Catalytic Degradation of Diatrizoate by Persulfate Activation with Peanut Shell Biochar-Supported Nano Zero-Valent Iron in Aqueous Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experiments
2.2.1. Synthesis Process of BC and nZVI/BC
2.2.2. Characterizations
2.2.3. DTZ Adsorption and Degradation
2.2.4. Analytical Methods
3. Results and Discussions
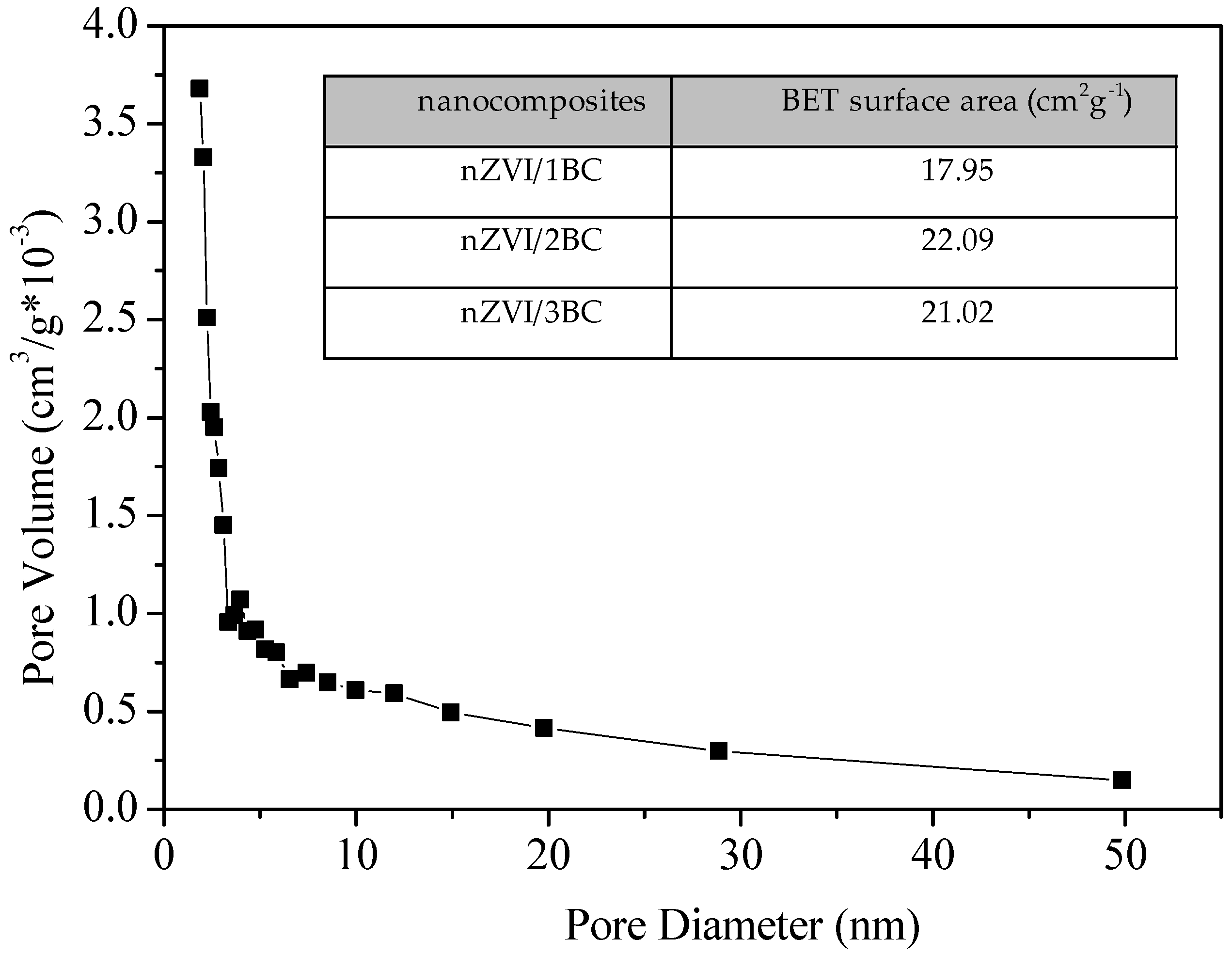
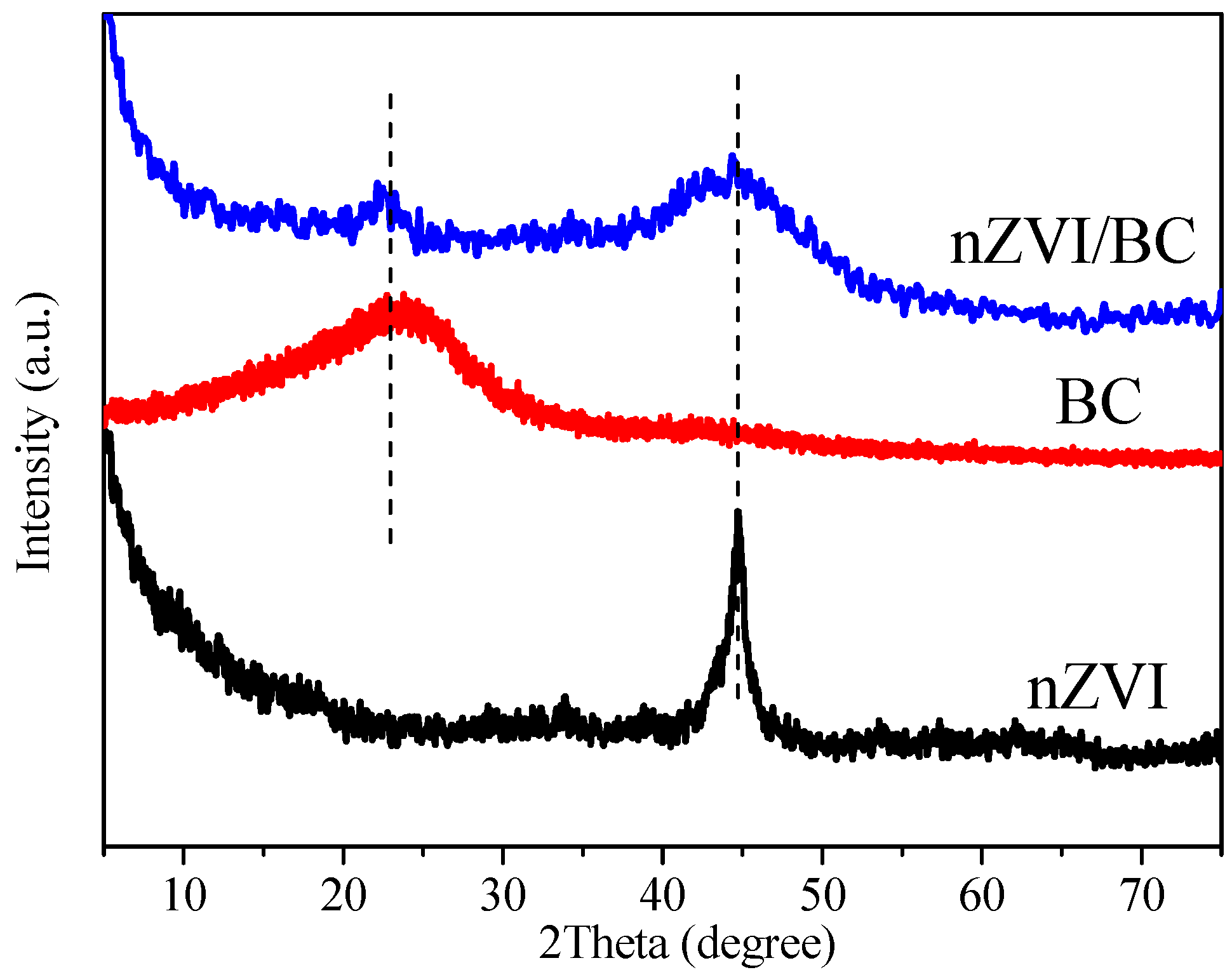
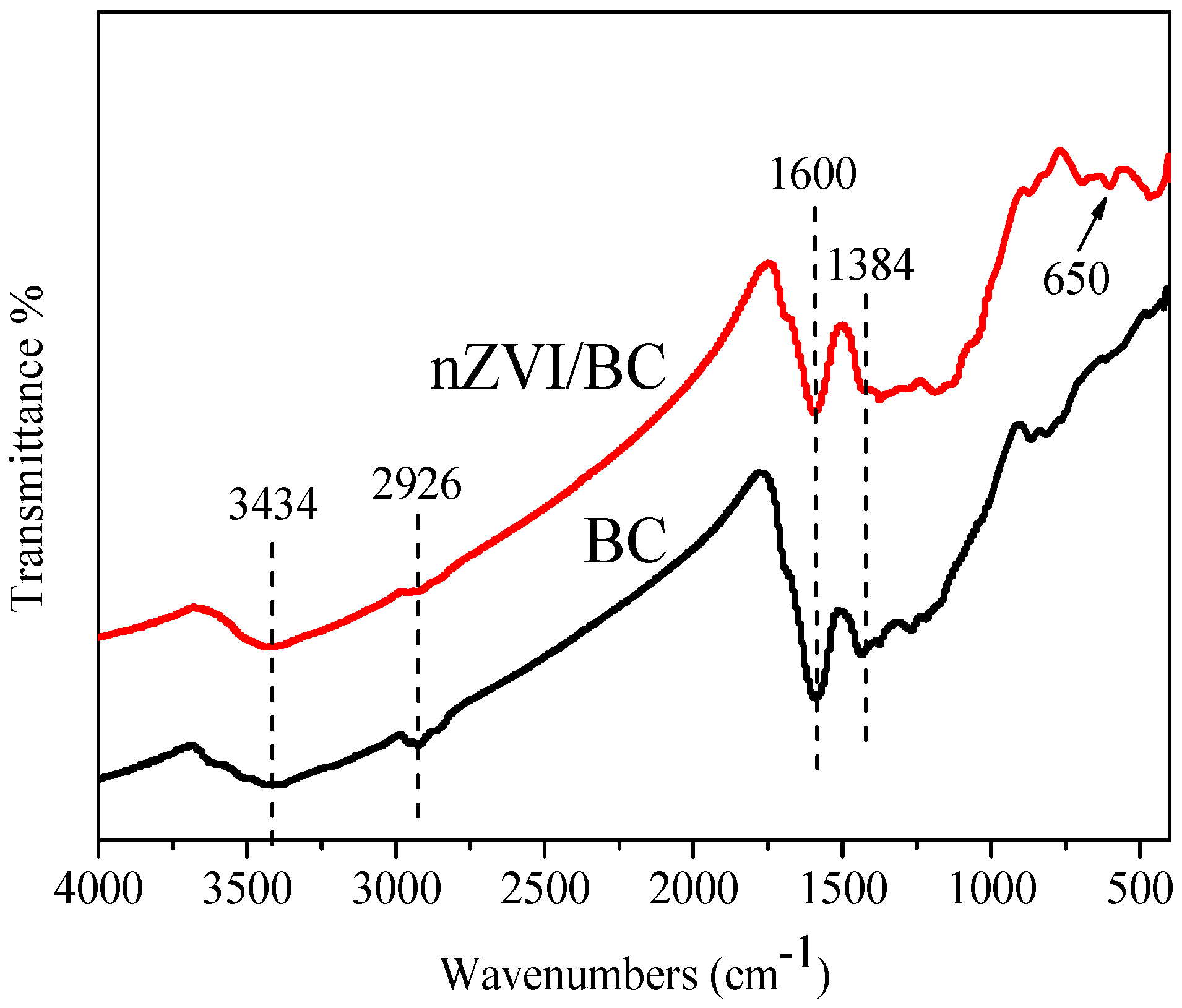
3.1. Characterization of nZVI/BC Composite
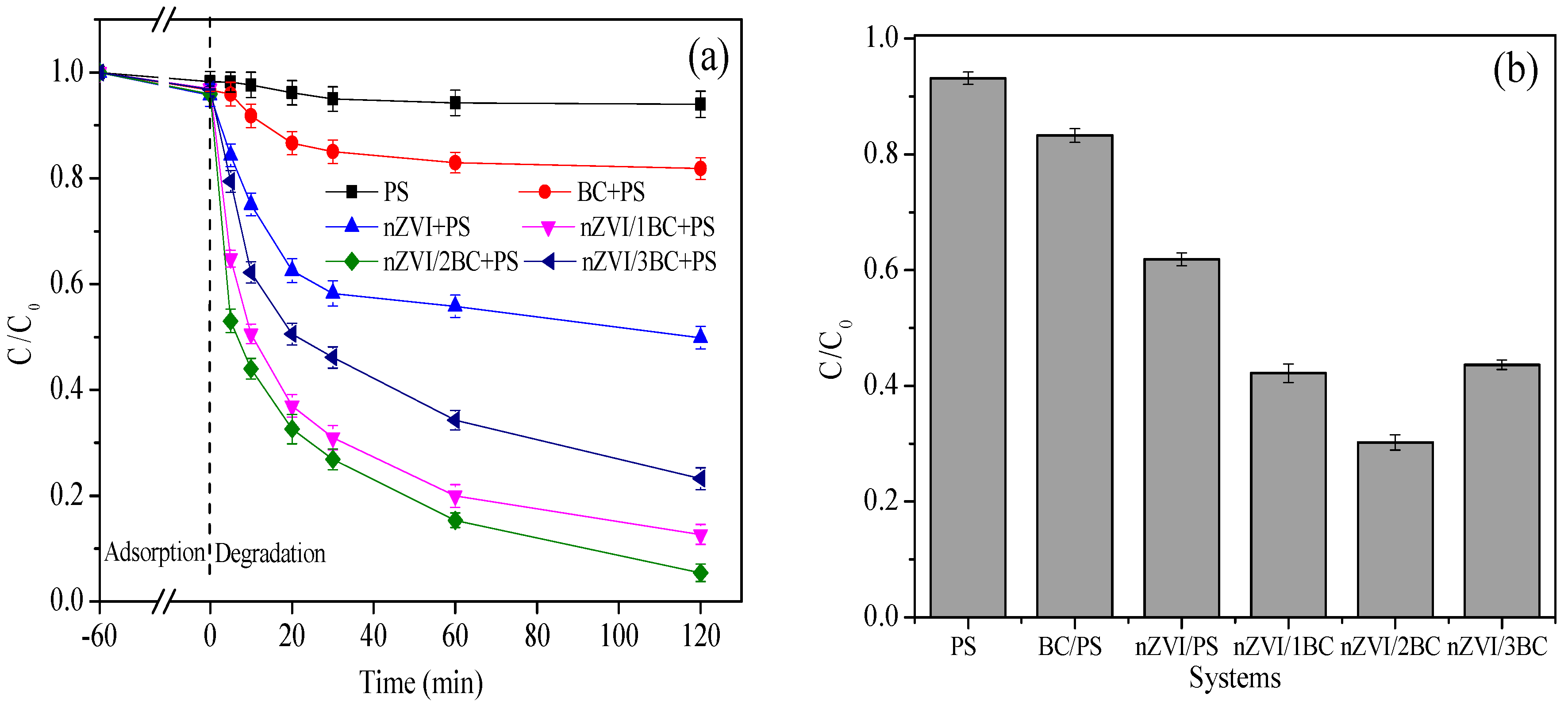
3.2. nZVI/BC Activation of PS for DTZ Removal
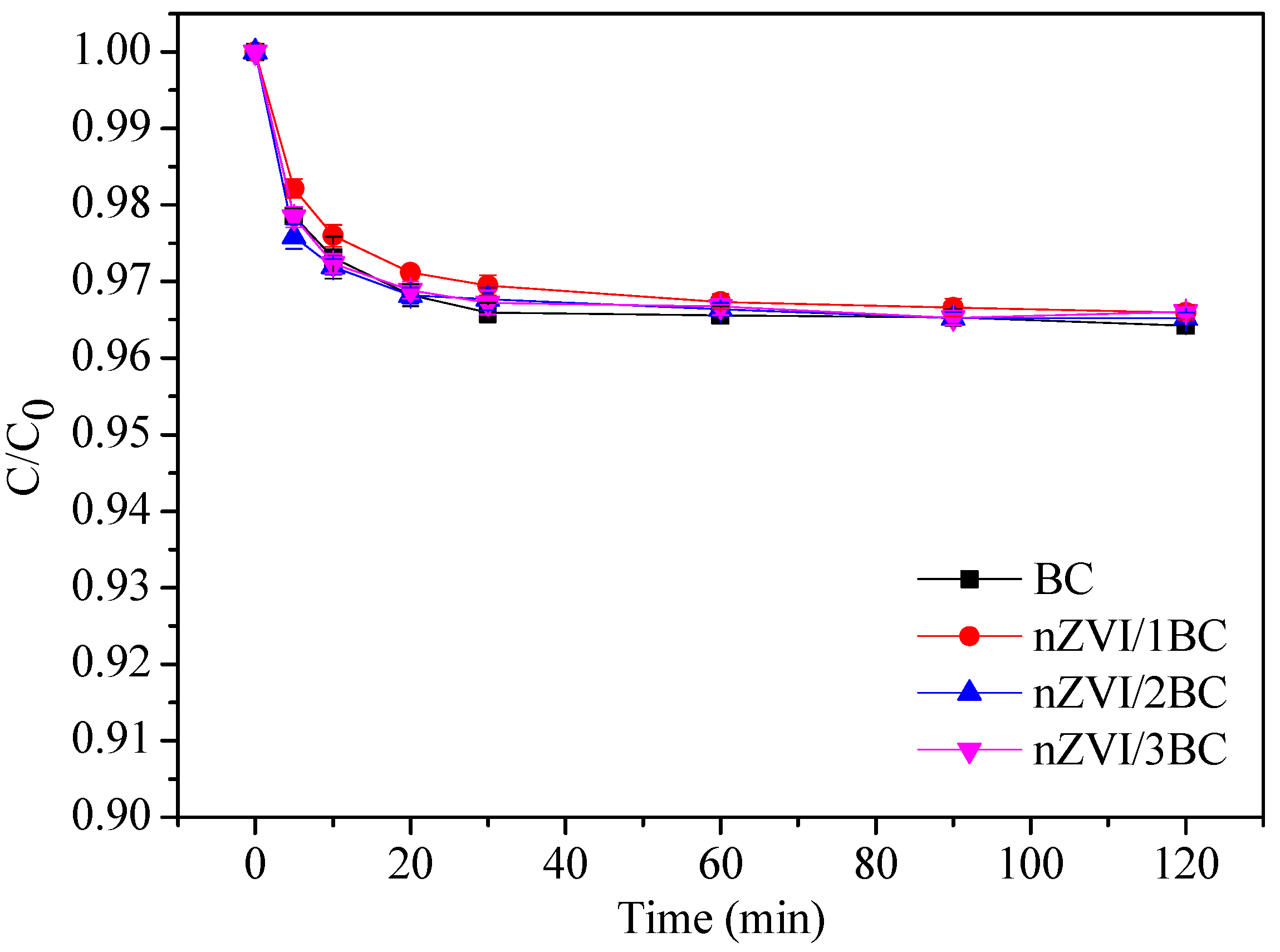
3.2.1. Adsorption
3.2.2. Degradation
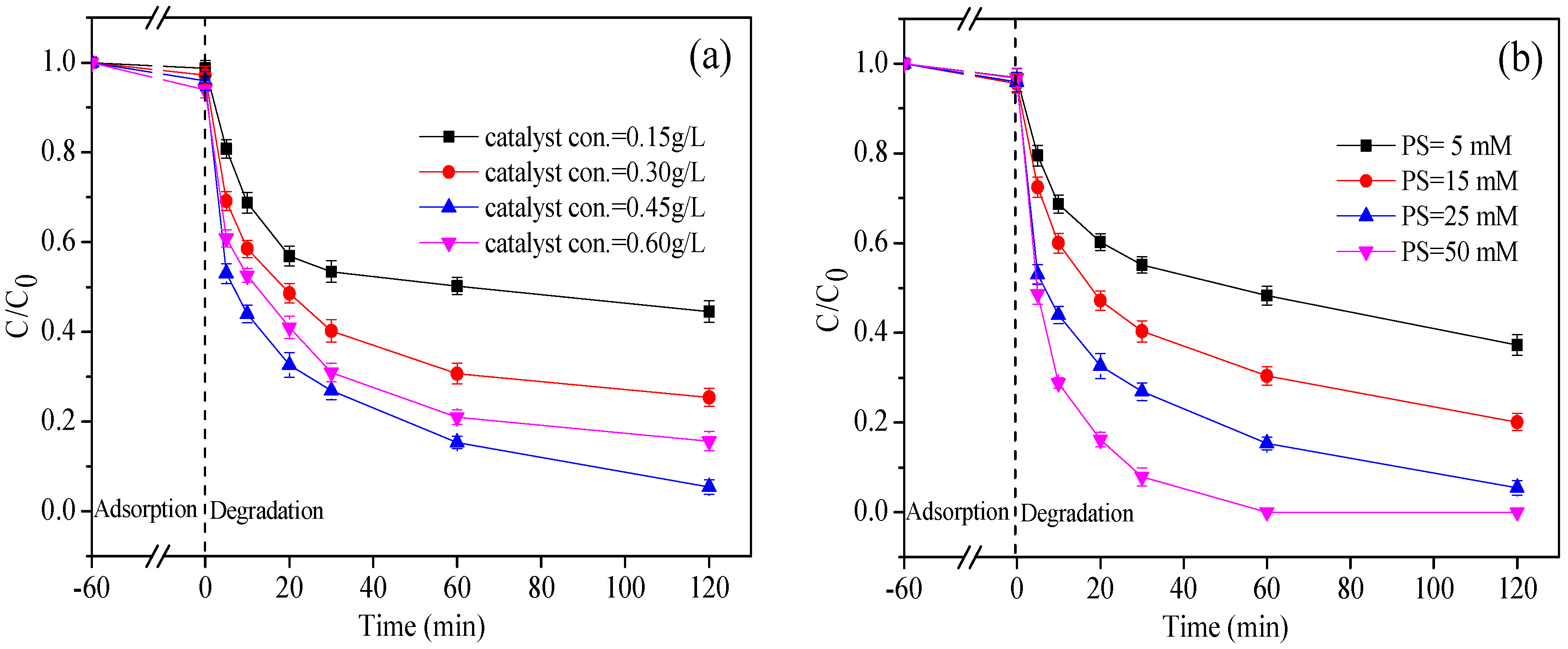
3.3. Effect of nZVI/2BC and PS Dosage
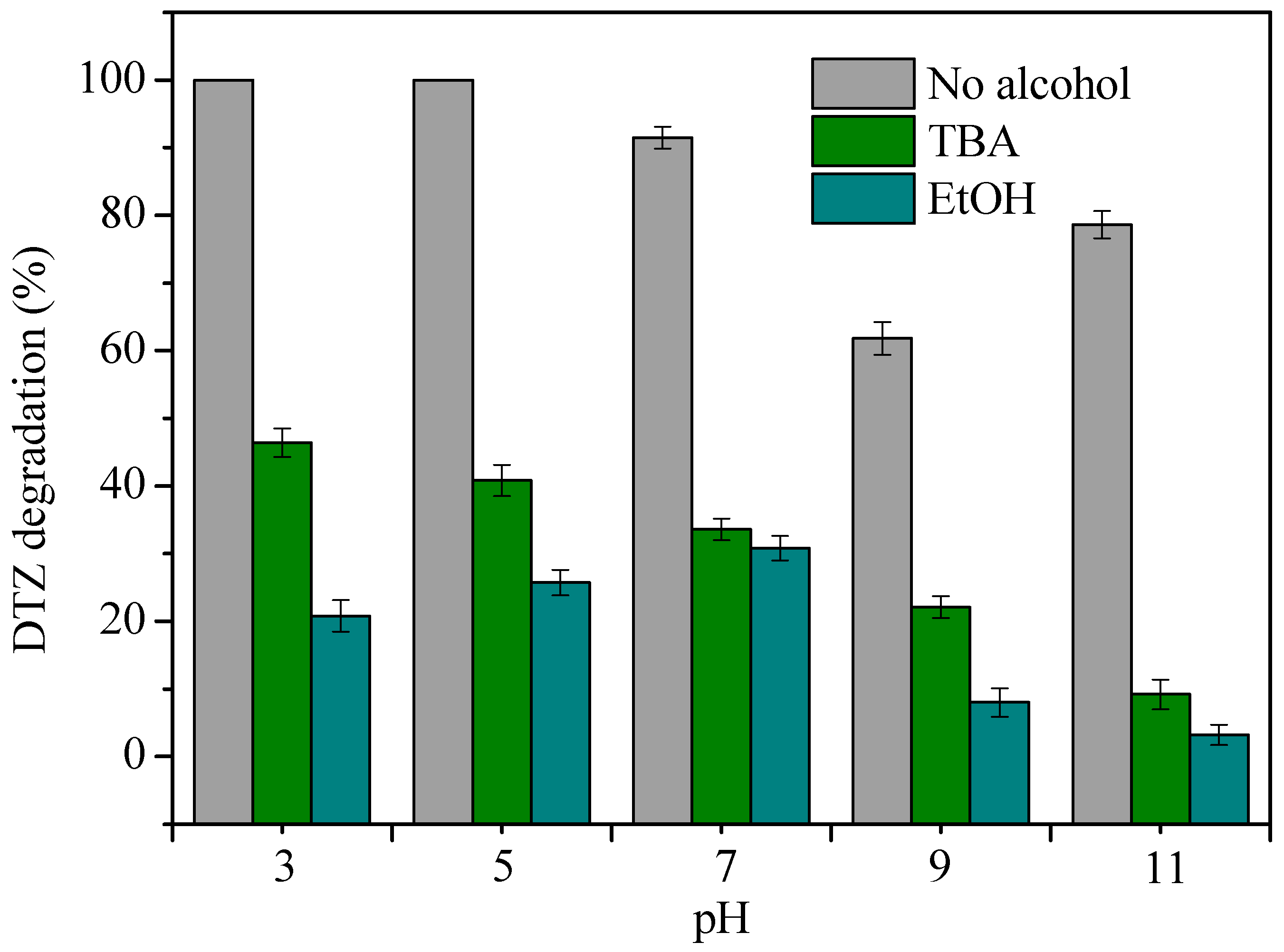
3.4. Effect of Reaction Temperature and Solution pH
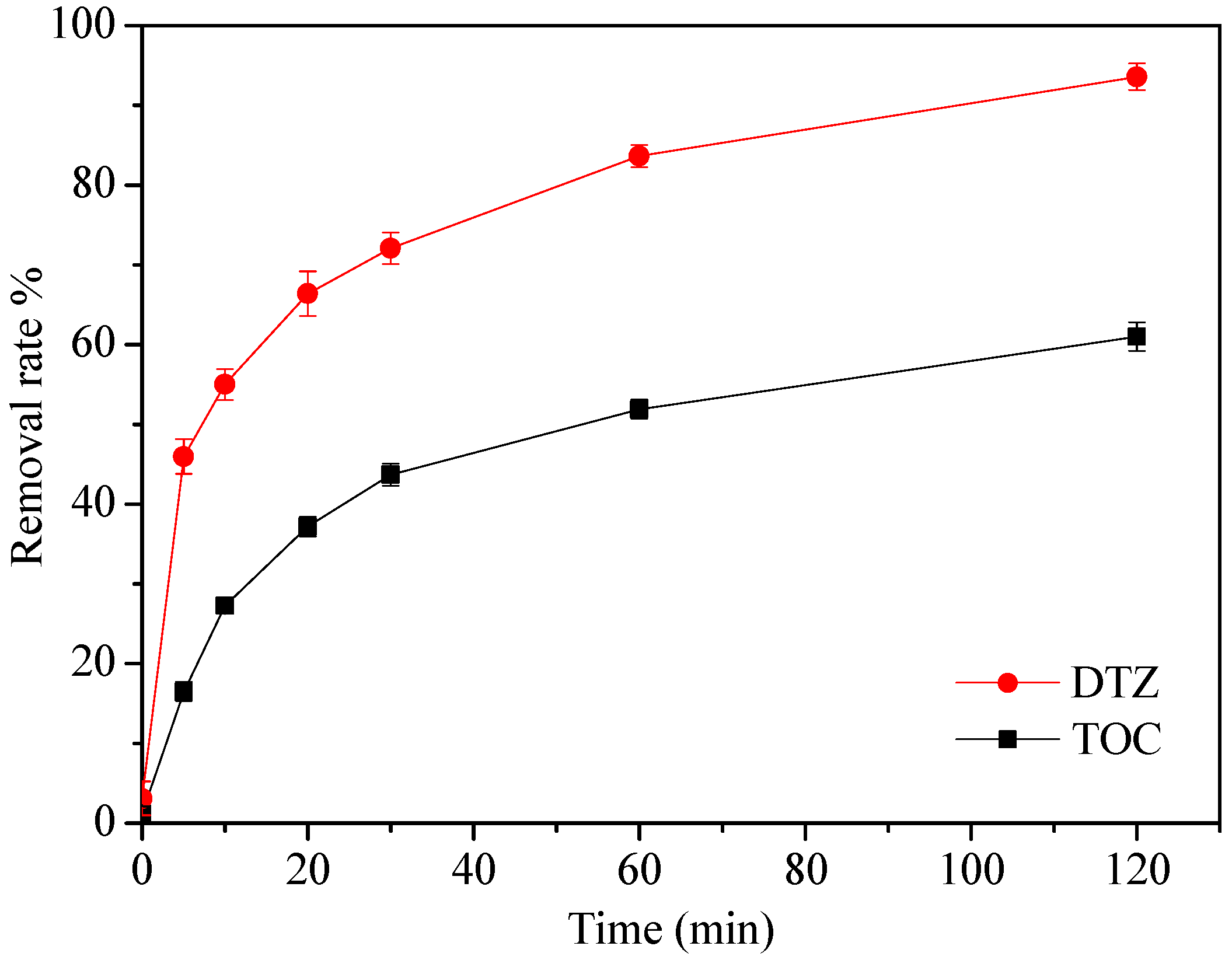
3.5. DTZ Mineralization
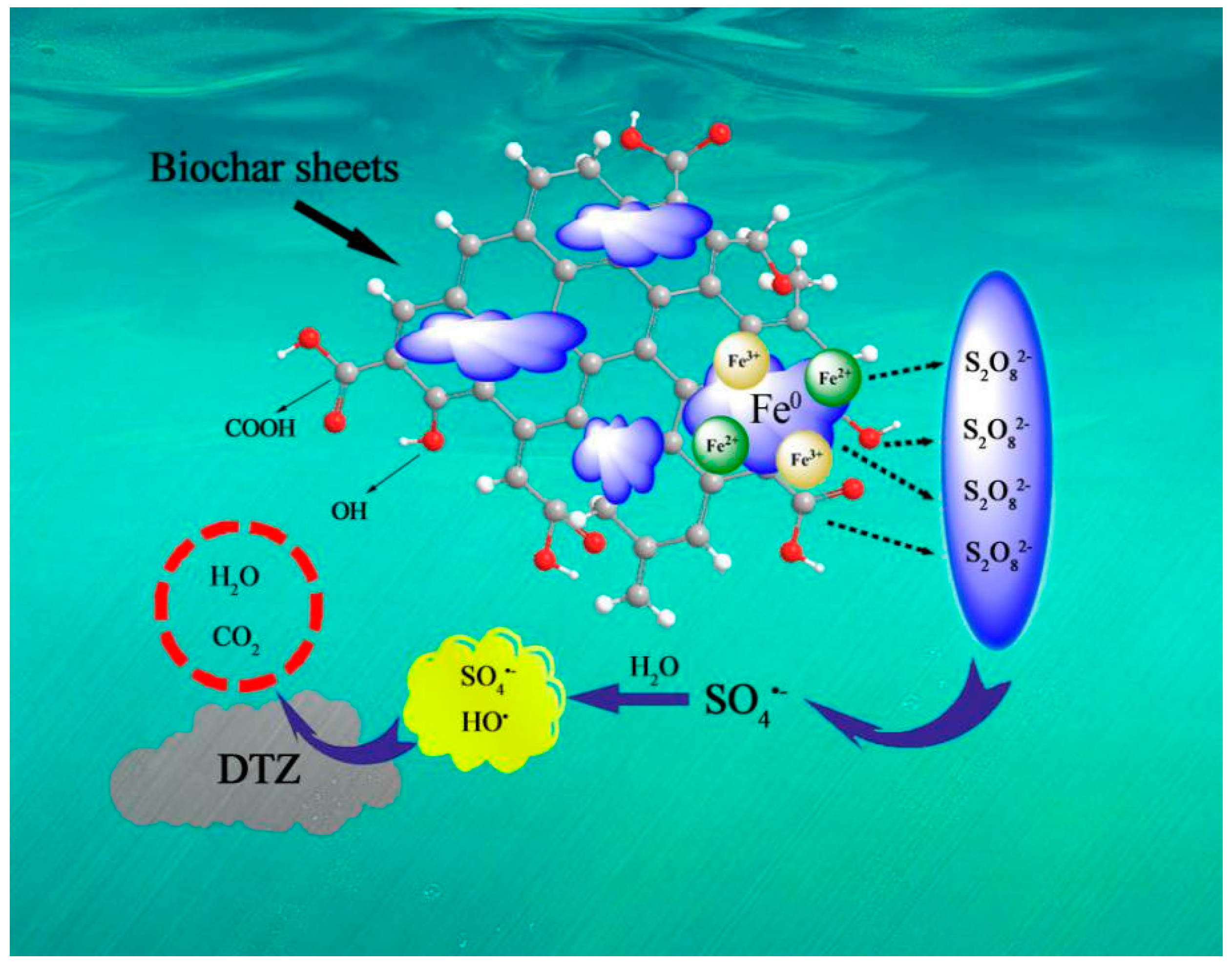
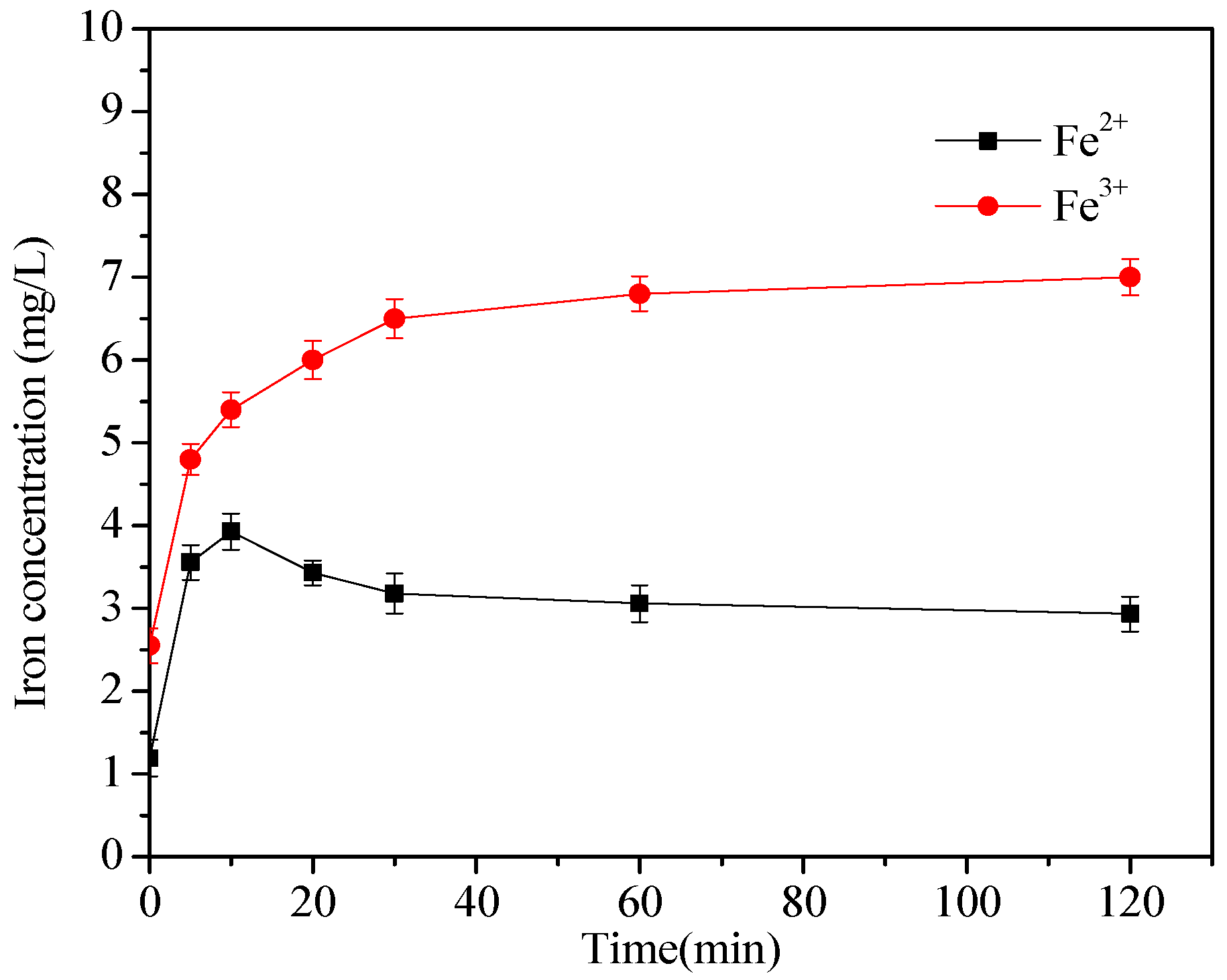
3.6. Activation Mechanism
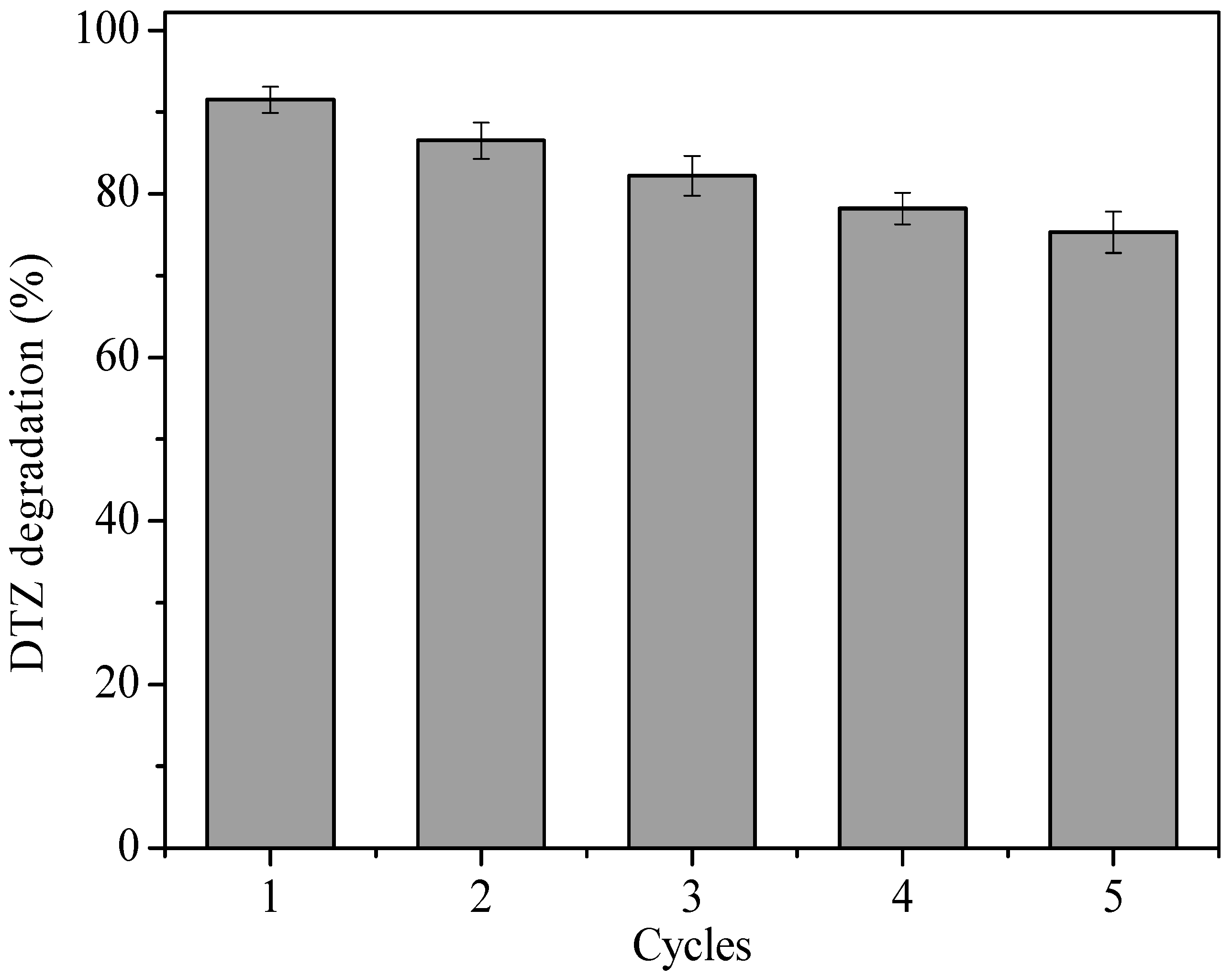
3.7. The Stability of the nZVI/BC
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.S.; Thomaidis, N.S.; Xu, J. Progress in the biological and chemical treatment technologies for emerging contaminant removal from wastewater: A critical review. J. Hazard. Mater. 2017, 323, 274–298. [Google Scholar] [CrossRef] [PubMed]
- Pantelaki, I.; Voutsa, D. Formation of iodinated THMs during chlorination of water and wastewater in the presence of different iodine sources. Sci. Total Environ. 2018, 613, 389–397. [Google Scholar] [CrossRef] [PubMed]
- Ens, W.; Senner, F.; Gygax, B.; Schlotterbeck, G. Development, validation, and application of a novel LC-MS/MS trace analysis method for the simultaneous quantification of seven iodinated X-ray contrast media and three artificial sweeteners in surface, ground, and drinking water. Anal. Bioanal. Chem. 2014, 406, 2789–2798. [Google Scholar] [CrossRef] [PubMed]
