The Effectiveness and Cost-Effectiveness of Hepatitis C Screening for Migrants in the EU/EEA: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Overall Approach and Key Questions
- What is the effectiveness of screening migrants arriving and living in the EU/EEA for HCV?
- What is the cost, resource utilization, and cost-effectiveness for screening migrants for HCV?
2.2. Search Strategy and Selection Criteria
2.3. Study Selection and Quality Assessment
2.4. Data Extraction and Synthesis
3. Results
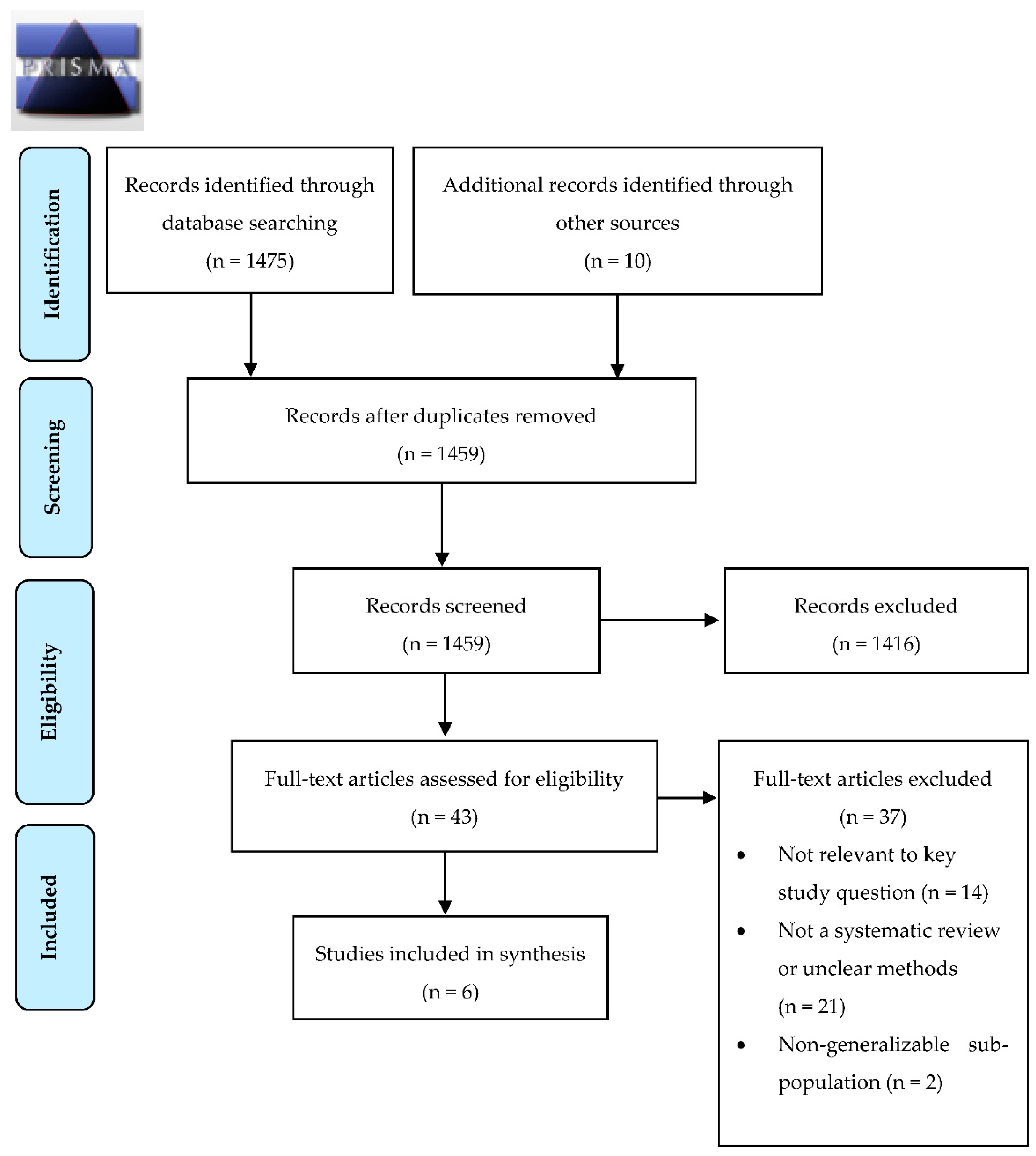
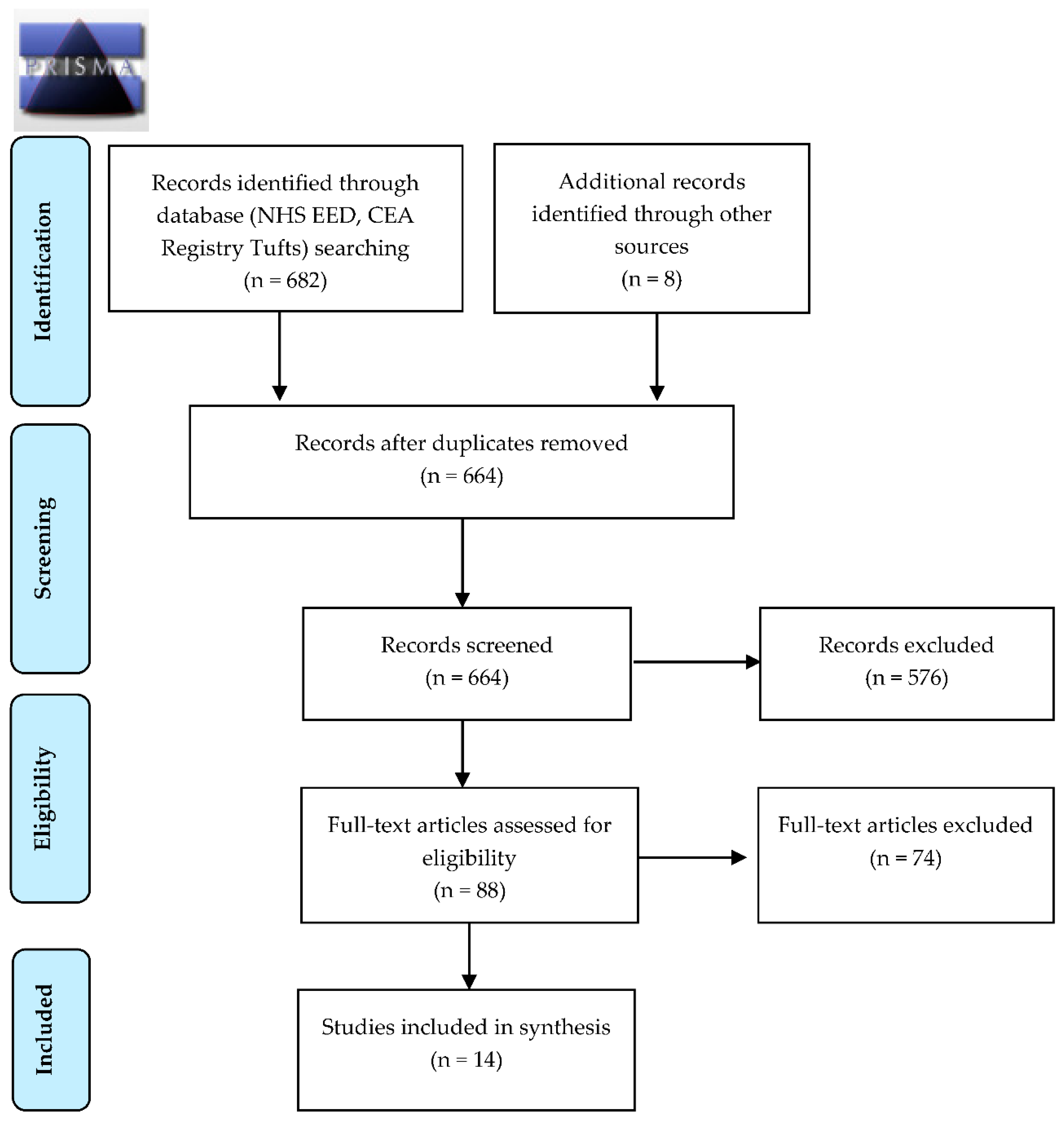
3.1. Search Results
3.2. Performance of Diagnostic Tests
3.3. Impact of Therapy on Long-Term Outcomes
3.4. The HCV Care Continuum and Pathway
3.5. Resource Use, Costs and Cost-Effectiveness
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- European Centre for Disease Prevention and Control. Systematic Review on Hepatitis B and C Prevalence in the EU/EEA; ECDC: Stockholm, Sweden, 2016.
