Automatic Classification of Sarcopenia Level in Older Adults: A Case Study at Tijuana General Hospital
Abstract
:1. Introduction
2. Materials and Methods
2.1. Description of the Database
2.2. Machine Learning Models for Classification of Sarcopenia Level Based on Patient Variables
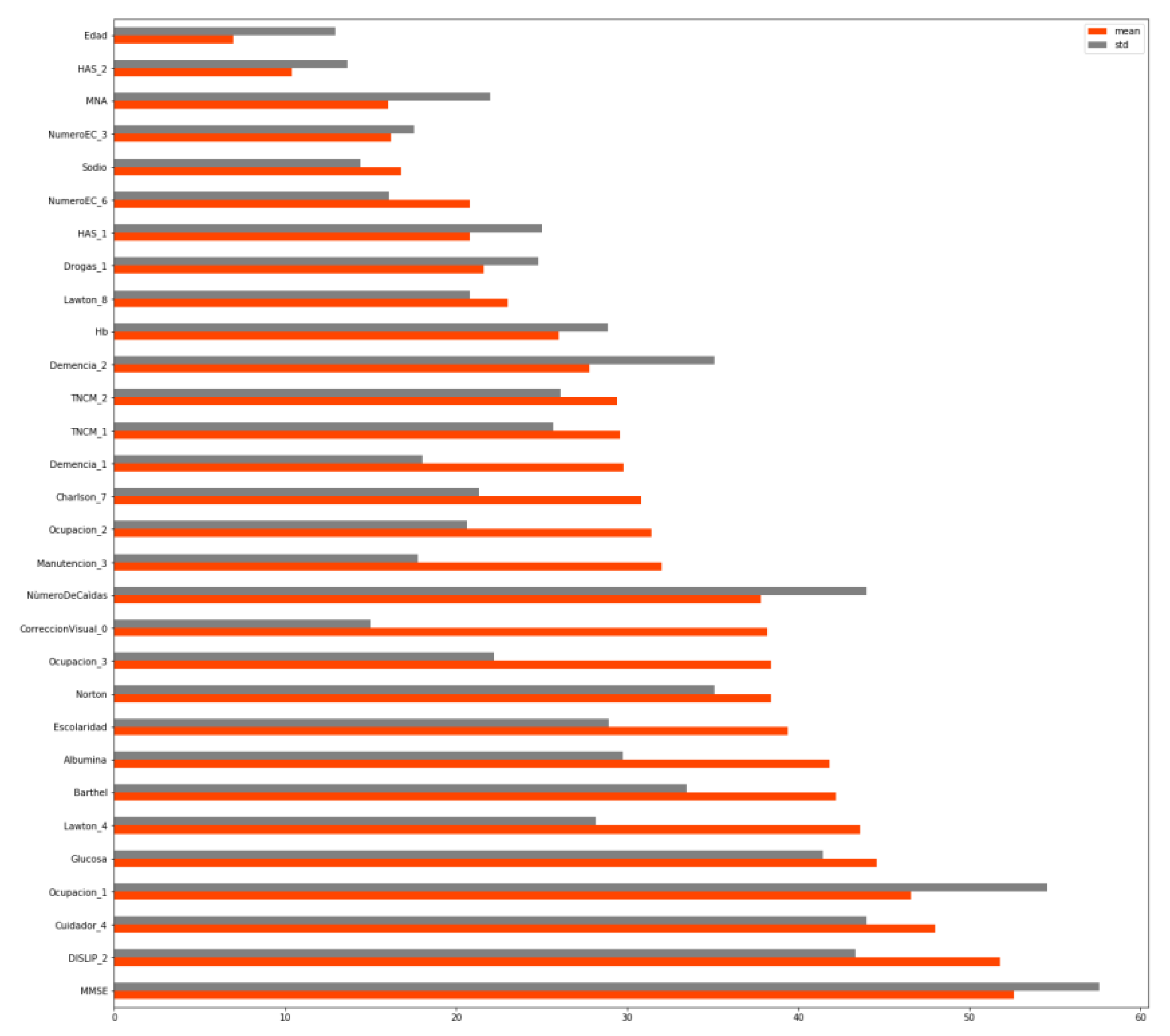
2.2.1. Classification of Variables
2.2.2. Classification of Models
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Goodpaster, B.H.; Park, S.W.; Harris, T.B.; Kritchevsky, S.B.; Nevitt, M.; Schwartz, A.V.; Simonsick, E.M.; Tylavsky, F.A.; Visser, M.; Newman, A.B. The loss of skeletal muscle strength, mass, and quality in older adults: The health, aging and body composition study. J. Gerontol. A Biol. Sci. Med. Sci. 2006, 61, 1059–1064. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.J. What Is Sarcopenia? Gerontol. Ser. A 1995, 50A, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Espinel-Bermudez, M.; Sanchez-Garcia, S.; Garcia-Peña, C.; Trujillo, X.; Huerta-Viera, M.; Granados-Garcia, V.; Hernández-González, S.; Arias-Merino, E.D. Associated factors with sarcopenia among Mexican elderly: 2012 National Health and Nutrition Survey. Rev. Med. Inst. Mex. Seguro Soc. 2018, 56, 46–53. [Google Scholar]
- Deutz, N.E.P.; Ashurst, I.; Ballesteros, M.D.; Bear, D.E.; Cruz-Jentoft, A.J.; Genton, L.; Landi, F.; Laviano, A.; Norman, K.; Prado, C.M. The Underappreciated Role of Low Muscle Mass in the Management of Malnutrition. J. Am. Med. Dir. Assoc. 2019, 1, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Michel, J.P.; Landi, F.; Martin, F.C.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: Consenso europeo sobre su definición y diagnóstico informe del grupo europeo de trabajo sobre la sarcopenia en personas de edad avanzada. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Bai, L. Sarcopenia in the elderly: Basic and clinical issues. Geriatr. Gerontol. Int. 2012, 3, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; Schneider, S.M.