Effectiveness of Screening and Treatment Approaches for Schistosomiasis and Strongyloidiasis in Newly-Arrived Migrants from Endemic Countries in the EU/EEA: A Systematic Review
Abstract
1. Introduction
2. Methods
- (i)
- What are the best diagnostic tests to detect these infections non-endemic settings?
- (ii)
- How effective are the drugs to treat them and what are the associated adverse events?
- (iii)
- What are the most cost-effective screening and treatment options for schistosomiasis and strongyloidiasis in migrant populations from endemic regions in the EU/EEA?
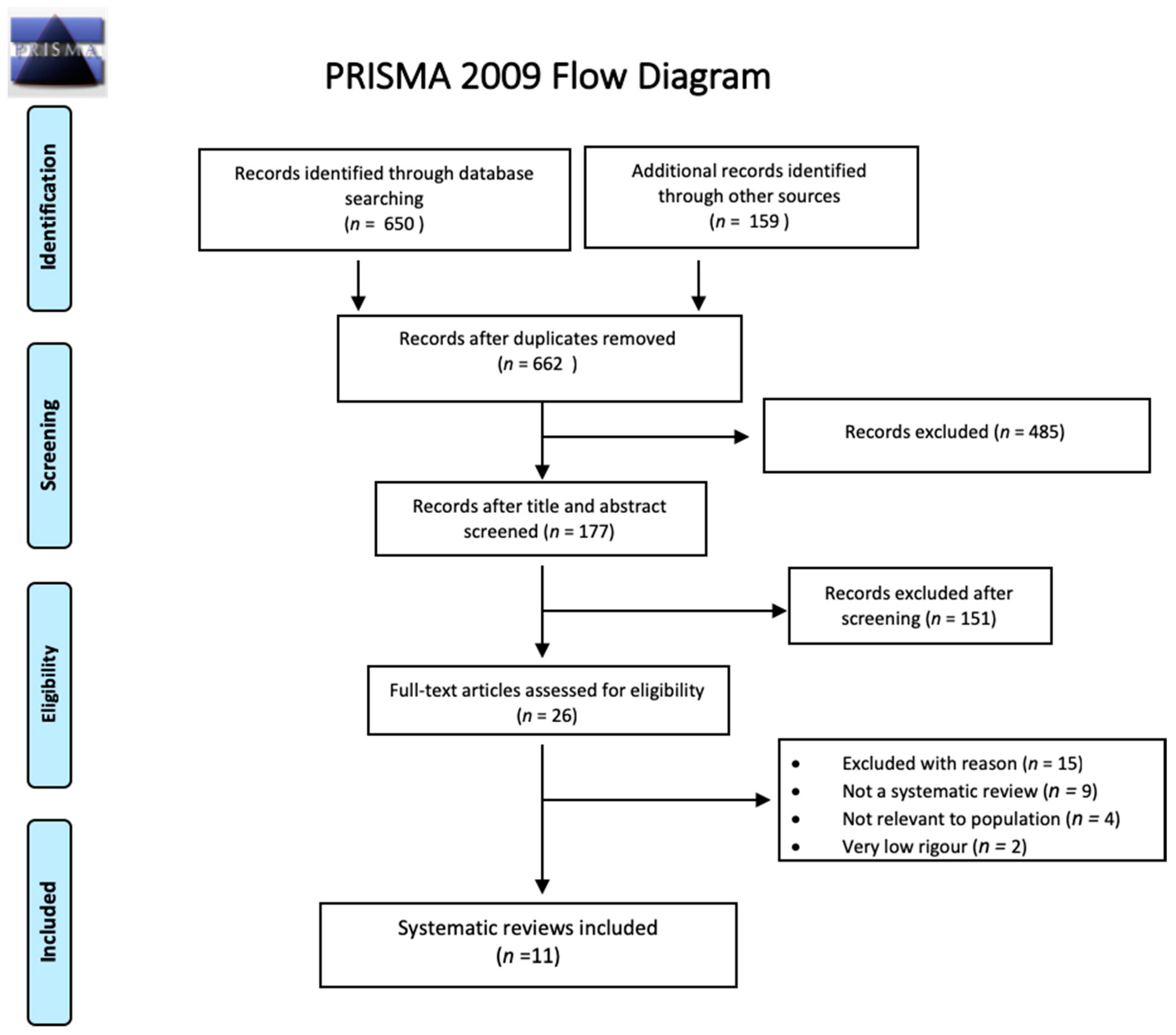
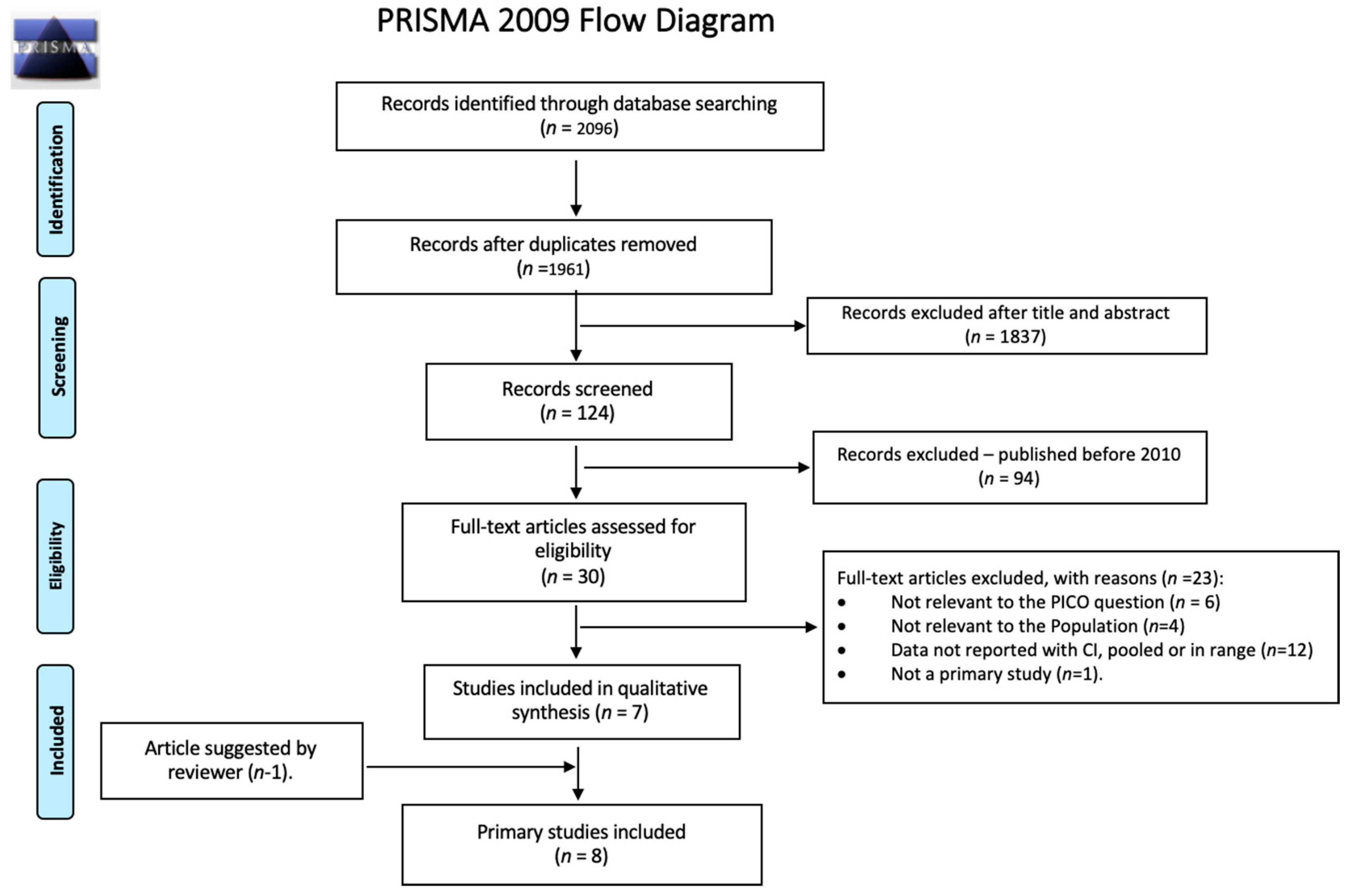
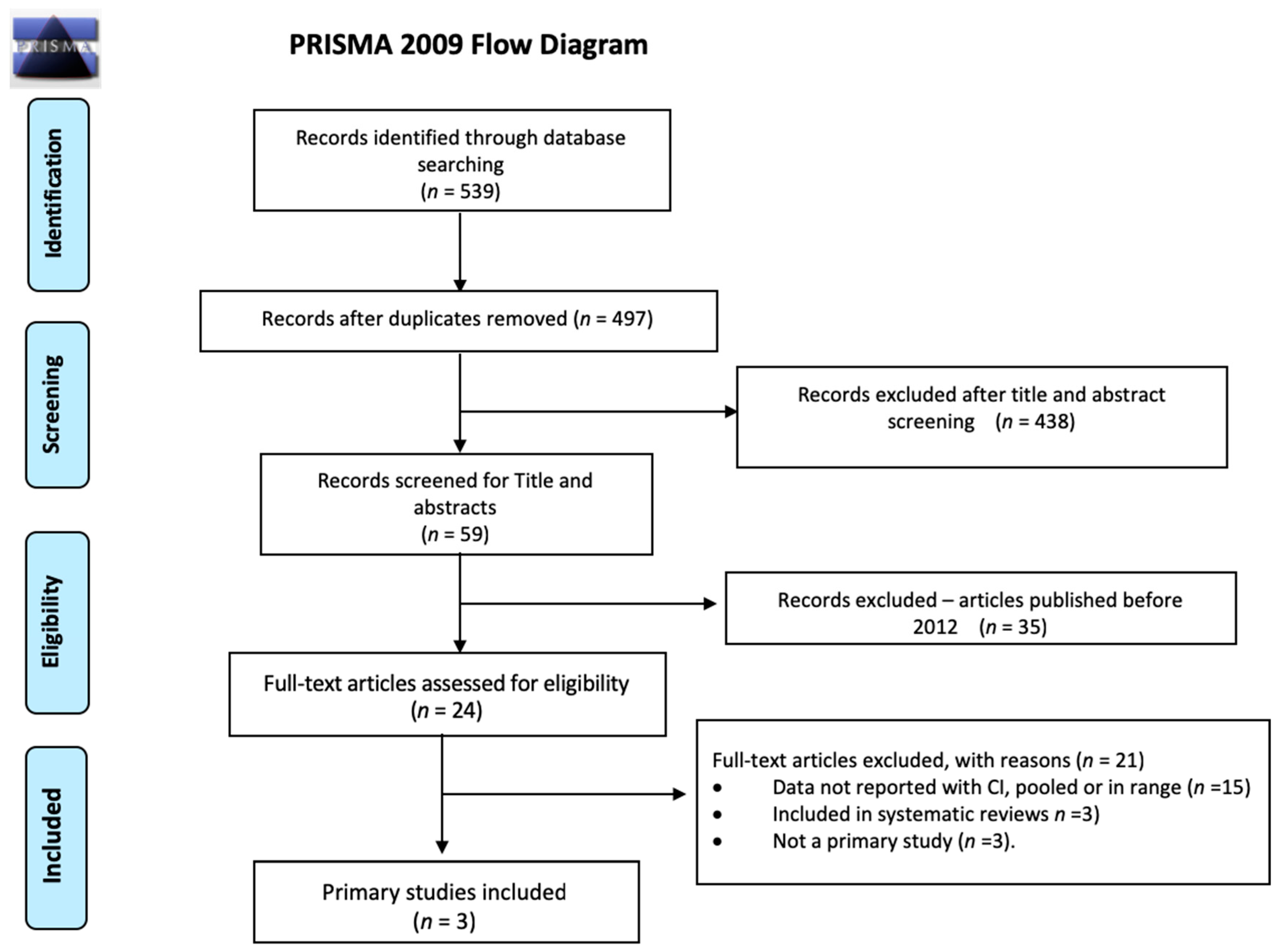
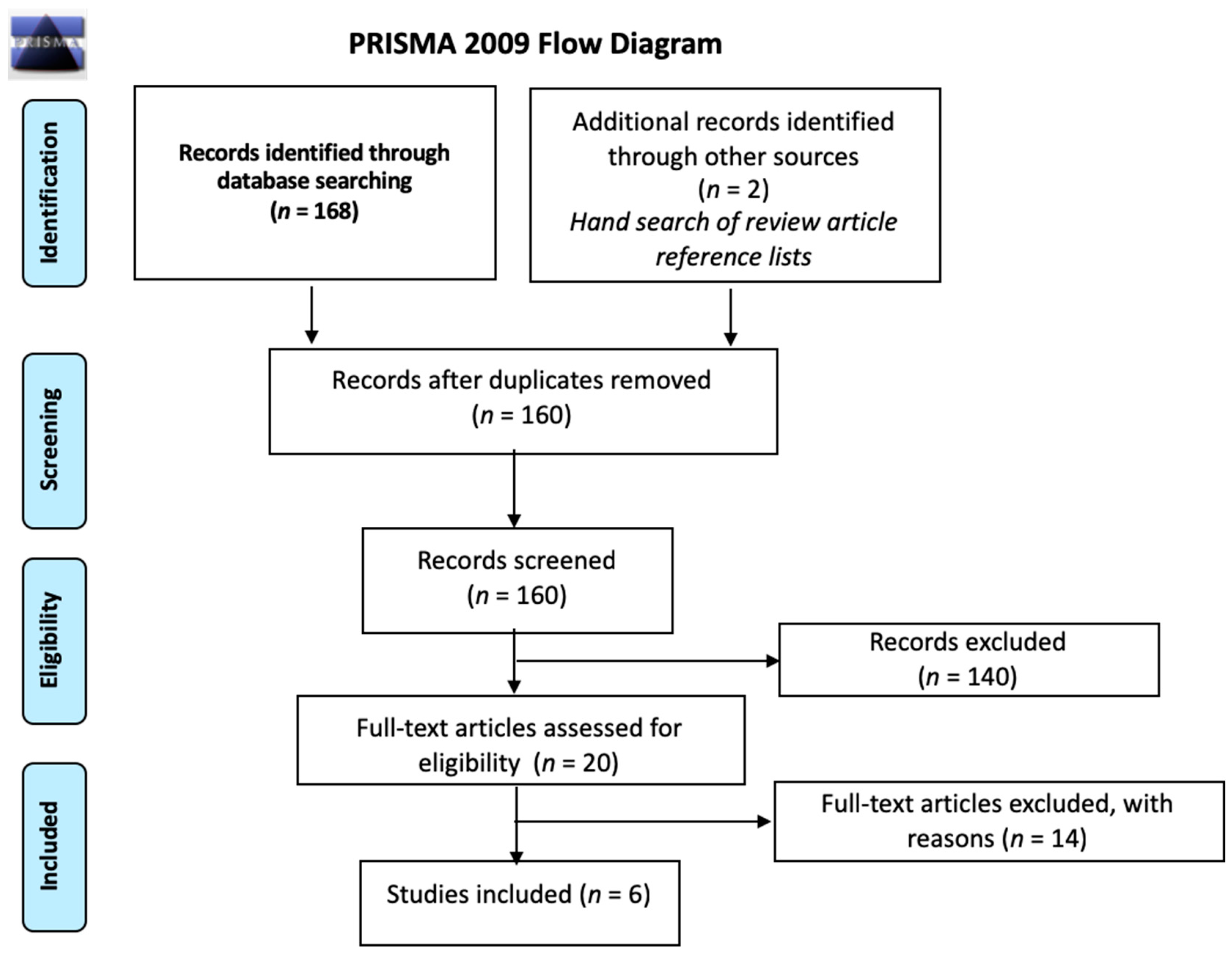
2.1. Search Strategy and Selection Criteria
2.2. Additional Included Studies
2.3. Study Selection, Quality Assessment, and Synthesis
3. Results
3.1. Screening: Diagnostic Test Accuracy for Schistosomiasis
3.1.1. Schistosoma Mansoni
3.1.2. Schistosoma Haematobium
3.1.3. Schistosoma Japonicum
3.2. Screening: Diagnostic Test Accuracy for Strongyloidiasis
3.3. Treatment Efficacy: Schistosomiasis and Strongyloidiasis
3.4. Resource use, Costs and Cost-Effectiveness
3.4.1. Strongyloidiasis
3.4.2. Schistosomiasis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
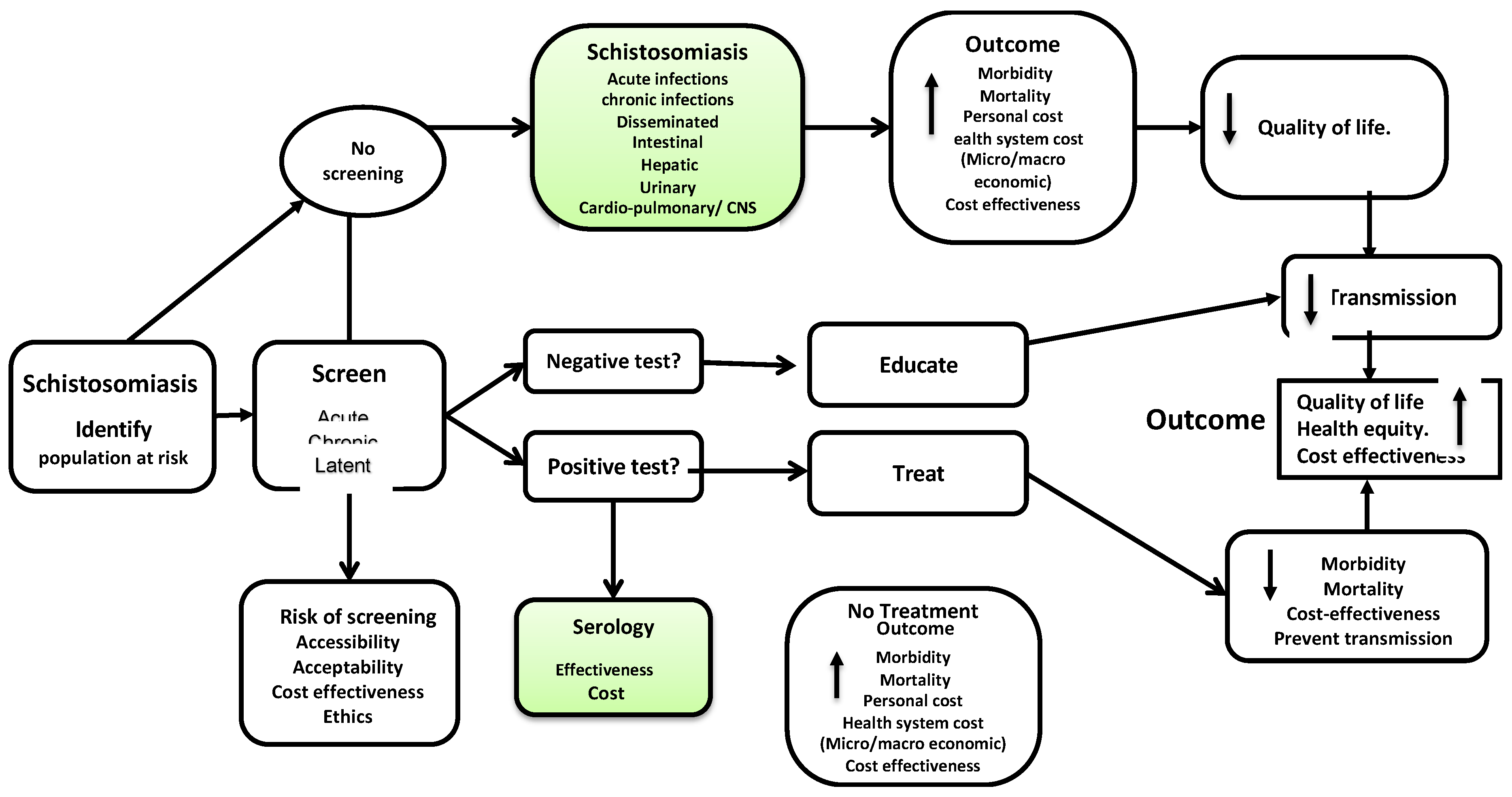
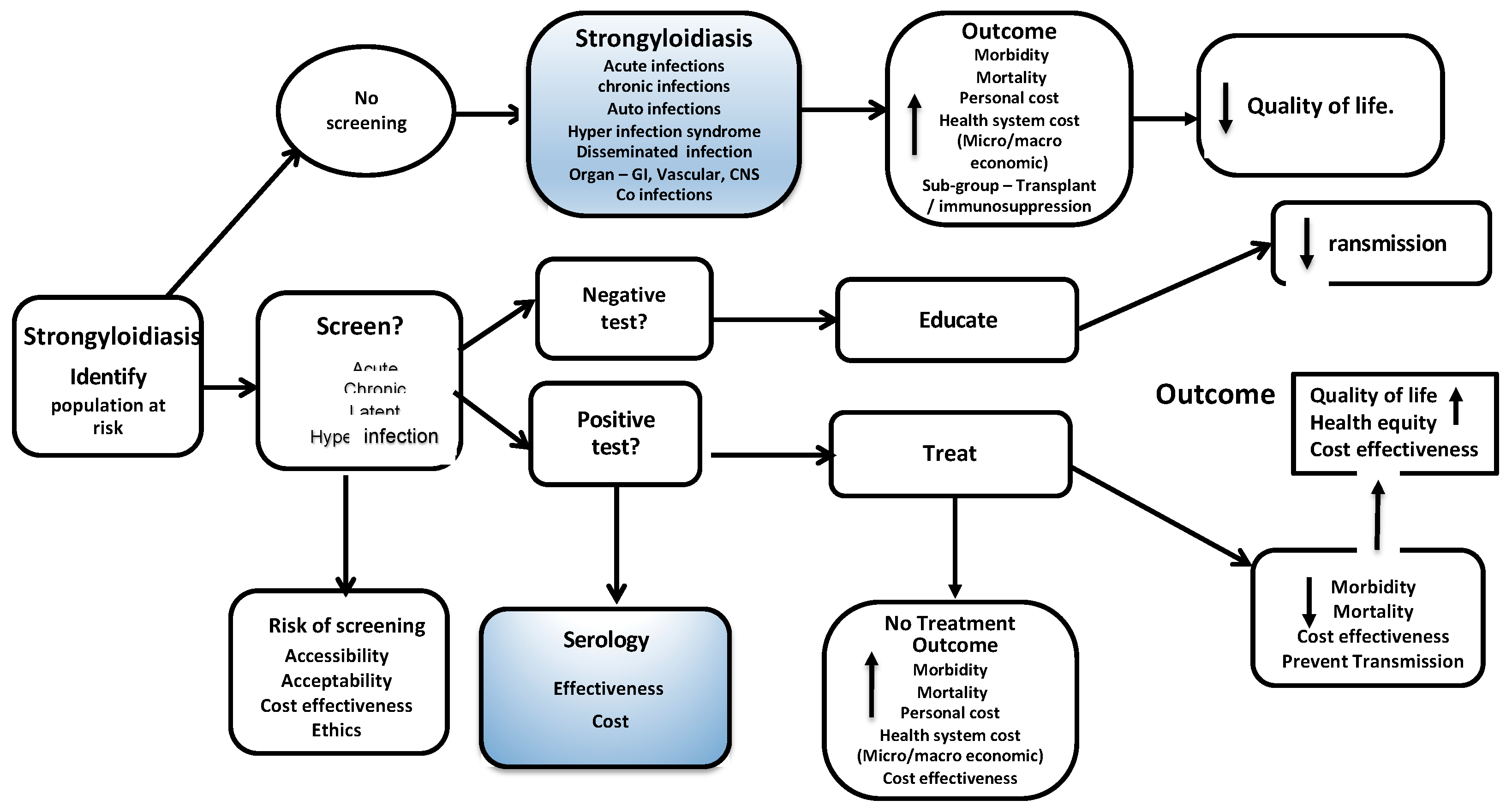
Appendix A. Logic Model—Analytic Framework for Screening and Treatment for Schistosomiasis and Strongyloidiasis in Migrants
Appendix B. List of Sites and Literature Search Strategy
- exp Schistosoma/ (15595)
- bilharzia$.tw. (2431)
- exp Schistosomiasis/ (21432)
- schistosom$.tw. (25367)
- katayama fever$.tw. (30)
- or/1–5 (30014)
- Strongyloides/ (985)
- Strongyloides stercoralis/ (1044)
- Strongyloidiasis/ (3301)
- strongyloid$.tw. (3988)
- or/7–10 (4959)
- 6 or 11 (34621)
- exp Mass Screening/ (107821)
- (screened or screening?) tw. (417896)
- Early Diagnosis/ (19041)
- (detected or detection? or diagnos$ or discover$ or indentif$) tw. (2972048)
- exp Population Surveillance/ (56090)
- (disease? adj2 surveillance) tw. (4053)
- Contact Tracing/ (3521)
- contact tracing tw. (1152)
- or/13–20 (3301561)
- meta analysis mp, pt. (91365)
- review pt. (2035657)
- search$ tw. (253765)
- or/22–24 (2222329)
- animals/ not (humans/ and animals/) (4194238)
- 25 not 26 (2065589)
- 12 and 21 and 27 (711)
- 28 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$) ed. (222)
- remove duplicates from 29 (218)
- exp Schistosoma/ (19846)
- bilharzia$.tw. (2115)
- exp schistosomiasis/ (20241)
- schistosom$.tw. (26744)
- katayama fever$.tw. (40)
- or/1–5 (33204)
- Strongyloides/ (1220)
- Strongyloides stercoralis/ (2315)
- strongyloidiasis/ (3835)
- strongyloid$.tw. (4704)
- or/7–10 (6600)
- 6 or 11 (39071)
- exp mass screening/ (178654)
- (screened or screening?).tw. (614882)
- early diagnosis/ (82347)
- parasite identification/ (13161)
- ((case? or early or parasit$) adj5 (detected or detection? or diagnos$ or discover$ or egg or indentif$)).tw. (385884)
- exp health survey/ (182738)
- (disease? adj2 surveillance).tw. (5156)
- contact examination/ (2830)
- contact tracing.tw. (1448)
- or/13–21 (1237076)
- meta analys$.mp. (167508)
- search$.tw. (362044)
- review.pt. (2131214)
- or/23–25 (2472677)
- (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans) ti.) (5499319)
- 26 not 27 (2251777)
- 12 and 22 and 28 (455)
- 29 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$) dd. (195)
- remove duplicates from 30 (190)
- exp Schistosoma/ (15714)
- bilharzia$.tw. (2438)
- exp Schistosomiasis/ (21583)
- schistosom$.tw. (25722)
- katayama fever$.tw. (30)
- or/1–5 (30381)
- Strongyloides/ (990)
- Strongyloides stercoralis/ (1056)
- Strongyloidiasis/ (3319)
- strongyloid$.tw. (4079)
- or/7–10 (5051)
- 6 or 11 (35067)
- exp Mass Screening/ (108535)
- (screened or screening? or tested or testing or tests).tw. (1734474)
- Early Diagnosis/ (19350)
- (detected or detection? or diagnos$ or discover$ or indentif$).tw. (3053822)
- exp Population Surveillance/ (56687)
- (disease? adj2 surveillance).tw. (4195)
- Contact Tracing/ (3563)
- contact tracing.tw. (1176)
- or/13–20 (4387118)
- meta analysis.mp,pt. (96759)
- review.pt. (2060867)
- search$.tw. (266775)
- guideline.pt. (15780)
- guideline/ (15780)
- guidelines as topic/ (34071)
- practice guideline.pt. (21216)
- practice guideline/ (21216)
- practice guidelines as topic/ (91792)
- (CPG or CPGs or guidance or guideline? or recommend$ or standard?).ti. (147179)
- exp clinical pathway/ (5273)
- exp clinical protocol/ (139345)
