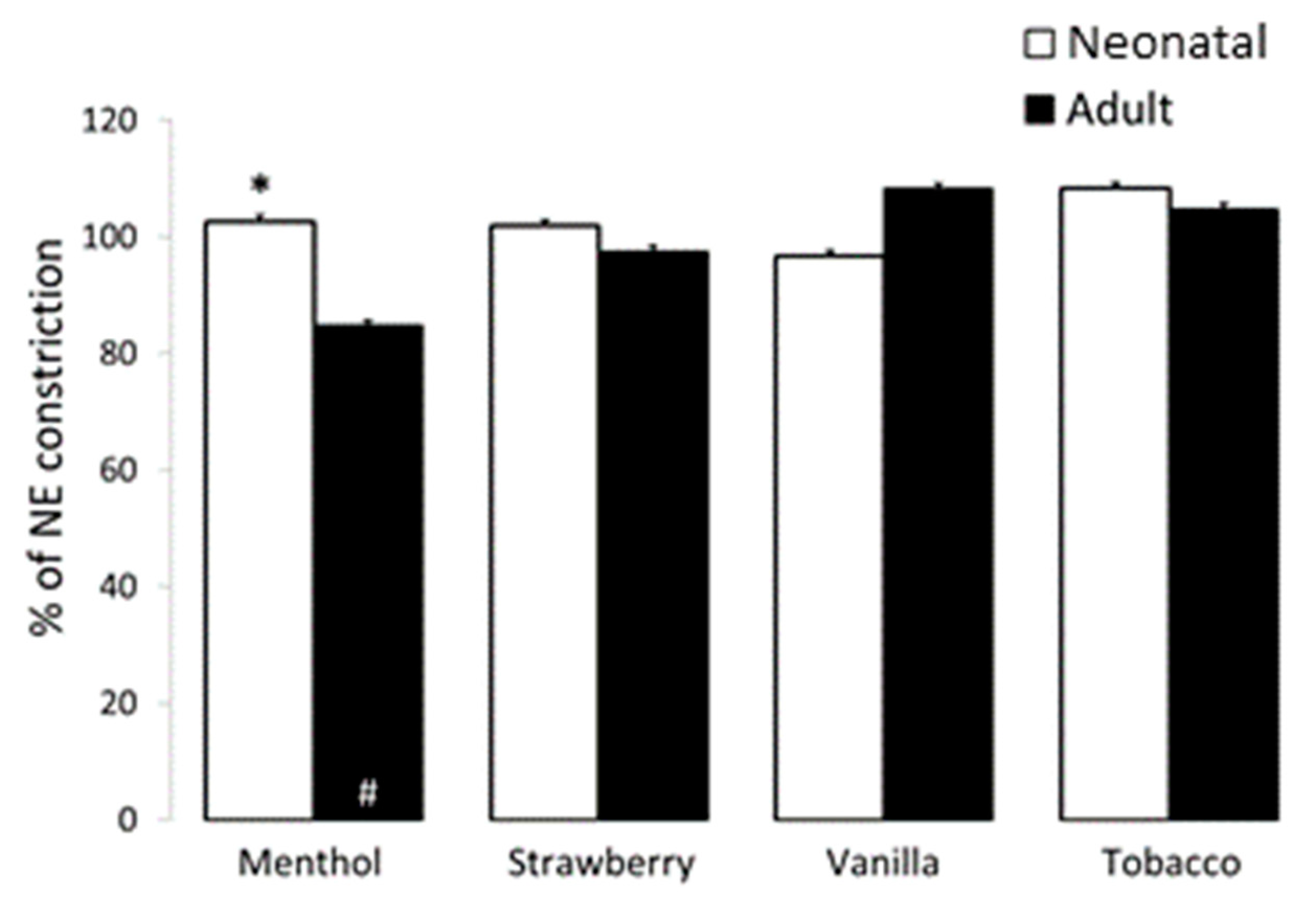
3.3. Airway and Vessel Reactivity Studies
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mangan, D. E-cigarette sales are smoking hot, set to hit $1.7 billion. CNBC Health and Science. 28 August 2013. Available online: https://www.cnbc.com/id/100991511 (accessed on 15 September 2019).