- Razavi, H.; Robbins, S.; Zeuzem, S.; Negro, F.; Buti, M.; Duberg, A.S.; Roudot-Thoraval, F.; Craxi, A.; Manns, M.; Marinho, R.T.; et al. Hepatitis C virus prevalence and level of intervention required to achieve the WHO targets for elimination in the European Union by 2030: A modelling study. Lancet Gastroenterol. Hepatol. 2017, 2, 325–336. [Google Scholar] [CrossRef]
- Mühlberger, N.; Schwarzer, R.; Lettmeier, B.; Sroczynski, G.; Zeuzem, S.; Siebert, U. HCV-related burden of disease in Europe: A systematic assessment of incidence, prevalence, morbidity, and mortality. BMC Public Health 2009, 9, 34. [Google Scholar] [CrossRef] [PubMed]
- Mathurin, P. HCV burden in Europe and the possible impact of current treatment. Dig. Liver Dis. 2013, 45 (Suppl. 5), S314–S317. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, A.C.; Wallace, C.; Klimack, W.K.; Razavi, H. Economic burden of hepatitis C-associated diseases: Europe, Asia Pacific, and the Americas. J. Med. Econ. 2012, 15, 887–896. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines on Hepatitis B and C Testing; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Falade-Nwulia, O.; Suarez-Cuervo, C.; Nelson, D.R.; Fried, M.W.; Segal, J.B.; Sulkowski, M.S. Oral direct-acting agent therapy for hepatitis c virus infection: A systematic review. Ann. Intern. Med. 2017, 166, 637–648. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Recommendations on Treatment of Hepatitis C 2018. J. Hepatol. 2018, 69, 461–511. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Combating Hepatitis B and C to Reach Elimination by 2030; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- World Health Organization Regional Office for Europe. Action Plan for the Health Sector Response to Viral Hepatitis in the WHO European Region; WHO/Europe: Copenhagen, Denmark, 2016. [Google Scholar]
- World Health Organisation. Global Health Sector Strategy on Viral Hepatitis 2016–2021. Towards Ending Viral Hepatitis; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- Falla, A.M.; Ahmad, A.A.; Duffell, E.; Noori, T.; Veldhuijzen, I.K. Estimating the scale of chronic hepatitis C virus infection in the EU/EEA: A focus on migrants from anti-HCV endemic countries. BMC Infect. Dis. 2018, 18, 42. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Epidemiological Assessment of Hepatitis B and C among Migrants in the EU/EEA; ECDC: Stockhlom, Sweden, 2016.
- Pepin, J.; Abou Chakra, C.; Pepin, E.; Nault, V.; Valiquette, L. Evolution of the global burden of viral infections from unsafe medical injections, 2000–2010. PLoS ONE 2014, 9, e99677. [Google Scholar] [CrossRef] [PubMed]
- Greenaway, C.; Thu Ma, A.; Kloda, L.A.; Klein, M.; Cnossen, S.; Schwarzer, G.; Shrier, I. The Seroprevalence of Hepatitis C Antibodies in Immigrants and Refugees from Intermediate and High Endemic Countries: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0141715. [Google Scholar]
- Seedat, F.; Hargreaves, S.; Nellums, L.B.; Ouyang, J.; Brown, M.; Friedland, J.S. How effective are approaches to migrant screening for infectious diseases in Europe? A systematic review. Lancet Infect. Dis. 2018. [Google Scholar] [CrossRef]
- Chen, W.; Tomlinson, G.; Krahn, M.; Heathcote, J. Immigrant patients with chronic hepatitis C and advanced fibrosis have a higher risk of hepatocellular carcinoma. J. Viral. Hepat. 2012, 19, 574–580. [Google Scholar] [CrossRef] [PubMed]
- Pottie, K.; Mayhew, A.; Morton, R.; Greenaway, C.; Akl, E.A.; Rahman, P.; Zenner, D.; Pareek, M.; Tugwell, P.; Welch, V.; et al. Prevention and assessment of infectious diseases among children and adult migrants arriving to the European Union/European Economic Association: A protocol for a suite of systematic reviews for public health and health systems. BMJ Open 2017, 7, e014608. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Woolf, S.; Schunemann, H.J.; Eccles, M.P.; Grimshaw, J.M.; Shekelle, P. Developing clinical practice guidelines: Types of evidence and outcomes; values and economics, synthesis, grading, and presentation and deriving recommendations. Implement. Sci. 2012, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Owens, D.K.; Whitlock, E.P.; Henderson, J.; Pignone, M.P.; Krist, A.H.; Bibbins-Domingo, K.; Curry, S.J.; Davidson, K.W.; Ebell, M.; Gillman, M.W.; et al. Use of Decision Models in the Development of Evidence-Based Clinical Preventive Services Recommendations: Methods of the U.S. Preventive Services Task Force. Ann. Intern. Med. 2016, 165, 501–508. [Google Scholar] [CrossRef] [PubMed]
- Shea, B.J.; Hamel, C.; Wells, G.A.; Bouter, L.M.; Kristjansson, E.; Grimshaw, J.; Henry, D.A.; Boers, M. AMSTAR is a reliable and valid measurement tool to assess the methodological quality of systematic reviews. J. Clin. Epidemiol. 2009, 62, 1013–1020. [Google Scholar] [CrossRef] [PubMed]
- Guyatt, G.; Oxman, A.D.; Akl, E.A.; Kunz, R.; Vist, G.; Brozek, J.; Norris, S.; Falck-Ytter, Y.; Glasziou, P.; Jaeschke, R. GRADE guidelines: 1. Introduction—GRADE evidence profiles and summary of findings tables. J. Clin. Epidemiol. 2011, 64, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.F.; Sculpher, M.J.; Claxton, K.; Stoddart, G.L.; Torrance, G.W. Methods for the Economic Evaluation of Health Care Programmes, 2nd ed.; Oxford University Press: New York, NY, USA, 1997; p. 396. [Google Scholar]
- Alonso-Coello, P.; Oxman, A.D.; Moberg, J.; Brignardello-Petersen, R.; Akl, E.A.; Davoli, A.; Treweek, S.; Mustafa, R.; Vandvik, P.; Meerpohl, J.; et al. GRADE Evidence to Decision (EtD) frameworks: A systematic and transparent approach to making well informed healthcare choices. 2: Clinical practice guidelines. Br. Med. J. 2016, 353, i2089. [Google Scholar] [CrossRef] [PubMed]
- Khuroo, M.S.; Khuroo, N.S.; Khuroo, M.S. Diagnostic accuracy of point-of-care tests for hepatitis C virus infection: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0121450. [Google Scholar] [CrossRef] [PubMed]
- Kimer, N.; Dahl, E.K.; Gluud, L.L.; Krag, A. Antiviral therapy for prevention of hepatocellular carcinoma in chronic hepatitis C: Systematic review and meta-analysis of randomised controlled trials. BMJ Open 2012, 2, e001313. [Google Scholar] [CrossRef] [PubMed]
- Simmons, B.; Saleem, J.; Heath, K.; Cooke, G.S.; Hill, A. Long-Term Treatment Outcomes of Patients Infected With Hepatitis C Virus: A Systematic Review and Meta-analysis of the Survival Benefit of Achieving a Sustained Virological Response. Clin. Infect. Dis. 2015, 61, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Public Health Agency of Canada. Treatment for Hepatitis C Virus: A Systematic Review and Meta-Analysis; Canadian Preventative Task Force: Ottawa, ON, Canada, 2016.