- Archer, E.; Petrie, B.; Kasprzyk-Hordern, B.; Wolfaardt, G.M. The fate of pharmaceuticals and personal care products (PPCPs), endocrine disrupting contaminants (EDCs), metabolites and illicit drugs in a WWTW and environmental waters. Chemosphere 2017, 174, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Seitz, W.; Weber, W.H.; Jiang, J.Q.; Lloyd, B.J.; Maier, M.; Maier, D.; Schulz, W. Monitoring of iodinated X-ray contrast media in surface water. Chemosphere 2006, 64, 1318–1324. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.F.; Li, X.; Hu, X.L.; Ying, D.Q. Distribution and relevance of iodinated X-ray contrast media and iodinated trihalomethanes in an aquatic environment. Chemosphere 2017, 184, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Polo, A.M.S.; López-Peñalver, J.J.; Sánchez-Polo, M.; Rivera-Utrilla, J.; Velo-Gala, I.; Salazar-Rabago, J.J. Oxidation of diatrizoate in aqueous phase by advanced oxidation processes based on solar radiation. J. Photochem. Photobiol. A Chem. 2016, 319, 87–95. [Google Scholar] [CrossRef]
- Meng, L.J.; Yang, S.G.; Sun, C.; He, H.; Xian, Q.M.; Li, S.Y.; Wang, G.X.; Zhang, L.M.; Jiang, D. A novel method for photo-oxidative degradation of diatrizoate in water via electromagnetic induction electrodeless lamp. J. Hazard. Mater. 2017, 2337, 34–46. [Google Scholar] [CrossRef] [PubMed]
- Gale, M.E.; Robbins, A.H.; Hamburger, R.J.; Widrich, W.C. Renal toxicity of contrast agents: Iopamidol, iothalamate, and diatrizoate. Am. J. Roentgenol. 1984, 142, 333–335. [Google Scholar] [CrossRef] [PubMed]
- Pfannenstiel, P.; Luhrbraun, A. Application of iodine-containing X-ray contrast media and thyroid-function. Med. Welt 1993, 44, 337–340. [Google Scholar]
- Zhou, D.N.; Zhang, H.; Chen, L. Sulfur-replaced Fenton systems: Can sulfate radical substitute hydroxyl radical for advanced oxidation technologies? J. Chem. Technol. Biotechnol. 2015, 5, 775–779. [Google Scholar] [CrossRef]
- Matzek, L.W.; Carter, K.E. Activated persulfate for organic chemical degradation: A review. Chemosphere 2016, 151, 178–188. [Google Scholar] [CrossRef] [PubMed]
- Boczkaj, G.; Fernandes, A. Wastewater treatment by means of advanced oxidation processes at basic pH conditions: A review. Chem. Eng. J. 2017, 320, 608–633. [Google Scholar] [CrossRef]