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Locquet, M.; Beaudart, C.; Hajaoui, M.; Petermans, J.; Reginster, J.Y.; Bruyère, O. Three-Year Adverse Health Consequences of Sarcopenia in Community-Dwelling Older Adults According to 5 Diagnosis Definitions. J. Am. Med. Dir. Assoc. 2019, 1, 43–46. [Google Scholar] [CrossRef]
- Ruiz, J.R.; Sui, X.; Lobelo, F.; Morrow, J.R.; Jackson, A.W.; Sjöström, M.; Blair, S.N. Association between muscular strength and mortality in men: Prospective cohort study. BMJ 2008, 337, a439. [Google Scholar] [CrossRef]
- Peterson, M.D.; Sen, A.; Gordon, P.M. Influence of resistance exercise on lean body mass in aging adults: A meta-analysis. Med. Sci. Sports Exerc. 2011, 2, 249–258. [Google Scholar] [CrossRef]
- Padilha, C.S.; Marinello, P.C.; Galvao, D.A.; Newton, R.U.; Borges, F.H.; Frajacomo, F.; Deminice, R. Evaluation of resistance training to improve muscular strength and body composition in cancer patients undergoing neoadjuvant and adjuvant therapy: A meta-analysis. J. Cancer Surviv. Res. 2017, 3, 339–349. [Google Scholar] [CrossRef] [PubMed]
- OMS. Informe Mundial Sobre el Envejecimiento y la Salud [Internet]. Available online: https://www.who.int/ageing/publications/world-report-2015/es/ (accessed on 15 May 2019).
- Bruyère, O.; Beaudart, C.; Ethgen, O.; Reginster, J.Y.; Locquet, M. The health economics burden of sarcopenia: A systematic review. Maturitas 2019, 119, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, J.; Rivera-Dommarco, J.; Shamah-Levy, T.; Villalpando, S.; Franco, A.; Cuevas-Nasu, L.; Hernández-Avila, M. Encuesta Nacional de Salud y Nutrición, Resultados Nacionales 2012; Instituto Nacional de Salud Pública: Cuernavaca, Morelos, 2012; Available online: https://ensanut.insp.mx/informes/ENSANUT2012ResultadosNacionales.pdf (accessed on 13 May 2019).
- Canto-Cetina, T. Sarcopenia: La epidemia del siglo XXl? Available online: https://www.sportlife.es/trainhard/ciencia-fuerza/articulo/sarcopenia-la-epidemia-del-siglo-xxi (accessed on 4 September 2019).