- Yehia, B.R.; Schranz, A.J.; Umscheid, C.A.; Lo Re, V., III. The treatment cascade for chronic hepatitis C virus infection in the United States: A systematic review and meta-analysis. PLoS ONE 2014, 9, e101554. [Google Scholar] [CrossRef] [PubMed]
- Deuffic-Burban, S.; Obach, D.; Canva, V.; Pol, S.; Roudot-Thoraval, F.; Dhumeaux, D.; Mathurin, P.; Yazdanpanah, Y. Cost-effectiveness and budget impact of interferon-free direct-acting antiviral-based regimens for hepatitis C treatment: The French case. J. Viral Hepat. 2016, 23, 767–779. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.W.; Tu, H.-A.; Feld, J.J.; Wong, T.; Krahn, M. Cost-effectiveness of screening for hepatitis C in Canada. CMAJ 2015, 187, E110–E121. [Google Scholar] [CrossRef] [PubMed]
- Najafzadeh, M.; Andersson, K.; Shrank, W.H.; Krumme, A.A.; Matlin, O.S.; Brennan, T.; Avorn, J.; Choudhry, N.K. Cost-effectiveness of novel regimens for the treatment of hepatitis C virus. Ann. Intern. Med. 2015, 162, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Chhatwal, J.; Kanwal, F.; Roberts, M.S.; Dunn, M.A. Cost-effectiveness and budget impact of hepatitis C virus treatment with sofosbuvir and ledipasvir in the United States. Ann. Intern. Med. 2015, 162, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.M.; Park, H.; Saab, S.; Ahmed, A.; Dieterich, D.; Gordon, S.C. Cost-effectiveness of all-oral ledipasvir/sofosbuvir regimens in patients with chronic hepatitis C virus genotype 1 infection. Aliment. Pharmacol. Ther. 2015, 41, 544–563. [Google Scholar] [CrossRef] [PubMed]
- Rein, D.B.; Wittenborn, J.S.; Smith, B.D.; Liffmann, D.K.; Ward, J.W. The cost-effectiveness, health benefits, and financial costs of new antiviral treatments for hepatitis C virus. Clin. Infect. Dis. 2015, 61, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Hagan, L.M.; Sulkowski, M.S.; Schinazi, R.F. Cost analysis of sofosbuvir/ribavirin versus sofosbuvir/simeprevir for genotype 1 hepatitis C virus in interferon-ineligible/intolerant individuals. Hepatology 2014, 60, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Saab, S.; Gordon, S.C.; Park, H.; Sulkowski, M.; Ahmed, A.; Younossi, Z. Cost-effectiveness analysis of sofosbuvir plus peginterferon/ribavirin in the treatment of chronic hepatitis C virus genotype 1 infection. Aliment. Pharmacol. Ther. 2014, 40, 657–675. [Google Scholar] [CrossRef] [PubMed]
- Linas, B.P.; Barter, D.M.; Morgan, J.R.; Pho, M.T.; Leff, J.A.; Schackman, B.R.; Horsburgh, C.R.; Assoumou, S.A.; Salomon, J.A.; Weinstein, M.C.; et al. The cost-effectiveness of sofosbuvir-based regimens for treatment of hepatitis c virus genotype 2 or 3 infection. Ann. Intern. Med. 2015, 162, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Leidner, A.J.; Chesson, H.W.; Xu, F.; Ward, J.W.; Spradling, P.R.; Holmberg, S.D. Cost-effectiveness of hepatitis C treatment for patients in early stages of liver disease. Hepatology 2015, 61, 1860–1869. [Google Scholar] [CrossRef] [PubMed]
- Brett-Major, D.M.; Frick, K.D.; Malia, J.A.; Hakre, S.; Okulicz, J.F.; Beckett, C.G.; Jagodinski, L.L.; Forgione, M.A.; Gould, P.L.; Harrison, S.A.; et al. Costs and consequences: Hepatitis C seroprevalence in the military and its impact on potential screening strategies. Hepatology 2016, 63, 398–407. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Li, K.; Roberts, M.S.; Spaulding, A.C.; Ayer, T.; Grefenstette, J.J.; Chhatwal, J. Prevention of Hepatitis C by Screening and Treatment in U.S. Prisons. Ann. Intern. Med. 2016, 164, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Orkin, C.; Flanagan, S.; Wallis, E.; Ireland, G.; Dhairyawan, R.; Fox, J.; Nandwani, R.; O’Connell, R.; Lascar, M.; Bulman, J.; et al. Incorporating HIV/hepatitis B virus/hepatitis C virus combined testing into routine blood tests in nine UK Emergency Departments: The “Going Viral” campaign. HIV Med. 2016, 17, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Selvapatt, N.; Ward, T.; Bailey, H.; Bennett, H.; Thorne, C.; See, L.M.; Tudor-Williams, G.; Thursz, M.; McEwan, P.; Brown, A. Is antenatal screening for hepatitis C virus cost-effective? A decade’s experience at a London centre. J. Hepatol. 2015, 63, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Ryen, L.; Svensson, M. The Willingness to Pay for a Quality Adjusted Life Year: A Review of the Empirical Literature. Health Econ. 2015, 24, 1289–1301. [Google Scholar] [CrossRef] [PubMed]
- Backus, L.I.; Belperio, P.S.; Shahoumian, T.A.; Mole, L.A. Direct-acting antiviral sustained virologic response: Impact on mortality in patients without advanced liver disease. Hepatology 2018. [Google Scholar] [CrossRef] [PubMed]
- Owiti, J.A.; Greenhalgh, T.; Sweeney, L.; Foster, G.R.; Bhui, K.S. Illness perceptions and explanatory models of viral hepatitis B & C among immigrants and refugees: A narrative systematic review. BMC Public Health 2015, 15, 151. [Google Scholar]
- Greenaway, C.; Azoulay, L.; Allard, R.; Cox, J.; Tran, V.A.; Abou Chakra, C.N.; Steele, R.; Klein, M. A population-based study of chronic hepatitis C in immigrants and non-immigrants in Quebec, Canada. BMC Infect. Dis. 2017, 17, 140. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.; Nguyen, M. Systematic review: Asian patients with chronic hepatitis C infection. Aliment. Pharmacol. Ther. 2013, 37, 921–936. [Google Scholar] [CrossRef] [PubMed]
- Tiittala, P.; Ristola, M.; Liitsola, K.; Ollgren, J.; Koponen, P.; Surcel, H.M.; Hiltunen-Back, E.; Davidkin, I.; Kivela, P. Missed hepatitis b/c or syphilis diagnosis among Kurdish, Russian, and Somali origin migrants in Finland: Linking a population-based survey to the national infectious disease register. BMC Infect. Dis. 2018, 18, 137. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.W.L.; Erman, A.; Feld, J.J.; Krahn, M. Model-based projection of health and economic effects of screening for hepatitis C in Canada. CMAJ Open 2017, 5, E662–E672. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Hepatitis B and C Testing Activities, Needs, and Priorities in the EU/EEA; ECDC: Stockholm, Sweden, 2017.
