Aeroallergens in Canada: Distribution, Public Health Impacts, and Opportunities for Prevention
Abstract
1. Introduction
2. Review
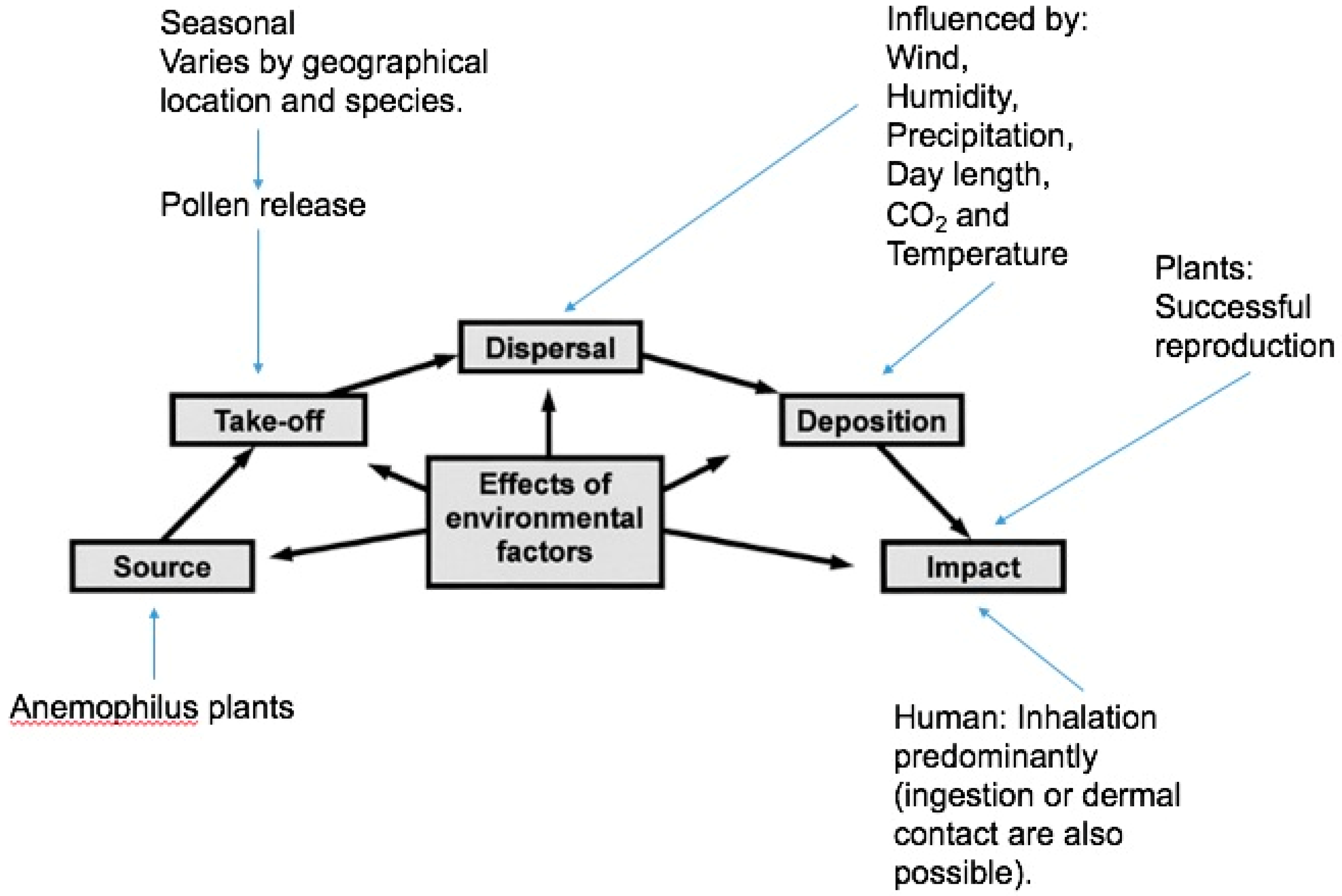
2.1. Introduction to Ambient Aeroallergens
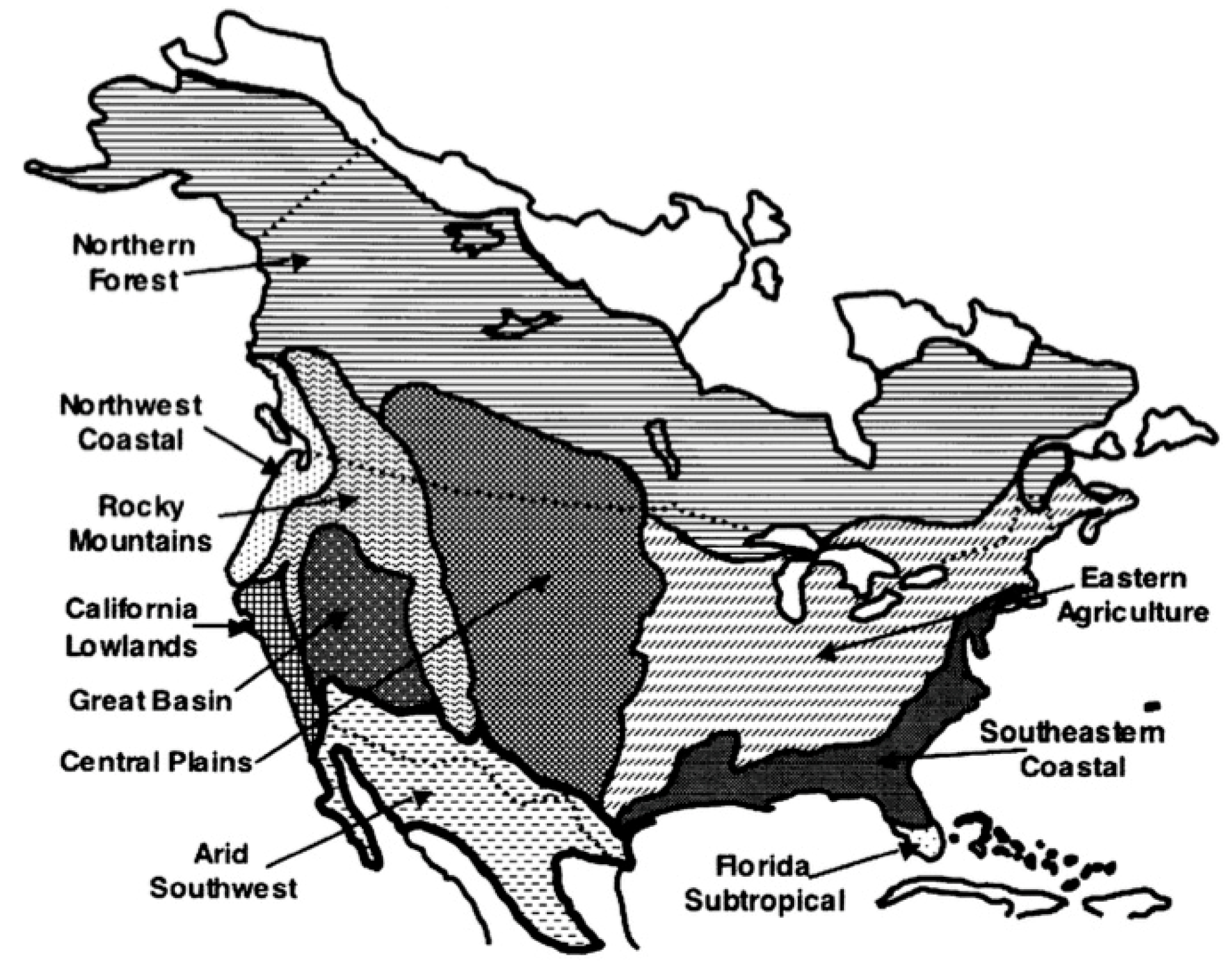
2.2. Canadian Distribution of Plant-Derived Aeroallergens
2.3. Fungal Aeroallergens
2.4. Aeroallergen Measurement and Prediction
2.5. Aeroallergen Impact on Humans
2.6. Aeroallergens and the Development of Respiratory Allergies
- (a)
- (b)
2.7. Variability in Sensitivity
2.8. Development of Respiratory Allergies: Risk/Protective Factors
2.9. Protective Factors
2.9.1. Animals
2.9.2. Breastfeeding
2.10. Risk Factors
2.10.1. Weather
2.10.2. Seasonality
2.10.3. Urbanization
2.10.4. Air Pollution
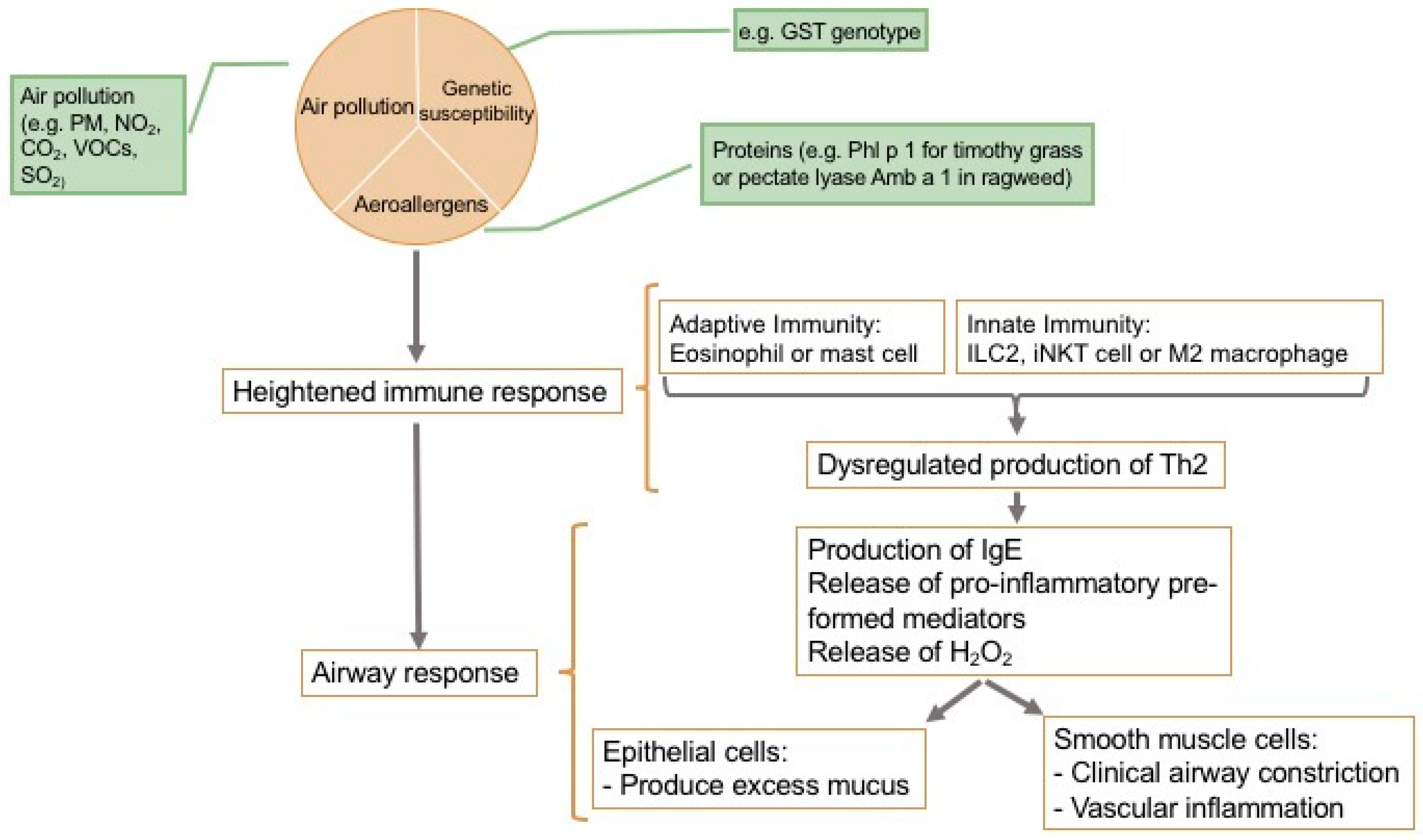
2.11. Fundamentals of the Allergic Response
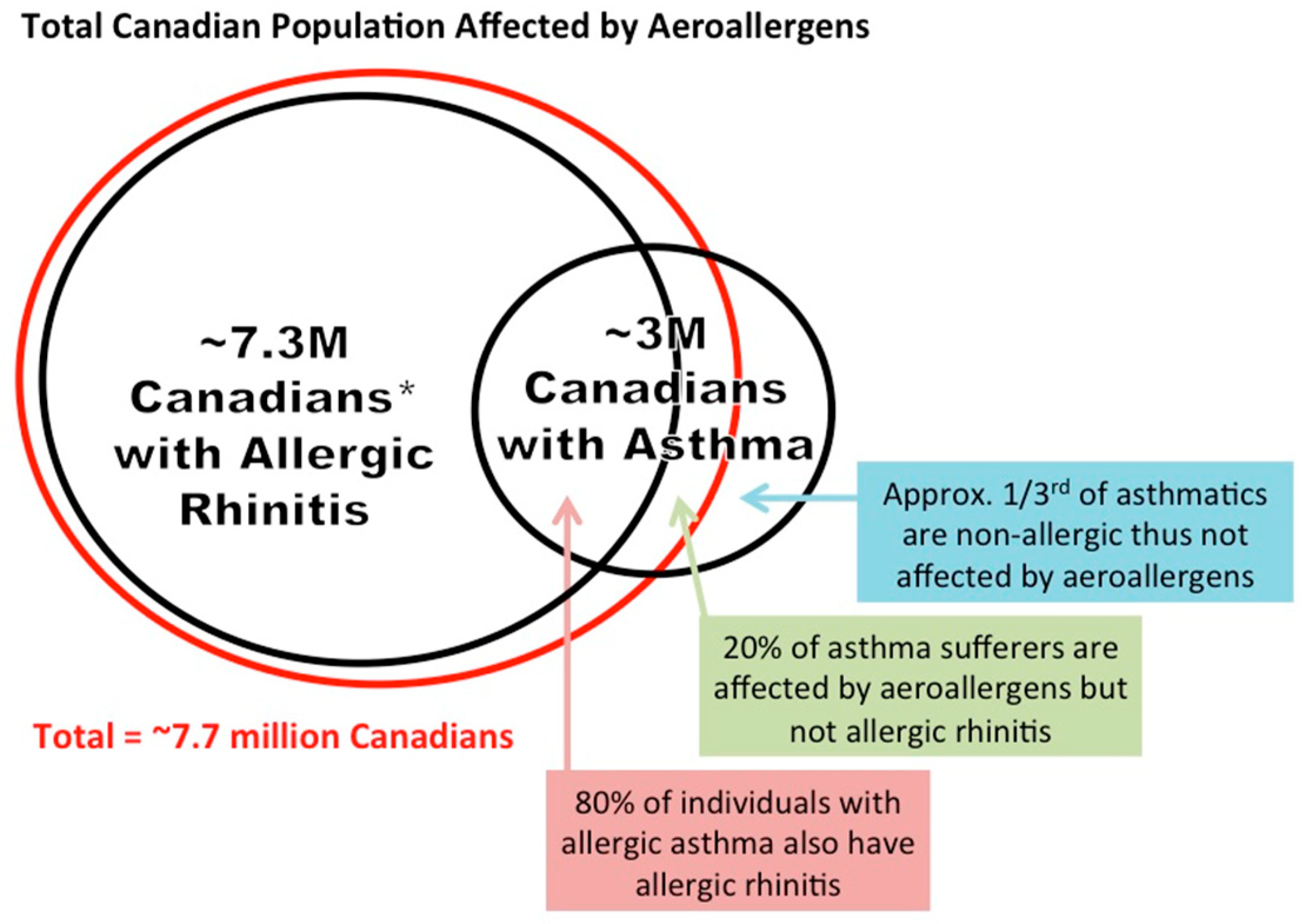
2.12. Prevalence of Respiratory Allergies in Canada
2.13. Economic Costs Associated with Respiratory Allergies
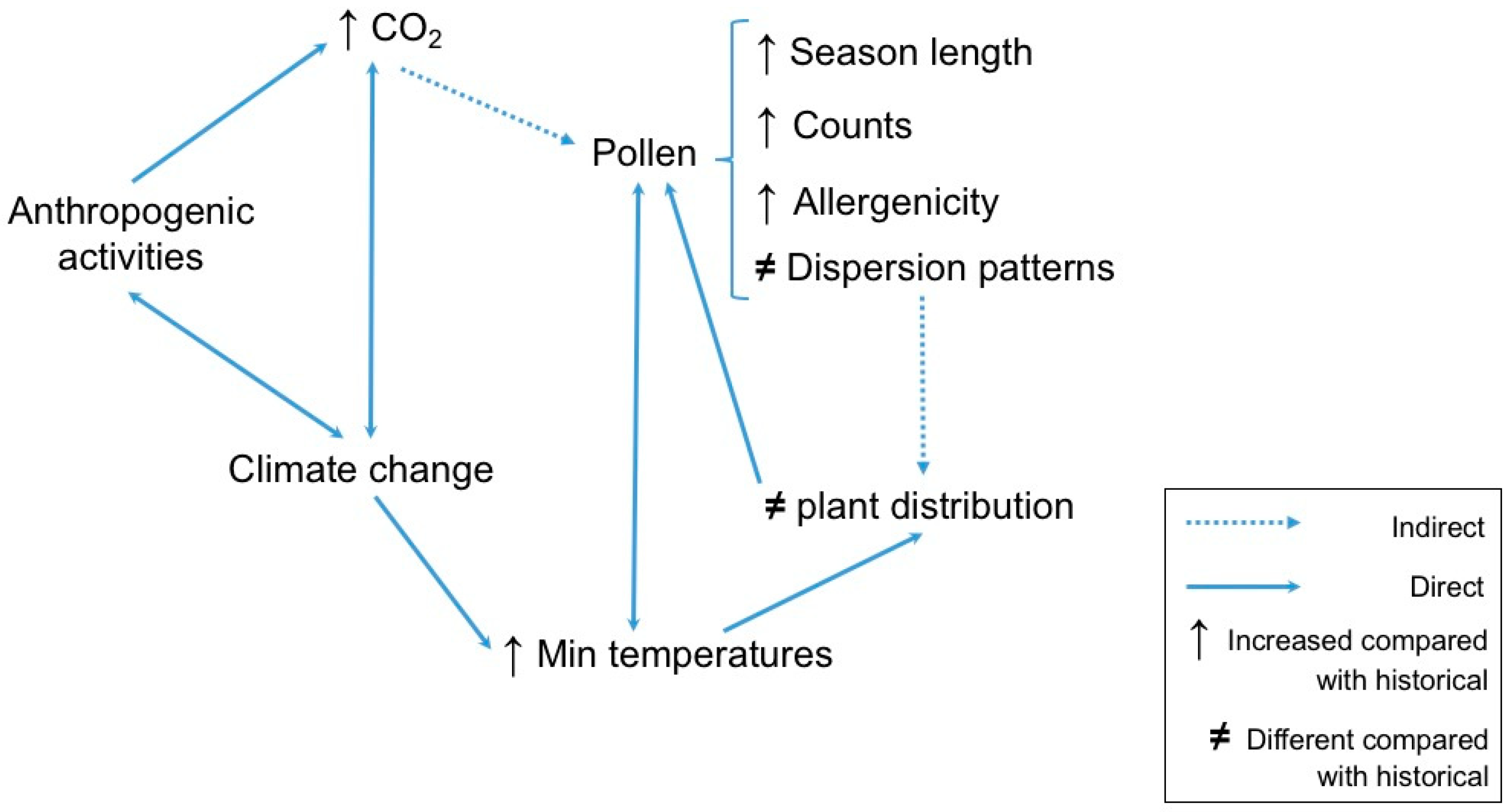
2.14. Effects of Climate Change on Aeroallergens
2.15. Environment and Health Interventions for Patients with Respiratory Allergies
3. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lau, S. Are asthma and allergies increasing in children and adolescents? Eur. J. Integr. Med. 2009, 1, 175. [Google Scholar] [CrossRef]