- Liang, C.J.; Bruell, C.J.; Marley, M.C.; Sperry, K.L. Persulfate oxidation for in situ remediation of TCE. I. Activated by ferrous ion with and without a persulfate–thiosulfate redox couple. Chemosphere 2004, 55, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.Y.; Kang, S.G.; Chiu, P.C. Degradation of 2,4-dinitrotoluene by persulfate activated with zero-valent iron. Sci. Total Environ. 2010, 408, 3464–3468. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.Y.; Kang, S.G.; Kim, D.W.; Chiu, P.C. Degradation of 2,4-dinitrotoluene by persulfate activated with iron sulfides. Chem. Eng. J. 2011, 172, 641–646. [Google Scholar] [CrossRef]
- Hussain, I.; Zhang, Y.Q.; Huang, S.B.; Du, X.Z. Degradation of p-chloroaniline by persulfate activated with zero-valent iron. Chem. Eng. J. 2012, 203, 269–276. [Google Scholar] [CrossRef]
- Wang, X.; Wang, L.G.; Li, J.B.; Qiu, J.J.; Cai, C.; Zhang, H. Degradation of Acid Orange 7 by persulfate activated with zero valent iron in the presence of ultrasonic irradiation. Sep. Purif. Technol. 2014, 122, 41–46. [Google Scholar] [CrossRef]
- Liang, C.J.; Lai, M.C. Trichloroethylene degradation by zero valent iron activated persulfate oxidation. Environ. Eng. Sci. 2008, 25, 1071–1078. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, S.Y.; Yang, X.; Huang, X.F.; Yang, Y.H.; He, E.K.; Wang, S.Q.; Qiu, R.L. Degradation of 2,2′,4,4′-tetrabromodiphenyl ether (BDE-47) by a nano zerovalent iron-activated persulfate process: The effect of metal ions. Chem. Eng. J. 2017, 317, 613–622. [Google Scholar] [CrossRef]
- Phenrat, T.; Saleh, N.; Sirk, K.; Tilton, R.D.; Lowry, G.V. Aggregation and sedimentation of aqueous nanoscale zerovalent iron dispersions. Environ. Sci. Technol. 2007, 41, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Hou, D.Y.; O’Connor, D.; Li, X.R.; Pehkonen, S.O.; Varma, R.S.; Wang, X. Green and Size-Specific Synthesis of Stable Fe-Cu Oxides as Earth-Abundant Adsorbents for Malachite Green Removal. ACS Sustain. Chem. Eng. 2018, 53, 9229–9236. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Lo, I.; O’Connor, D.; Pehkonen, S.; Cheng, H.F.; Hou, D.Y. High efficiency removal of methylene blue using SDS surface-modified ZnFe2O4 nanoparticles. J. Colloid Interface Sci. 2017, 508, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Park, Y.M.; Yoo, K.M.; Lee, S.H. Reduction of nitrate by resin-supported nanoscale zero-valent iron. Water Sci. Technol. 