- Greenaway, C.; Makarenko, I.; Tanveer, F.; Janjua, N. Addressing Hepatitis C in the Foreign-Born Population: A key to HCV Elimination in Canada. Can. Liver J. 2018, 1, 34–50. [Google Scholar] [CrossRef]
- Noret, I.; Frydryszak, D.; Macherey, A.; Simonnot, N. European Network to Reduce Vulnerabilities in Health. Available online: https://mdmeuroblog.wordpress.com/about/ (accessed on 14 September 2018).
- Institute of Medicine Committee on the Prevention Control of Viral Hepatitis Infection. Hepatitis and Liver Cancer: A National Strategy for Prevention and Control of Hepatitis B and C; Colvin, H.M., Mitchell, A.E., Eds.; National Academies Press (US), National Academy of Sciences: Washington, DC, USA, 2010.
- Ferrante, J.M.; Winston, D.G.; Chen, P.H.; de la Torre, A.N. Family physicians’ knowledge and screening of chronic hepatitis and liver cancer. Fam. Med. 2008, 40, 345–351. [Google Scholar] [PubMed]
- Blondell, S.J.; Kitter, B.; Griffin, M.P.; Durham, J. Barriers and Facilitators to HIV Testing in Migrants in High-Income Countries: A Systematic Review. AIDS Behav. 2015, 19, 2012–2024. [Google Scholar] [CrossRef] [PubMed]
- Pareek, M.; Noori, T.; Hargreaves, S.; van den Muijsenbergh, M. Linkage to Care Is Important and Necessary When Identifying Infections in Migrants. Int. J. Environ. Res. Public Health 2018, 15, 1550. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.; Manzanares, S.; Jacques, C.; Caylá, J.; Kunkel, J.; Foster, G. Screening for Chronic viral Hepatitis in Migrant Populations—Report on Four HEPscreen Pilot Studies. Available online: http://hepscreen.eu/wp-content/uploads/2014/12/HEPscreen_Final-WP6-report_Pilot-studies.pdf (accessed on 14 September 2018).
- Jafferbhoy, H.; Miller, M.H.; McIntyre, P.; Dillon, J.F. The effectiveness of outreach testing for hepatitis C in an immigrant Pakistani population. Epidemiol. Infect. 2012, 140, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Perumalswami, P.; DiClemente, F.M.; Kapelusznik, L.; Pan, C.; Chang, C.; Friedman, S.L.; Vachon, M.-L.C.; Standen, M.; Khaitova, V.; Factor, S.H.; et al. Hepatitis outreach network (HONE): HBV and HCV screening of ethnic urban populations of New York city with linkage to care. In Hepatology (Baltimore, Md.); John Wiley and Sons Inc.: New York, NY, USA, 2011; Volume 54, pp. 594A–595A. [Google Scholar]
- Perumalswami, P.V.; DeWolfe Miller, F.; Orabee, H.; Regab, A.; Adams, M.; Kapelusznik, L.; Aljibawi, F.; Pagano, W.; Tong, V.; Dieterich, D.T.; et al. Hepatitis C screening beyond CDC guidelines in an Egyptian immigrant community. Liver Int. Off. J. Int. Assoc. Stud. Liver 2014, 34, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Perumalswami, P.V.; Factor, S.H.; Kapelusznik, L.; Friedman, S.L.; Pan, C.Q.; Chang, C.; Di Clemente, F.; Dieterich, D.T. Hepatitis Outreach Network: A practical strategy for hepatitis screening with linkage to care in foreign-born communities. J. Hepatol. 2013, 58, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Zuure, F.R.; Bouman, J.; Martens, M.; Vanhommerig, J.W.; Urbanus, A.T.; Davidovich, U.; van Houdt, R.; Speksnijder, A.G.C.L.; Weegink, C.J.; van den Hoek, A.; et al. Screening for hepatitis B and C in first-generation Egyptian migrants living in the Netherlands. Liver Int. 2013, 33, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Bottero, J.; Boyd, A.; Gozlan, J.; Carrat, F.; Nau, J.; Pauti, M.-D.; Rougier, H.; Girard, P.-M.; Lacombe, K. Simultaneous Human Immunodeficiency Virus-Hepatitis B-Hepatitis C Point-of-Care Tests Improve Outcomes in Linkage-to-Care: Results of a Randomized Control Trial in Persons Without Healthcare Coverage. Open Forum Infect. Dis. 2015, 2, ofv162. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.D.; Cunningham, E.B.; Nielsen, S.; Aghemo, A.; Alho, H.; Backmund, M.; Bruggmann, P.; Dalgard, O.; Seguin-Devaux, C.; Flisiak, R.; et al. Restrictions for reimbursement of interferon-free direct-acting antiviral drugs for HCV infection in Europe. Lancet Gastroenterol. Hepatol. 2018, 3, 125–133. [Google Scholar] [CrossRef]
- Marshall, A.D.; Pawlotsky, J.M.; Lazarus, J.V.; Aghemo, A.; Dore, G.J.; Grebely, J. The removal of DAA restrictions in Europe—One step closer to eliminating HCV as a major public health threat. J. Hepatol. 2018, in press. [Google Scholar] [CrossRef] [PubMed]
- Duffell, E.; Hedrich, D.; Mardh, O.; Mozalevskis, A. Towards Elimination of hepatitsi B and C in European Union and European Economic Area Countries: Monitiroing the World Health Organization’s global health sector strategy core indicators and scaling up key interventions. Eurosurveillance 2017, 22, 30476. [Google Scholar] [CrossRef] [PubMed]
| Study | Quality of Systematic Review/GRADE Certainty of Evidence | Design | Population | Intervention/Outcomes | Results |
|---|---|---|---|---|---|
| Khuroo 2015 [26] | Quality of systematic Review AMSTAR: 8/11 GRADE Certainty of Evidence Very Low | Systematic Review Up to March 2012. N = 30 studies: 25—full-text article 2—WHO reports 1—WHO draft report 2—letters to editor | Adults > 18 years 17,151 participants 16 studies were conducted in low/middle income countries (India n = 4, Brazil n = 2, Cameroon n = 2, China n = 2, Egypt n = 1, Malawi n = 1, South Korea n = 2, Thailand n = 1, and Zimbabwe n = 1) 14 studies conducted in high income countries (United States n = 8, Germany n = 1, Italy n = 1, Spain n = 1) (Report for WHO n = 3) | Intervention: Point-of-care test: any commercially available assay at or near the site of patient care with <30 min turn-around time. Reference standard: third generation EIA, microenzyme immunoassay, CIA, RIBA, NAT Outcome: Sensitivity, specificity, LR+/−, Diagnostic OR (95% CI) | Pooled sensitivity: 97.4% (95% CI = 95.9–98.4) specificity: 99.5% (99.2–99.7) +LR: 80.17 (55.35–116.14) −LR: 0.03 (0.02–0.04) Diagnostic OR: 3032.85 (1595.86–5763.78) OraQuick test had the highest sensitivity and specificity: sensitivity: 99.5 (98.9–99.8) specificity: 99.8 (99.6–99.9) |