- Rowell, T.R.; Tarran, R. Will chronic e-cigarette use cause lung disease? Am. J. Phys. Lung Cell Mol. Phys. 2015, 309, L1398–L1409. [Google Scholar] [CrossRef]
- King, B.A.; Patel, R.; Nguyen, K.H.; Dube, S.R. Trends in awareness and use of electronic cigarettes among US adults, 2010–2013. Nicotine Tob. Res. 2015, 17, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.H.; Gamst, A.; Lee, M.; Cummins, S.; Yin, L.; Zoref, L. The use and perception of electronic cigarettes and snus among the U.S. population. PLoS ONE 2013, 8, e79332. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.H.; Tong, V.T.; Marynak, K.L.; King, B.A. US Adults’ Perceptions of the Harmful Effects During Pregnancy of Using Electronic Vapor Products Versus Smoking Cigarettes, Styles Survey, 2015. Prev. Chronic. Dis. 2016, 13, E175. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kahr, M.K.; Padgett, S.; Shope, C.D.; Griffin, E.N.; Xie, S.S.; Gonzalez, P.J.; Levison, J.; Mastrobattista, J.; Abramovici, A.R.; Northrup, T.F.; et al. A qualitative assessment of the perceived risks of electronic cigarette and hookah use in pregnancy. BMC Public Health 2015, 15, 1273. [Google Scholar] [CrossRef] [PubMed]
- Wagner, N.J.; Camerota, M.; Propper, C. Prevalence and Perceptions of Electronic Cigarette Use during Pregnancy. Matern. Child Health J. 2017, 21, 1655–1661. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, N.R.; Day, K.D.; Payakachat, N.; Franks, A.M.; McCain, K.R.; Ragland, D. Use and Risk Perception of Electronic Nicotine Delivery Systems and Tobacco in Pregnancy. Womens Health Issues 2018, 21, 1655–1661. [Google Scholar] [CrossRef]
- Oncken, C.; Ricci, K.A.; Kuo, C.L.; Dornelas, E.; Kranzler, H.R.; Sankey, H.Z. Correlates of Electronic Cigarettes Use Before and During Pregnancy. Nicotine Tob. Res. 2017, 19, 585–590. [Google Scholar] [CrossRef]
- Waldum, H.L.; Nilsen, O.G.; Nilsen, T.; Rorvik, H.; Syversen, V.; Sanvik, A.K.; Haugen, O.A.; Torp, S.H.; Brenna, E. Long-term effects of inhaled nicotine. Life Sci. 1996, 58, 1339–1346. [Google Scholar] [CrossRef]
- Husari, A.; Shihadeh, A.; Talih, S.; Hashem, Y.; El Sabban, M.; Zaatari, G. Acute Exposure to Electronic and Combustible Cigarette Aerosols: Effects in an Animal Model and in Human Alveolar Cells. Nicotine Tob. Res. 2016, 18, 613–619. [Google Scholar] [CrossRef]
- England, L.J.; Bunnell, R.E.; Pechacek, T.F.; Tong, V.T.; McAfee, T.A. Nicotine and the Developing Human: A Neglected Element in the Electronic Cigarette Debate. Am. J. Prev. Med. 2015, 49, 286–293. [Google Scholar] [CrossRef] [PubMed]
- McGrath-Morrow, S.A.; Hayashi, M.; Aherrera, A.; Lopez, A.; Malinina, A.; Collaco, J.M.; Neptune, E.; Klein, J.D.; Winickoff, J.P.; Breysse, P.; et al. The effects of electronic cigarette emissions on systemic cotinine levels, weight and postnatal lung growth in neonatal mice. PLoS ONE 2015, 10, e0118344. [Google Scholar] [CrossRef] [PubMed]
- Zelikoff, J.T.; Parmalee, N.L.; Corbett, K.; Gordon, T.; Klein, C.B.; Aschner, M. Microglia Activation and Gene Expression Alteration of Neurotrophins in the Hippocampus Following Early-Life Exposure to E-Cigarette Aerosols in a Murine Model. Toxicol. Sci. 2018, 162, 276–286. [Google Scholar] [CrossRef] [PubMed]