- ((care or clinical) adj2 pathway?).tw. (5129)
- or/22–34 (2572065)
- 12 and 21 and 35 (960)
- animals/ not (humans/ and animals/) (4215704)
- 36 not 37 (838)
- 38 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).ed. (271)
- remove duplicates from 39 [reviews and guidelines] (261)
- exp "costs and cost analysis"/ (197942)
- cost$.mp. (467877)
- cost effective$.tw. (83090)
- cost benefit analys$.mp. (67319)
- health care costs.mp. (37157)
- or/41–45 (477217)
- 12 and 21 and 46 (260)
- animals/ not (humans/ and animals/) (4215704)
- 47 not 48 (222)
- 49 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).ed. (82)
- remove duplicates from 50 (78)
- exp Schistosoma/ (21727)
- bilharzia$.tw. (2492)
- exp schistosomiasis/ (21930)
- schistosom$.tw. (29047)
- katayama fever$.tw. (42)
- or/1–5 (36157)
- Strongyloides/ (1229)
- Strongyloides stercoralis/ (2447)
- strongyloidiasis/ (3986)
- strongyloid$.tw. (4977)
- or/7–10 (6962)
- 6 or 11 (42352)
- exp mass screening/ (182895)
- (screened or screening? or tested or testing or tests).tw. (2429856)
- early diagnosis/ (83110)
- parasite identification/ (13222)
- ((case? or early or parasit$) adj5 (detected or detection? or diagnos$ or discover$ or egg or indentif$)).tw. (405389)
- exp health survey/ (184236)
- (disease? adj2 surveillance).tw. (5253)
- contact examination/ (2867)
- contact tracing.tw. (1512)
- or/13-21 (2999272)
- meta analys$.mp. (170914)
- search$.tw. (371898)
- review.pt. (2163187)
- guideline.pt. (0)
- guideline/ (144)
- guidelines as topic/ (229895)
- practice guideline.pt. (0)
- practice guideline/ (275502)
- practice guidelines as topic/ (171091)
- (CPG or CPGs or guidance or guideline? or recommend$ or standard?).ti. (203285)
- exp clinical pathway/ (6983)
- exp clinical protocol/ (75932)
- ((care or clinical) adj2 pathway?).tw. (9455)
- or/23–35 (2900847)
- 12 and 22 and 36 (824)
- (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans).ti.) (5865460)
- 37 not 38 (678)
- 39 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).dd. (304)
- remove duplicates from 40 [reviews and guidelines] (295)
- cost effectiveness analysis/ (114264)
- cost.tw. (387431)
- costs.tw. (208732)
- or/42–44 (544771)
- 12 and 22 and 45 (274)
- (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans).ti.) (5865460)
- 46 not 47 (223)
- 48 and (2010$ or 2011$ or 2012$ or 2013$ or 2014$ or 2015$ or 2016$).dd. (115)
- remove duplicates from 49 [costing] (111)
- Schistosomiasis/ (13485)
- Schistosomiasis.mp. (24533)
- snail fever.mp. (10)
- schistosome *.mp. (5528)
- exp "Sensitivity and Specificity"/ (495027)
- sensitivity.tw. (638974)
- specificity.tw. (379605)
- ((pre-test or pretest) adj probability).tw. (1695)
- post-test probability.tw. (441)
- predictive value$.tw. (85102)
- likelihood ratio$.tw. (11639)
- or/5–11 (1217873)
- or/1–4 (26340)
- 12 and 13 (1493)
- limit 14 to humans (1112)
- from 15 keep 1001–1112 (112)
- Strongyloidiasis/ (3403)
- Strongyloidiasis.mp. (3747)
- Strongyloides stercoralis/ (1098)
- Strongyloides stercoralis.mp. (2142)
- or/1–4 (4376)
- exp “Sensitivity and Specificity”/ (494358)
- sensitivity.tw. (637846)
- specificity.tw. (379066)
- ((pre-test or pretest) adj probability).tw. (1689)
- post-test probability.tw. (438)
- predictive value$.tw. (84929)
- likelihood ratio$.tw. (11613)
- or/6–12 (1216076)
- 5 and 13 (247)
- limit 14 to humans (207)
References
- Puthiyakunnon, S.; Boddu, S.; Li, Y.; Zhou, X.; Wang, C.; Li, J.; Chen, X. Strongyloidiasis—An insight into its global prevalence and management. PLoS Negl. Trop. Dis. 2014, 8, e3018. [Google Scholar] [CrossRef] [PubMed]
- Riccardi, N.; Nosenzo, F.; Peraldo, F.; Sarocchi, F.; Taramasso, L.; Traverso, P.; Viscoli, C.; Di Biagio, A.; Derchi, L.E.; De Maria, A. Increasing prevalence of genitourinary schistosomiasis in Europe in the Migrant Era: Neglected no more? PLoS Negl. Trop. Dis. 2017, 11, e0005237. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, M.; Shibuya, K.; Salomon, J.A.; Abdalla, S.; et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef]
- King, C.H. Parasites and poverty: The case of schistosomiasis. Acta Trop. 2010, 113, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Zoni, A.C.; Catalá, L.; Ault, S.K. Schistosomiasis Prevalence and Intensity of Infection in Latin America and the Caribbean Countries, 1942–2014: A Systematic Review in the Context of a Regional Elimination Goal. PLoS Negl. Trop. Dis. 2016, 10, e0004493. [Google Scholar] [CrossRef] [PubMed]