- Breton, M.-C.; Garneau, M.; Fortier, I.; Guay, F.; Louis, J. Relationship between climate, pollen concentrations of Ambrosia and medical consultations for allergic rhinitis in Montreal, 1994–2002. Sci. Total Environ. 2006, 370, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Rider, C.F.; Yamamoto, M.; Günther, O.P.; Hirota, J.A.; Singh, A.; Tebbutt, S.J.; Carlsten, C. Controlled diesel exhaust and allergen coexposure modulates microRNA and gene expression in humans: Effects on inflammatory lung markers. J. Allergy Clin. Immunol. 2016, 138, 1690–1700. [Google Scholar] [CrossRef] [PubMed]
- Smits, H.H.; van der Vlugt, L.E.; von Mutius, E.; Hiemstra, P.S. Childhood allergies and asthma: New insights on environmental exposures and local immunity at the lung barrier. Curr. Opin. Immunol. 2016, 42, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Cardaba, B.; Llanes, E.; Chacartegui, M.; Sastre, B.; Lopez, E.; Molla, R.; Del Pozo, V.; Florido, F.; Quiralte, J.; Palomino, P.; et al. Modulation of allergic response by gene-environment interaction: Olive pollen allergy. J. Investig. Allergol. Clin. Immunol. 2007, 17 (Suppl. 1), 31–35. [Google Scholar] [PubMed]
- Huang, F.; Yin, J.-N.; Wang, H.-B.; Liu, S.-Y.; Li, Y.-N. Association of imbalance of effector T cells and regulatory cells with the severity of asthma and allergic rhinitis in children. Allergy Asthma Proc. 2017, 38, 70–77. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.; Tang, X.