- Ferreira-Mesquita, A.; Cruz-da-Silva, E.; Eickemberg, M.; Carneiro-Roriz, A.-K.; Barreto-Medeiros, J.-M.; Barbosa-Ramos, L. Factors associated with sarcopenia in institutionalized elderly. Nutr. Hosp. 2017, 34, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Hamrioui, S.; De la Torre Díez, I.; Garcia-Zapirain, B.; Saleem, K.; Rodrigues, J.J.P.C. A Systematic Review of Security Mechanisms for Big Data in Health and New Alternatives for Hospitals. Wirel. Commun. Mob. Comput. 2017, 2017, 6. [Google Scholar] [CrossRef]
- Mugueta-Aguinaga, I.; Garcia-Zapirain, B. Is Technology Present in Frailty? Technology a Back-up Tool for Dealing with Frailty in the Elderly: A Systematic Review. Aging Dis. 2017, 2, 176–195. [Google Scholar]
- De la Torre, I.; Cosgaya, H.M.; García-Zapirain, B.; López-Coronado, M. Big data in health: A literature review from the year 2005. J. Med. Syst. 2016, 40, 209. [Google Scholar] [CrossRef] [PubMed]
- Coplade, B.C. Tijuana; COPLADE: Tijuana, Mexico, 2017; p. 10. Available online: http://www.copladebc.gob.mx/publicaciones/2017/Mensual/Tijuana%202017.pdf (accessed on 21 May 2019).
- Steffl, M.; Bohannon, R.W.; Sontakova, L.; Tufano, J.J.; Shiells, K.; Holmerova, I. Relationship between sarcopenia and physical activity in older people: A systematic review and meta-analysis. Clin. Interv. Aging 2017, 12, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Hao, Q.; Hai, S.; Wang, H.; Cao, L.; Dong, B. Sarcopenia as a predictor of all-cause mortality among community-dwelling older people: A systematic review and meta-analysis. Maturitas 2017, 103, 16–22. [Google Scholar] [CrossRef]
- Bianchi, L.; Abete, P.; Bellelli, G.; Bo, M.; Cherubini, A.; Corica, F.; Di Bari, M.; Maggio, M.; Manca, G.M.; Rizzo, M.R.; et al. Prevalence and Clinical Correlates of Sarcopenia, Identified According to the EWGSOP Definition and Diagnostic Algorithm, in Hospitalized Older People: The GLISTEN Study. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 11, 1575–1581. [Google Scholar] [CrossRef]
- Polan, D.F.; Brady, S.L.; Kaufman, R.A. Tissue segmentation of computed tomography images using a Random Forest algorithm: A feasibility study. Phys. Med. Biol. 2016, 17, 6553–6569. [Google Scholar] [CrossRef]
- Dreder, A. Machine Learning Based Approaches for Identifying Sarcopenia-Related Genomic Biomarkers in Ageing Males [Internet]; Northumbria University: London, UK, 2017; Available online: http://nrl.northumbria.ac.uk/36184/ (accessed on 21 August 2018).
- Wong, A.; Young, A.T.; Liang, A.S.; Gonzales, R.; Douglas, V.C.; Hadley, D. Development and Validation of an Electronic Health Record–Based Machine Learning Model to Estimate Delirium Risk in Newly Hospitalized Patients Without Known Cognitive Impairment EHR-Based Machine Learning to Estimate Delirium Risk in Newly Hospitalized PatientsEHR-Based Machine Learning to Estimate Delirium Risk in Newly Hospitalized Patients. JAMA Netw. Open 2018, 4, e181018. [Google Scholar]
- Lee, H.; Troschel, F.M.; Tajmir, S.; Fuchs, G.; Mario, J.; Fintelmann, F.J.; Do, S. Pixel-Level Deep Segmentation: Artificial Intelligence Quantifies Muscle on Computed Tomography for Body Morphometric Analysis. J. Digit. Imaging 2017, 4, 487–498. [Google Scholar] [CrossRef] [PubMed]
