Salt Reduction Intervention in Families Investigating Metabolic, Behavioral and Health Effects of Targeted Intake Reductions: Study Protocol for a Four Months Three-Armed, Randomized, Controlled “Real-Life” Trial
Abstract
1. Introduction
- To examine the effects of two different salt reduction strategies ((A) gradually lowering salt content in bread; (B) intervention combining salt reduced bread and dietary counseling) on intake of salt, sodium and potassium intake, the sodium/potassium ratio and the overall dietary intake,
- To examine the effects of different strategies of salt reduction on selected cardiovascular risk factors (blood pressure, blood lipids, renin, aldosterone, norepinephrine, epinephrine),
- To test effects of different levels of salt reduction on salt sensitivity and preference.
2. Materials and Methods
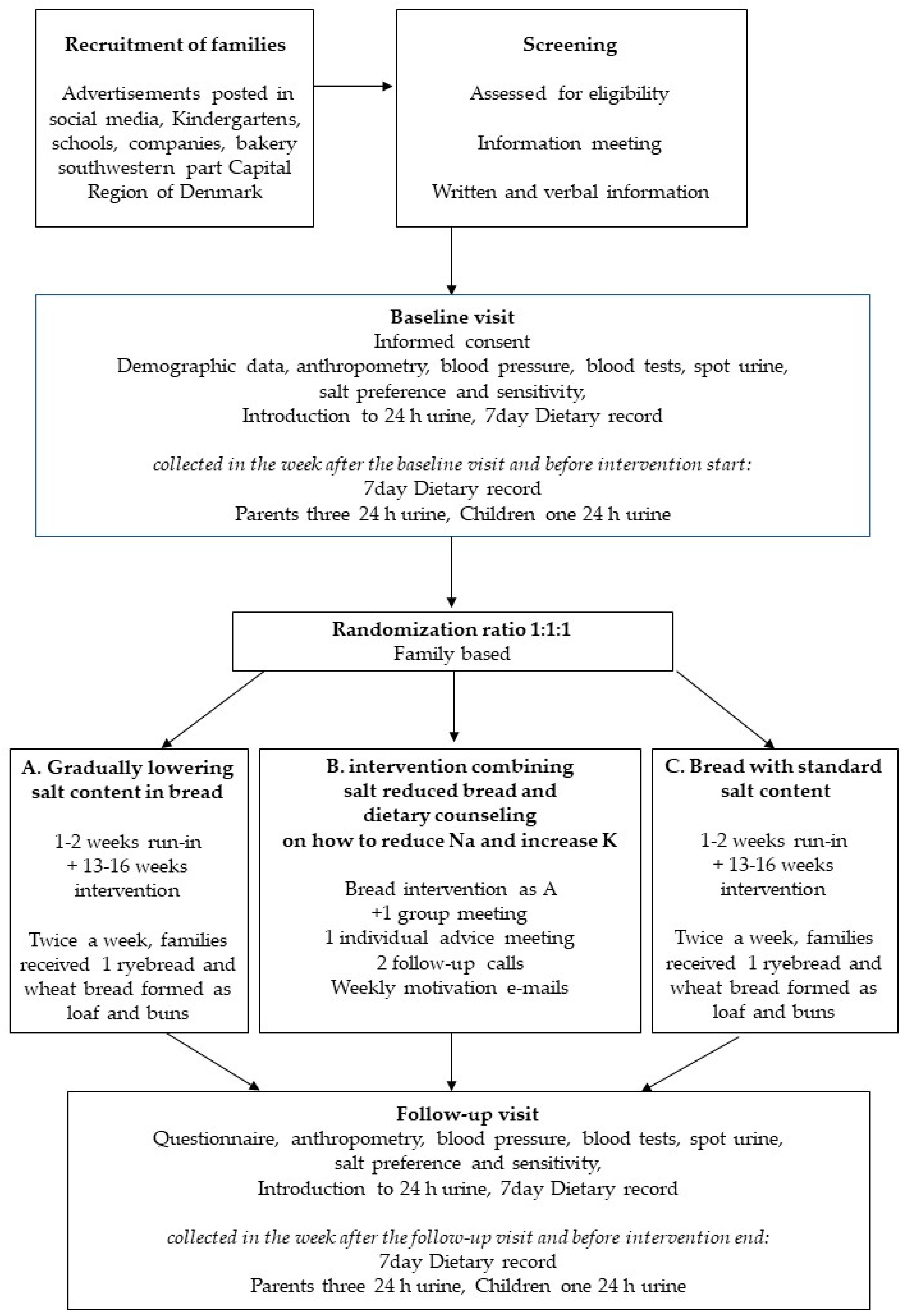
2.1. Study Design
2.2. Study Setting, Sample and Recruitment Procedure
2.3. Ethics
2.4. Randomization
2.5. Blinding
2.6. Sample Size
2.7. Study Principles
2.8. Intervention Overview
2.8.1. Intervention A: Salt Reduced Bread
2.8.2. Intervention B: Salt Reduced Bread Combined with Dietary Counseling
2.8.3. Control Group
2.9. Bread Production
2.10. Theoretical Framework of the Dietary Counseling Intervention
2.10.1. Salt Intake Screener
2.10.2. Standardization of Dietary Counseling
2.11. Outcome Measures
2.11.1. Primary Outcome Measure
Urine Collection
2.11.2. Secondary Outcome Measures
Assessment of Blood Pressure and Biochemistry
Salt Sensitivity and Preference
Dietary Assessment
2.11.3. Additional Variables
Anthropometry
Covariates
2.12. Data Management
2.13. Statistical Analysis Plan
2.13.1. Recruitment and Withdrawals
2.13.2. Baseline Data
2.13.3. Compliance
2.13.4. Primary Analyses of the Primary Outcome
Sensitivity Analyses of Primary Outcome
Subgroup Analyses of Primary Outcome
2.13.5. Analyses of Secondary Outcomes
- (1)
- To examine the effects of different levels of salt reduction on selected cardiovascular risk factors:
- (a)
- Potassium and sodium to potassium ratio (measured in 24 urine)
- (b)
- Blood pressure, blood lipids (triglyceride, total cholesterol, HDL and LDL)
- (c)