-Y.; Yang, Y. Immune imbalance of regulatory T/type 2 helper cells in the pathogenesis of allergic rhinitis in children. J. Laryngol. Otol. 2016, 130, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Sears, M.R.; Greene, J.M.; Willan, A.R.; Taylor, D.R.; Flannery, E.M.; Cowan, J.O.; Herbison, G.P.; Poulton, R. Long-term relation between breastfeeding and development of atopy and asthma in children and young adults: A longitudinal study. Lancet 2002, 360, 901–907. [Google Scholar] [CrossRef]
- Bach, J.-F. The Effect of Infections on Susceptibility to Autoimmune and Allergic Diseases. N. Engl. J. Med. 2002, 347, 911–920. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.K.; Ratz, J.D.; Day, A.G.; Day, J.H. Factors that affect the allergic rhinitis response to ragweed allergen exposure. Ann. Allergy Asthma Immunol. 2010, 104, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.; Lee, J.S.; Wilson, D.; Camuso, N.; Salapatek, A. Repeated Low-dose Aerosolized Dust Mite Allergen Exposure in Asthmatic and Non-asthmatic Dust Mite Allergic patients in An Environmental Exposure Chamber Induces Specific Asthma Symptoms as well as Allergic Rhinoconjunctivitis Symptoms. J. Allergy Clin. Immunol. 2011, 127, AB20. [Google Scholar] [CrossRef]