- Maritz, G.S.; Rayise, S.S. Effect of maternal nicotine exposure on neonatal rat lung development: Protective effect of maternal ascorbic acid supplementation. Exp. Lung Res. 2011, 37, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Maritz, G.S.; Thomas, R.A. The influence of maternal nicotine exposure on the interalveolar septal status of neonatal rat lung. Cell Biol. Int. 1994, 18, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Maritz, G.S.; Dennis, H. Maternal nicotine exposure during gestation and lactation interferes with alveolar development in the neonatal lung. Reprod. Fertil. Dev. 1998, 10, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Petre, M.A.; Petrik, J.; Ellis, R.; Inman, M.D.; Holloway, A.C.; Labiris, N.R. Fetal and neonatal exposure to nicotine disrupts postnatal lung development in rats: Role of VEGF and its receptors. Int. J. Toxicol. 2011, 30, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Rahman, A.; Dechkovskaia, A.M.; Sutton, J.M.; Chen, W.C.; Guan, X.; Khan, W.A.; Abou-Donia, M.B. Maternal exposure of rats to nicotine via infusion during gestation produces neurobehavioral deficits and elevated expression of glial fibrillary acidic protein in the cerebellum and CA1 subfield in the offspring at puberty. Toxicology 2005, 209, 245–261. [Google Scholar] [CrossRef]
- Pauly, J.R.; Sparks, J.A.; Hauser, K.F.; Pauly, T.H. In utero nicotine exposure causes persistent, gender-dependant changes in locomotor activity and sensitivity to nicotine in C57Bl/6 mice. Int. J. Dev. Neurosci. 2004, 22, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Chung, S.; Noh, J. Maternal Nicotine Exposure During Late Gestation and Lactation Increases Anxiety-Like and Impulsive Decision-Making Behavior in Adolescent Offspring of Rat. Toxicol. Res. 2016, 32, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Bahl, V.; Lin, S.; Xu, N.; Davis, B.; Wang, Y.H.; Talbot, P. Comparison of electronic cigarette refill fluid cytotoxicity using embryonic and adult models. Reprod. Toxicol. 2012, 34, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Berkelhamer, S.K.; Farrow, K.N. Developmental regulation of antioxidant enzymes and their impact on neonatal lung disease. Antioxid. Redox Signal. 2014, 21, 1837–1848. [Google Scholar] [CrossRef] [PubMed]
- Melville, J.M.; Moss, T.J. The immune consequences of preterm birth. Front. Neurosci. 2013, 7, 79. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, V. Developmental differences in the role of interleukins in hyperoxic lung injury in animal models. Front. Biosci. 2002, 7, d1624–d1633. [Google Scholar] [CrossRef] [PubMed]
- Berkelhamer, S.K.; Kim, G.A.; Radder, J.E.; Wedgwood, S.; Czech, L.; Steinhorn, R.H.; Schumacker, P.T. Developmental differences in hyperoxia-induced oxidative stress and cellular responses in the murine lung. Free Radic. Biol. Med. 2013, 61, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Datta, A.; Kim, G.A.; Taylor, J.M.; Gugino, S.F.; Farrow, K.N.; Schumacker, P.T.; Berkelhamer, S.K. Mouse lung development and NOX1 induction during hyperoxia are developmentally regulated and mitochondrial ROS dependent. Am. J. Phys. Lung Cell Mol. Phys. 2015, 309, L369–L377. [Google Scholar] [CrossRef] [PubMed]
- Makri, A.; Goveia, M.; Balbus, J.; Parkin, R. Children’s susceptibility to chemicals: A review by developmental stage. J. Toxicol. Environ. Health B Crit Rev. 2004, 7, 417–435. [Google Scholar] [CrossRef] [PubMed]