- Schar, F.; Trostdorf, U.; Giardina, F.; Khieu, V.; Muth, S.; Marti, H.; Vounatsou, P.; Odermatt, P. Strongyloides stercoralis: Global Distribution and Risk Factors. PLoS Negl. Trop. Dis. 2013, 7, e2288. [Google Scholar] [CrossRef] [PubMed]
- Bisoffi, Z.; Buonfrate, D.; Montresor, A.; Requena-Mendez, A.; Munoz, J.; Krolewiecki, A.J.; Gotuzzo, E.; Mena, M.A.; Chiodini, P.L.; Anselmi, M.; et al. Strongyloides stercoralis: A plea for action. PLoS Negl. Trop. Dis. 2013, 7, e2214. [Google Scholar] [CrossRef]
- Adenowo, A.F.; Oyinloye, B.E.; Ogunyinka, B.I.; Kappo, A.P. Impact of human schistosomiasis in sub-Saharan Africa. Braz. J. Infect. Dis. 2015, 19, 196–205. [Google Scholar] [CrossRef]
- Hotez, P.J.; Alvarado, M.; Basanez, M.G.; Bolliger, I.; Bourne, R.; Boussinesq, M.; Brooker, S.J.; Brown, A.S.; Buckle, G.; Budke, C.M.; et al. The global burden of disease study 2010: Interpretation and implications for the neglected tropical diseases. PLoS Negl. Trop. Dis. 2014, 8, e2865. [Google Scholar] [CrossRef] [PubMed]
- Beltrame, A.; Buonfrate, D.; Gobbi, F.; Angheben, A.; Marchese, V.; Monteiro, G.B.; Bisoffi, Z. The hidden epidemic of schistosomiasis in recent African immigrants and asylum seekers to Italy. Eur. J. Epidemiol. 2017. [Google Scholar] [CrossRef]
- Khan, K.; Sears, J.; Chan, A.; Rashid, M.; Greenaway, C.; Stauffer, W.; Narasiah, L.; Pottie, K. Canadian Collaboration for Immigrant and Refugee Health (CCIRH). Strongyloides and Schistosoma: Evidence review for newly arriving immigrants and refugee. In The Canadian Collaboration for Immigrant and Refugee Health. Appendix 8: Intestinal Parasites; Canadian Medical Association Journal: Ottawa, ON, Canada, 2011. [Google Scholar]
- Wilson, J.M.G.; Jungner, G.; Organization, W.H. Principles and Practice of Screening for Disease; World Health Organization: Geneva, Switzerland, 1968. [Google Scholar]
- Colley, D.G.; Bustinduy, A.L.; Secor, W.E.; King, C.H. Human schistosomiasis. Lancet 1969, 383, 2253–2264. [Google Scholar] [CrossRef]
- Deniaud, F.; Rouesse, C.; Collignon, A.; Domingo, A.; Rigal, L. Failure to offer parasitology screening to vulnerable migrants in France: Epidemiology and consequences. Sante (Montrouge, France) 2010, 20, 201–208. (In French) [Google Scholar]
- Ross, A.G.; McManus, D.P.; Farrar, J.; Hunstman, R.J.; Gray, D.J.; Li, Y.S. Neuroschistosomiasis. J. Neurol. 2012, 259, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Buonfrate, D.; Requena-Mendez, A.; Angheben, A.; Munoz, J.; Gobbi, F.; Van Den Ende, J.; Bisoffi, Z. Severe strongyloidiasis: A systematic review of case reports. BMC Infect. Dis. 2013, 13, 78. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Kim, D.S.; Yoon, Y.K.; Sohn, J.W.; Kim, M.J. Donor-Derived Strongyloidiasis Infection in Solid Organ Transplant Recipients: A Review and Pooled Analysis. Transp. Proc. 2016, 48, 2442–2449. [Google Scholar] [CrossRef] [PubMed]
- Berry, A.; Paris, L.; Boissier, J.; Caumes, E. Schistosomiasis Screening of Travelers to Corsica, France. Emerg. Infect. Dis. 2016, 22, 159. [Google Scholar] [CrossRef] [PubMed]
- Requena-Mendez, A.; Chiodini, P.; Bisoffi, Z.; Buonfrate, D.; Gotuzzo, E.; Munoz, J. The laboratory diagnosis and follow up of strongyloidiasis: A systematic review. PLoS Negl. Trop. Dis. 2013, 7, e2002. [Google Scholar] [CrossRef]
- Greaves, D.; Coggle, S.; Pollard, C.; Aliyu, S.H.; Moore, E.M. Strongyloides stercoralis infection. BMJ 2013, 347, f4610. [Google Scholar] [CrossRef]
- Deniaud, F.; Legros, P.; Collignon, A.; Prevot, M.; Domingo, A.; Ayache, B. Targeted screening proposed in 6 migrant worker housing units in Paris in 2005: Feasibility and impact study. Sante Publique 2008, 20, 547–559. (In French) [Google Scholar] [CrossRef]
- Chernet, A.; Kling, K.; Sydow, V.; Kuenzli, E.; Hatz, C.; Utzinger, J.; van Lieshout, L.; Marti, H.; Labhardt, N.D.; Neumayr, A. Accuracy of diagnostic tests for Schistosoma mansoni infection in asymptomatic Eritrean refugees: Serology and POC-CCA against stool microscopy. Clin. Infect. Dis. 2017. [Google Scholar] [CrossRef]
- Weerakoon, K.G.; Gobert, G.N.; Cai, P.; McManus, D.P. Advances in the Diagnosis of Human Schistosomiasis. Clin. Microbiol. Rev. 2015, 28, 939–967. [Google Scholar] [CrossRef] [PubMed]
- Agbata, E.N.; Padilla, P.F.; Agbata, I.N.; Armas, L.H.; Sola, I.; Pottie, K.; Alonso-Coello, P. Migrant Healthcare Guidelines: A Systematic Quality Assessment. J. Immigr. Minor. Health 2018. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Eurostat migr_resfirst, m.r. Residence permits statistics. Available online: https://ec.europa.eu/eurostat/documents/2995521/9333446/3-25102018-AP-EN.pdf/3fa5fa53-e076-4a5f-8bb5-a8075f639167 (accessed on 19 December 2018).