| Kimer 2012 [27] | Quality of systematic Review AMSTAR: 7/11 GRADE Certainty of Evidence Very Low | Systematic Review Up to 2012 N = eight RCTs, five prospective cohorts | RCTs conducted in France, Italy, Spain, Japan, and USA in patients with HCV-related cirrhosis or fibrosis and treated with antiviral therapy 1156 patients with therapy 1074 controls Prospective cohorts: Patients with HCV-related cirrhosis | Intervention: Antiviral therapy (PR, IFN, PEG-IFN) Outcome: RR, 95% CI of HCC development; number needed to treat to prevent 1 case of HCC = 1/risk difference overall mortality liver-related mortality liver-related morbidity | Risk of HCC among received antiviral therapy vs. did not receive: Absolute number of HCC: 81/1156 vs. 129/1174 RR (95% CI): 0.53 (0.34–0.81) SVR and non-SVR compared to no therapy—RR (95% CI): SVR: RR = 0.15 (0.05–0.45) Non-SVR: RR = 0.57 (0.37–0.85) Number needed to treat to prevent one case of HCC: eight patients. |
| Simmons 2015 [28] | Quality of systematic Review AMSTAR: 8/11 GRADE Certainty of Evidence Very low to low | Systematic Review 1990–2014 N = 31 studies: General population: 17 studies; Cirrhotic: nine studies; HIV co-infected: five studies | Adults (>18 years old) chronically infected with HCV of any genotype treated with any antiviral regimen stratified into 3 groups:
General population: 28,398 Cirrhotic: 2604 HIV co-infected: 2358 | Intervention: PR; IFN; PEG-IFN; IFN-beta Outcome: all-cause mortality; pooled adjusted HR (95% CI); pooled estimates for the 5-year mortality | Mortality of achieved SVR vs. non-SVR, aHR (95% CI): General population: 0.50 (0.37–0.67) Cirrhotic group: 0.26 (0.18–0.74) HIV co–infected group: 0.21 (0.10–0.45) Pooled 5-year mortality rates for SVR vs. non–SVR, IR (95% CI): General population: 1.98 (1.00–3.45) vs. 7.75 (5.86–10.98) Cirrhotic group: 4.90 (3.45–7.28) vs. 15.88 (11.44–21.80) Co–infected group: 1.49 (0.50–2.96) vs. 11.44 (6.33–19.3) |
| Public Health Agency of Canada, Canadian Task Force on Preventative Health Care 2016 [29] | Quality of systematic Review AMSTAR: 11/11 GRADE Certainty of Evidence Very low to moderate | Systematic Review Up to November 2015 N = Benefits of treatment: 11 studies; Harms of treatment: 7 | Treatment-naïve nonpregnant HIV/HBV negative adults Wide range of fibrosis scores +80% noncirrhotic RCTs (n = 7) 6/7 RCTs all patients were Genotype 1 2431 participants ranged from 121 to 499 participants in a study. Recruitment sites included: United States, Australia, Austria, Belgium, Canada, Denmark, France, Germany, New Zealand, Norway, Poland, Russia, Spain, Japan, Italy, Mexico, Puerto Rico, Romania, Ukraine, United Kingdom, Sweden, the Netherlands, Bulgaria, Portugal, Slovakia, China, and the Republic of Korea. | Intervention: DAA-based vs. PR regimens. DDA therapies included those that were approved at the time of the study and those anticipated to be approved by February 2016 for all HCV genotypes. Outcomes: All-cause mortality; hepatic mortality; hepatic decompensation; hepatocellular carcinoma; need for liver transplantation. | Hepatic mortality: 60 fewer/1000 (95% CI 59–62) Hepatocellular carcinoma: 18 fewer/1000 (17–19) Decompensated cirrhosis: 46 fewer/1000 (46–47) Need for liver transplantation: 4 fewer/1000 (4–6) In cirrhotic individuals DAA–based regimens compared to PR resulted in 30 fewer/1000 people affected by hepatic mortality. |
| Yehia 2014 [30] | Quality of systematic Review AMSTAR: 3/11 Data quality not formally assessed | Systematic Review 2003–2013 N = 10 studies | Only studies from the US that collected data after 2000 were included. Studies of the general population excluded those with only a single study site, exclusively focused on specific populations (e.g., only immigrants, injection drug users, those with HIV/HCV co-infection) Study subjects for each question Chronic infection: 15,079 Diagnosed/Aware: 203 Access to Care: 101 HCV RNA confirmed: 8810 Liver biopsy: 180,703 Prescribed HCV treatment: 46,452 Achieved SVR: 18,105 | Examined data addressing seven key steps along the HCV care and treatment cascade | Care/Treatment cascade: 100% Chronic HCV infected (3,500,000) 50% Diagnosed and aware 43% Access to outpatient care 27% HCV RNA confirmed 17% Underwent liver biopsy 16% Prescribed HCV treatment 9% Achieved SVR |
| Study | Quality/Certainty of Economic Evidence | Design/Population | Intervention(s) | Cost-Effectiveness (ICER or INB) | Resource Requirements |
|---|---|---|---|---|---|
| Cost-effectiveness of HCV Screening and DAA therapy | |||||
| Brett-Major 2016 [41] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs, HCV rates, and ranges were provided. Threshold sensitivity analysis undertaken. PSA not performed. Justification for choice of ranges was provided for all parameters. Cost offsets (and net savings) rather than cost-effectiveness was reported | Design: Decision-analytic costing model; results reported in US dollars Population: Applicants to US military service HCV prevalence 0.48–0.98/1000 | Three strategies:
| Not applicable (Costing study) | High costs. With no screening, the cost to the Department of Defence of treating the estimated 93 cases of chronic HCV cases from a single year’s accession cohort was $9.3 million [€7,293,134]. Screening with the HCV antibody test followed by the nucleic acid test for confirmation yielded a net annual savings and a $3.1 million dollar [€2,431,044] advantage over not screening. |