- Rondón, C.; Fernández, J.; López, S.; Campo, P.; Doña, I.; Torres, M.J.; Mayorga, C.; Blanca, M. Nasal inflammatory mediators and specific IgE production after nasal challenge with grass pollen in local allergic rhinitis. J. Allergy Clin. Immunol. 2009, 124, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
- Gieras, A.; Focke-Tejkl, M.; Ball, T.; Verdino, P.; Hartl, A.; Thalhamer, J.; Valenta, R. Molecular determinants of allergen-induced effector cell degranulation. J. Allergy Clin. Immunol. 2007, 119, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Weber, R.W. Floristic zones and aeroallergen diversity. Immunol. Allergy Clin. N. Am. 2003, 23, 357–369. [Google Scholar] [CrossRef]
- Lafeuille, M.-H.; Gravel, J.; Figliomeni, M.; Zhang, J.; Lefebvre, P. Burden of illness of patients with allergic asthma versus non-allergic asthma. J. Asthma 2013, 50, 900–907. [Google Scholar] [CrossRef] [PubMed]
- Grant-Downton, R.; Hafidh, S.; Twell, D.; Dickinson, H.G. Small RNA Pathways Are Present and Functional in the Angiosperm Male Gametophyte. Mol. Plant 2009, 2, 500–512. [Google Scholar] [CrossRef] [PubMed]
- Farré-Armengol, G.; Filella, I.; Llusià, J.; Peñuelas, J. Pollination mode determines floral scent. Biochem. Syst. Ecol. 2015, 61, 44–53. [Google Scholar] [CrossRef]
- Park, H.J.; Lee, J.-H.; Park, K.H.; Kim, K.R.; Han, M.J.; Choe, H.; Oh, J.W.; Hong, C.S. A Six-Year Study on the Changes in Airborne Pollen Counts and Skin Positivity Rates in Korea: 2008–2013. Yonsei Med. J. 2016, 57, 714–720. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Vitale, C.; De Martino, A.; Viegi, G.; Lanza, M.; Molino, A.; Sanduzzi, A.; Vatrella, A.; Annesi-Maesano, I.; D’amato, M. Effects on asthma and respiratory allergy of Climate change and air pollution. Multidiscip. Respir. Med. 2015, 10, 39. [Google Scholar] [CrossRef] [PubMed]
- Katelaris, C.H.; Beggs, P.J. Climate change: Allergens and allergic diseases. Intern. Med. J. 2018, 48, 129–134. [Google Scholar] [CrossRef] [PubMed]
- Lacey, J. Spore dispersal—Its role in ecology and disease: The British contribution to fungal aerobiology. Mycol. Res. 1996, 100, 641–660. [Google Scholar] [CrossRef]
- Pablos, I.; Wildner, S.; Asam, C.; Wallner, M.; Gadermaier, G. Pollen Allergens for Molecular Diagnosis. Curr. Allergy Asthma Rep. 2016, 16, 31. [Google Scholar] [CrossRef] [PubMed]
- Gunawan, H.; Takai, T.; Kamijo, S.; Wang, X.L.; Ikeda, S.; Okumura, K.; Ogawa, H. Characterization of proteases, proteins, and eicosanoid-like substances in soluble extracts from allergenic pollen grains. Int. Arch. Allergy Immunol. 2008, 147, 276–288. [Google Scholar] [CrossRef] [PubMed]
- Skjøth, C.A.; Šikoparija, B.; Jäger, S. EAN-Network. Pollen Sources. In Allergenic Pollen; Springer: Dordrecht, The Netherlands, 2013; pp. 9–27. Available online: http://link.springer.com/chapter/10.1007/978-94-007-4881-1_2 (accessed on 31 May 2018).
