Aluminium in Brain Tissue in Multiple Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissues
2.2. Quantitative Measurements
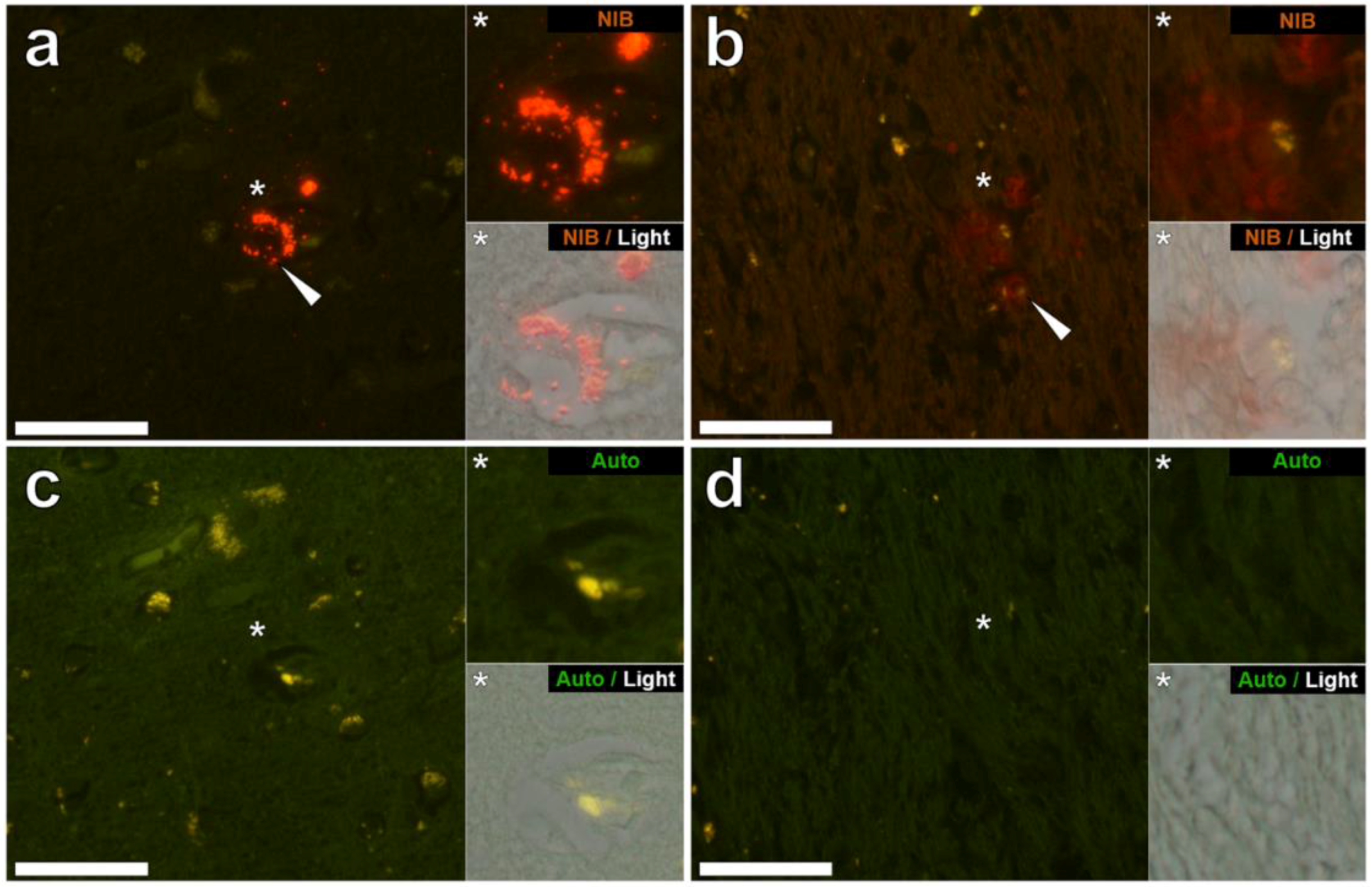
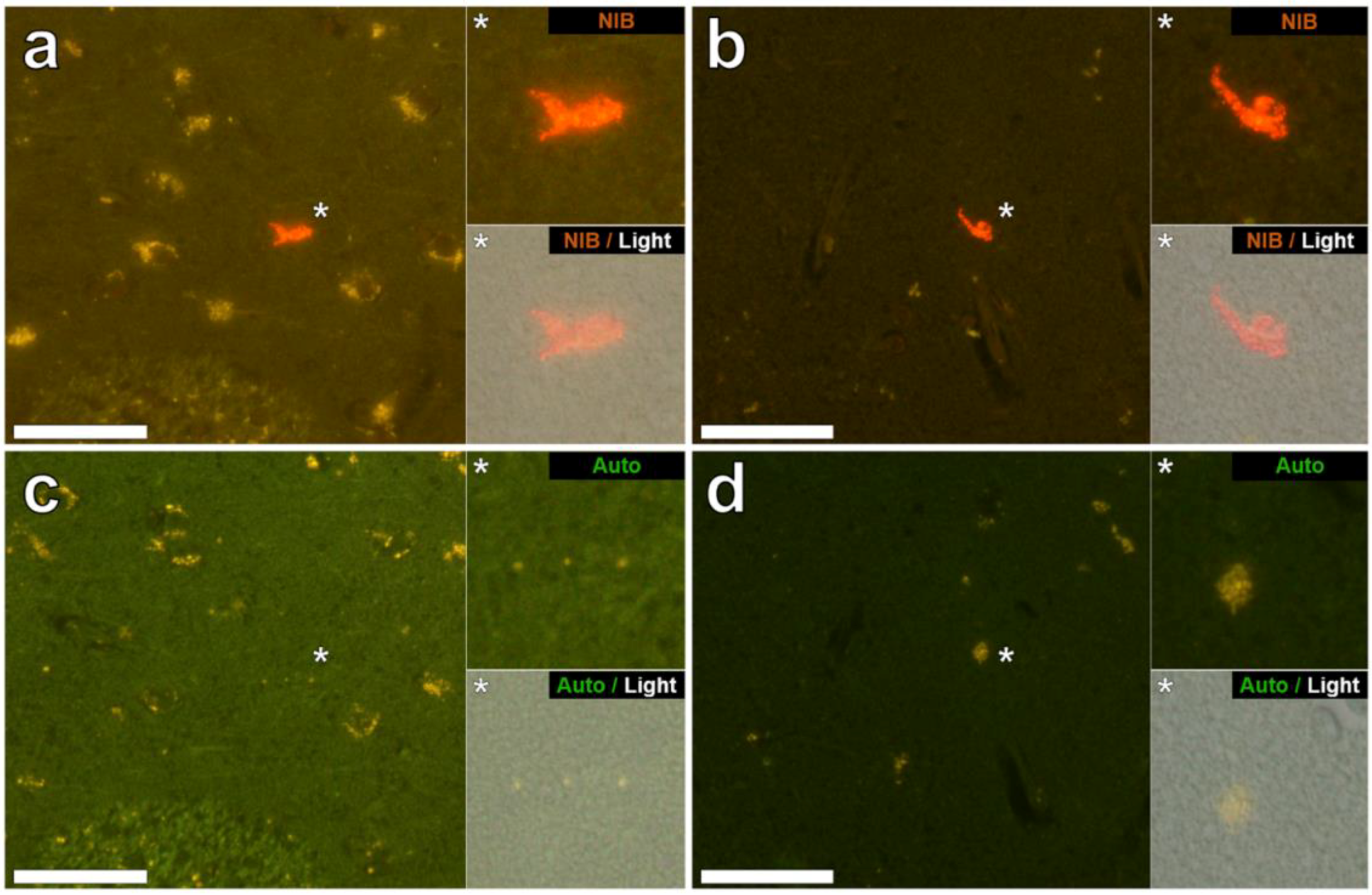
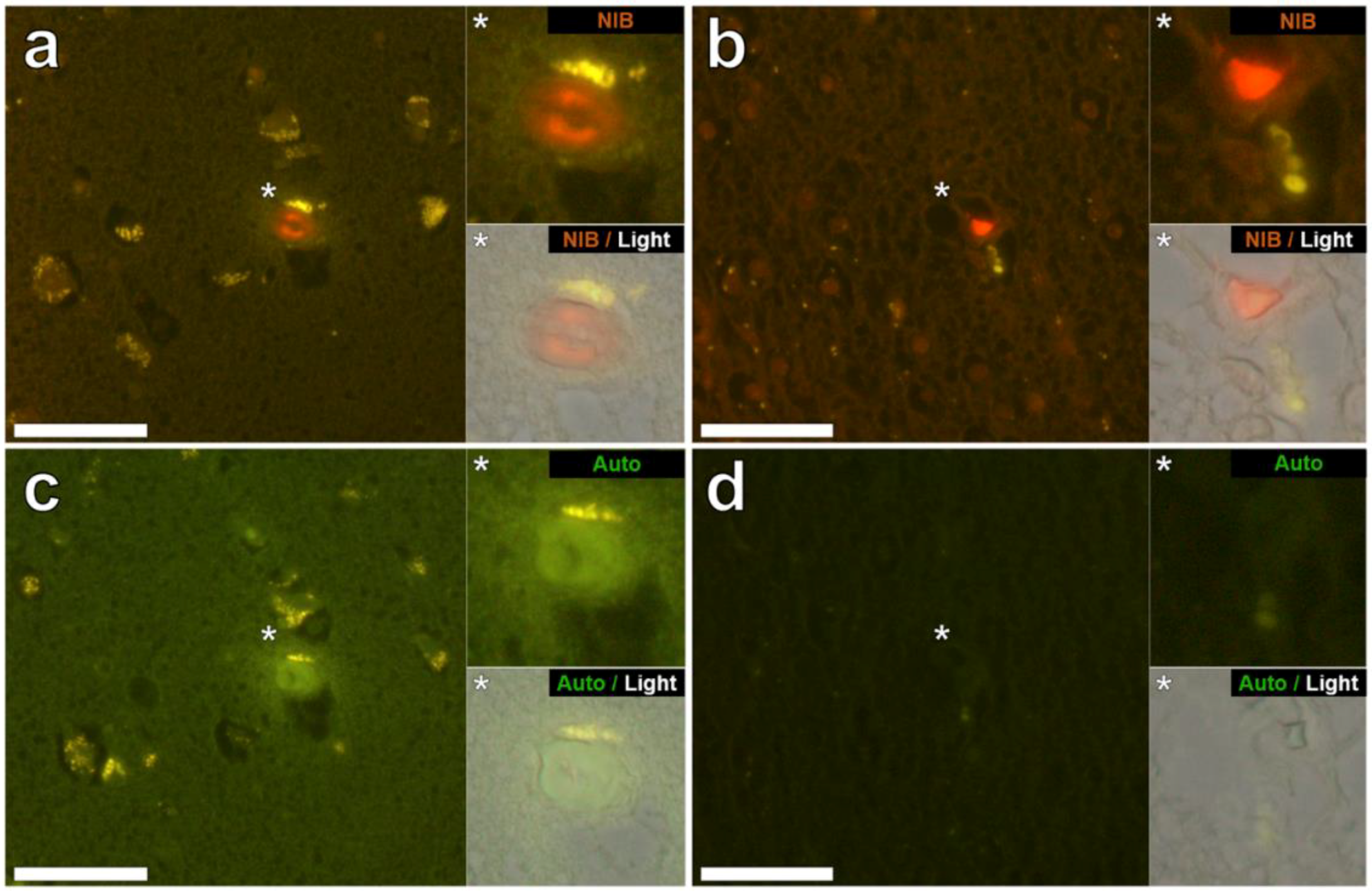
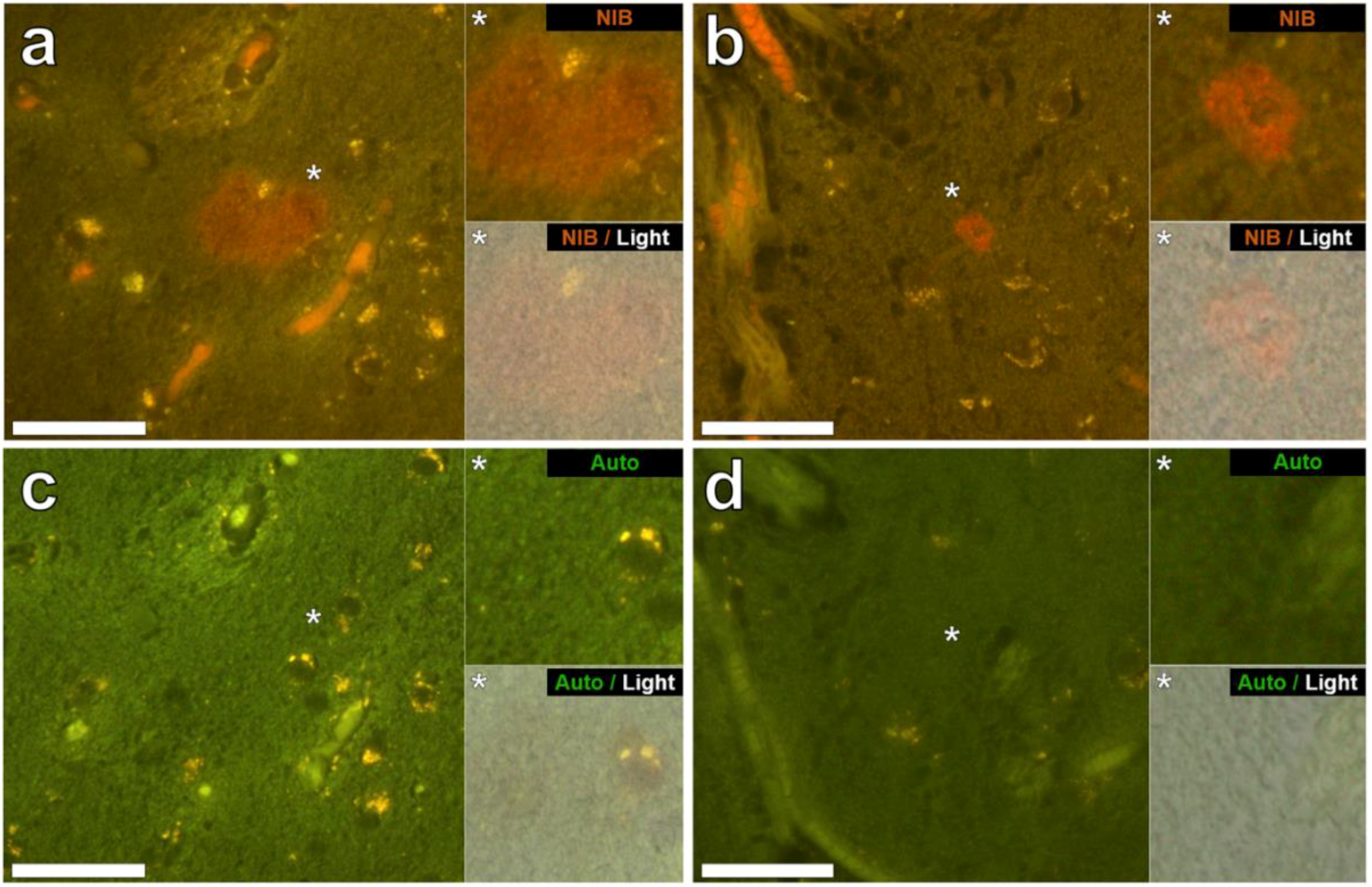
2.3. Fluorescence Microscopy
2.4. Statistical Analyses
3. Results
3.1. Aluminium Content of Brain Tissues
3.2. Aluminium Fluorescence in Brain Tissues
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Ontaneda, D.; Thompson, A.J.; Fox, R.J.; Cohen, J.A. Progressive multiple sclerosis: Prospects for disease therapy, repair and restoration of function. Lancet 2017, 389, 1357–1366. [Google Scholar] [CrossRef]
- Thompson, A.J. Challenge of progressive multiple sclerosis therapy. Curr. Opin. Neurol. 2017, 30, 237–240. [Google Scholar] [CrossRef] [PubMed]
- Montalban, X.; Hauser, S.L.; Kappos, L.; Arnold, D.L.; Bar-Or, A.; Comi, G.; de Sèze, J.; Giovannoni, G.; Hartung, H.P.; Hemmer, B.; et al. Ocrelizumab versus placebo in primary progressive multiple sclerosis. N. Engl. J. Med. 2017, 376, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Exley, C. Human exposure to aluminium. Environ. Sci. Process. Impacts 2013, 15, 1807–1816. [Google Scholar] [CrossRef] [PubMed]