| He 2016 [42] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA not performed. Justification for choice of ranges was not provided for all parameters. Cost-effectiveness results were sensitive to the time horizon. | Design: Dynamic microsimulation model of transmission/progression of HCV, and cost-effectiveness and budget impact analysis; results reported in US dollars Population: Population in US prisons HCV prevalence 25% and 50% undiagnosed | Three strategies:
| ICER ($/QALY gained): 1 year risk-based vs. no screening: $19,635 [€15,552] 1 year universal vs. no screening: $20,571 [€16,293] 5 year universal vs. no screening: $24,046 [€19,046] 10 year universal vs. no screening: $29,234 [€23,155] | Low to moderate costs. Screening cost per 2 million prisoners: 1year risk-based vs. no screening: +$37M [€29M] 1year universal vs. no screening: +$107M [€84M] 5year universal vs. no screening: +$178M [€140M] 10year universal vs. no screening: +$249M [€197M] Treatment cost per 2 million prisoners: No screening: $59,035M [€46,759M] 1year risk-based vs. no screening: +$816M [€646M] 1year universal vs. no screening: +$1480M [€1172M] 5year universal vs. no screening: +$1951M [€1545M] 10year universal vs. no screening: +$2190M [€1734M] |
| Orkin 2016 [43] | Certainty of evidence: low Allowance was not made for uncertainty in the estimates of costs and consequences. No source for unit prices (costs) was given. PSA not performed. Justification for choice of ranges was not provided for all parameters. Cost-effectiveness results were not reported. | Design: Prospective 1 week-long snapshot observational study with assumed costs for testing and treating; results reported in British pounds Population: People visiting emergency departments in the UK HCV prevalence 1.84% | One strategy: Routine combined HIV, HCV, and HBV testing | Not applicable | Low to moderate costs. Assuming the cost per diagnosis is £7 [€8], the cost per new case detected would be £988 [€1109] for HCV, £1351 [€1517] for HBV. and £2478 [€2783] for HIV. |
| Rein 2015 [36] | Certainty of evidence: high Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were sensitive to treatment cost, SVR probability, QALY post SVR, fibrosis rate | Design: Monte Carlo simulation model; results presented in US dollars. Population: General population aged ≥20, and patients with chronic HCV genotype 1, 2, 3, and 4 in US HCV prevalence rate: varies by birth decade, race, and sex Heavy alcoholics 0.089 HIV+ 0.02 | Screening followed by treatment Five strategies:
| Genotype ¼—ICER ($/QALY): PR vs. no treatment: $59,792 [€47,359] PR extensively dominated by PI+PR PI + PR vs. no treatment:$43,530 [€34,478] SOF+PR vs. PI+PR: $47,237 [€37,414] SOF+SIM vs. SOF+PR: $72,169 [€57,162] | Treatment costs: Genotype 1&4: PR: $61,224 [€48,493] PI+PR: $78,812 [€62,424] SOF+PR: $99,306 [€78,656] SOF+SIM: $150,360 [€119,094] Genotype 2: PR: $30,612 [€24,246] SOF+RBV: $88,158 [€69,826] Genotype 3: PR: $30,612 [€24,246] SOF+RBV: $176,316 [€139,653] Other costs: Testing: antibody: $25 [€19] RNA: $59 [€45] Post-diagnostic evaluation: if coordinated with treatment: $832 [€658] if not treated: $869 [€688] |
| Selvapatt 2015 [44] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA not performed. Justification for choice of ranges was not provided for all parameters. Cost-effectiveness results were sensitive to the prevalence of HCV infection among the screened women and the proportion of identified women treated. | Design: Markov cohort simulation model; results reported in British pounds Population: Pregnant women attending antenatal clinics in the UK HCV prevalence 0.38% | Two strategies:
Additional scenarios:
| ICER (£/QALY): Screening + PR vs. no screening + no treatment: £2400 [€2745] (screening + newer direct-acting antiviral regimens vs. no screening + no treatment: £9139 [€10,455]) | Moderate costs. Total costs of screening and confirmation of 44 new diagnoses: £240,641 [€275,299] Cost per newly diagnosed individual: £5469 [€6256] |
| Wong 2015 [32] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were sensitive to rates of chronic HCV infection, seroprevalence, costs (excluding the cost of antiviral therapy), treatment uptake and quality of life (utilities). | Design: Decision-analytic Markov model; results reported in Canadian dollars Population: General Canadian population, 2 age groups: 25–64 and 45–64 years old HCV prevalence assumed is 0.5% | Four strategies:
| ICER (CAN $/QALY) of screening and treatment vs. no screening: Age 25–64 years: PR: $38,117 [€25,502] IFN-free DAA (genotype 1), SOF+RBV (genotype 2/3),or PR (genotype 4/5/6): $34,783 [€23,271] PR+RBV (genotype 1), SOF+RBV (genotype 2/3), or PR (genotype 4/5/6): $42,398 [€28,366] Age 45–64 years: PR: $34,359 [€22,988] SIM+PR (G1), SOF+RBV (G2/3), or PR (G4/5/6): $44,034 [€29,461] IFN-free DAA (G1), SOF+RBV (G2/3), or PR (G4/5/6): $35,562 [€23,793] | Moderate to high costs: CAN$70,000 [€46,834] $84,000 [€56,201]/person. Costs of antiviral therapies (CAN $): SIM-based: 24 weeks: $46,157 [€30,881]; 48 wk: $55,811 [€37,340] SOF-based: 12 wk: $55,000 [€36,798] ABT-based: 48 wk: $19,948 [€13,346]; 24 wk: $9974 [€6673] Costs of adverse events (weekly): anemia: $107 [€71], depression: $73 [€48], pruritus: $12 [€8], rash: $12 [€8] HCV-tests: anti-HCV: $14 [€9], HCV RNA: $100 [€66] For age 25–64 years, No screening: $71,327 [€47,722]; Screen and treat: PR: $71,450 [€47,804] IFN-free DAA (G1), SOF+RBV (G2/3) or PR (G4/5/6): $71,593 [€47,900] SIM+PR (G1), SOF+RBV (G2/3) or PR (G4/5/6): $71,593 [€47,900] For age 45–64: No screening: $83,335 [€55,756] Screen and treat: PR: $83,476 [€55,850] IFN-free DAA (G1), SOF+RBV (G2/3) or PR (G4/5/6): $83,672 [€55,981] SIM+PR (G1), SOF+RBV (G2/3) or PR (G4/5/6): $83,673 [€55,982] |
| Cost-effectiveness of DAA therapy | |||||