- European Centre for Disease Prevention and Control. Monitoring implementation of the Dublin Declaration on Partnership to Fight HIV/AIDS in Europe and Central Asia: 2017 Progress Report Stockholm; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2017.
- Eurostat. Eurostat migr_asydcfsta, t. Asylum quarterly report. Available online: https://ec.europa.eu/eurostat/statistics-explained/pdfscache/13562.pdf (accessed on 19 December 2018).
- European Parliament. EU Migrant Crisis: Facts and Figures. 2017. Available online: http://www.europarl.europa.eu/news/en/headlines/society/20170629STO78630/eu-migrant-crisis-facts-and-figures (accessed on 19 December 2018).
- Gushulak, B.D.; MacPherson, D.W. Population mobility and health: An overview of the relationships between movement and population health. J. Travel Med. 2004, 11, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Beknazarova, M.; Whiley, H.; Ross, K. Strongyloidiasis: A disease of socioeconomic disadvantage. Int. J. Environ. Res. Public Health 2016, 13, 517. [Google Scholar] [CrossRef] [PubMed]
- Seedat, F.; Hargreaves, S.; Nellums, L.B.; Ouyang, J.; Brown, M.; Friedland, J.S. How effective are approaches to migrant screening for infectious diseases in Europe? A systematic review. Lancet Infect. Dis. 2018, 18, e259–e271. [Google Scholar] [CrossRef]
- Kortas, A.; Polenz, J.; von Hayek, J.; Rüdiger, S.; Rottbauer, W.; Storr, U.; Wibmer, T. Screening for infectious diseases among asylum seekers newly arrived in Germany in 2015: A systematic single-centre analysis. Public Health 2017, 153, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Aldridge, R.W.; Yates, T.A.; Zenner, D.; White, P.J.; Abubakar, I.; Hayward, A.C. Pre-entry screening programmes for tuberculosis in migrants to low-incidence countries: A systematic review and meta-analysis. Lancet Infect. Dis. 2014, 14, 1240–1249. [Google Scholar] [CrossRef]
- Carballo, M.; Hargreaves, S.; Gudumac, I.; Maclean, E.C. Evolving migrant crisis in Europe: Implications for health systems. Lancet Glob. Health 2017, 5, e252–e253. [Google Scholar] [CrossRef]
- Karki, T.; Napoli, C.; Riccardo, F.; Fabiani, M.; Dente, M.G.; Carballo, M.; Noori, T.; Declich, S. Screening for infectious diseases among newly arrived migrants in EU/EEA countries-varying practices but consensus on the utility of screening. Int. J. Environ. Res. Public Health 2014, 11, 11004–11014. [Google Scholar] [CrossRef]
- Semenza, J.C.; Carrillo-Santisteve, P.; Zeller, H.; Sandgren, A.; van der Werf, M.J.; Severi, E.; Pastore Celentano, L.; Wiltshire, E.; Suk, J.E.; Dinca, I.; et al. Public Health needs of migrants, refugees and asylum seekers in Europe, 2015: Infectious disease aspects. Eur. J. Public Health 2016, 26, 372–373. [Google Scholar] [CrossRef]
- Schunemann, H.J.; Wiercioch, W.; Brozek, J.; Etxeandia-Ikobaltzeta, I.; Mustafa, R.A.; Manja, V.; Brignardello-Petersen, R.; Neumann, I.; Falavigna, M.; Alhazzani, W.; et al. GRADE Evidence to Decision (EtD) frameworks for adoption, adaptation, and de novo development of trustworthy recommendations: GRADE-ADOLOPMENT. J. Clin. Epidemiol. 2017, 81, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Pottie, K.; Mayhew, A.D.; Morton, R.L.; Greenaway, C.; Akl, E.A.; Rahman, P.; Zenner, D.; Pareek, M.; Tugwell, P.; Welch, V.; et al. Prevention and assessment of infectious diseases among children and adult migrants arriving to the European Union/European Economic Association: A protocol for a suite of systematic reviews for public health and health systems. BMJ Open 2017, 7, e014608. [Google Scholar] [CrossRef] [PubMed]
- Shemilt, I.; Thomas, J.; Morciano, M. A web-based tool for adjusting costs to a specific target currency and price year. Evid. Policy A J. Res. Debate Pract. 2010, 6, 51–59. [Google Scholar] [CrossRef]
- Shea, B.J.; Grimshaw, J.M.; Wells, G.A.; Boers, M.; Andersson, N.; Hamel, C.; Porter, A.C.; Tugwell, P.; Moher, D.; Bouter, L.M. Development of AMSTAR: A measurement tool to assess the methodological quality of systematic reviews. BMC Med. Res. Methodol. 2007, 7, 10. [Google Scholar] [CrossRef] [PubMed]
- The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 19 December 2018).
- Whiting, P.F.; Rutjes, A.W.; Westwood, M.E.; Mallett, S.; Deeks, J.J.; Reitsma, J.B.; Leeflang, M.M.; Sterne, J.A.; Bossuyt, P.M. QUADAS-2: A revised tool for the quality assessment of diagnostic accuracy studies. Ann. Intern. Med. 2011, 155, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Ochodo, E.A.; Gopalakrishna, G.; Spek, B.; Reitsma, J.B.; van Lieshout, L.; Polman, K.; Lamberton, P.; Bossuyt, P.M.M.; Leeflang, M.M.G. Circulating antigen tests and urine reagent strips for diagnosis of active schistosomiasis in endemic areas. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef]
- King, C.H.; Bertsch, D. Meta-analysis of Urine Heme Dipstick Diagnosis of Schistosoma haematobium Infection, Including Low-Prevalence and Previously-Treated Populations. PLoS Negl. Trop. Dis. 2013, 7, e2431. [Google Scholar] [CrossRef]
- Wang, W.; Li, Y.; Li, H.; Xing, Y.; Qu, G.; Dai, J.; Liang, Y. Immunodiagnostic efficacy of detection of Schistosoma japonicum human infections in China: A meta analysis. Asian Pac. J. Trop. Med. 2012, 5, 15–23. [Google Scholar] [CrossRef]
- Danso-Appiah, A.; Olliaro, P.L.; Donegan, S.; Sinclair, D.; Utzinger, J. Drugs for treating Schistosoma mansoni infection. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Kramer, C.V.; Zhang, F.; Sinclair, D.; Olliaro, P.L. Drugs for treating urinary schistosomiasis. Cochrane Database Syst. Rev. 2014. [Google Scholar] [CrossRef] [PubMed]
- Pérez del Villar, L.; Burguillo, F.J.; López-Abán, J.; Muro, A. Systematic Review and Meta-Analysis of Artemisinin Based Therapies for the Treatment and Prevention of Schistosomiasis. PLoS ONE 2012, 7, e45867. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Tan, X.D.; Liu, B.; Yang, C.; Ni, Z.L.; Gao, X.D.; Wang, Y. Meta-analysis of the diagnostic efficiency of the questionnaires screening for schistosomiasis. Parasitol. Res. 2015, 114, 3509–3519. [Google Scholar] [CrossRef] [PubMed]
- Campo Polanco, L.; Gutierrez, L.A.; Cardona Arias, J. Diagnosis of Strongyloides Stercoralis infection: Meta-analysis on evaluation of conventional parasitological methods (1980–2013). Rev. Esp. Salud Publica 2014, 88, 581–600. (In French) [Google Scholar] [CrossRef] [PubMed]
- Henriquez-Camacho, C.; Gotuzzo, E.; Echevarria, J.; White, A.C., Jr.; Terashima, A.; Samalvides, F.; Pérez-Molina, J.A.; Plana, M.N. Ivermectin versus albendazole or thiabendazole for Strongyloides stercoralis infection. Cochrane Database Syst. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Danso-Appiah, A.; Minton, J.; Boamah, D.; Otchere, J.; Asmah, R.H.; Rodgers, M.; Bosompem, K.M.; Eusebi, P.; De Vlas, S.J. Accuracy of point-of-care testing for circulatory cathodic antigen in the detection of schistosome infection: Systematic review and meta-analysis. Bull. World Health Organ. 2016, 94, 522–533. [Google Scholar] [CrossRef] [PubMed]
- Kinkel, H.F.; Dittrich, S.; Baumer, B.; Weitzel, T. Evaluation of eight serological tests for diagnosis of imported schistosomiasis. Clin. Vaccine Immunol. 2012, 19, 948–953. [Google Scholar] [CrossRef]
- Lodh, N.; Mwansa, J.C.; Mutengo, M.M.; Shiff, C.J. Diagnosis of Schistosoma mansoni without the stool: Comparison of three diagnostic tests to detect Schistosoma [corrected] mansoni infection from filtered urine in Zambia. Am. J. Trop. Med. Hyg. 2013, 89, 46–50. [Google Scholar] [CrossRef]
- Espirito-Santo, M.C.; Alvarado-Mora, M.V.; Dias-Neto, E.; Botelho-Lima, L.S.; Moreira, J.P.; Amorim, M.; Pinto, P.L.; Heath, A.R.; Castilho, V.L.; Goncalves, E.M.; et al. Evaluation of real-time PCR assay to detect Schistosoma mansoni infections in a low endemic setting. BMC Infect. Dis. 2014, 14, 558. [Google Scholar] [CrossRef]
- Espirito-Santo, M.C.; Alvarado-Mora, M.V.; Pinto, P.L.; Sanchez, M.C.; Dias-Neto, E.; Castilho, V.L.; Goncalves, E.M.; Chieffi, P.P.; Luna, E.J.; Pinho, J.R.; et al. Comparative Study of the Accuracy of Different Techniques for the Laboratory Diagnosis of Schistosomiasis Mansoni in Areas of Low Endemicity in Barra Mansa City, Rio de Janeiro State, Brazil. Biomed. Res. Int. 2015, 2015, 135689. [Google Scholar] [CrossRef]
- da Frota, S.M.; Carneiro, T.R.; Queiroz, J.A.; Alencar, L.M.; Heukelbach, J.; Bezerra, F.S. Combination of Kato-Katz faecal examinations and ELISA to improve accuracy of diagnosis of intestinal schistosomiasis in a low-endemic setting in Brazil. Acta Trop. 2011, 120 (Suppl. 1), S138–S141. [Google Scholar] [CrossRef] [PubMed]
- Silveira, A.M.; Costa, E.G.; Ray, D.; Suzuki, B.M.; Hsieh, M.H.; Fraga, L.A.; Caffrey, C.R. Evaluation of the CCA Immuno-Chromatographic Test to Diagnose Schistosoma mansoni in Minas Gerais State, Brazil. PLoS Negl. Trop. Dis. 2016, 10, e0004357. [Google Scholar] [CrossRef] [PubMed]