- Sertel, O.; Dogdas, B.; Chiu, C.S.; Gurcan, M.N. Muscle histology image analysis for sarcopenia: Registration of successive sections with distinct atpase activity. In Proceedings of the 2010 IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Rotterdam, The Netherlands, 14–17 April 2010; pp. 1423–1426. [Google Scholar]
- Lerouge, J.; Herault, R.; Chatelain, C.; Jardin, F.; Modzelewski, R. IODA: An input/output deep architecture for image labeling. Pattern Recognit. 2015, 9, 2847–2858. [Google Scholar] [CrossRef]
- Jauk, S.; Kramer, D.; Leodolter, W. Cleansing and Imputation of Body Mass Index Data and Its Impact on a Machine Learning Based Prediction Model. Stud. Health Technol. Inform. 2018, 248, 116–123. [Google Scholar] [PubMed]
- Mamoshina, P.; Volosnikova, M.; Ozerov, I.V.; Putin, E.; Skibina, E.; Cortese, F.; Zhavoronkov, A. Machine Learning on Human Muscle Transcriptomic Data for Biomarker Discovery and Tissue-Specific Drug Target Identification. Front. Genet. 2018, 9, 242. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.X.; Chong, M.S.; Lim, W.S.; Tay, L.; Yew, S.; Yeo, A.; Tan, C.H. Validity of estimating muscle and fat volume from a single MRI section in older adults with sarcopenia and sarcopenic obesity. Clin. Radiol. 2017, 5, e9–e427. [Google Scholar] [CrossRef]
- Katakis, S.; Barotsis, N.; Kastaniotis, D.; Theoharatos, C.; Tsourounis, D.; Fotopoulos, S.; Panagiotopoulos, E. Muscle Type Classification on Ultrasound Imaging Using Deep Convolutional Neural Networks. In Proceedings of the 2018 IEEE 13th Image, Video, and Multidimensional Signal Processing Workshop (IVMSP), Zagori, Aristi Village, Greece, 10–12 June 2018; pp. 1–5. [Google Scholar]
- Thondoo, M.; Rojas-Rueda, D.; Gupta, J.; de Vries, D.H.; Nieuwenhuijsen, M.J. Systematic Literature Review of Health Impact Assessments in Low and Middle-Income Countries. Int. J. Environ. Res. Public Health 2019, 16, 2018. [Google Scholar] [CrossRef] [PubMed]
- Saenz-de-Urturi, Z.; Garcia-Zapirain Soto, B. Kinect-based virtual game for the elderly that detects incorrect body postures in real time. Sensors 2016, 15, 704. [Google Scholar] [CrossRef]
- Ortiz-Vigon Uriarte Ide, L.; Garcia-Zapirain, B.; Garcia-Chimeno, Y. Game design to measure reflexes and attention based on biofeedback multi-sensor interaction. Sensors 2015, 3, 6520–6548. [Google Scholar] [CrossRef]
- Sáenz-de-Urturi, Z.; Zapirain, B.G.; Zorrilla, A.M. Kinect-based Virtual Game for Motor and Cognitive Rehabilitation: A Pilot Study for Older Adults. In Proceedings of the 8th International Conference on Pervasive Computing Technologies for Healthcare [Internet]. ICST, Brussels, Belgium, 20–23 May 2014; pp. 262–265. Available online: http://dx.doi.org/10.4108/icst.pervasivehealth.2014.255328 (accessed on 21 August 2018).
| Risk Factors | Chronic Diseases |
|---|---|
| Constitutional | Cognitive impairment |
| Female gender | Mood disorders |
| Low weight at birth | Diabetes mellitus |
| Genetic predisposition | Heart failure |
| Lifestyle | Liver failure |
| Malnutrition | Kidney failure |
| Low protein intake | Shortage of breath |
| Smoking habit | Osteoarthritis |
| Physical inactivity | Chronic pain |