2009, 59, 2153–2157. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, K.; Bleyl, S.; Georgi, A.; Kopinke, F.D. Carbo-Iron-An Fe/AC composite-As alternative to nano-iron for groundwater treatment. Water Res. 2012, 46, 3817–3826. [Google Scholar] [CrossRef] [PubMed]
- Fukuchi, S.; Nishimoto, R.; Fukushima, M.; Zhu, Q.Q. Effects of reducing agents on the degradation of 2,4,6-tribromophenol in a heterogeneous Fenton-like system with an iron-loaded natural zeolite. Appl. Catal. B Environ. 2014, 147, 411–419. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, A.K.; Dasmahapatra, A.K.; Mandal, T.K.; Bandyopadhyay, D. Graphene based multifunctional superbots. Carbon 2015, 89, 31–40. [Google Scholar] [CrossRef]
- O’Connor, D.; Peng, T.Y.; Zhang, J.L.; Tsang, D.C.; Alessi, D.S.; Shen, Z.T.; Bolan, N.S.; Hou, D.Y. Biochar application for the remediation of heavy metal polluted land: A review of in situ field trials. Sci. Total Environ. 2018, 619, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; O’Connor, D.; Zhang, J.L.; Peng, T.Y.; Shen, Z.T.; Tsang, D.; Hou, D.Y. Effect of pyrolysis temperature, heating rate, and residence time on rapeseed stem derived biochar. J. Clean. Prod. 2018, 174, 977–987. [Google Scholar] [CrossRef]
- O’Connor, D.; Peng, T.Y.; Li, G.H.; Wang, S.X.; Duan, L.; Mulder, J.; Cornelissen, G.; Cheng, Z.L.; Yang, S.M.; Hou, D.Y. Sulfur-modified rice husk biochar: A green method for the remediation of mercury contaminated soil. Sci. Total Environ. 2017, 621, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.T.; Hou, D.Y.; Zhao, B.; Xu, W.D.; Ok, Y.S.; Bolan, N.S.; Alessi, D.S. Stability of heavy metals in soil washing residue with and without biochar addition under accelerated ageing. Sci. Total Environ. 2018, 619, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.M.; Gao, B.; Zimmerman, A.R.; Chen, H.; Zhang, M.; Cao, X.D. Biochar-supported zerovalent iron for removal of various contaminants from aqueous solutions. Bioresour. Technol. 