- Espirito-Santo, M.C.; Sanchez, M.C.; Sanchez, A.R.; Alvarado-Mora, M.V.; Castilho, V.L.; Goncalves, E.M.; Luna, E.J.; Gryschek, R.C. Evaluation of the sensitivity of IgG and IgM ELISA in detecting Schistosoma mansoni infections in a low endemicity setting. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 2275–2284. [Google Scholar] [CrossRef] [PubMed]
- Beltrame, A.; Guerriero, M.; Angheben, A.; Gobbi, F.; Requena-Mendez, A.; Zammarchi, L.; Formenti, F.; Perandin, F.; Buonfrate, D.; Bisoffi, Z. Accuracy of parasitological and immunological tests for the screening of human schistosomiasis in immigrants and refugees from African countries: An approach with Latent Class Analysis. PLoS Negl. Trop. Dis. 2017, 11, e0005593. [Google Scholar] [CrossRef] [PubMed]
- Bisoffi, Z.; Buonfrate, D.; Sequi, M.; Mejia, R.; Cimino, R.O.; Krolewiecki, A.J.; Albonico, M.; Gobbo, M.; Bonafini, S.; Angheben, A.; et al. Diagnostic accuracy of five serologic tests for Strongyloides stercoralis infection. PLoS Negl. Trop. Dis. 2014, 8, e2640. [Google Scholar] [CrossRef] [PubMed]
- Rascoe, L.N.; Price, C.; Shin, S.H.; McAuliffe, I.; Priest, J.W.; Handali, S. Development of Ss-NIE-1 recombinant antigen based assays for immunodiagnosis of strongyloidiasis. PLoS Negl. Trop. Dis. 2015, 9, e0003694. [Google Scholar] [CrossRef] [PubMed]
- Knopp, S.; Salim, N.; Schindler, T.; Karagiannis Voules, D.A.; Rothen, J.; Lweno, O.; Mohammed, A.S.; Singo, R.; Benninghoff, M.; Nsojo, A.A.; et al. Diagnostic accuracy of Kato-Katz, FLOTAC, Baermann, and PCR methods for the detection of light-intensity hookworm and Strongyloides stercoralis infections in Tanzania. Am. J. Trop. Med. Hyg. 2014, 90, 535–545. [Google Scholar] [CrossRef] [PubMed]
- King, C.H.; Olbrych, S.K.; Soon, M.; Singer, M.E.; Carter, J.; Colley, D.G. Utility of Repeated Praziquantel Dosing in the Treatment of Schistosomiasis in High-Risk Communities in Africa: A Systematic Review. PLoS Negl. Trop. Dis. 2011, 5, e1321. [Google Scholar] [CrossRef] [PubMed]
- Muennig, P.; Pallin, D.; Sell, R.L.; Chan, M.-S. The Cost Effectiveness of Strategies for the Treatment of Intestinal Parasites in Immigrants. N. Engl. J. Med. 1999, 340, 773–779. [Google Scholar] [CrossRef]
- Muennig, P.; Pallin, D.; Challah, C.; Khan, K. The cost-effectiveness of ivermectin vs. albendazole in the presumptive treatment of strongyloidiasis in immigrants to the United States. Epidemiol. Infect. 2004, 132, 1055–1063. [Google Scholar] [CrossRef]
- Maskery, B.; Coleman, M.S.; Weinberg, M.; Zhou, W.; Rotz, L.; Klosovsky, A.; Cantey, P.T.; Fox, L.M.; Cetron, M.S.; Stauffer, W.M. Economic Analysis of the Impact of Overseas and Domestic Treatment and Screening Options for Intestinal Helminth Infection among US-Bound Refugees from Asia. PLoS Negl. Trop. Dis. 2016, 10, e0004910. [Google Scholar] [CrossRef] [PubMed]
- Worrell, C.M.; Bartoces, M.; Karanja, D.M.; Ochola, E.A.; Matete, D.O.; Mwinzi, P.N.; Montgomery, S.P.; Secor, W.E. Cost analysis of tests for the detection of Schistosoma mansoni infection in children in western Kenya. Am. J. Trop. Med. Hyg. 2015, 92, 1233–1239. [Google Scholar] [CrossRef] [PubMed]
- Libman, M.D.; MacLean, J.D.; Gyorkos, T.W. Screening for schistosomiasis, filariasis, and strongyloidiasis among expatriates returning from the tropics. Clin. Infect. Dis. 1993, 17, 353–359. [Google Scholar] [CrossRef] [PubMed]
- CDC. Guidelines for Overseas Presumptive Treatment of Strongyloidiasis, Schistosomiasis, and Soil-Transmitted Helminth Infections; CDC: Atlanta, GA, USA, 2013.
- Buonfrate, D.; Sequi, M.; Mejia, R.; Cimino, R.O.; Krolewiecki, A.J.; Albonico, M.; Degani, M.; Tais, S.; Angheben, A.; Requena-Mendez, A.; et al. Accuracy of five serologic tests for the follow up of Strongyloides stercoralis infection. PLoS Negl. Trop. Dis. 2015, 9, e0003491. [Google Scholar] [CrossRef] [PubMed]
- Zammarchi, L.; Bonati, M.; Strohmeyer, M.; Albonico, M.; Requena-Méndez, A.; Bisoffi, Z.; Nicoletti, A.; García, H.H.; Bartoloni, A. Screening, diagnosis and management of human cysticercosis and T. solium taeniasis: Technical recommendations by the COHEMI project study group. Trop. Med. Int. Health 2017, 2, 881–894. [Google Scholar] [CrossRef] [PubMed]
- Jonas, D.E.; Ferrari, R.M.; Wines, R.C.; Vuong, K.T.; Cotter, A.; Harris, R.P. Evaluating evidence on intermediate outcomes: Considerations for groups making healthcare recommendations. Am. J. Prev. Med. 2018, 54, S38–S52. [Google Scholar] [CrossRef] [PubMed]
- Atkins, D.; Best, D.; Briss, P.A.; Eccles, M.; Falck-Ytter, Y.; Flottorp, S.; Guyatt, G.H.; Harbour, R.T.; Haugh, M.C.; Henry, D. Grading quality of evidence and strength of recommendations. BMJ 2004, 328, 1490. [Google Scholar]
| Study | Quality | Design | Population | Intervention/Outcomes | Results |
|---|---|---|---|---|---|
| Included systematic reviews of diagnostic tests to detect schistosomiasis | |||||
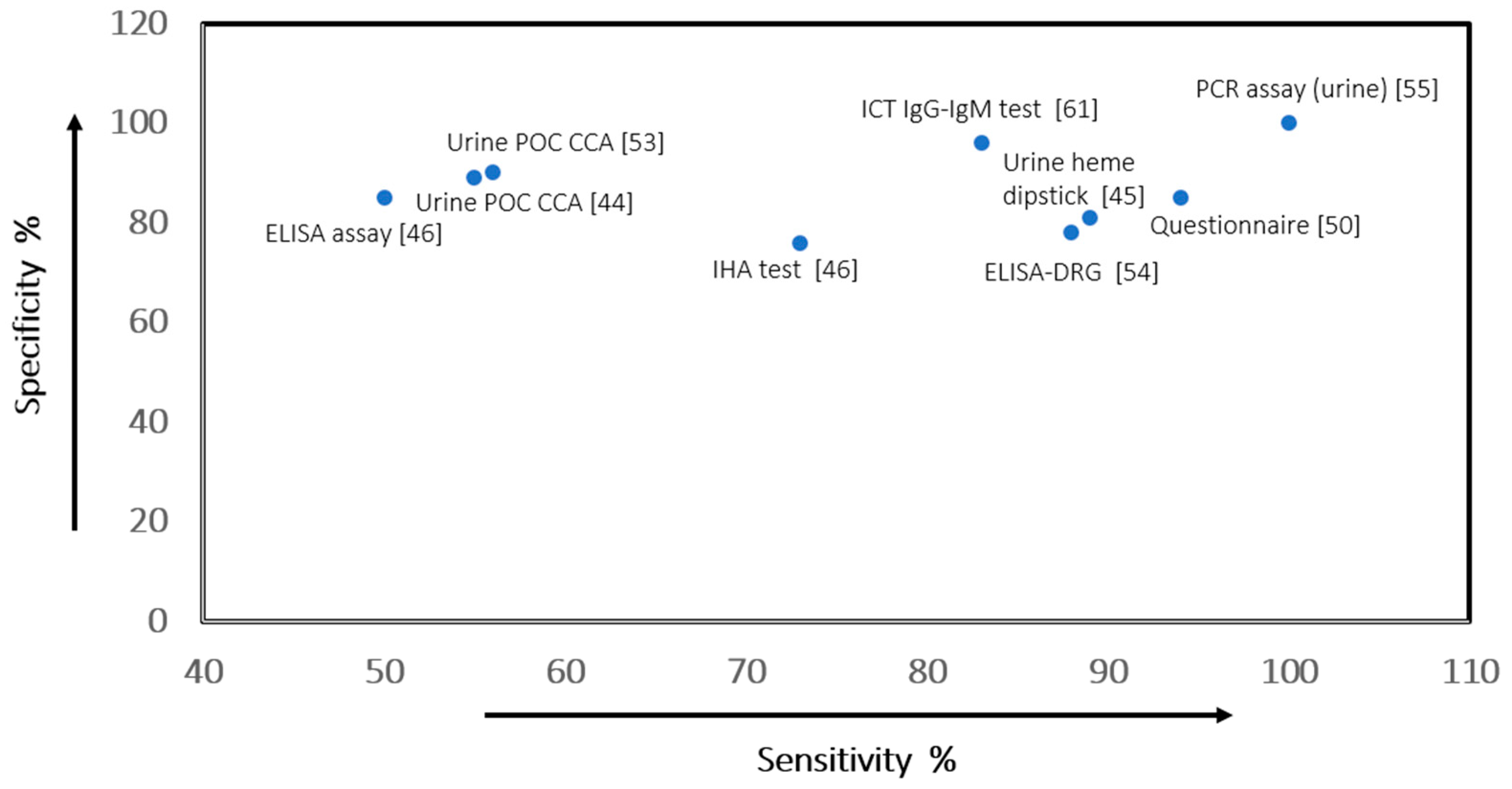
| Danso Appiah et al., 2016 [53] | AMSTAR: 11/11 GRADE: low to moderate-quality evidence | Systematic review and meta-analysis | Preschool children and infants, school-aged children or adults from high-/low-prevalence locations | Intervention: POC CCA for Sc. mansoni Outcomes: detection of egg-positive urine—sensitivity/specificity (95% CI:) | Sensitivity/specificity (95% CI) POC CCA (single standard) 90% (84–94)/56% (39–71); POC CCA (duplicate standard) 85% (80–88)/66% (53–76); POC CCA (triplicate standard) 91% (84–95)/56% (39–72) |
| Yang et al., 2015 [50] | AMSTAR: 11/11 GRADE: low to moderate-quality evidence | Meta-analysis | Patients infected with schistosomiasis in endemic areas; mainly school children, Africa and China | Intervention: questionnaire screening for Schistosoma species. Outcomes: sensitivity/specificity (95% CI:) | Sensitivity/specificity (95%CI:) Sc. haematobium 85% (84–86)/94% (94–94); Sc. mansoni 46% (45–47)/81% (80–82); Sc. japonicum 82% (79–85)/59% (57–60) |
| Ochodo et al., 2015 [44] | AMSTAR: 11/11 GRADE: very low to low-quality evidence | Systematic review and meta-analysis of RCTs | Individuals with active infection with S. haematobium | Intervention: urine reagent strip tests; circulating antigen tests in urine/serum Outcomes: sensitivity/specificity (95% CI:) | Sensitivity/specificity (95% CI) Sc. haematobuim: microhaematuria 75% (71–79)/87% (84–90); proteinuria 61% (53–68)/82% (77–88); leukocyturia 58% (44–71)/61% (34–88); Sc. mansoni (CCA test) 89% (86–92)/55% (46–65) |
| King and Bertsch, 2013 [45] | AMSTAR: 11/11 GRADE: low-quality evidence | Systematic review and meta-analysis of surveys | Schools, communities with high/low prevalence, low intensity groups in Africa | Intervention: dipstick test Sc. haematobium. Outcomes: sensitivity and specificity (95% CI:), diagnostic odds ratio (DOR) | Sensitivity/specificity (95% CI) Detection of egg-positive urine 81% (79–83)/89% (87–92). In high-prevalence settings 80% (78–83)/86% (82–90); lower in treated population 72% (61–78)/87% (81–94); in lower intensity population subgroups 65% (58–72)/82% (76–90) |
| Wang, et al., 2012 [46] | AMSTAR: 7/11 GRADE: very low- to low-quality evidence | Systematic review and meta-analysis of RCTs, retro-/pro-observational studies | Infected patients with schistosomiasis in control programmes in China | Intervention: IHA and ELISA. Outcomes: true positive rates, sensitivity/specificity (95% CI:), DOR | Sensitivity/specificity (95% CI) IHA 75.6% (74–77)/73% (72–74) ELISA 84.9% (83–87)/50.4% (49.2–51.6) The DOR of IHA was 9.41 (95% CI: 5–18), and ELISA 4.78 (95% CI: 3.21–7.13) |
| Included primary studies of diagnostic tests to detect schistosomiasis | |||||
| Espirito-Santo et al., 2015 [57] | QUADAS-2-11/14 GRADE: very low- to low-quality evidence | Cross-sectional epidemiological survey in areas of low prevalence of Sc. Mansoni | The estimated sample size required was 650 individuals; Barra Mansa City, Rio de Janeiro State, Brazil | Intervention: diagnostic assays: ELISA-IgG/ELISA-IgM/IFT-IgM/qPCR in faeces. Outcomes: sensitivity/specificity (95% CI:) | Sensitivity/specificity (95% CI) KK 13.8% (4–32)/99.8% (99.0–100); ELISA-IgG 66.7% (48–82)/91.5% (89–94); ELISA-IgM 81.8% (64–93)/82% (79–85); IFT-IgM 78.8% (61–91)/87.7% (84.8–90); qPCR in faeces 51.7% (32–71)/92.6% (90–95); qPCR in serum 12.1% (3–28)/99.1% (98–99) |
| Espirito-Santo et al., 2014a [60] | QUADAS-2-12/14 GRADE: very low- to low-quality evidence | Cross-sectional study | City of Barra Mansa, Rio de Janeiro State, Brazil, with an estimated prevalence of 1% | Intervention: diagnostic assays: ELISA-IgG and ELISA-IgM. Outcomes: sensitivity/specificity (95% CI:); PPV, NPV | Sensitivity/specificity (95%CI) ELISA-IgG 60.0% (15–95) /89.1% (86.2–91.5); ELISA-IgM 60.0(15–95)/79.2% (75.6–82.5) PPV/NPV (95%CI): ELISA-IgG 4.6% (1–13) /99.6% (98–100); ELISA-IgM 2.5% (0.5–7); NPV 99.6% (98.4–100.0) |