- Ellis, A.K.; Soliman, M.; Steacy, L.; Boulay, M.-È.; Boulet, L.-P.; Keith, P.K.; Vliagoftis, H.; Waserman, S.; Neighbour, H. The Allergic Rhinitis–Clinical Investigator Collaborative (AR-CIC): Nasal allergen challenge protocol optimization for studying AR pathophysiology and evaluating novel therapies. Allergy Asthma Clin. Immunol. 2015, 11, 16. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.K.; North, M.L.; Walker, T.; Steacy, L.M. Environmental exposure unit: A sensitive, specific, and reproducible methodology for allergen challenge. Ann. Allergy Asthma Immunol. 2013, 111, 323–328. [Google Scholar] [CrossRef] [PubMed]
- White, J.F.; Bernstein, D.I. Key pollen allergens in North America. Ann. Allergy Asthma Immunol. 2003, 91, 425–435. [Google Scholar] [CrossRef]
- Léonard, R.; Wopfner, N.; Pabst, M.; Stadlmann, J.; Petersen, B.O.; Duus, J.Ø.; Himly, M.; Radauer, C.; Gadermaier, G.; Razzazzi-Fazeli, E.; et al. A new allergen from ragweed (Ambrosia artemisiifolia) with homology to art v 1 from mugwort. J. Biol. Chem. 2010, 285, 27192. [Google Scholar] [CrossRef] [PubMed]
- Oberhuber, C.; Ma, Y.; Wopfner, N.; Gadermaier, G.; Dedic, A.; Niggemann, B.; Maderegger, B.; Gruber, P.; Ferreira, F.; Scheiner, O.; et al. Prevalence of IgE-Binding to Art v 1, Art v 4 and Amb a 1 in Mugwort-Allergic Patients. Int. Arch. Allergy Immunol. 2008, 145, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Asero, R.; Wopfner, N.; Gruber, P.; Gadermaier, G.; Ferreira, F. Artemisia and Ambrosia hypersensitivity: Co-sensitization or co-recognition? Clin. Exp. Allergy 2006, 36, 658–665. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, N.E.; Wihl, J.A.; Arrendal, H.; Strandhede, S.O. Tree pollen allergy. II. Sensitization to various tree pollen allergens in Sweden. A multi-centre study. Allergy 1984, 39, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, S. News—Pollen Season Is upon US. Who’s Getting it Bad This Year? The Weather Network. Available online: https://www.theweathernetwork.com/news/articles/pollen-season-is-upon-us-whos-getting-it-bad-this-year/66940 (accessed on 31 May 2018).
- Weinberger, K.R.; Kinney, P.L.; Robinson, G.S.; Sheehan, D.; Kheirbek, I.; Matte, T.D.; Lovasi, G.S. Levels and determinants of tree pollen in New York City. J. Expo. Sci. Environ. Epidemiol. 2018, 28, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Hugg, T.T.; Hjort, J.; Antikainen, H.; Rusanen, J.; Tuokila, M.; Korkonen, S.; Weckström, J.; Jaakkola, M.S.; Jaakkola, J.J. Urbanity as a determinant of exposure to grass pollen in Helsinki Metropolitan area, Finland. PLoS ONE 2017, 12, e0186348. [Google Scholar] [CrossRef] [PubMed]
- Rogers, C.A. An aeropalynological study of metropolitan Toronto. Aerobiologia 1997, 13, 243–257. [Google Scholar] [CrossRef]
- Solomon, W.R. Airborne pollen: A brief life. J. Allergy Clin. Immunol. 2002, 109, 895–900. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, M.K.; Swanson, M.C.; Reed, C.E.; Yunginger, J.W. Immunochemical quantitation of airborne short ragweed, Alternaria, antigen, E.; and Alt-I allergens: A two-year prospective study. J. Allergy Clin. Immunol. 1983, 72, 40–45. [Google Scholar] [CrossRef]
- Agarwal, M.K.; Swanson, M.C.; Reed, C.E.; Yunginger, J.W. Airborne ragweed allergens: Association with various particle sizes and short ragweed plant parts. J. Allergy Clin. Immunol. 1984, 74, 687–693. [Google Scholar] [CrossRef]
- Grote, M.; Vrtala, S.; Niederberger, V.; Wiermann, R.; Valenta, R.; Reichelt, R. Release of allergen-bearing cytoplasm from hydrated pollen: A mechanism common to a variety of grass (Poaceae) species revealed by electron microscopy. J. Allergy Clin. Immunol. 2001, 108, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Portnoy, J.; Barnes, C. Clinical relevance of spore and pollen counts. Immunol. Allergy Clin. N. Am. 2003, 23, 389–410. [Google Scholar] [CrossRef]