2014, 152, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.F.; Liu, Y.G.; Gu, Y.L.; Xu, Y.; Zeng, G.M.; Hu, X.J.; Liu, S.B.; Wang, X.; Liu, S.M.; Li, J. Biochar-based nano-composites for the decontamination of wastewater: A review. Bioresour. Technol. 2016, 212, 318–333. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.Y.; Yang, X.; Shao, X.T.; Niu, R.; Wang, L.L. Activated carbon catalyzed persulfate oxidation of Azo dye acid orange 7 at ambient temperature. J. Hazard. Mater. 2011, 186, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Pu, M.J.; Ma, Y.W.; Wan, J.Q.; Wang, Y.; Huang, M.Z.; Chen, Y.M. Fe/S doped granular activated carbon as a highly active heterogeneous persulfate catalyst toward the degradation of Orange G and diethyl phthalate. J. Colloid Interface Sci. 2014, 418, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Zhai, J.P.; Zhang, W.Y.; Wang, M.M.; Zhou, J. Kinetic studies of adsorption of Pb(II), Cr(III) and Cu(II) from aqueous solution by sawdust and modified peanut husk. J. Hazard. Mater. 2017, 141, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Tanyildizi, M.S. Modeling of adsorption isotherms and kinetics of reactive dye from aqueous solution by peanut hull. Chem. Eng. J. 2011, 168, 1234–1240. [Google Scholar] [CrossRef]
- Cheng, Q.M.; Huang, Q.; Khan, S.; Liu, Y.J.; Liao, Z.N.; Li, G.; Ok, Y.S. Adsorption of Cd by peanut husks and peanut husk biochar from aqueous solutions. Ecol. Eng. 2016, 87, 240–245. [Google Scholar] [CrossRef]
- Han, X.Y.; Chu, L.; Liu, S.M.; Chen, T.M.; Cheng, D.; Yan, J.L.; Cui, L.Q.; Quan, G.X. Removal of methylene blue from aqueous solution using porous biochar obtained by KOH activation of peanut shell biochar. BioResources 2015, 10, 2836–2849. [Google Scholar] [CrossRef]
- Hou, D.Y.; Li, F.S. Complexities Surrounding China’s Soil Action Plan. Land Degrad. Dev. 2017, 28, 2315–2320. [Google Scholar] [CrossRef]
- Hou, D.Y.; O’Connor, D.; Nathanail, P.; Tian, L.; Ma, Y. Integrated GIS and multivariate statistical analysis for regional scale assessment of heavy metal soil contamination: A critical review. Environ. Pollut. 2017, 231, 1188–1200. [Google Scholar] [CrossRef] [PubMed]
- Hou, D.Y.; Al-Tabbaa, A. Sustainability: A new imperative in contaminated land remediation. Environ. Sci. Policy 2014, 39, 25–34. [Google Scholar] [CrossRef]