- Exley, C.; Mamutse, G.; Korchazhkina, O.; Pye, E.; Strekopytov, S.; Polwart, A.; Hawkins, C. Elevated urinary excretion of aluminium and iron in multiple sclerosis. Mult. Scler. 2006, 12, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.; Linhart, C.; Hawkins, C.; Exley, C. Urinary excretion of aluminium and silicon in secondary progressive multiple sclerosis. EBioMedicine 2017, 26, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Verstraeten, S.V.; Golub, M.S.; Keen, C.L.; Oteiza, P.I. Myelin is a preferential target of aluminium-mediated oxidative damage. Arch. Biochem. Biophys. 1997, 344, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Golub, M.S.; Tarrara, R.P. Morphometric studies of myelination in the spinal cord of mice exposed developmentally to aluminium. Neurotoxicology 1999, 20, 953–960. [Google Scholar] [PubMed]
- Mirza, A.; King, A.; Troakes, C.; Exley, C. Aluminium in brain tissue in familial Alzheimer’s disease. J. Trace Elem. Med. Biol. 2017, 40, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Mold, M.; Umar, D.; King, A.; Exley, C. Aluminium in brain tissue in autism. J. Trace Elem. Med. Biol. 2018, 46, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Lublin, F.D.; Rheingold, S.C.; Cohen, J.A.; Cutter, G.R.; Sørensen, P.S.; Thompson, A.J.; Wolinsky, J.S.; Balcer, L.J.; Banwell, B.; Barkhof, F.; et al. Defining the clinical course of multiple sclerosis. Neurology 2014, 83, 278–286. [Google Scholar] [CrossRef] [PubMed]
- House, E.; Esiri, M.; Forster, G.; Ince, P.G.; Exley, C. Aluminium, iron and copper in human brain tissues donated to the medical research council’s cognitive function and ageing study. Metallomics 2012, 4, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Mold, M.; Eriksson, H.; Siesjö, P.; Darabi, A.; Shardlow, E.; Exley, C. Unequivocal identification of intracellular aluminium adjuvant in a monocytic THP-1 cell line. Sci. Rep. 2014, 4, 6287. [Google Scholar] [CrossRef] [PubMed]
- Mirza, A.; King, A.; Troakes, C.; Exley, C. The identification of aluminium in human brain tissue using lumogallion and fluorescence microscopy. J. Alzheimer’s Dis. 2016, 54, 1333–1338. [Google Scholar] [CrossRef] [PubMed]