| Living conditions | Obesity |
| Inanition | Catabolic effects of drugs |
| Being bedridden | Cancer |
| Weightlessness | Chronic inflammatory diseases |
| Gender | Body Mass Index (BMI) | Grip Strength | Walking Speed | |
|---|---|---|---|---|
| Women | 65% | <6.1 kg/m2 | <20 | <0.8 |
| Men | 35% | <8.5 kg/m2 | <30 | <0.8 |
| Metric | Formula |
|---|---|
| Accuracy | |
| Precision | |
| F1 |
| Classifier | Description | |
|---|---|---|
| 1 | Nearest Neighbors (3) | 3-Nearest Neighbours |
| 2 | Linear SVM (C = 0.025) | Linear Support Vector Machine |
| 3 | RBF SVM (gamma = 2, C = 1) | Radial Basis Support Vector Machine |
| 4 | Gaussian Process (RBF (1.0)) | Gaussian Support Vector Machine |
| 5 | Decision Tree (max_depth = 3) | Decision Tree of Depth 3 |
| 6 | Random Forest (max_depth = 3, n_estimators = 10) | Random Forest of 10 trees and depth 3 |
| 7 | MPL (alpha = 1) | Multi-Layer Perceptron |
| 8 | AdaBoost | AdaBoost classifier |
| 9 | Gaussian Naive Bayes | Naive Bayes classifier |
| 10 | QDA | Quadratic Discriminant classifier |
| Dataset 1 | Classifier | Accuracy | F1 | Precision |
| 1 | Nearest Neighbors (3) | 0.819 | 0.895 | 0.843 |
| 1 | Linear SVM (C = 0.025) | 0.813 | 0.897 | 0.813 |
| 1 | RBF SVM (gamma = 2, C = 1) | 0.825 | 0.902 | 0.828 |
| 1 | Gaussian Process (RBF (1.0)) | 0.813 | 0.897 | 0.813 |
| 1 | Decision Tree (max_depth = 3) | 0.831 | 0.900 | 0.864 |
| 1 | Random Forest (max_depth = 3, n_estimators = 10) | 0.825 | 0.901 | 0.836 |
| 1 | MPL (alpha = 1) | 0.807 | 0.888 | 0.836 |
| 1 | AdaBoost | 0.783 | 0.871 | 0.841 |
| 1 | Gaussian Naive Bayes | 0.801 | 0.883 | 0.844 |
| 1 | QDA | 0.789 | 0.876 | 0.833 |
| dataSET 2 | ||||
| 2 | Nearest Neighbors (3) | 0.795 | 0.879 | 0.840 |
| 2 | Linear SVM (C = 0.025) | 0.813 | 0.897 | 0.813 |
| 2 | RBF SVM (gamma = 2, C = 1) | 0.813 | 0.897 | 0.813 |
| 2 | Gaussian Process (RBF (1.0)) | 0.813 | 0.897 | 0.813 |
| 2 | Decision Tree (max_depth = 3) | 0.795 | 0.879 | 0.844 |
| 2 | Random Forest (max_depth = 3, n_estimators = 10) | 0.825 | 0.902 | 0.827 |
| 2 | MPL (alpha = 1) | 0.819 | 0.892 | 0.864 |
| 2 | AdaBoost | 0.789 | 0.874 | 0.847 |
| 2 | Gaussian Naive Bayes | 0.814 | 0.886 | 0.867 |
| 2 | QDA | 0.826 | 0.894 | 0.875 |
| dataSET 3 | ||||
| 3 | Nearest Neighbors (3) | 0.783 | 0.874 | 0.824 |
| 3 | Linear SVM (C = 0.025) | 0.813 | 0.897 | 0.813 |
| 3 | RBF SVM (gamma = 2, C = 1) | 0.813 | 0.897 | 0.813 |
| 3 | Gaussian Process (RBF (1.0)) | 0.813 | 0.897 | 0.813 |
| 3 | Decision Tree (max_depth = 3) | 0.819 | 0.897 | 0.840 |
| 3 | Random Forest (max_depth = 3, n_estimators = 10) | 0.795 | 0.886 | 0.810 |
| 3 | MPL (alpha = 1) | 0.814 | 0.890 | 0.852 |
| 3 | AdaBoost | 0.777 | 0.868 | 0.837 |
| 3 | Gaussian Naive Bayes | 0.765 | 0.855 | 0.863 |
| 3 | QDA | 0.635 | 0.708 | 0.791 |
| dataSET 4 | ||||
| 4 | Nearest Neighbors (3) | 0.783 | 0.878 | 0.807 |
| 4 | Linear SVM (C = 0.025) | 0.777 | 0.873 | 0.810 |
| 4 | RBF SVM (gamma = 2 C = 1) | 0.813 | 0.897 | 0.813 |
| 4 | Gaussian Process (RBF (1.0)) | 0.789 | 0.881 | 0.813 |
| 4 | Decision Tree (max_depth = 3) | 0.765 | 0.842 | 0.866 |
| 4 | Random Forest (max_depth = 3, n_estimators = 10) | 0.801 | 0.890 | 0.811 |