- O’Connor, D.; Hou, D.; Ok, Y.S.; Song, Y.; Sarmah, A.; Li, X.; Tack, F.M. Sustainable in situ remediation of recalcitrant organic pollutants in groundwater with controlled release materials: A review. J. Control. Release 2018, 283, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Hussain, I.; Li, M.Y.; Zhang, Y.Q.; Li, Y.C.; Huang, S.B.; Du, X.D.; Liu, G.Q.; Hayat, W.; Anwar, N. Insights into the mechanism of persulfate activation with nZVI/BC nanocomposite for the degradation of nonylphenol. Chem. Eng. J. 2017, 311, 163–172. [Google Scholar] [CrossRef]
- American Public Health Association; American Water Works Association; Water Environment Federation. Standards Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1998. [Google Scholar]
- Liang, C.J.; Huang, C.F.; Mohanty, N.; Kurakalva, R.M. A rapid spectrophotometric determination of persulfate anion in ISCO. Chemosphere 2008, 73, 1540–1543. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.C.; Xiao, F.; Ching, C.B.; Duan, H.W. One-step electrochemical synthesis of PtNi nanoparticle-graphene nanocomposites for nonenzymatic amperometric glucose detection. ACS Appl. Mater. Interface 2011, 3, 3049–3057. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Zhang, C.; Hou, K.J.; Cheng, Y.J.; Deng, J.M.; Jiang, Z.; Tang, L.; Zeng, G.M. Removal of trichloroethylene by biochar supported nanoscale zero-valent iron in aqueous solution. Sep. Purif. Technol. 2017, 188, 188–196. [Google Scholar] [CrossRef]
- Yan, J.C.; Han, L.; Gao, W.G.; Xue, S.; Chen, M.F. Biochar supported nanoscale zerovalent iron composite used as persulfate activator for removing trichloroethylene. Bioresour. Technol. 2015, 175, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.C.; Lv, H.H.; Gong, Y.Y.; Huang, Y. Preparation and characterization of a novel graphene/biochar composite for aqueous phenanthrene and mercury removal. Bioresour. Technol. 2015, 196, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Inyang, M.; Gao, B.; Zimmerman, A.; Zhou, Y.M.; Cao, X.D. Sorption and cosorption of lead and sulfapyridine on carbon nanotube-modified biochars. Environ. Sci. Pollut. Res. 2015, 22, 1868–1876. [Google Scholar] [CrossRef] [PubMed]