- Lambers, D.S.; Clark, K.E. The maternal and fetal physiologic effects of nicotine. Semin. Perinatol. 1996, 20, 115–126. [Google Scholar] [CrossRef]
- Leigh, N.J.; Lawton, R.I.; Hershberger, P.A.; Goniewicz, M.L. Flavourings significantly affect inhalation toxicity of aerosol generated from electronic nicotine delivery systems (ENDS). Tob. Control. 2016, 25, ii81–ii87. [Google Scholar] [CrossRef]
- Gerloff, J.; Sundar, I.K.; Freter, R.; Sekera, E.R.; Friedman, A.E.; Robinson, R.; Pagano, T.; Rahman, I. Inflammatory Response and Barrier Dysfunction by Different e-Cigarette Flavoring Chemicals Identified by Gas Chromatography-Mass Spectrometry in e-Liquids and e-Vapors on Human Lung Epithelial Cells and Fibroblasts. Appl. In Vitro Toxicol. 2017, 3, 28–40. [Google Scholar] [CrossRef]
- Lerner, C.A.; Sundar, I.K.; Yao, H.; Gerloff, J.; Ossip, D.J.; McIntosh, S.; Robinson, R.; Rahman, I. Vapors produced by electronic cigarettes and e-juices with flavorings induce toxicity, oxidative stress, and inflammatory response in lung epithelial cells and in mouse lung. PLoS ONE 2015, 10, e0116732. [Google Scholar] [CrossRef] [PubMed]
- Tierney, P.A.; Karpinski, C.D.; Brown, J.E.; Luo, W.; Pankow, J.F. Flavour chemicals in electronic cigarette fluids. Tob. Control. 2016, 25, e10–e15. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhan, Y.; Li, Q.; Zeng, D.D.; Leischow, S.J.; Okamoto, J. An Examination of Electronic Cigarette Content on Social Media: Analysis of E-Cigarette Flavor Content on Reddit. Int. J. Environ. Res. Public Health 2015, 12, 14916–14935. [Google Scholar] [CrossRef] [PubMed]
- Czogala, J.; Goniewicz, M.L.; Fidelus, B.; Zielinska-Danch, W.; Travers, M.J.; Sobczak, A. Secondhand exposure to vapors from electronic cigarettes. Nicotine Tob. Res. 2014, 16, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.W.; Marynak, K.L.; Agaku, I.T.; King, B.A. Secondhand Exposure to Electronic Cigarette Aerosol Among US Youths. JAMA Pediatr. 2017, 171, 490–492. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.M.; Naeher, L.P.; Yu, X.; Rathbun, S.L.; Muilenburg, J.L.; Wang, J.S. Air monitoring at large public electronic cigarette events. Int. J. Hyg. Environ. Health 2018, 221, 541–547. [Google Scholar] [CrossRef]
- Hutzler, C.; Paschke, M.; Kruschinski, S.; Henkler, F.; Hahn, J.; Luch, A. Chemical hazards present in liquids and vapors of electronic cigarettes. Arch. Toxicol. 2014, 88, 1295–1308. [Google Scholar] [CrossRef]
- Zahedi, A.; Phandthong, R.; Chaili, A.; Leung, S.; Omaiye, E.; Talbot, P. Mitochondrial Stress Response in Neural Stem Cells Exposed to Electronic Cigarettes. Science 2019, 16, 250–269. [Google Scholar] [CrossRef]
- Omaiye, E.E.; McWhirter, K.J.; Luo, W.; Pankow, J.F.; Talbot, P. High-Nicotine Electronic Cigarette Products: Toxicity of JUUL Fluids and Aerosols Correlates Strongly with Nicotine and Some Flavor Chemical Concentrations. Chem. Res. Toxicol. 2019, 32, 1058–1069. [Google Scholar] [CrossRef]
- Hua, M.; Omaiye, E.E.; Luo, W.; McWhirter, K.J.; Pankow, J.F.; Talbot, P. Identification of Cytotoxic Flavor Chemicals in Top-Selling Electronic Cigarette Refill Fluids. Sci. Rep. 2019, 9, 2782. [Google Scholar] [CrossRef]
- Omaiye, E.E.; McWhirter, K.J.; Luo, W.; Tierney, P.A.; Pankow, J.F.; Talbot, P. High concentrations of flavor chemicals are present in electronic cigarette refill fluids. Sci. Rep. 2019, 9, 2468. [Google Scholar] [CrossRef] [PubMed]