| Espirito-Santo et al., 2014b [56] | QUADAS-2-13/14 GRADE: very low- to low-quality evidence | Cross-sectional epidemiological survey | 7000 inhabitants located in the outskirts of Barra Mansa, Rio de Janeiro, Brazil | Intervention: qPCR in serum or faeces. Outcomes: sensitivity/specificity (95% CI:); PPV, NPV | Sensitivity/specificity (95% CI) qPCR in faeces 80.0% (28–99)/92.4% (90–94); qPCR in serum 20.0% (0.5–71.6)/98.8 (97–99) PPV/NPV (95% CI:): qPCR in faeces 8.0% (2–19)/99.8% (99–100); qPCR in serum 12.5% (0.3–52.7)/99.3% (98.2–99.8) |
| Lodh et al., 2013 [55] | QUADAS-2-12/14 GRADE: very low- to low-quality evidence | Cross-sectional case study | Filtered urine specimens from infected and not-infected patients in Zambia | Intervention: qPCR ELISA IgG in serum or faeces; filtered Urine PCR. Outcomes: sensitivity/specificity (95% CI:); PPV, NPV | Sensitivity/specificity (95%CI) KK test 57% (45–68)/100% (69–100); CCA rapid test 65% (56–77)/60% (26–88); PCR 100% (95–100)/100% (69–100) PPV/NPV: KK test 100%/23%; CCA rapid test 93%/19%; PCR 100%/100%. |
| Kinkel et al., 2012 [54] | QUADAS-2-12/14 GRADE: very low- to low-quality evidence | Retrospective comparative diagnostic study: performance of 8 serological tests for Schistosoma spp | Serum specimens from infected patients and those without the infection in low-prevalence locations or non-endemic settings (Germany) | Intervention: serological assays: IFAT, ELISA-CA, ELISA-AWA, ELISA-SEA, IHA, ELISA-NovaTec, ELISA-DRG and ELISA-Viramed. Outcomes: sensitivity and specificity (95% CI:) | Sensitivity/specificity-(95% CI): IFAT 75.7% (58–98)/98.1% (92–99); ELISA-CA 40.5% (25–59)/95.2% (89–98); ELISA-AWA 54.1% (37–70)/100% (95.6–100); ELISA-SEA-75.7% (58–98)/97.1% (91–99); IHA 73.0% (55.6–85.6)/99.0% (94.0–100); ELISA-NovaTec 64.9% (47–79)/99 (94–100); ELISA-DRG 78.3% (61.3–89.6)/88.4 (80–94); ELISA-Viramed 67.6% (50–81)/76.9% (67–84). |
| De Frotas et al., 2011 [58] | QUADAS-2-12/14 GRADE: very low- to low-quality evidence | Cross-sectional survey | Stool and serum specimens from infected and not infected patients, low-endemic setting in Brazil | Intervention: serological assays, ELISA IgG Outcomes: sensitivity and specificity (95%CI) | Sensitivity/specificity (95% CI): ELISA-IgG 100% (68–100)/72.9% (67–78). PPV/NPV (95% CI): ELISA-IgG 26.0% (18–36) /100% (97–100). |
| Silveira et al., 2016 [59] | QUADAS-2-12/14 GRADE: very low- to low-quality evidence | Evaluation of the CCA test to diagnose Sc. mansoni in Minas Gerais State, Brazil. | Infected individuals in regions with moderate to high prevalence | Intervention: CCA-immuno-chromatographic test. Outcomes: sensitivity/specificity (95% CI:) | Sensitivity/specificity (95% CI): CCA-ICT 68.7% (54–81)/97.6% (87–99) |
| Beltrame et al., 2017 [61] | QUADAS-2-12/14 GRADE: very low- to low-quality evidence | Accuracy of parasitological and immunological tests for the screening of human schistosomiasis in immigrants and refugees from African countries | Frozen serum specimens from recent African asylum seekers that were routinely screened for schistosomiasis in Italy | Intervention: urine CCA; Bordier-ELISA, Western Blot IgG, ICT IgG-IgM, microscopy compared with composite reference standard. Outcomes: sensitivity/specificity (95% CI:) | Sensitivity/specificity (95% CI): Urine CCA 29% (22–37)/95% (91–97); Bordier-ELISA 71% (63–78)/99.6% (98–100); Western blot IgG 92% (86–96)/94% (90–97); ICT IgG-IgM 96% (91–99)/83% (77–87); microscopy 45% (37–54)/100% |
| Included systematic reviews for diagnostic effectiveness for strongyloidiasis | |||||
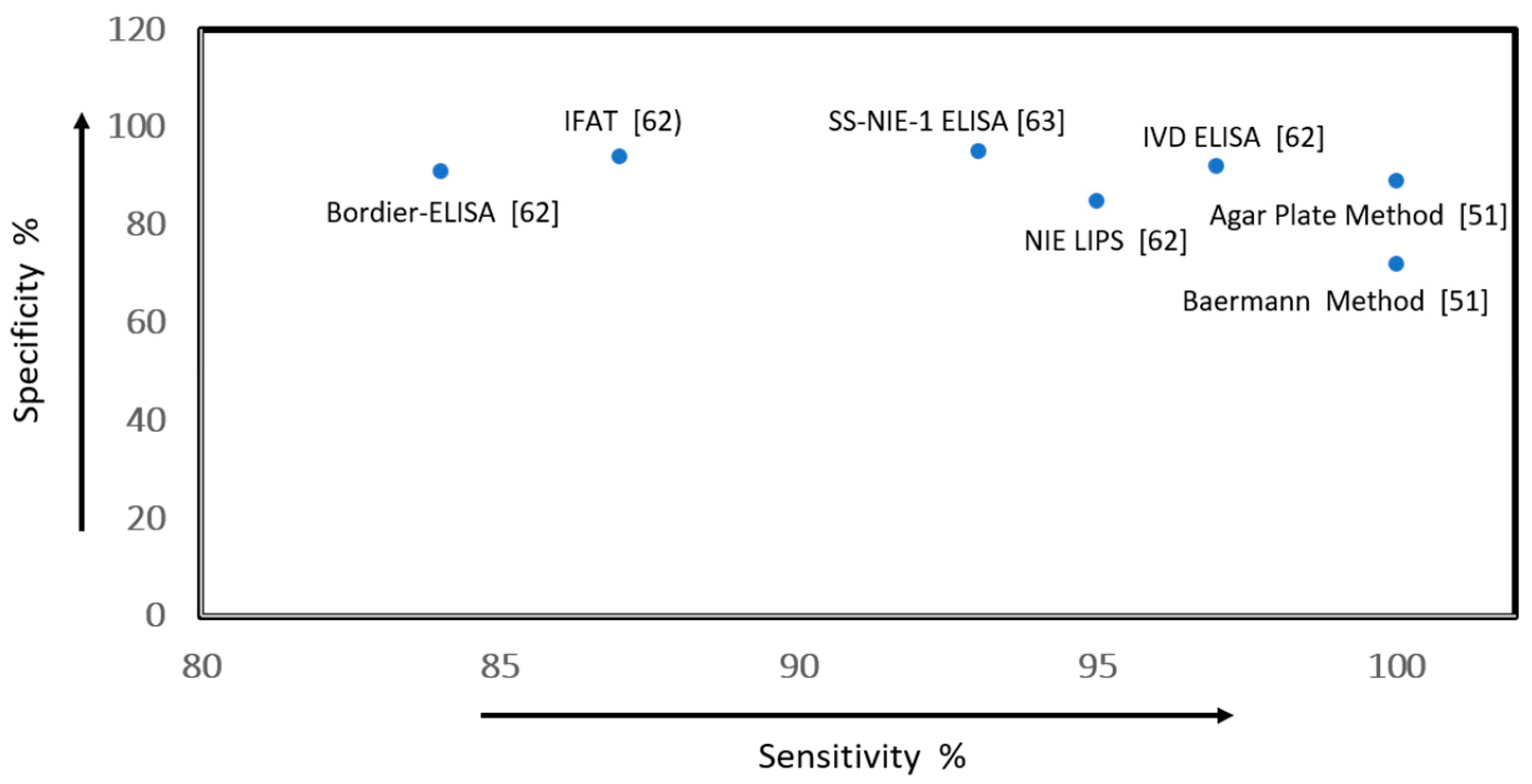
| Campo Polanco et al., 2014 [51] | AMSTAR: 11/11 GRADE: moderate-quality evidence | Systematic review and meta-analysis | Individuals with active/chronic infection | Intervention: Baermann method, agar plate, direct faecal smear examination and formol-ether concentration technique. Outcome: sensitivity and specificity (95% CI:) | Sensitivity: Baermann method (72%) with LR+228 and LR−0.32; APC 89%, LR+341 and LR−0.11; stool microscopy 21%, LR + 67 and LR−0.67; formol-ether concentration 48%, LR + 110 and LR−0.59. Specificity: 100% in all four tests. APC and Baermann method are best. |
| Requena-Méndez et al., [19] | AMSTAR: 7/11 GRADE: low- to moderate-quality evidence | Systematic review | Individuals with active/chronic infection | Intervention: Baermann method, agar plate, direct faecal smear examination and formol-ether concentration technique, serological techniques. Outcome: sensitivity and specificity (95% CI:) | No meta-analysis was undertaken. Sensitivity and specificity of different techniques were individually reported. |
| Included primary studies for diagnostic effectiveness for strongyloidiasis | |||||
| Bisofi et al., 2014 [62] | QUADAS-2: 13/14 GRADE: low-quality evidence | Retrospective comparative diagnostic study to evaluate the performance of 5 tests for St. stercoralis. | Serum specimens from subjects with St. stercoralis; healthy people and patients with previous exposure | Intervention: IFAT, NIE-LIPS NIE-ELISA, IVD-ELISA- and Bordier-ELISA Outcome: sensitivity and specificity (95% CI:) | Sensitivity/specificity (95% CI): NIE-ELISA 75.4% (67–83)/94.8% (91–99); NIE-LIPS 85.1% (78–92)/100% (100–100); IFAT 93.9% (89–98)/92.2% (87–97); IVD-ELISA 91.2% (86–96)/99.1% (97.4–100.0); Bordier-ELISA 89.5% (84–95) 98.3% (96–100). |
| Rascoe et al., 2015 [63] | QUADAS-2: 10/14 GRADE: low-quality evidence | Retrospective comparative diagnostic study of 5 tests for the follow-up of patients infected with St. stercoralis | Serum samples positive for St. stercoralis and negative samples from United States residents with no history of foreign travel | Intervention: Ss-NIE-1 ELISA, Ss-NIE-1 Luminex. Outcome: sensitivity and specificity (95% CI:) | Sensitivity/specificity (95% CI): SS-NIE-1 ELISA 95% (92–97)/93% (90–96); Ss-NIE-1 Luminex 93% (88–96)/95% (93–97). The inter-assay coefficient of variation was determined to be 22% for the low-positive control serum and 10% for the medium-positive control serum. |
| Knopp et al., 2014 [64] | QUADAS-2: 11/14 GRADE: low-quality evidence | International standard randomised controlled trial | Children and adults residing in rural villages in the Baga moyo District, Tanzania (endemic areas) | Intervention: Real-time PCR, FLOTAC technique, KK method. Outcome: sensitivity and specificity (95% CI:) | Sensitivity/specificity (95% CI): PCR + pseudo-standard PCR 17.4 (8–31)/3.9 (89–97); Baermann + pseudo-standard 47 (23–72)/78.4 (72 -84); PCR + multiple gold standard 30.9 (19.1–44.8)/100 (100–100); Baermann + multiple gold standard 83.6 (71.2–92.2)/100 (100–100) |
| Study | Quality | Design | Population | Intervention/Outcomes | Results |
|---|---|---|---|---|---|
| Treatment efficacy of anti-Schistosoma drugs | |||||
| Kramer et al., 2014 [48] | AMSTAR: 11/11 Data in study: GRADE: high-quality evidence | Systematic review, fixed effects meta-analysis; Embase, MEDLINE (1966 to 2014), LILACS, Cochrane library, Cochrane infectious disease (1980–2014) | School-aged and young adults: 6–20 years (16 trials); 2–23 years (5 trials); Adults (2 trials). Participants setting: Rural areas in 15 sub-Saharan African countries; an urban setting in Saudi Arabia | Interventions: drugs used to treat urinary schistosomiasis: praziquantel, metrifonate, artesunate and/or in combination Outcome: parasitological cure or failure at 4 weeks; % egg reduction rate at 4 weeks | Praziquantel (single dose 40 mg/kg), egg reduction (60%) in urine achieved in 4–8 weeks (38 per 100 (95% CI: 26–54). Treatment failure: RR 0.42, (95% CI: 0.29–0.59), 864 participants, 7 trials Metrifonate (single dose 10 mg/kg) reduced egg excretion only marginally in comparison to placebo (RR 0.63, 95% CI: 0.54 to 0.73) 210 participants, 1 trial, at 8 months |
| Danso-Appiah et al., 2013 [47] | AMSTAR: 11/11 Data in study: GRADE: low- to moderate-quality evidence | Systematic review and meta-narrative of RCTs, RTCs of anti-Schistosoma drugs | Trials conducted in Africa (n = 36), South America (n = 15; all in Brazil) and the Middle East (n = 1). 52 trials enrolling 10,269 participants in endemic areas | Intervention: praziquantel 40 mg/kg, oxamniquine 40 mg/kg | Praziquantel (single dose 40 mg/kg) vs. placebo: reduced parasitological treatment failure at 1 month (69/100; RR = 3.13, 2 trials, 414 participants). Praziquantel (single dose 30 mg/kg): RR = 1.52, 3 trials, 521 participants. Higher doses: no significant difference. Oxamniquine (single dose 40 mg/kg) vs. Placebo: reduced parasitological treatment failure at 3 months in 2 trials (68/100; RR = 8.74). |