- Fang, G.D.; Liu, C.; Gao, J.; Dionysiou, D.D.; Zhou, D.M. Manipulation of Persistent Free Radicals in Biochar To Activate Persulfate for Contaminant Degradation. Environ. Sci. Technol. 2015, 9, 5645–5653. [Google Scholar] [CrossRef] [PubMed]
- Nie, M.H.; Yang, Y.; Zhang, Z.J.; Yan, C.X.; Wang, X.N.; Li, H.J.; Dong, W.B. Degradation of chloramphenicol by thermally activated persulfate in aqueous solution. Chem. Eng. J. 2014, 246, 373–382. [Google Scholar] [CrossRef]
- Potakis, N.; Frontistis, Z.; Antonopoulou, M.; Konstantinou, L.; Mantzavinos, D. Oxidation of bisphenol A in water by heat-activated persulfate. J. Environ. Manag. 2016, 195, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.J.; Su, H.W. Identification of sulfate and hydroxyl radicals in thermally activated persulfate. Ind. Eng. Chem. Res. 2009, 48, 5558–5562. [Google Scholar] [CrossRef]
- Furman, O.S.; Teel, A.L.; Ahmad, M.; Merker, M.C.; Watts, R.J. Effect of basicity on persulfate reactivity. J. Environ. Eng. 2011, 137, 241–247. [Google Scholar] [CrossRef]
- Furman, O.S.; Teel, A.L.; Watts, R.J. Mechanism of base activation of persulfate. Environ. Sci. Technol. 2010, 44, 6423–6428. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.F.; Chang, Y.C.; Liu, K.Y. A kinetic and mechanistic study of the degradation of 1,2-dichloroethane and methyl tert-butyl ether using alkaline-activated persulfate oxidation. RSC Adv. 2016, 6, 75578–75587. [Google Scholar] [CrossRef]
- Zhou, L.C.; Ma, J.J.; Zhang, H.; Shao, Y.M.; Li, Y.F. Fabrication of magnetic carbon composites from peanut shells and its application as a heterogeneous Fenton catalyst in removal of methylene blue. Appl. Surf. Sci. 2015, 324, 490–498. [Google Scholar] [CrossRef]
- Ouyang, D.; Yan, J.C.; Qian, L.B.; Chen, Y.; Han, L.; Su, A.Q.; Zhang, W.Y.; Ni, H.; Chen, M.F. Degradation of 1,4-Dioxane by biochar supported nano magnetite particles activating persulfate. Chemosphere 2017, 184, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Peng, X.Z.; Liu, J.H.; Li, J.J.; Wu, F. Decolorization of Orange II in Aqueous Solution by an Fe(II)/sulfite System: Replacement of Persulfate. Ind. Eng. Chem. Res. 2012, 42, 13632–13638. [Google Scholar] [CrossRef]