- Exley, C.; Esiri, M. Severe cerebral congophilic angiopathy coincident with increased brain aluminium in a resident of Camelford, Cornwall, UK. J. Neurol. Neurosurg. Psychiatry 2006, 77, 877–879. [Google Scholar] [CrossRef] [PubMed]
- Exley, C.; Vickers, T. Elevated brain aluminium and early onset Alzheimer’s disease in an individual occupationally exposed to aluminium: A case report. J. Med. Case Rep. 2014, 8, 41. [Google Scholar] [CrossRef] [PubMed]
- Exley, C.; House, E. Aluminium in the human brain. Monatsh. Chem. Chem. Mon. 2011, 142, 357–363. [Google Scholar] [CrossRef]
- Selmaj, K.; Pawlowska, Z.; Walczak, A.; Koziolkiewicz, W.; Raine, C.S.; Cierniewski, C.S. Corpora amylacea from multiple sclerosis brain tissue consists of aggregated neuronal cells. Acta Biochim. Pol. 2008, 55, 43–49. [Google Scholar] [PubMed]
- Augé, E.; Cabezón, I.; Pelegri, C.; Vilaplana, J. New perspectives on corpora amylacea in the human brain. Sci. Rep. 2017, 7, 41807. [Google Scholar] [CrossRef] [PubMed]
| Donor Id. | Gender | Age | MS | Frontal | Temporal | Occipital | Parietal |
|---|---|---|---|---|---|---|---|