- The renin-angiotensin system (renin, aldosterone measured in blood)
- (d)
- The sympathetic nervous response (metanefrines measured in blood)
- (2)
- To test effects of different levels of salt reduction on salt taste sensitivity and preference
- (3)
- To test effects of different levels of salt reduction on the overall dietary intake and identify possible explanations of salt intake and salt excretion levels.
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Data Category | Information |
|---|---|
| Primary registry and trial identification number | Clinical Trials.gov: NCT03810885 |
| Date of registration in primary registry | January 2019 |
| Secondary identifying numbers | The Ethics Committee of the Capital Region of Denmark:H-17030995 |
| The Danish Data Protection Agency: CSU-FCFS-2017-017 | |
| Source(s) of monetary or material support | The Danish Heart Foundation, The Research Fond of the Capital Region of Denmark, The Toyota Foundation, Ministry of Environment and Food of Denmark, Axel Muusfeldt’s Foundation and Sofus Friis’ Foundation and the Technical University of Denmark |
| Primary sponsor | The Danish Heart Foundation |
| Secondary sponsor(s) | The Research Fond of the Capital Region of Denmark, The Toyota Foundation, Ministry of Environment and Food of Denmark, Axel Muusfeldt’s Foundation and Sofus Friis’ Foundation and the Technical University of Denmark |
| Contact for public queries | MSc, PhD kirsten.bjoernsbo@regionh.dk |
| Contact for scientific queries | MSc, PhD ulla.toft@regionh.dk |
| Public title | Saltintervention: Bread and Health |
| Scientific title | SalT Reduction InterVEntion (STRIVE): Examination of the Metabolic, Behavioral and Health Consequences of Reducing Salt Intake. A Randomized Controlled Trial in a Real Life Setting |
| Country of recruitment | Denmark |
| Health condition(s) or problems studied | Salt intake |
| Intervention(s) | Salt reduced bread |
| Salt reduced bread + Dietary Counselling | |
| Control: Normal bread | |
| Key inclusion and exclusion criteria | Inclusion Criteria:
|
| Study type | Interventional |
| Allocation: Randomized | |
| Blinding: Blinded to participants and outcome assessors | |