| Chhatwal 2015 [34] | Certainty of evidence: high Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were most sensitive to quality of life after successful treatment, cost of SOF, drug efficacy | Design: Decision-analytic Markov model; results reported in US dollars Population: Treatment naïve and treatment-experienced HCV population in US | Two strategies:
| SOF-LDV vs. IFN-based therapy—ICER ($/QALY): Treatment naïve patients: No cirrhosis: $61,517 [€48,725.] Cirrhosis: $20,673 [€16,374] Treatment experienced patients: No cirrhosis: $69,707 [€55,212] Cirrhosis: $92,302 [€73,109] | Treating eligible HCV patient would cost an additional $65 billion [€51.5 billion] over a 5 year period The weekly costs by third-party payer SOF: $7000 [€5544] LDV: $875 [€693] PEG-IFN: $587 [€464] RBV: $309 [€244] BOC: $1100 [€871] TEL: $4100 [€3247] |
| Deuffic-Burban 2016 [31] * | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA not performed. Limited justification for choice of ranges. Cost-effectiveness results were sensitive to the price of new DAAs particularly for treating genotype 1 | Design: Decision-analytic Markov model; results reported in Euros Population: Patients with chronic HCV aged ≥18, aware of their infection, in fibrosis stage F0–F4 or decompensated cirrhosis, treated in France. HCV prevalence | Three strategies:
| Genotype 1: IFN-free was a cost-effective vs. IFN-based: ICER: €40,400 to €88,300/QALY QALY/person: 12.59 vs. 12.11 for IFN-based therapy Genotypes 2 or 3: IFN-based was the most cost-effective: ICER: €21,300/QALY for genotype 2 ICER: €19,400/QALY for genotype 3 Genotype 4: IFN-free regimens was cost-effective: ICER: €23,000 to €58,200/QALY | Moderate to high resource Treating all CHC-screened patients over 5 years would cost: €3.5–7.2 billion Cost of treatment/week: SOF: €3417 OBV/PTV-r: €3259 DCV: €2125 SIM: €1750 TEL: €1042 LDV: €417 BOC: €378 DAV: €284 PEG-IFN: €158 RBV: €55 Costs related to adverse events (cost per event): Severe anaemia: €2564 Severe depression: €1619 Severe rash: €2942 Moderate anaemia: €4200 |
| Hagan 2014 [37] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA not performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were sensitive to SVR rates | Design: Decision-analytic Markov model; results reported in US dollars Population: Chronic HCV genotype 1, in 50 years old in US HCV prevalence 1.6% | Two strategies:
| SOF-SIM dominated SOF-RBV: yielded lower costs and more QALYs SOF-SIM: $165,336 [€133,108] and 14.69 QALYs SOF-RBV: $243,586 [€196,106] and 14.45 QALYs | Costs of drugs per course: 24-weeks SOF/RBV: $169,000 12-weeks SOF/SIM: $150,000 Treatment-associated medical care: SOF/RBV: $2100 (1890–2310) [€1690 (€1521–€1859)] SOF/SIM: $1160 (1044–1276) [€933 (€840–€1027)] |
| Leidner 2015 [40] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA not performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were sensitive to post-treatment quality of life (utilities) and treatment costs. | Design: Decision-analytic Markov model; results reported in US dollars Population: 55-year old patient in US with genotype 1 HCV infection | Two strategies:
| ICER ($/QALY): Patients diagnosed at F0: treatment at F2 vs. F3: $97,900 [€80,102] treatment at F0 vs. F2: $242,900 [€198,741] Patients diagnosed at F1: treatment at F2 vs. F3: $59,500 [€48,683] treatment at F1 vs. F2: $174,100 [€142,449] Patients diagnosed at F2: treatment at F2 vs. F3: $37,300 [€30,518] The threshold of treatment costs: for ICER $50,000/QALY: $20,200 [€16,527] for ICER $100,000/QALY: $42,400 [€34,691] | Moderate to high costs. Larger costs for patients with advanced or end stage liver disease, compared to early stage liver disease. Nontreatment and treatment costs: Patients starting at F0: treatment at F3: $33,600 [€27,491] treatment at F2: $45,000 [€36,819] treatment at F1: $70,800 [€57,928] treatment at F0: $11,100 [€9082] Patients starting at F1: treatment at F3: $59,200 [€48,437] treatment at F2: $77,400 [€63,328] treatment at F1: $113,200 [€92,620] Patients starting at F2: treatment at F3: $91,000 [€74,456] treatment at F2: $113,600 [€92,947] |
| Linas 2015 [39] | Certainty of evidence: high Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were sensitive to cost of SOF | Design: Monte Carlo simulation. Results reported in US dollars Population: Chronic HCV genotype 2 or 3 in the US | Three strategies:
| ICER ($/QALY) Genotype 2 No cirrhosis (naïve): 24 wk PR vs. no therapy: $3000 [€2415] 12 wk SOF-RBV vs. 24 wk PR: $238,000 [€191,609] No cirrhosis (treatment experienced): 12 wk SOF-RBV vs. no therapy: $63,700 [€51,283] 16 wk SOF-RBV vs. 12 wk SOF-RBV: $468,000 [€376,777] Cirrhosis (treatment naïve): 24 wk PR vs. no therapy: $8700 [€7004] 12 wk SOF-RBV vs. 24 wk PR: $35,500 [€28,580] Cirrhosis (treatment experienced): 12 wk SOF-RBV dominated by no therapy SOF-RBV 16 wk vs. 12 wk: $27,300 [€21,978] Genotype 3 No cirrhosis (treatment-naïve): 24 wk PR vs. no treatment: $4800 [€3864] 12 wk SOF-RBV dominated by 24 wk PR 12 wk PR-SOF vs. 24 wk PR: $263,000 [€211,736] 24 wk SOF-RBV vs. 12 wk PR-SOF: $266,000 [€214,151] No cirrhosis (treatment-experienced): 12 wk PR-SOF vs. no treatment: $82,000 [€66,016] 12 wk SOF-RBV and 16 wk SOF-RBV both dominated by 12 wk PR-SOF SOF-RBV 24 wk vs. 16 wk: $1,100,000 [€805,080] Cirrhosis (treatment-naïve): 24 wk PR vs. no treatment: $13,600 [€10,949] 12 wk SOF-RBV dominated by 12 wk PR 12 wk PR-SOF vs. 24 wk PR: $22,600 [€18,194] 24 wk SOF-RBV vs. 12 wk PR-SOF: $107,000 [€86,143] Cirrhosis (treatment-experienced): 12 wk PR-SOF vs. no treatment: $22,300 [€17,953] 12 wk, 16 wk and 24 wk SOF-RBV all dominated by 12 wk PR-SO | Total HCV therapy costs per number of weeks (base case value and range from sensitivity analysis): 24 wk PR: $25,300 (12,800–37,800) [€20,368 (10,305–30,432)] 12 wk SOF–RBV: $91,500 (2000–97,500) [€73,664 (1610–78,495)] 16 wk SOF–RBV: $121,900 (30,000–129,900) [€98,139 (24,152–104,579)] 24 wk SOF–RBV: $182,900 (4900–194,900) [€147,249 (3944–156,910)] 12 wk PR-SOF: $9000 (3000–$105,000) [€7245 (2415–84,533)] |