| MS307 | Male | 55 | PPMS | 1.75(0.91) (5) | 0.46(0.60) (13) | NA | 1.76(1.28) (6) |
| MS107 | Male | 38 | RPMS | 3.41(3.54) (5) | 0.59(0.64) (3) | 0.58(0.04) (2) | 9.84(16.70) (17) |
| MS245 | Male | 64 | SPMS | 2.86(0.80) (4) | NA | NA | NA |
| MS274 | Male | 56 | RRMS | 29.14(57.92) (5) | 3.53(2.55) (13) | 0.50(0.57) (10) | 0.36(0.20) (3) |
| MS304 | Male | 52 | SPMS | 1.82(1.40) (5) | NA | 3.98(9.84) (28) | NA |
| MS313 | Male | 66 | PPMS | 0.47(0.63) (5) | NA | 3.64(4.41) (20) | 1.28(3.22) (20) |
| MS330 | Female | 59 | SPMS | 2.02(1.64) (5) | NA | NA | NA |
| MS317 | Female | 48 | SPMS | 5.44(5.73) (5) | NA | NA | NA |
| MS356 | Female | 45 | SPMS | 1.84(2.85) (5) | 1.81(1.78) (5) | 1.40(1.86) (16) | 1.51(2.44) (8) |
| MS401 | Female | 82 | SPMS | 0.65(0.65) (4) | 1.55(1.96) (16) | 5.66(9.27) (20) | 2.36(1.65) (8) |
| MS180 | Female | 44 | SPMS | 3.34(3.71) (5) | NA | NA | NA |
| MS114 | Female | 52 | SPMS | 1.68(1.25) (5) | 3.45(4.38) (17) | NA | 0.59(0.55) (6) |
| MS203 | Female | 53 | SPMS | 2.39(1.34) (5) | 0.81(0.41) (7) | NA | 3.31(4.00) (2) |