| Data of first enrollment | February 2018 |
| Target sample size | 120 families |
| Recruitment status | 96 families |
| Primary outcome(s) | Change from Baseline sodium level in 24 h urine at 4 months (follow-up) |
| Key secondary outcomes | Change from baseline levels regarding BP, Blood lipids, potassium, Na/K ratio, salt sensitivity and preference, dietary intake |
References
- Campbell, N.; Correa-Rotter, R.; Neal, B.; Cappuccio, F. New evidence to the health impact of reducing salt intake. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 617–619. [Google Scholar] [CrossRef] [PubMed]
- Aburto, N.J.; Ziolkovska, A.; Hooper, L.; Elliot, P.; Cappuccio, F.P.; Meerpohl, J.J. Effect of lower sodium intake on health: Systematic review and meta-analyses. BMJ 2013, 346, 1–20. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Report of the Formal Meeting of Member States to Conclude the Work on the Comprehensive Global Monitoring Framework, Including Indicators, and a set of Voluntary global Targets for the Prevention and Control of non-Communicable Diseases; Worlds Health Organization Press: Geneva, Switzerland, 2012. [Google Scholar]
- Scientific Report of the 2015 Dietary Guidelines Advisory Committee. Advisory Report to the Secretary of Health and Human Services and Secretary of Agriculture; U.S. Department of Agriculture, Agricultural Research Servicer: Washington DC, USA, 2015.
- Cobiac, L.; Vos, T.; Veerman, J. Cost-effectiveness of interventions to reduce dietary salt intake. Heart 2010, 96, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; MacGregor, G.A. Importance of salt in determining blood pressure in children: Meta-analysis of controlled trials. Hypertension 2006, 48, 861–869. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, C.; Jørgensen, T.; Andreasen, A.H.; Siganos, G.; Toft, U. Konsekvensberegninger for Reduktion af Danskernes Saltindtag. Forskningscenter for Forebyggelse og Sund. Rapport Udarbejdet for FøDevarestyrelsen, Februar 2012. Available online: https://www.frederiksberghospital.dk/ckff/pnw/Documents/Konsekvensberegninger%20for%20reduktion%20af%20danskernes%20saltindtag.pdf (accessed on 18 September 2019).