| Najafzadeh 2015 [33] | Certainty of evidence: high Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was provided for all parameters. Cost-effectiveness results were sensitive to treatment cost | Design: Discrete event simulation Population: Treatment-naive patients infected with chronic HCV genotype 1, 2, or 3 in the US. | Five strategies (genotype 1)
| ICER ($/QALY): Genotype 1 BOC+PR: reference SOF+PR: $21,528 [€17,051] SOF+SIM: $71,445 [€56,589] SOF+DCV: $63,355 [€50,181] SOF+LDV: $12,825 [€10,158] Genotype 2: PR: reference SOF+RBV: $110,168 [€87,260] SOF+DCV: $691,574 [€48,770] Genotype 3: PR: reference SOF+RBV: dominated by PR SOF+DCV: $396,229 [€313,839] SOF+LDV+RBV: $73,236 [€58,007] | Drug costs: Genotype 1: BOC+PR: $100,926 [€79,940] SOF+PR: $120,648 [€95,561] SOF+SIM: $171,023 [€135,461] SOF+DCV: $169,747 [€134,450] SOF+LDV: $115,358 [€91,371] Genotype 2: PR: $54,005 [€42,775] SOF+RBV: $109,958 [€87,093] SOF+DCV: $316,845 [€250,962] Genotype 3: PR: $58,323 [€46,195] SOF+RBV: $207,872 [€164,648] SOF+DCV: $317,830 [€251,742] SOF+LDV+RBV: $120,464 [€95,415] |
| Saab 2014 [38] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was not provided for all parameters. Cost-effectiveness results were most sensitive to cirrhosis prevalence and fibrosis rate, recurrence rates in patients achieving SVR. | Design: Decision-analytic Markov model; results reported in US dollars Population: Patients with chronic HCV genotype 1 in US | Five Strategies:
| ICER ($/QALY): Treatment naïve (without cirrhosis) SOF+PR compared with PR: ≤$29,271 [€23,565] No treatment: $2071 [€1667] SOF+PR dominated BOC+PR, TEL+PR and SIM+PR Treatment naïve (with cirrhosis) SOF+PR compared with PR: ≤$16,939 [€13,637] BOC+PR: $8450 [€6802] SIM+PR: $1899 [€1528] No treatment: $17,299 [€13,927] SOF+PR dominated TEL+PR Treatment experienced (all patients) SOF+PR compared with PR: ≤$4290 [€3453] No treatment: $16,617 [€13,378] SOF+PR dominated BOC+PR, TEL+PR and SIM+PR | Total lifetime costs: Treatment-naïve without cirrhosis: SOF+PR: $116,715 [€93,964] PR: ≤$95,333 [€76,750] BOC+PR: $124,229 [€100,014] TEL+ PR: $128,879 [€103,757] SIM+PR: $120,318 [€96,865] No treatment: $112,093 [€90,243] Treatment-naïve with cirrhosis: SOF+PR: $209,923 [€169,004] PR: ≤$172,814 [€139,129] BOC+PR: $199,192 [€160,365] TEL+ PR: $211,996 [€170,673] SIM+PR: $207,758 [€167,261] No treatment: $140,210 [€112,880] Treatment-experienced all patients: SOF+PR: $148,812 [€119,805] PR: ≤$145,009 [€116,743] BOC+PR: $165,983 [€133,629] TEL+ PR: $165,428 [€133,182] SIM+PR: $168,251 [€135,455] No treatment: $115,911 [€93,317] |
| Younossi 2015 [35] | Certainty of evidence: moderate Allowance was made for uncertainty in the estimates of costs and consequences, and ranges were provided. PSA was performed. Justification for choice of ranges was not provided for patient distribution, regimen efficacy, costs, or utilities. Cost-effectiveness results were robust across the limited ranges tested. | Design: Decision-analytic Markov model; results reported in US dollars Population: Patients with chronic HCV genotype 1 in US. | Six strategies:
| LDV/SOF (ICER): Treatment-naïve patients: dominant over no treatment dominant over SOF+PR (12/24 weeks) less expensive and less effective than SOF+SIM dominant over SOF+RBV dominant over BOC+PR Results similar for patients with and without cirrhosis; and for treatment experienced patients with PR or Protease inhibitor (PI) + RBV | Drug costs/pack: BOC: $6687 [€5296] LDV/SOF: $31,500 [€24,950] PEG-IFN: $3310 [€2621] SIM: $22,120 [€17,520] SOF: $28,000 [€22,177] RBV: $1153 [€913]; Generic: $238 [€188] Total lifetime costs by strategy (treatment naïve): No treatment: $141,856 [€112,359] LDV/SOF: $90,127 [€71,386] SOF+PR: $119,846 [€94,925] SIM +PR: $128,793 [€102,012] SOF+SIM: $191,631 [€151,784] SOF+RBV: $229,200 [€181,541] BOC+PR: $127,759 [€101,193] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greenaway, C.; Makarenko, I.; Abou Chakra, C.N.; Alabdulkarim, B.; Christensen, R.; Palayew, A.; Tran, A.; Staub, L.; Pareek, M.; Meerpohl, J.J.; et al. The Effectiveness and Cost-Effectiveness of Hepatitis C Screening for Migrants in the EU/EEA: A Systematic Review. Int. J. Environ. Res. Public Health 2018, 15, 2013. https://doi.org/10.3390/ijerph15092013
Greenaway C, Makarenko I, Abou Chakra CN, Alabdulkarim B, Christensen R, Palayew A, Tran A, Staub L, Pareek M, Meerpohl JJ, et al. The Effectiveness and Cost-Effectiveness of Hepatitis C Screening for Migrants in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health. 2018; 15(9):2013. https://doi.org/10.3390/ijerph15092013
Chicago/Turabian StyleGreenaway, Christina, Iuliia Makarenko, Claire Nour Abou Chakra, Balqis Alabdulkarim, Robin Christensen, Adam Palayew, Anh Tran, Lukas Staub, Manish Pareek, Joerg J. Meerpohl, and et al. 2018. "The Effectiveness and Cost-Effectiveness of Hepatitis C Screening for Migrants in the EU/EEA: A Systematic Review" International Journal of Environmental Research and Public Health 15, no. 9: 2013. https://doi.org/10.3390/ijerph15092013
APA StyleGreenaway, C., Makarenko, I., Abou Chakra, C. N., Alabdulkarim, B., Christensen, R., Palayew, A., Tran, A., Staub, L., Pareek, M., Meerpohl, J. J., Noori, T., Veldhuijzen, I., Pottie, K., Castelli, F., & Morton, R. L. (2018). The Effectiveness and Cost-Effectiveness of Hepatitis C Screening for Migrants in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health, 15(9), 2013. https://doi.org/10.3390/ijerph15092013