| MS234 | Female | 39 | RPMS | 6.35(8.27) (5) | NA | 0.74(0.50) (5) | 2.79(2.83) (19) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mold, M.; Chmielecka, A.; Rodriguez, M.R.R.; Thom, F.; Linhart, C.; King, A.; Exley, C. Aluminium in Brain Tissue in Multiple Sclerosis. Int. J. Environ. Res. Public Health 2018, 15, 1777. https://doi.org/10.3390/ijerph15081777
Mold M, Chmielecka A, Rodriguez MRR, Thom F, Linhart C, King A, Exley C. Aluminium in Brain Tissue in Multiple Sclerosis. International Journal of Environmental Research and Public Health. 2018; 15(8):1777. https://doi.org/10.3390/ijerph15081777
Chicago/Turabian StyleMold, Matthew, Agata Chmielecka, Maria Raquel Ramirez Rodriguez, Femia Thom, Caroline Linhart, Andrew King, and Christopher Exley. 2018. "Aluminium in Brain Tissue in Multiple Sclerosis" International Journal of Environmental Research and Public Health 15, no. 8: 1777. https://doi.org/10.3390/ijerph15081777
APA StyleMold, M., Chmielecka, A., Rodriguez, M. R. R., Thom, F., Linhart, C., King, A., & Exley, C. (2018). Aluminium in Brain Tissue in Multiple Sclerosis. International Journal of Environmental Research and Public Health, 15(8), 1777. https://doi.org/10.3390/ijerph15081777