- Liem, D.G. Infants’ and Children’s Salt Taste Perception and Liking: A Review. Nutrients 2017, 9, 1011. [Google Scholar] [CrossRef] [PubMed]
- Brown, I.; Tzoulaki, I.; Candeias, V.; Elliot, P. Salt intakes around the world: Implications for public health. Int. J. Epidemiol. 2009, 38, 791–813. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, Y. Tracking of blood pressure from childhood to adulthood: A systematic review and meta-regression analysis. Circulation 2008, 117, 3171–3180. [Google Scholar] [CrossRef] [PubMed]
- Smith-Spangler, C.; Juusole, J.; Enns, E.; Owens, D.; Garber, A. Population strategies to decrease sodium intake and the burden of cardiovascular disease: A cost-effectiveness analysis. Ann. Int. Med. 2010, 152, 481–483. [Google Scholar] [CrossRef] [PubMed]
- He, F.J.; Pombo-Rodrigues, S.; MacGregor, G.A. Salt reduction in England from 2003 to 2011: Its relationship to blood pressure, stroke and ischaemic heart disease mortality. BMJ Open 2014, 4, e004549. [Google Scholar] [CrossRef]
- Institute of Medicine (IOM). Sodium Intake in Populations: Assessment of Evidence; The National Academies Press: Washington, DC, USA, 2013. [Google Scholar]
- Graudal, N.; Hubeck-Graudal, T.; Jurgens, G.; McCarron, D.A. The significance of duration and amount of sodium reduction intervention in normotensive and hypertensive individuals: A meta-analysis. Adv. Nutr. 2015, 6, 169–177. [Google Scholar] [CrossRef]
- Cobb, L.K.; Anerson, C.A.M.; Elliot, P.; Hu, F.B.; Liu, K.; Neaton, J.D.; Whelton, P.K.; Woodward, M.; Appel, L.J. Methodological issues in cohort studies that relate sodium intake to cardiovascular disease outcomes: A science advisory form the American Heart Association. Circulation 2014, 129, 1173–1186. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.; Dunford, E.; Hawkes, C.; Neal, B. Salt reduction initiatives around the world. J. Hypertens. 2011, 29, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Graudal, N.A.; Hubeck-Graudal, T.; Jurgens, G. Effects of low-sodium diet vs. high-sodium diet on blood pressure, renin, aldosterone, catecholamines, cholesterol, and triglyceride (Cochrane Review). Am. J. Hypertens. 2012, 25, 1–15. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.; Mente, A.; Rangarajan, S.; McQueen, M.B.; Wang, X.Y.; Liu, X.S.; Yan, H.; Lee, S.F.; Mony, P.; Devanath, A.; et al. Urinary sodium and potassium excretion, mortality, and cardiovascular events. N. Engl. J. 2014, 371, 612–623. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Liu, T.; Kuklina, E.V.; Flanders, D.; Hong, Y.; Gillespie, C.; Chang, M.-H.; Gwinn, M.; Dowling, N.; Khoury, M.J.; et al. Sodium and potassium intake and mortality among US adults. Arch. Int. Med. 2011, 171, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-Y.; Hu, Y.-W.; Yue, C.-S.J.; Wen, Y.-W.; Yeh, W.-T.; Hsu, L.-S.; Tsai, S.-Y.; Pan, W.-H. Effect of potassium-enriched salt on cardiovascular mortality and medical expenses of elderly men. Am. J. Lin. Nutr. 2006, 83, 1289–1296. [Google Scholar] [CrossRef] [PubMed]
- Perez, V.; Chang, E.T. Sodium-to-Potassium Ratio and Blood Pressure, Hypertension, and Related Factors. American Society for Nutrition. Adv. Nutr. 2014, 5, 712–741. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Macgregor, G. Reducing population salt intake worldwide: From evidence to implementation. Prog. Cardiovasc. Dis. 2010, 52, 363–382. [Google Scholar] [CrossRef]
- Kasey, J.C.; Abigail, S.; Huffman, M.D.; Jenner, K.; Xavier, D.; Dunford, E.K. Differences in the sodium content of bread products in the USA-and UK: Implications for policy. Public Health Nutr. 2018, 21, 632–636. [Google Scholar] [CrossRef]
- Taylor, C.; Doyle, M.; Webb, D. The safety of sodium reduction in the food supply: A cross-discipline balancing act—Workshop proceedings. Crit. Rev. Food Sci. Nutr. 2018, 58, 1650–1659. [Google Scholar] [CrossRef]
- Hendrie, G.A.; Brindal, E.; Corsini, N.; Gardner, C.; Baird, D.; Golley, R.K. Combined home and school appendi prevention interventions for children: What behavior change strategies and intervention characteristics are associated with effectiveness? Health Educ. Behav. 2012, 39, 159–171. [Google Scholar] [CrossRef] [PubMed]
- Danish National Survey on Diet and Physical Activity (DANSDA), 2011–2013, Personal Communication, Ellen Trolle. Available online: https://www.food.dtu.dk/english/service/about-the-institute/facilities-and-infrastructure/data-on-danes-dietary-habits (accessed on 18 September 2019).