- Levetin, E.; Horner, W.E.; Scott, J.A. Taxonomy of Allergenic Fungi. J. Allergy Clin. Immunol. Pract. 2016, 4, 375–385. [Google Scholar] [CrossRef] [PubMed]
- Hibbett, D.S.; Binder, M.; Bischoff, J.F.; Blackwell, M.; Cannon, P.F.; Eriksson, O.E.; Huhndorf, S.; James, T.; Kirk, P.M.; Lücking, R.; et al. A higher-level phylogenetic classification of the Fungi. Mycol. Res. 2007, 111, 509–547. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, D.J.; Hibbett, D.S.; Lutzoni, F.; Spatafora, J.W.; Vilgalys, R. The search for the fungal tree of life. Trends Microbiol. 2009, 17, 488–497. [Google Scholar] [CrossRef] [PubMed]
- Aukrust, L. Mold allergy. Introduction. Clin. Rev. Allergy 1992, 10, 147–151. [Google Scholar] [PubMed]
- Burge, H.A. Classification of the fungi. Clin. Rev. Allergy 1992, 10, 153–163. [Google Scholar] [PubMed]
- Taylor, J.; Jacobson, D.; Fisher, M. The Evolution of Asexual Fungi: Reproduction, Speciation and Classification. Annu. Rev. Phytopathol. 1999, 37, 197–246. [Google Scholar] [CrossRef] [PubMed]
- Crameri, R.; Zeller, S.; Glaser, A.G.; Vilhelmsson, M.; Rhyner, C. Cross-reactivity among fungal allergens: A clinically relevant phenomenon? Mycoses 2009, 52, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.B. Pollen and Fungal Aeroallergens Associated with Allergy and Asthma in India. Glob. J. Immunol. Allerg. Dis. 2014, 2, 19–28. [Google Scholar] [CrossRef]
- Akiyama, K. The role of fungal allergy in bronchial asthma. Nihon Ishinkin Gakkai Zasshi Jpn. J. Med. Mycol. 2000, 41, 149–155. [Google Scholar] [CrossRef]
- Weber, R.W. Outdoor aeroallergen sampling: Not all that simple. Ann. Allergy Asthma Immunol. 2007, 98, 505–506. [Google Scholar] [CrossRef]
- Weber, R.W. Pollen Identification. Ann. Allergy Asthma Immunol. 1998, 80, 141–148. [Google Scholar] [CrossRef]
- Crouzy, B.; Stella, M.; Konzelmann, T.; Calpini, B.; Clot, B. All-Optical automatic pollen identification: Towards an operational system. Atmos. Environ. 2016, 140, 202–212. [Google Scholar] [CrossRef]
- Levetin, E.; Van de Water, P.K. Pollen count forecasting. Immunol. Allergy Clin. N. Am. 2003, 23, 423–442. [Google Scholar] [CrossRef]
- Hjort, J.; Hugg, T.T.; Antikainen, H.; Rusanen, J.; Sofiev, M.; Kukkonen, J.; Jaakkola, M.S.; Jaakkola, J.J. Fine-Scale Exposure to Allergenic Pollen in the Urban Environment: Evaluation of Land Use Regression Approach. Environ. Health Perspect. 2016, 124, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Esser, C. Principles of the Immune System: Players and Organization. In Environmental Influences on the Immune System; Springer: Vienna, Austria, 2016; pp. 1–17. Available online: http://link.springer.com/chapter/10.1007/978-3-7091-1890-0_1 (accessed on 31 May 2018).
- Graham-Rowe, D. Lifestyle: When allergies go west. Nature 2011, 479, S2–S4. [Google Scholar] [CrossRef]
- Cecchi, L. Introduction. In Allergenic Pollen; Springer: Dordrecht, The Netherlands, 2013; pp. 1–7. Available online: http://link.springer.com/chapter/10.1007/978-94-007-4881-1_1 (accessed on 31 May 2018).
- De Weger, L.A.; Bergmann, K.C.; Rantio-Lehtimäki, A.; Dahl, Å.; Buters, J.; Déchamp, C.; Belmonte, J.; Thibaudon, M.; Cecchi, L.; Besancenot, J.P.; et al. Impact of Pollen. In Allergenic Pollen; Sofiev, M., Bergmann, K.-C., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 161–215. Available online: http://link.springer.com/10.1007/978-94-007-4881-1_6 (accessed on 31 May 2018).
- Cantani, A. Allergic Rhinitis. In Pediatric Allergy, Asthma and Immunology; Springer: Berlin, Germany, 2008; pp. 875–910. Available online: http://link.springer.com.proxy.lib.sfu.ca/chapter/10.1007/978-3-540-33395-1_12 (accessed on 18 July 2016).