- Zare, S.; Nemati, M.; Zheng, Y. A systematic review of consumer preference for e-cigarette attributes: Flavor, nicotine strength, and type. PLoS ONE 2018, 13, e0194145. [Google Scholar] [CrossRef] [PubMed]
- Farrow, K.N.; Groh, B.S.; Schumacker, P.T.; Lakshminrusimha, S.; Czech, L.; Gugino, S.F.; Russell, J.A.; Steinhorn, R.H. Hyperoxia increases phosphodiesterase 5 expression and activity in ovine fetal pulmonary artery smooth muscle cells. Circ. Res. 2008, 102, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Lakshminrusimha, S.; Steinhorn, R.H. Pulmonary vascular biology during neonatal transition. Clin. Perinatol. 1999, 26, 601–619. [Google Scholar] [CrossRef]
- Hanouni, M.; Bernal, G.; McBride, S.; Narvaez, V.R.; Ibe, B.O. Hypoxia and hyperoxia potentiate PAF receptor-mediated effects in newborn ovine pulmonary arterial smooth muscle cells: Significance in oxygen therapy of PPHN. Physiol. Rep. 2016, 4, e12840. [Google Scholar] [CrossRef] [PubMed]
- Black, S.M.; Field-Ridley, A.; Sharma, S.; Kumar, S.; Keller, R.L.; Kameny, R.; Maltepe, E.; Datar, S.A.; Fineman, J.R. Altered Carnitine Homeostasis in Children With Increased Pulmonary Blood Flow Due to Ventricular Septal Defects. Pediatr. Crit. Care Med. 2017, 18, 931–934. [Google Scholar] [CrossRef]
- Lakshminrusimha, S.; Swartz, D.D.; Gugino, S.F.; Ma, C.X.; Wynn, K.A.; Ryan, R.M.; Russell, J.A.; Steinhorn, R.H. Oxygen concentration and pulmonary hemodynamics in newborn lambs with pulmonary hypertension. Pediatr. Res. 2009, 66, 539–544. [Google Scholar] [CrossRef]
- Fineman, J.R.; Wong, J.; Soifer, S.J. Hyperoxia and alkalosis produce pulmonary vasodilation independent of endothelium-derived nitric oxide in newborn lambs. Pediatr. Res. 1993, 33, 341–346. [Google Scholar] [CrossRef][Green Version]
- Lakshminrusimha, S.; Russell, J.A.; Steinhorn, R.H.; Ryan, R.M.; Gugino, S.F.; Morin, F.C., 3rd; Swartz, D.D.; Kumar, V.H. Pulmonary arterial contractility in neonatal lambs increases with 100% oxygen resuscitation. Pediatr. Res. 2006, 59, 137–141. [Google Scholar] [CrossRef]
- Zayek, M.; Cleveland, D.; Morin, F.C., 3rd. Treatment of persistent pulmonary hypertension in the newborn lamb by inhaled nitric oxide. J. Pediatr. 1993, 122, 743–750. [Google Scholar] [CrossRef]
- Pearl, J.M.; Nelson, D.P.; Raake, J.L.; Manning, P.B.; Schwartz, S.M.; Koons, L.; Shanley, T.P.; Wong, H.R.; Duffy, J.Y. Inhaled nitric oxide increases endothelin-1 levels: A potential cause of rebound pulmonary hypertension. Crit. Care Med. 2002, 30, 89–93. [Google Scholar] [CrossRef] [PubMed]
- McMullan, D.M.; Bekker, J.M.; Johengen, M.J.; Hendricks-Munoz, K.; Gerrets, R.; Black, S.M.; Fineman, J.R. Inhaled nitric oxide-induced rebound pulmonary hypertension: Role for endothelin-1. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H777–H785. [Google Scholar] [CrossRef] [PubMed]
- Thebaud, B.; Abman, S.H. Bronchopulmonary dysplasia: Where have all the vessels gone? Roles of angiogenic growth factors in chronic lung disease. Am. J. Respir. Crit. Care Med. 2007, 175, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Misra, M.; Leverette, R.D.; Cooper, B.T.; Bennett, M.B.; Brown, S.E. Comparative in vitro toxicity profile of electronic and tobacco cigarettes, smokeless tobacco and nicotine replacement therapy products: E-liquids, extracts and collected aerosols. Int. J. Environ. Res. Public Health 2014, 11, 11325–11347. [Google Scholar] [CrossRef] [PubMed]