| Pérez del Villar et al., 2012 [49] | AMSTAR: 11/11 Data in study: not reported. GRADE: Moderate-quality evidence | Quantitative systematic review and meta-analysis | Healthy villagers who live in areas in Africa endemic for Sc. haematobium and Sc. mansoni and in China for Sc. Japonicum | Intervention: prophylactic effect of artesunate or artemether vs. placebo against Sc. haematobium, Sc. mansoni and Sc. japonica infections. Outcomes: parasitological cure rate at 3–8 weeks; infection rate at 3–4 weeks after treatment. | Artesunate treatment (single dose: significantly lower cure rates than with praziquantel. Combined therapy of artesunate plus sulfadoxine-pyrimethamine: significantly less effective than praziquantel treatment Combination of artemisinin derivatives and praziquantel: higher cure rate than praziquantel monotherapy Artesunate or artemether: significantly better than a placebo. |
| Treatment efficacy of drugs for strongyloidiasis | |||||
| Henriquez-Camacho et al., 2016 [52] | AMSTAR: 11/11 GRADE: Moderate-quality evidence | Systematic review of RCTs, controlled or uncontrolled interventional studies. | Individuals with chronic infections of St. stercoralis; Immuno-competent patients. All ages | Intervention: ivermectin (single/double dose) vs. albendazole or thiabendazole. Outcome: elimination of infection; parasitological cure (>2 negative stool samples, 5 weeks). | Ivermectin (single/double dose) vs. albendazole: parasitological cure was higher with ivermectin, 84/100 vs. 48/100 ivermectin (RR = 1.79). Ivermectin vs. thiabendazole: little or no difference in parasitological cure, 74/100 vs. 68/100), but adverse events were less common with ivermectin (RR = 0.31) than albendazole. No serious adverse events or death reported |
| Study | Quality | Design | Population | Intervention/Outcomes | Results |
|---|---|---|---|---|---|
| Libman et al., 1993 [70] | NA | Retrospective-cross-sectional study with cost analysis | Cohort of individuals returning from the tropics and screened in a Canadian clinic 1981–1987 Costs in 1988 CAD | Stool examination + eosinophil count + serological studies for filariasis and schistosomiasis (gold standard); vs. stool examination + eosinophil count; vs. stool examination alone; vs. stool examination + serological studies; vs. eosinophil counts only Outcome: difference in cost or resource use/cost effectiveness | Difference in resource use/costs: high-/low-prevalence locations Costs per case of schistosomiasis and/or strongyloidiasis diagnosed for each strategy: (i) CAN$4674 [€3693]; (ii) CAN$6111 [€4829]; (iii) CAN$4788 [€3783]; (iv) CAN$3737 [€2953]; (v) CAN$3307 [€2613] Cost-effectiveness (ICER or INB): no ICER calculated. Study did not include a decision analytic model |
| Muennig et al., 1999 [66] | NA | Decision analytic model | Large immigrant populations in which St. stercoralis is not endemic (one third of the sample population was from the state of New York) Costs in 1997 USD | No preventive intervention (watchful waiting) vs. universal screening vs. presumptive treatment with albendazole Outcome: difference in cost or resource use/cost effectiveness (ICER or INB) per DALY averted | Difference in resource use/costs: gross costs: USD 11,086,181 [€7,228,785] for no intervention, USD 7,290,624 [€40,203,726] per year for treatment with albendazole, USD 40,547,651 [€40,203,726] for universal screening Cost-effectiveness (ICER or INB): treatment with albendazole was cost saving compared with no intervention, universal screening had ICER of USD 159,236/DALY [€157,885/DALY] averted |
| Muennig et al., 2004 [67] | NA | Decision analytic model (Markov) | California and New York, two states with large immigrant populations in which St. stercoralis is not endemic Costs in 2000 USD | Intervention: no intervention (watchful waiting) vs. 3 or 5 days of albendazole vs. eosinophil screening vs. ivermectin Outcome: difference in cost or resource use/cost effectiveness (ICER or INB) | Difference in resource use/costs: costs per person: no intervention USD 1666 [€1611], albendazole 3 days USD 1674 [€1618], albendazole 5 days USD 1680 [€1624], screening USD 1684 [€1628], ivermectin USD 1688 [€1632] Cost-effectiveness (ICER or INB): ICERs varied based on prevalence: albendazole USD 155–1584/QALY gained [€150–1531], albendazole 5 days USD 314–3175/QALY gained [€304-3069], ivermectin USD 848–8514/QALY gained [€820-8231]. Eosinophil was documented among all prevalence groups |
| King et al., 2011 [65] | AMSTAR | Systematic review of efficacy of schistosomiasis treatment with praziquantel (by dose), with a Markov model estimating cost-effectiveness of various dosing strategies | Non-migrants in endemic setting; population-based or sub-population-based (e.g., schools) drug treatment of Sc. haematobium or Sc. Mansoni. Costs in 2002 & 2008 USD | Intervention: No treatment vs. single dose of praziquantel per annual treatment vs. double dose Outcome: difference in cost or resource use/cost effectiveness (ICER or INB) | Difference in resource use/costs: single dose lifetime cost: USD 23 [€19] per person; double dose: USD 46 [€35] per person. Cost-effectiveness (ICER or INB): single dose: ICER of USD 48 [€39] and USD 46 [€37] per QALY gained for Sc. mansoni and Sc. haematobium, respectively, compared with no treatment; double dose: ICERs of USD 291 [€236] and USD 433 [€351] per QALY gained respectively compared with single dose |
| Worrell et al., 2015 [69] | NA | Cost analysis study | Cohort of children in Kenya assessed 2010–2011. Non-migrant settings. Costs in 2010 USD | Intervention: single KK (stool examination) vs. triplicate KK vs. POC CCA (urine dipstick) Outcome: difference in cost or resource use/cost effectiveness (ICER or INB) | Difference in resource use/costs: total costs per test: single KK USD 6.89 [€5], triplicate KK USD 17.54 [€14], POC CCA USD 7.26 [€6] Cost-effectiveness (ICER or INB): no ICER calculated, this was not a decision analysis study. |
| Maskery et al., 2016 [68] | NA | Cost analysis study; Markov model: discount rate of 3% over 60-year time horizon; costs in 2013 USD | Average annual cohort of 27,700 Asian refugees based on Department of Homeland Security data for 2002–2011, primarily from south-east Asia and the Middle East | Intervention: no screening or treatment vs. overseas albendazole and ivermectin treatment vs. domestic screening and treatment vs. overseas albendazole and domestic screening for strongyloidiasis. Outcome: difference in cost or resource use/cost effectiveness (ICER or INB) | Difference in resource use/costs, total costs per migrant (strongyloidiasis.): no treatment USD 5.99 [€5], overseas albendazole and ivermectin USD 15.12 [€12], domestic screening and treatment USD 138.36 [€108], overseas albendazole and domestic screening for Strongyloides infection USD 78.79 [€61]. Cost-effectiveness: ICERs per QALY gained: USD 2219 for “overseas albendazole and ivermectin”, USD 32,706 [€25,422] for domestic screening and treatment, USD 18,167 [€14,121] for overseas albendazole followed by domestic screening for strongyloidiasis. All vs. no screening or treatment [€1723] |
| Index Test at Median Test Prevalence in Study * | Sensitivity (95% CI) | Specificity (95% CI) | Post-Test Probability of a Positive Result (95% CI) | Post-Test Probability of a Negative Result (95% CI) | Number of Studies/ Participants | Certainty of Evidence (GRADE) | Reference Standard |
|---|---|---|---|---|---|---|---|
| PCR assay (filtered urine) at 89% prevalence—Sc. mansoni [55] | 1.00 (0.95–1.00) | 1.00 (0.69–1.00) | 100% (96–100) | 0% (37–0) | 1/89 | Very Low a,b,c | KK test—duplicate smears |
| Urine POC CCA test at 36% prevalence—Sc. mansoni [44] | 0.89 (0.86–0.92) | 0.55 (0.46–0. 65) | 53% (47–60) | 10% (15–7) | 15/6091 | Very Low a,b,c | Stool microscopy |
| Urine POC CCA test at 30% prevalence—Sc. mansoni [53] | 0.90 (0.84–0.94) d | 0.56 (0.39–0.71) d | 47% (37–58) | 7% (15–3) | 7/4584 | Moderate a,b | KK test |
| Questionnaire screening 30% prevalence—Sc. haematobium [50] | 0.85 (0.84–0.86) d | 0.94 (0.94–0.94) d | 86% (86–86) | 6% (7–6) | 12/41,412 | Low c,e | Urine filtration/microscopy |
| ELISA-DRG (commercial kit) at 26% prevalence—All cases [54] | 0.78 (0.61–0.90) | 0.88 (0.80–0.94) | 85% (65–95) | 7% (13–4) | 1/37 | Very Low c,e,f | Stool/urine microscopy |
| Urine heme dipstick at 27% prevalence—Sc. haematobium [45] | 0.81 (0.73–0.83) d | 0.89 (0.87–0.92) d | 73% (67–79) | 7% (10–6) | 98/126,119 | Low a,f,g | Urine microscopy |
| ELISA at 24% prevalence—Sc. japonicum [46] | 0.85 (0.83–0.87) | 0.50 (0.49–0.52) | 35% (34–36) | 9% (10–7) | 10/9014 | Low a,f,g | KK and Miracidium hatching test |
| IHA at 12% prevalence—Sc. japonicum [46] | 0.76 (0.72–0.74) d | 0.73 (0.72–0.74) d | 28% (26–28) | 4% (5–5) | 15/23,411 | Low a,b | KK and Miracidium hatching test |
| ICT IgG-IgM test at 17% prevalence Sc. mansoni and Sc. haematobium [61] | 0.96 (0. 91–0.99) | 0.83 (0.77–0.87) | 13% (9–16) | 0% (0–0) | 1/373 | Low b,c | Stool/urine microscopy/composite standard. |
- a
- Heterogeneity across similar studies because of several factors; downgraded because of serious inconsistency.