| 4 | MPL (alpha = 1) | 0.753 | 0.854 | 0.818 |
| 4 | AdaBoost | 0.729 | 0.831 | 0.831 |
| 4 | Gaussian Naive Bayes | 0.234 | 0.178 | 0.412 |
| 4 | QDA | 0.784 | 0.878 | 0.807 |
| Dataset | Variables |
|---|---|
| 1 | ‘Age’, ‘HAS’, ‘MNA’, ‘ECNumber’, ‘Sodium’ |
| 2 | ‘Age’, ‘HAS’, ‘MNA’, ‘ECNumber’, ‘Sodium’, ‘Drugs’, ‘Lawton’ |
| 3 | ‘Age’, ‘HAS’, ‘MNA’, ‘ECNumber’, ‘Sodium’, ‘Drugs’, ‘Lawton’, ‘Hb’, ‘Dementia’, ‘TNCM’, ‘Charlson’, ‘Profession’, ‘FinSupport’ |
| 4 | ‘Status’, ‘Gender’, ‘Age’, ‘Schooling’, ‘LevelofStudies’, ‘MaritalStatus’, ‘Carer’, ‘Religion’, ‘Residence’, ‘Profession’, ‘Income’, ‘FinSupport’, ‘Sight’, ‘VisualCorrection’, ‘Hearing’, ‘HearingCorrection’, ´ECNumber’, ‘HAS’, ‘DMII’, ‘OA’, ‘OSTEOP’, ‘GASTRITIS’, ‘DEPRE’, ‘CARDIO’, ‘TNCM’, ‘PARKIN’, ‘HIPOT’, ‘HIPERT’, ‘CANCER’, ‘EPOC’, ‘DISLIP’, ‘IRC’, ‘OTHERS’, ‘LiverFailure’, ‘SmokingHabit’, ‘Alcoholism’, ‘Drugs’, ‘ExpBiomass’, ‘MMSE’, ‘GDS’, ‘Depression’, ‘Barthel’, ‘Falls’, ‘NumberofFalls’, ‘Ulcers’, ‘Norton’, ‘Lawton’, ‘MNA’, ‘Charlson’, ‘TallaMts’, ‘Dementia’, ‘Cognition’, ‘EVC’, ‘Infection’, ‘Pain’, ‘Cancer’, ‘Hb’, ‘Urea’, ‘Creatinine’, ‘Albumin’, ‘Glucose’, ‘Sodium’ |
| Classifiers | DataSET 1 | DataSET 2 | DataSET 3 | DataSET 4 | DataSET | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ACC | F1 | P | ACC | F1 | P | ACC | F1 | P | ACC | F1 | P | Final | |
| RBF SVM (gamma = 2, C = 1) | 0.825 | 0.902 | 0.828 | 0.813 | 0.897 | 0.813 | 0.813 | 0.897 | 0.813 | 0.813 | 0.897 | 0.813 | 1, 2, 3, 4 |
| Decision Tree (max_depth = 3) | 0.831 | 0.9 | 0.864 | 0.795 | 0.879 | 0.844 | 0.819 | 0.897 | 0.84 | 0.765 | 0.842 | 0.866 | 1, 3 |
| Random Forest (max_depth = 3, n_estimators = 10) | 0.825 | 0.901 | 0.836 | 0.825 | 0.902 | 0.827 | 0.795 | 0.886 | 0.810 | 0.801 | 0.89 | 0.811 | 1, 2, 4 |
| Linear SVM (C = 0.025) | 0.813 | 0.897 | 0.813 | 0.813 | 0.897 | 0.813 | 0.813 | 0.897 | 0.813 | 0.765 | 0.842 | 0.866 | 2, 3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castillo-Olea, C.; García-Zapirain Soto, B.; Carballo Lozano, C.; Zuñiga, C. Automatic Classification of Sarcopenia Level in Older Adults: A Case Study at Tijuana General Hospital. Int. J. Environ. Res. Public Health 2019, 16, 3275. https://doi.org/10.3390/ijerph16183275
Castillo-Olea C, García-Zapirain Soto B, Carballo Lozano C, Zuñiga C. Automatic Classification of Sarcopenia Level in Older Adults: A Case Study at Tijuana General Hospital. International Journal of Environmental Research and Public Health. 2019; 16(18):3275. https://doi.org/10.3390/ijerph16183275
Chicago/Turabian StyleCastillo-Olea, Cristián, Begonya García-Zapirain Soto, Christian Carballo Lozano, and Clemente Zuñiga. 2019. "Automatic Classification of Sarcopenia Level in Older Adults: A Case Study at Tijuana General Hospital" International Journal of Environmental Research and Public Health 16, no. 18: 3275. https://doi.org/10.3390/ijerph16183275
APA StyleCastillo-Olea, C., García-Zapirain Soto, B., Carballo Lozano, C., & Zuñiga, C. (2019). Automatic Classification of Sarcopenia Level in Older Adults: A Case Study at Tijuana General Hospital. International Journal of Environmental Research and Public Health, 16(18), 3275. https://doi.org/10.3390/ijerph16183275