- Trolle, E.; Saxholt, E.; Knuthsen, P. Saltindhold I brød og morgencerealier. E-artikel fra DTU Fødevareinstituttet, nr. 4. 2016. Available online: https://orbit.dtu.dk/en/publications/saltindhold-i-broed-og-morgenmadscerealier(38ec212d-412a-4d22-9e9f-b08dd7565cd9).html (accessed on 18 September 2019).
- The Danish Study of Functional Disorders (DanFunD), 2014, Personal Communication Ulla Toft. Available online: https://www.frederiksberghospital.dk/ckff/sektioner/SSF/saltogsundhed/monitorering/Sider/default.aspx (accessed on 18 September 2019).
- Chan, A.-W.; Tetzlaff, J.M.; Gøtzsche, P.C.; Altman, D.G.; Mann, H.; Berlin, J.A.; Dickersin, K.; Hróbjartsson, A.; Schulz, K.F.; Parulekar, W.R.; et al. SPIRIT 2013 explanation and elaboration: Guidance for protocols of clinical trials. BMJ 2013, 346, e7586. [Google Scholar] [CrossRef] [PubMed]
- Boutron, I.; Moher, D.; Altman, D.G.; Schulz, K.F.; Ravaud, P. Methods and processes of the CONSORT Group: Example of an extension for trials assessing nonpharmacologic treatments. Ann. Int. Med. 2008, 148, W60–W66. [Google Scholar] [CrossRef] [PubMed]
- Madsen, K.H.; Rasmussen, L.B.; Andersen, R.; Mølgaard, C.; Jakobsen, J.; Bjerrum, P.; Andersen, E.W.; Mejborn, H.; Tetens, I. Randomized controlled trial of the effects of vitamin D–fortified milk and bread on serum 25-hydroxyvitamin D concentrations in families in Denmark during winter: The VitmaD study1–3. Am. J. Clin. Nutr. 2013, 98, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Keyhole Label. Available online: https://altomkost.dk/english/ (accessed on 18 September 2019).
- The Danish Whole Grain Partnership. Available online: https://fuldkorn.dk/english/ (accessed on 18 September 2019).
- Cobb, L.K.; Appel, L.J.; Anderson, C.A.M. Strategies to Reduce Dietary Sodium Intake. Cardiovasc. Med. 2012, 14, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Glanz, K.; Rimer, B.; Viswanath, K. Health Behavior: Theory, Research, and Practice, 5th ed.; Jossey-Bass: San Francisco, CA, USA, 2015. [Google Scholar]
- van Stralen, M.M.; Yildirim, M.; te Velde, S.J.; Brug, J.; van Mechelen, W.; Chinapaw, M.J.; ENERGY-consortium. What works in school-based energy balance behaviour interventions and what does not? A systematic review of mediating mechanisms. Int. J. Obes. (Lond.) 2011, 35, 1251–1265. [Google Scholar] [CrossRef] [PubMed]
- Cerin, E.; Barnett, A.; Baranowski, T. Testing theories of dietary behavior change in youth using the mediating variable model with intervention programs. J. Nutr. Educ. Behav. 2009, 41, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Cullen, K.W.; Baranowski, T.; Smith, S.P. Using goal setting as a strategy for dietary behavior change. J. Am. Diet. Assoc. 2001, 101, 562–566. [Google Scholar] [CrossRef]
- McClain, A.D.; Chappuis, C.; Nguyen-Rodriguez, S.T.; Yaroch, A.L.; Spruijt-Metz, D. Psychosocial correlates of eating behavior in children and adolescents: A review. Int. J. Behav. Nutr. Phys. Act. 2009, 6, 54. [Google Scholar] [CrossRef] [PubMed]
- Lowe, C.F.; Horne, P.J.; Tapper, K.; Bowdery, M.; Egerton, C. Effects of a peer modelling and rewards-based intervention to increase fruit and vegetable consumption in children. Eur. J. Clin. Nutr. 2004, 58, 510–522. [Google Scholar] [CrossRef]
- Shopping guide from Danish Heart Foundation. Available online: https://shop.hjerteforeningen.dk/Product/Static/52319/indkobsguide-lommekort-ba3 (accessed on 18 September 2019).