- b
- Use of intermediate or surrogate outcomes rather than health outcomes, hence a source of serious indirectness.
- c
- Single study design, not a randomised control trial.
- d
- Sensitivity and specificity values obtained from multiple-field study.
- e
- Use of indirect comparisons; sample population not migrants, another source of indirectness.
- f
- Very low-quality of evidence (downgraded by 1) because of serious indirectness.
- g
- Studies were insufficient to provide summary estimates for CAA tests.
| Index Test | True Positives Pre-Test Probability * | False Positives Pre-Test Probability * | True Negative Pre-Test Probability * | False Negative Pre-Test Probability | % Infected Correctly Diagnosed | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Test % Prevalence a | 2.5% | 10% | 30% | 2.5% | 10% | 30% | 2.5% | 10% | 30% | 2.5% | 10% | 30% | |
| PCR assay (filtered urine)—Sc. mansoni [55] | 25 | 100 | 300 | 0 | 0 | 0 | 975 | 900 | 700 | 0 | 0 | 0 | 100% |
| ICT IgG-IgM test—Sc. haematobium/Sc. mansoni [61] | 24 | 96 | 288 | 166 | 153 | 119 | 809 | 747 | 581 | 1 | 4 | 12 | 96% |
| Urine POC CCA test—Sc. mansoni [53] | 23 | 90 | 270 | 429 | 396 | 308 | 546 | 504 | 392 | 2 | 10 | 30 | 90% |
| Questionnaire screening—Sc. haematobium [50] | 21 | 85 | 255 | 58 | 54 | 42 | 917 | 846 | 658 | 4 | 15 | 45 | 85% |
| ELISA-DRG (commercial kit)—Sc. haematobuim/Sc. mansoni [54] | 20 | 78 | 235 | 47 | 43 | 34 | 928 | 857 | 666 | 5 | 22 | 65 | 78.3% |
| Urine heme dipstick—Sc. haematobium infections [45] | 20 | 81 | 243 | 107 | 99 | 77 | 868 | 801 | 623 | 5 | 19 | 57 | 81.0% |
| ELISA—Sc. japonicum [46] | 21 | 85 | 255 | 484 | 446 | 347 | 491 | 454 | 353 | 4 | 15 | 45 | 84.9% |
| IHA—Sc. japonicum [46] | 19 | 76 | 227 | 263 | 243 | 189 | 712 | 657 | 511 | 6 | 24 | 73 | 75.6% |
| Index Test—at 10% Prevalence * | Sensitivity (95% CI) | Specificity (95% CI) | Post-Test Probability of a Positive Result (95% CI) | Post-Test Probability of a Negative Result (95% CI) | Number of Studies/ Participants | Certainty of Evidence (GRADE) | Reference Standard |
|---|---|---|---|---|---|---|---|
| Baermann method [51] | 0.72 (0.67–0.76) a | 1.00 (1.00–1.00) a | 100% (100–100) | 3% (4–3) | 9/2459 | Moderate b,c | Combination of diagnostic tests |
| Agar plate—10% prevalence [51] | 0.89 (0.86–0.92) a | 1.00 (1.00–1.00) a | 100% (100–100) | 1% (2–1) | 10/3563 | Moderate b,c | Combination of diagnostic tests |
| NIE LIPS [62] d | 0.85 (0.79–0.92) | 0.95 (0.93–0.98) | 65% (56–84) | 2% (2–1) | 1/399 | Low e,f,g | Stool microscopy or culture |
| IVD ELISA—commercial test [62] | 0.92 (0.87–0.97) | 0.97 (0.96–0.99) | 77% (71–92) | 1% (1–0) | 1/399 | Low e,f,h | Stool microscopy |
| IFAT [62] | 0.94 (0.90–0.98) | 0.87 (0.83–0.91) | 45% (37–55) | 1% (1–0) | 1/399 | Low e,f,h | Stool microscopy and culture |
| Bordier-ELISA—commercial kit [62] | 0.91 (0.86–0.96) | 0.94 (0.91–0.96) | 63% (52–77) | 1% (2–0) | 1/193 | Low e,f,h | Kato–Katz, Flotac, and Baermann method |
| SS-NIE-1 ELISA [63] | 0.95 (0.92–0.97) | 0.93 (0.90–0.96) | 60% (71–73%) | 1% (1–0) | 1/583 | Low f,g,i | Stool microscopy and culture |
- a
- Sensitivity and specificity values obtained from a multiple-field study.
- b
- Evidence was downgraded because of serious inconsistencies and heterogeneity.
- c
- Heterogeneity between studies; use of intermediate or surrogate outcomes rather than health outcomes.
- d
- Test result with a primary standard.
- e
- Absence of a reliable gold standard for diagnosis of S. stercoralis infection. The review did not describe the specific gold standard used in the included studies for each test.
- f
- Single study design.
- g
- Samples were classified according to a composite reference standard, a procedure suggested for evaluation of diagnostic tests when there is no gold standard.
- h
- Use of intermediate or surrogate outcomes rather than health outcomes.
- i
- The inter-assay coefficient of variation was determined to be 22% for the low-positive control serum and 10% for the medium-positive control serum.
| Index tests | True-Positives Pre-Test Probability a | False-Positives Pre-Test Probability a | True-Negatives Pre-Test Probability a | False-Negatives Pre-Test Probability a | % Infected Correctly Diagnosed | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Test % Prevalence b | 2.5% | 10% | 30% | 2.5% | 10% | 30% | 2.5% | 10% | 30% | 2.5% | 10% | 30% | |
| Baermann method [51] | 18 | 72 | 216 | 0 | 0 | 0 | 975 | 900 | 700 | 7 | 28 | 84 | 72% |
| Agar plate [51] | 22 | 89 | 267 | 0 | 0 | 0 | 975 | 900 | 700 | 3 | 11 | 33 | 89% |
| NIE-LIPS [62] | 21 | 85 | 255 | 49 | 45 | 35 | 926 | 855 | 665 | 4 | 15 | 45 | 85.1% |
| IVD-ELISA (commercial test) [62] | 23 | 92 | 276 | 29 | 27 | 21 | 946 | 873 | 679 | 2 | 8 | 24 | 92% |
| IFAT [62] | 23 | 94 | 282 | 127 | 117 | 91 | 848 | 783 | 609 | 2 | 6 | 18 | 93.8% |
| Bordier-ELISA (commercial kit) [62] | 23 | 91 | 272 | 58 | 54 | 42 | 917 | 846 | 658 | 2 | 9 | 28 | 90.7% |
| SS-NIE-1 ELISA [63] | 24 | 95 | 285 | 68 | 63 | 49 | 907 | 837 | 651 | 1 | 5 | 15 | 95% |
- a
- Data reported as effect per 1000 migrants tested.
- b
- pre-test prevalence or probability of having schistosomiasis in an at-risk population.
| Outcomes | Anticipated Absolute Effects a (95% CI) | Relative Chance of Cure (95% CI) | Number of Participants/Studies | Certainty of the Evidence (GRADE) | |
|---|---|---|---|---|---|
| Risk with Placebo per 1000 | Cure with Intervention Drug | ||||
| Parasitological failure at 1 to 2 months (praziquantel 40 mg/kg single dose) [48] | 908 | 381 (263–562) | RR 0.42 (0.29 to 0.58) | 864/7 RCTs | High |
| Parasitological cure at 1 month b—Sc. mansoni infections (praziquantel 40 mg/kg single dose) [47] | 337 | 1000 (347–1000) | RR 3.13 (1.03–9.53) | 414/2 RCTs | Moderate c |
| Microhaematuria at 8 weeks (praziquantel 40 mg/kg single dose) [48] | 281 | 149 (93–236) | RR 0.53 (0.33–0.84) | 119/1 RCT | Low d,e,f |
| Infection rate of Sc. japonicum (artemether monotherapy 6 mg/kg) [49] | 175 | 44 (28–70) | RR 0.25 (0.16–0.40) | 8051/13 RCTs | Moderate c |
| Parasitological cure rate of Schistosoma species. (Artesunate—monotherapy (4 mg/kg daily for three consecutive days)) [49] | 615 * | 302 (172–459) | RR 0.49 (0.28–0.75) | 800/7 RCTS | Moderate c |
| Adverse events, minor (praziquantel 40 mg/kg single dose) [49] | None | None | Not estimable | 1591/9 RCTs | Low d |
- a
- The risk in the intervention group per 1000 persons treated (95% CI) was based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
- b
- Treatment of only Sc. mansoni infections reported.
- c
- Downgraded by 1 for indirectness: only two trials from limited settings evaluated this comparison.
- d
- The trial was under-powered; downgraded by 1.
- e
- Only a single trial reported this outcome.
- f
- Publication bias was unclear.
| Outcomes | Anticipated Absolute Effects (95% CI) | Relative Chance of Cure (95% CI) b | Number of Participants/Studies | Certainty of the Evidence (GRADE) | |
|---|---|---|---|---|---|
| Cure with Comparator Drug per 1000 a | Cure with Intervention Drug—Ivermectin (200 mg/kg) b | ||||
| Cure overall assessed at 5 weeks—albendazole [52] | 480 | 840 (720–980) | RR 1.79 (1.55–2.08) | 478/4 RCTs | Moderate d |
| Adverse events assessed at 5 weeks—albendazole [52] | 260 | 210 (150–290) | RR 0.80 (0.59–1.09) | 518/4 RCTs | Low c,g |
| Cure overall assessed at 11 weeks—thiabendazole [52] | 690 | 740 (660–820) | RR 1.07 (0.96–1.20) | 467/3 RCTs | Moderate e |
| Adverse events assessed at 11 weeks—thiabendazole [52] | 730 | 230 (150–360) | RR 0.31 (0.20–0.50) | 507/3 RCTs | Moderate f |
- a
- Albendazole or thiabendazole.
- b
- The risk in the intervention group per 1000 persons treated (95% CI) was based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI).
- c
- No method of allocation concealment in two trials and no method of allocation described.
- d
- Two trials did not conceal allocation and no method of allocation was described.
- e
- Two trials did not conceal allocation and no method of allocation was described in one trial.
- f
- Two trials did not conceal allocation and no method of allocation was described.
- g
- Wide range of estimates in three trials could include substantive fewer events.
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agbata, E.N.; Morton, R.L.; Bisoffi, Z.; Bottieau, E.; Greenaway, C.; Biggs, B.-A.; Montero, N.; Tran, A.; Rowbotham, N.; Arevalo-Rodriguez, I.; et al. Effectiveness of Screening and Treatment Approaches for Schistosomiasis and Strongyloidiasis in Newly-Arrived Migrants from Endemic Countries in the EU/EEA: A Systematic Review. Int. J. Environ. Res. Public Health 2019, 16, 11. https://doi.org/10.3390/ijerph16010011
Agbata EN, Morton RL, Bisoffi Z, Bottieau E, Greenaway C, Biggs B-A, Montero N, Tran A, Rowbotham N, Arevalo-Rodriguez I, et al. Effectiveness of Screening and Treatment Approaches for Schistosomiasis and Strongyloidiasis in Newly-Arrived Migrants from Endemic Countries in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health. 2019; 16(1):11. https://doi.org/10.3390/ijerph16010011
Chicago/Turabian StyleAgbata, Eric N., Rachael L. Morton, Zeno Bisoffi, Emmanuel Bottieau, Christina Greenaway, Beverley-A. Biggs, Nadia Montero, Anh Tran, Nick Rowbotham, Ingrid Arevalo-Rodriguez, and et al. 2019. "Effectiveness of Screening and Treatment Approaches for Schistosomiasis and Strongyloidiasis in Newly-Arrived Migrants from Endemic Countries in the EU/EEA: A Systematic Review" International Journal of Environmental Research and Public Health 16, no. 1: 11. https://doi.org/10.3390/ijerph16010011
APA StyleAgbata, E. N., Morton, R. L., Bisoffi, Z., Bottieau, E., Greenaway, C., Biggs, B.-A., Montero, N., Tran, A., Rowbotham, N., Arevalo-Rodriguez, I., Myran, D. T., Noori, T., Alonso-Coello, P., Pottie, K., & Requena-Méndez, A. (2019). Effectiveness of Screening and Treatment Approaches for Schistosomiasis and Strongyloidiasis in Newly-Arrived Migrants from Endemic Countries in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health, 16(1), 11. https://doi.org/10.3390/ijerph16010011