- Rowell, T.R.; Reeber, S.L.; Lee, S.L.; Harris, R.A.; Nethery, R.C.; Herring, A.H.; Glish, G.L.; Tarran, R. Flavored e-cigarette liquids reduce proliferation and viability in the CALU3 airway epithelial cell line. Am. J. Phys. Lung Cell Mol. Phys. 2017, 313, L52–L66. [Google Scholar] [CrossRef] [PubMed]
- Ito, S.; Kume, H.; Shiraki, A.; Kondo, M.; Makino, Y.; Kamiya, K.; Hasegawa, Y. Inhibition by the cold receptor agonists menthol and icilin of airway smooth muscle contraction. Pulm. Pharmacol. Ther. 2008, 21, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.W.; Liu, S.C.; Chao, P.Z.; Lee, F.P. Menthol inhibiting parasympathetic function of tracheal smooth muscle. Int. J. Med. Sci. 2016, 13, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Bitzer, Z.T.; Goel, R.; Reilly, S.M.; Elias, R.J.; Silakov, A.; Foulds, J.; Muscat, J.; Richie, J.P., Jr. Effect of flavoring chemicals on free radical formation in electronic cigarette aerosols. Free Radic. Biol. Med. 2018, 120, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Budinger, G.R.; Mutlu, G.M.; Urich, D.; Soberanes, S.; Buccellato, L.J.; Hawkins, K.; Chiarella, S.E.; Radigan, K.A.; Eisenbart, J.; Agrawal, H.; et al. Epithelial cell death is an important contributor to oxidant-mediated acute lung injury. Am. J. Respir. Crit. Care Med. 2011, 183, 1043–1054. [Google Scholar] [CrossRef]
- Korsmeyer, S.J.; Yin, X.M.; Oltvai, Z.N.; Veis-Novack, D.J.; Linette, G.P. Reactive oxygen species and the regulation of cell death by the Bcl-2 gene family. Biochim. Biophys. Acta 1995, 1271, 63–66. [Google Scholar] [CrossRef]
- Kjaeve, J.; Ingebrigtsen, T.; Naess, L.; Bjertnaes, L.; Vaage, J. Methylprednisolone attenuates airway and vascular responses induced by reactive oxygen species in isolated, plasma-perfused rat lungs. Free Radic. Res. 1996, 25, 407–414. [Google Scholar] [CrossRef] [PubMed]
- National Cancer Institute Tobacco Control Research. Research Topic: Menthol and Tobacco; U.S. Department of Health and Human Services, National Institute of Health: Bethesda, MD, USA, 2009.
- Villanti, A.C.; Collins, L.K.; Niaura, R.S.; Gagosian, S.Y.; Abrams, D.B. Menthol cigarettes and the public health standard: A systematic review. BMC Public Health 2017, 17, 983. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Foreman, M.G.; Demeo, D.L.; Bhatt, S.P.; Hansel, N.N.; Wise, R.A.; Soler, X.; Bowler, R.P. Menthol cigarette smoking in the COPDGene cohort: Relationship with COPD, comorbidities and CT metrics. Respirology 2015, 20, 108–114. [Google Scholar] [CrossRef] [PubMed]
- Famele, M.; Ferranti, C.; Abenavoli, C.; Palleschi, L.; Mancinelli, R.; Draisci, R. The chemical components of electronic cigarette cartridges and refill fluids: Review of analytical methods. Nicotine Tob. Res. 2015, 17, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Behar, R.Z.; Luo, W.; McWhirter, K.J.; Pankow, J.F.; Talbot, P. Analytical and toxicological evaluation of flavor chemicals in electronic cigarette refill fluids. Sci. Rep. 2018, 8, 8288. [Google Scholar] [CrossRef] [PubMed]