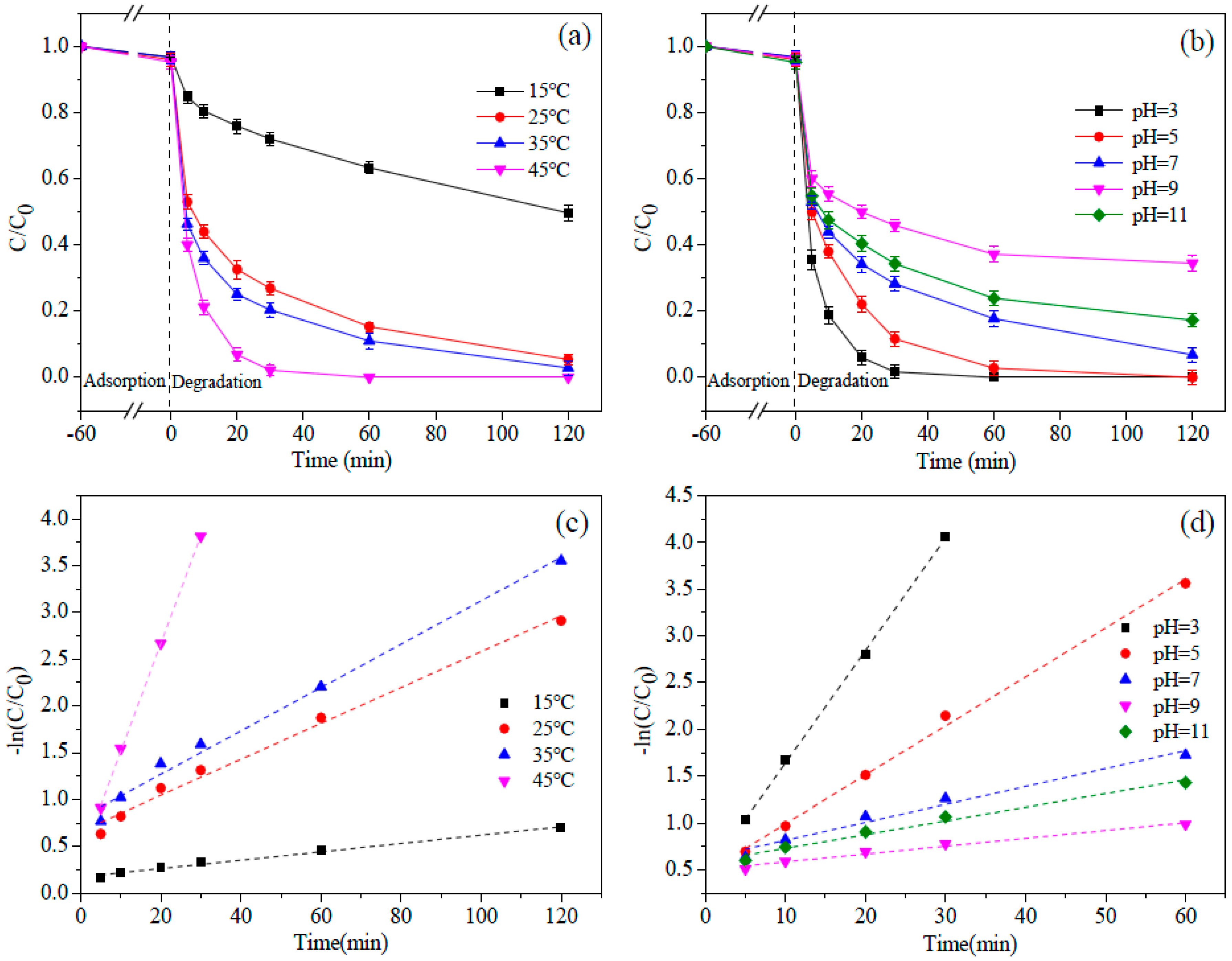
| Factors | Gradient | Kinetic Equations | R2 | k/(min−1) |
|---|---|---|---|---|
| Temperature | 15 °C | −ln(C/C0) = 0.0045 t + 0.1745 | 0.9912 | 0.0045 |
| 25 °C | −ln(C/C0) = 0.0192 t + 0.6622 | 0.9903 | 0.0192 | |
| 35 °C | −ln(C/C0) = 0.0232 t + 0.8061 | 0.9912 | 0.0232 | |
| 45 °C | −ln(C/C0) = 0.1154 t + 0.3617 | 0.9997 | 0.1154 | |
| pH | 3 | −ln(C/C0) = 0.1294 t + 0.2391 | 0.9912 | 0.1294 |
| 5 | −ln(C/C0) = 0.0523 t + 0.4676 | 0.9970 | 0.0523 | |
| 7 | −ln(C/C0) = 0.0171 t + 0.6687 | 0.9901 | 0.0171 | |
| 9 | −ln(C/C0) = 0.0084 t + 0.5021 | 0.9808 | 0.0084 | |
| 11 | −ln(C/C0) = 0.0146 t + 0.5854 | 0.9822 | 0.0146 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, J.; Zhang, X.; Sun, C.; He, H.; Dai, Y.; Yang, S.; Lin, Y.; Zhan, X.; Li, Q.; Zhou, Y. Catalytic Degradation of Diatrizoate by Persulfate Activation with Peanut Shell Biochar-Supported Nano Zero-Valent Iron in Aqueous Solution. Int. J. Environ. Res. Public Health 2018, 15, 1937. https://doi.org/10.3390/ijerph15091937
Xu J, Zhang X, Sun C, He H, Dai Y, Yang S, Lin Y, Zhan X, Li Q, Zhou Y. Catalytic Degradation of Diatrizoate by Persulfate Activation with Peanut Shell Biochar-Supported Nano Zero-Valent Iron in Aqueous Solution. International Journal of Environmental Research and Public Health. 2018; 15(9):1937. https://doi.org/10.3390/ijerph15091937
Chicago/Turabian StyleXu, Jian, Xueliang Zhang, Cheng Sun, Huan He, Yuxuan Dai, Shaogui Yang, Yusuo Lin, Xinhua Zhan, Qun Li, and Yan Zhou. 2018. "Catalytic Degradation of Diatrizoate by Persulfate Activation with Peanut Shell Biochar-Supported Nano Zero-Valent Iron in Aqueous Solution" International Journal of Environmental Research and Public Health 15, no. 9: 1937. https://doi.org/10.3390/ijerph15091937
APA StyleXu, J., Zhang, X., Sun, C., He, H., Dai, Y., Yang, S., Lin, Y., Zhan, X., Li, Q., & Zhou, Y. (2018). Catalytic Degradation of Diatrizoate by Persulfate Activation with Peanut Shell Biochar-Supported Nano Zero-Valent Iron in Aqueous Solution. International Journal of Environmental Research and Public Health, 15(9), 1937. https://doi.org/10.3390/ijerph15091937