- Rollnick, S.; Miller, W.R.; Butler, C.C. Motivational Interviewing in Healthcare: Helping Patients Change Behavior; Guilford Press: New York, NY, USA, 2008. [Google Scholar]
- Guan, V.X.; Probst, Y.C.; Neale, E.P.; Batterham, M.J.; Tapsell, L.C. Identifying usual food choices at meals in overweight and obese study volunteers: Implications for dietary advice. Br. J. Nutr. 2018, 120, 472–480. [Google Scholar] [CrossRef] [PubMed]
- Leech, R.M.; Worsley, A.; Timperio, A.; McNaughton, S.A. Understanding meal patterns: Definitions, methodology and impact on nutrient intake and diet quality. Nutr. Res. Rev. 2015, 28, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, K.M.; Chan, L.; Moores, C.J.; Miller, J.; Thomas, J.; Yaxley, A.; Jackson, K.; Mehta, K.; Matwiejczyk, L.; Wray, A.; et al. Eating occasions and the contribution of foods to sodium and potassium intakes in adults. Public Health Nutr. 2018, 21, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, L.B.; Lassen, A.D.; Hansen, K.; Knuthsen, P.; Saxholt, E.; Fagt, S. Salt content in canteen and fast food meals in Denmark. Food Nutr. Res. 2010, 54. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, J.; Ovesen, L.; Fagt, S.; Pedersen, A.N. Para-aminobenzoic acid used as a marker for completeness of 24 h urine: Assessment of control limits for a specific HPLC method. Eur. J. Clin. Nutr. 1997, 51, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Bingham, S.; Cummings, J.H. The use of 4-aminobenzoic acid as a marker to validate the completeness of 24 h urine collections in man. Clin. Sci. 1983, 64, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Bingham, S.A.; Cassidy, A.; Cole, T.J.; Welch, A.; Runswick, S.A.; Black, A.E.; Thurnham, D.; Bates, C.; Khaw, K.T.; Key, T.J.A.; et al. Validation of weighed records and other methods of dietary assessment using the 24 h urine nitrogen technique and other biological markers. Br. J. Nutr. 1995, 73, 531–550. [Google Scholar] [CrossRef]
- Black, A.E.; Bingham, S.A.; Johansson, G.; Coward, W.A. Validation of dietary intakes of protein and energy against 24 h urinary N and DLWenergy expenditure in middle-aged women, retired men and post-obese subjects: Comparisons with validation against presumed energy requirements. Eur. J. Clin. Nutr. 1997, 51, 405–413. [Google Scholar] [CrossRef]
- He, F.J.; Wu, Y.; Feng, X.X.; Wang, H.J.; Zhang, J.; Lin, C.P.; Yuan, J.H.; Ma, Y.; Yang, Y.D.; Nowson, C.; et al. School based education programme to reduce salt intake in children and their families (School-EduSalt): Cluster randomized controlled trial. BMJ 2015, 350, h770. [Google Scholar] [CrossRef]
- International Standard ISO 3972: Sensory Analysis-Methodology-Method of Investigating Sensitivity of Taste; ISO: Geneva, Switzerland, 2011.