- Shields, P.G.; Berman, M.; Brasky, T.M.; Freudenheim, J.L.; Mathe, E.; McElroy, J.P.; Song, M.A.; Wewers, M.D. A Review of Pulmonary Toxicity of Electronic Cigarettes in the Context of Smoking: A Focus on Inflammation. Cancer Epidemiol. Biomarkers Prev. 2017, 26, 1175–1191. [Google Scholar] [CrossRef] [PubMed]
- Behrsing, H.; Hill, E.; Raabe, H.; Tice, R.; Fitzpatrick, S.; Devlin, R.; Pinkerton, K.; Oberdorster, G.; Wright, C.; Wieczorek, R.; et al. In vitro exposure systems and dosimetry assessment tools for inhaled tobacco products: Workshop proceedings, conclusions and paths forward for in vitro model use. Altern. Lab. Anim. 2017, 45, 117–158. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.H.; Sun, J.Y.; Bonnevie, E.; Cummins, S.E.; Gamst, A.; Yin, L.; Lee, M. Four hundred and sixty brands of e-cigarettes and counting: Implications for product regulation. Tob. Control. 2014, 23, iii3–iii9. [Google Scholar] [CrossRef]
- Sosnowski, T.R.; Kramek-Romanowska, K. Predicted Deposition of E-Cigarette Aerosol in the Human Lungs. J. Aerosol. Med. Pulm. Drug Deliv. 2016, 29, 299–309. [Google Scholar] [CrossRef]
| Menthol | Strawberry | Tobacco | Vanilla | |
|---|---|---|---|---|
| All chemicals | ||||
| Batch 1 | 76 | 83 | 37 | 77 |
| Batch 2 | 72 | 72 | 39 | 66 |
| Propylene glycol (PG) | Positive | Positive | Positive | Positive |
| Vegetable glycerin (VG) | Positive | Positive | Positive | Positive |
| Nicotine | Negative | Negative | Negative | Negative |
| Flavoring chemicals | ||||
| Batch 1 | 44 | 39 | 12 | 43 |
| Batch 2 | 35 | 34 | 14 | 34 |
| % flavorings | 53% | 47% | 34% | 54% |
| Major flavoring chemicals detected | (±)-Menthol Pulegone Alpha-pinene Anethol (E)-Cinnamyl acetate Linalool | Methyl cinnamate Benzyaldehyde Isovaleraldehyde Linalool | Beta-damascene Isoamyl butyrate | Dihydrojasmone lactone Para-anisaldehyde Acetoin Fruity ketal Vanillin |
| Fetal | Neonatal | Adult | |
|---|---|---|---|
| Menthol | 0.62 | 0.59 | 1.62 |
| Strawberry | 1.14 | 1.34 | 2.15 |
| Tobacco | 2.23 | 2.95 | 5.60 |
| Vanilla | >10 | 6.61 | >10 |
| PG | >10 | >10 | >10 |
| VG | >10 | 7.19 | >10 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berkelhamer, S.K.; Helman, J.M.; Gugino, S.F.; Leigh, N.J.; Lakshminrusimha, S.; Goniewicz, M.L. In Vitro Consequences of Electronic-Cigarette Flavoring Exposure on the Immature Lung. Int. J. Environ. Res. Public Health 2019, 16, 3635. https://doi.org/10.3390/ijerph16193635
Berkelhamer SK, Helman JM, Gugino SF, Leigh NJ, Lakshminrusimha S, Goniewicz ML. In Vitro Consequences of Electronic-Cigarette Flavoring Exposure on the Immature Lung. International Journal of Environmental Research and Public Health. 2019; 16(19):3635. https://doi.org/10.3390/ijerph16193635
Chicago/Turabian StyleBerkelhamer, Sara K., Justin M. Helman, Sylvia F. Gugino, Noel J. Leigh, Satyan Lakshminrusimha, and Maciej L. Goniewicz. 2019. "In Vitro Consequences of Electronic-Cigarette Flavoring Exposure on the Immature Lung" International Journal of Environmental Research and Public Health 16, no. 19: 3635. https://doi.org/10.3390/ijerph16193635
APA StyleBerkelhamer, S. K., Helman, J. M., Gugino, S. F., Leigh, N. J., Lakshminrusimha, S., & Goniewicz, M. L. (2019). In Vitro Consequences of Electronic-Cigarette Flavoring Exposure on the Immature Lung. International Journal of Environmental Research and Public Health, 16(19), 3635. https://doi.org/10.3390/ijerph16193635