- Biltoft-Jensen, A.; Bysted, A.; Trolle, E.; Christensen, T.; Knuthsen, P.; Damsgaard, C.T.; Andersen, L.F.; Brockhoff, P.; Tetens, I. Evaluation of Web-based Dietary Assessment Software for Children: Comparing reported fruit, juice and vegetable intakes with plasma carotenoid concentration and school lunch observations. Br. J. Nutr. 2013, 110, 186–195. [Google Scholar] [CrossRef]
- Biltoft-Jensen, A.; Trolle, E.; Christensen, T.; Islam, N.; Andersen, L.F.; Egenfeldt-Nielsen, S.; Tetens, I. WebDASC: A web-based dietary assessment software for 8–11- year-old Danish children. J. Hum. Nutr. Diet. 2014, 27, 43–53. [Google Scholar] [CrossRef]
- Biltoft-Jensen, A.; Matthiessen, J.; Rasmussen, L.B.; Fagt, S.; Groth, M.V.; Hels, V. Validation of the Danish 7-day pre-coded food diary among adults: Energy intake v. energy expenditure and recording length. Br. J. Nutr. 2009, 102, 1838–1846. [Google Scholar] [CrossRef] [PubMed]
- The Danish National Health Survey. Available online: http://www.danskernessundhed.dk/ (accessed on 18 September 2019).
- Goldberg, G.; Black, A.; Jebb, S.; Cole, T.; Murgatroyd, P.; Coward, W.; Prentice, A. Critical evaluation of energy intake data using fundamental principles of energy physiology: 1. Derivation of cut-off limits to identify under-recording. Eur. J. Clin. Nutr. 1991, 45, 569–581. [Google Scholar] [PubMed]
- Black, A.E. Critical evaluation of energy intake using the Goldberg cut-off for energy intake: Basal metabolic rate. A practical guide to its calculation, use and limitations. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1119–1130. [Google Scholar] [CrossRef] [PubMed]
| Week from Intervention Start | Bread, Reduced Salt Content | Bread, Standard Salt Content | ||
|---|---|---|---|---|
| Rye Salt (g/100 g) | Wheat Salt (g/100 g) | Rye Salt (g/100 g) | Wheat Salt (g/100 g) | |
| 1 | 1.2 | 1.2 | 1.2 | 1.2 |
| 2 | 1.2 | 1.2 | 1.2 | 1.2 |
| 3 | 1.0 | 1.0 | 1.2 | 1.2 |
| 4 | 0.8 | 0.8 | 1.2 | 1.2 |
| 5 | 0.6 | 0.6 | 1.2 | 1.2 |
| 6 | 0.6 | 0.4 | 1.2 | 1.2 |
| Activity | Social Cognitive Theory | Advice and Skill Development | Tools | Measurements |
|---|---|---|---|---|
| General introduction | Knowledge | Salt and Health | Shopping guide | Salt intake screener |
| How to reduce salt and increase potassium | Keyhole label | |||
| Nutrition facts labels | Workshops | |||
| More taste with less salt | ||||
| Individual advice | Self-efficacy | Rethink food choice alternatives with less salt | Traffic light: foods with lower salt content | Motivation, Barriers, Focus points/goal setting |
| Promote keyhole, nutrition declarations | ||||
| Two follow-up calls | Re-enforcement | Encouragement | Follow-up calls | |
| Guidance | Motivation, Barriers, focus points | |||
| Weekly motivation e-mails | Re-enforcement | Information, recipes |
| Assessment | Outcome | Method |
|---|---|---|
| Demographics | Gender, age, family, marital status, education, work | Questionnaire |
| Lifestyle | Physical activity, smoking, alcohol, salt and bread consumption, food habits | Questionnaire |
| Medical history | Hypertension, heart disease, cancer, diabetes, cholesterol, coeliac disease | Questionnaire, Interview |
| Anthropometry | Height, weight, BMI, arm, hip and waist circumferences, fat percentage | Standardized measurements by nurse, impedance |
| Blood pressure | Pulse, diastolic Blood Pressure (BP) Systolic BP | Three resting BP’s, 5 min rest–average of last 2 BP’s |
| Biochemistry | P-glucose, glycated haemoglobin (HbA1C), Triglyceride, Cholesterol, high-density lipoproteins (HDL), low-density lipoproteins (LDL), | Semi fasting (min 2 h) blood samples after 30 min rest |
| renin, aldosterone, catecholamine metabolites | 24 h urine | |
| U-albumin, U-sodium, U-potassium, U Creatinine | ||
| U-albumin | Spot urine, micral test | |
| Salt sensitivity/preference | Salt taste detection- and recognition thresholds staircase procedure: eight salt concentrations | Taste of salt solutions |
| Preference bread 0.4/0.8/1.2 g salt/ 100 g | Taste of bread | |
| 24 h urine collection | 24 h-sodium, 24 h-potassium, 24 h-Creatinine, para-aminobenzoic acid (PABA) | Adults: 3 days 24 h urine + PABA |
| Children: 1 day 24 h urine | ||
| 7-day dietary assessment | Energy, macro- and micronutrients, foods, meal pattern, eating at home/out | Validated web-based dietary record, estimated portion sizes from photos |
| Bread intervention | Bread collection twice a week registration | Four months of bread intervention: |
| Weekly test of salt content in bread | Intro week 1–2 after completion baseline dietary record and urine collection | |
| Six weeks gradual reduction in salt | ||
| Participants evaluation of bread consumed | End after completion follow-up dietary record and urine collection | |
| Dietary counseling | Salt intake, potassium | Salt intake screener, interview, follow-up telephone interviews |
| Motivation, barriers, focus points | ||
| Actions applied to reduce salt intake | Evaluation questionnaire |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bjoernsbo, K.S.; Riis, N.L.; Andreasen, A.H.; Petersen, J.; Dahl Lassen, A.; Trolle, E.; Frederiksen, A.K.S.; Munk, J.K.; Toft, U. Salt Reduction Intervention in Families Investigating Metabolic, Behavioral and Health Effects of Targeted Intake Reductions: Study Protocol for a Four Months Three-Armed, Randomized, Controlled “Real-Life” Trial. Int. J. Environ. Res. Public Health 2019, 16, 3532. https://doi.org/10.3390/ijerph16193532
Bjoernsbo KS, Riis NL, Andreasen AH, Petersen J, Dahl Lassen A, Trolle E, Frederiksen AKS, Munk JK, Toft U. Salt Reduction Intervention in Families Investigating Metabolic, Behavioral and Health Effects of Targeted Intake Reductions: Study Protocol for a Four Months Three-Armed, Randomized, Controlled “Real-Life” Trial. International Journal of Environmental Research and Public Health. 2019; 16(19):3532. https://doi.org/10.3390/ijerph16193532
Chicago/Turabian StyleBjoernsbo, Kirsten Schroll, Nanna Louise Riis, Anne Helms Andreasen, Janne Petersen, Anne Dahl Lassen, Ellen Trolle, Amalie Kruse Sigersted Frederiksen, Jens Kristian Munk, and Ulla Toft. 2019. "Salt Reduction Intervention in Families Investigating Metabolic, Behavioral and Health Effects of Targeted Intake Reductions: Study Protocol for a Four Months Three-Armed, Randomized, Controlled “Real-Life” Trial" International Journal of Environmental Research and Public Health 16, no. 19: 3532. https://doi.org/10.3390/ijerph16193532
APA StyleBjoernsbo, K. S., Riis, N. L., Andreasen, A. H., Petersen, J., Dahl Lassen, A., Trolle, E., Frederiksen, A. K. S., Munk, J. K., & Toft, U. (2019). Salt Reduction Intervention in Families Investigating Metabolic, Behavioral and Health Effects of Targeted Intake Reductions: Study Protocol for a Four Months Three-Armed, Randomized, Controlled “Real-Life” Trial. International Journal of Environmental Research and Public Health, 16(19), 3532. https://doi.org/10.3390/ijerph16193532