- Vaitla, P.M.; Drewe, E. Identifying the culprit allergen in seasonal allergic rhinitis. Practitioner 2011, 255, 27–31. [Google Scholar] [PubMed]
- Hesse, M.; Halbritter, H.; Zetter, R.; Weber, M.; Buchner, R.; Frosch-Radivo, A.; Ulrich, S.; Zetter, R. Pollen Morphology. In Pollen Terminology an Illustrated Handbook; Springer: Wien, Austria, 2009. [Google Scholar]
- Szema, A.M. Asthma, Hay Fever, Pollen, and Climate Change. In Global Climate Change and Public Health; Pinkerton, K.E., Rom, W.N., Eds.; Springer: New York, NY, USA, 2014; pp. 155–165. Available online: http://link.springer.com.proxy.lib.sfu.ca/chapter/10.1007/978-1-4614-8417-2_9 (accessed on 15 July 2016).
- Green, B.J.; Beezhold, D.H.; Gallinger, Z.; Barron, C.S.; Melvin, R.; Bledsoe, T.A.; Kashon, M.L.; Sussman, G.L. Allergic sensitization in Canadian chronic rhinosinusitis patients. Allergy Asthma Clin. Immunol. 2014, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Cakmak, S.; Dales, R.E.; Coates, F. Does air pollution increase the effect of aeroallergens on hospitalization for asthma? J. Allergy Clin. Immunol. 2011, 129, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Sandin, A.; Bjorksten, B.; Braback, L. Development of atopy and wheezing symptoms in relation to heredity and early pet keeping in a Swedish birth cohort. Pediatr. Allergy Immunol. 2004, 15, 316–322. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Vitale, C.; Lanza, M.; Molino, A.; D’Amato, M. Climate change, air pollution, and allergic respiratory diseases: An update. Curr. Opin. Allergy Clin. Immunol. 2016, 16, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Eder, W.; Ege, M.J.; von Mutius, E. The Asthma Epidemic. N. Engl. J. Med. 2006, 355, 2226–2235. [Google Scholar] [CrossRef] [PubMed]
- Armentia, A.; Banuelos, C.; Arranz, M.L.; Del Villar, V.; Martin-Santos, J.M.; Gil, F.J.; Gil, F.M.; Vega, J.M.; Callejo, A.; Paredes, C. Early introduction of cereals into children’s diets as a risk-factor for grass pollen asthma. Clin. Exp. Allergy 2001, 31, 1250–1255. [Google Scholar] [CrossRef] [PubMed]
- Halken, S. Prevention of allergic disease in childhood: Clinical and epidemiological aspects of primary and secondary allergy prevention. Pediatr. Allergy Immunol. 2004, 15, 9–32. [Google Scholar] [CrossRef] [PubMed]
- Bardei, F.; Bouziane, H.; Kadiri, M.; Rkiek, B.; Tebay, A.; Saoud, A. Skin sensitisation profiles to inhalant allergens for patients in Tetouan city (North West of Morocco). Rev. Pneumol. Clin. 2016, 72, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Duffy, D.L.; Mitchell, C.A.; Martin, N.G. Genetic and environmental risk factors for asthma: A cotwin-control study. Am. J. Respir. Crit. Care Med. 1998, 157, 840–845. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Balashazy, I.; Farkas, A.; Sarkany, Z.; Hofmann, W.; Czitrovszky, A.; Dobos, E. Quantification of airway deposition of intact and fragmented pollens. Int. J. Environ. Health Res. 2011, 21, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, G.; Sun, Y.; Li, J.; Tang, N.; Dong, L. Airway wall thickness of allergic asthma caused by weed pollen or house dust mite assessed by computed tomography. Respir. Med. 2015, 109, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Von Mutius, E. Gene-environment interactions in asthma. J. Allergy Clin. Immunol. 2009, 123, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Vinhas, R.; Cortes, L.; Cardoso, I.; Mendes, V.M.; Manadas, B.; Todo-Bom, A.; Pires, E.; Verissimo, P. Pollen proteases compromise the airway epithelial barrier through degradation of transmembrane adhesion proteins and lung bioactive peptides. Allergy 2011, 66, 1088–1098. [Google Scholar] [CrossRef] [PubMed]
- Tran, M.M.; Lefebvre, D.L.; Dharma, C.; Dai, D.; Lou, W.Y.W.; Subbarao, P.; Becker, A.B.; Mandhane, P.J.; Turvey, S.E.; Sears, M.R.; et al. Predicting the atopic march: Results from the Canadian Healthy Infant Longitudinal Development Study. J. Allergy Clin. Immunol. 2017, 141, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, R.L.; Wahn, U.; Bergmann, K.E. The allergy march: From food to pollen. Environ. Toxicol. Pharmacol. 1997, 4, 79–83. [Google Scholar] [CrossRef]
- Warner, J.A.; Jones, C.A.; Jones, A.C.; Warner, J.O. Prenatal origins of allergic disease. J. Allergy Clin. Immunol. 2000, 105, S493–S498. [Google Scholar] [CrossRef]
- Leung, R.C.; Carlin, J.B.; Burdon, J.G.; Czarny, D. Asthma, allergy and atopy in Asian immigrants in Melbourne. Med. J. Aust. 1994, 161, 418–425. [Google Scholar] [PubMed]
- Ventura, M.T.; Munno, G.; Giannoccaro, F.; Accettura, F.; Chironna, M.; Lama, R.; Hoxha, M.; Panetta, V.; Ferrigno, L.; Rosmini, F.; et al. Allergy, asthma and markers of infections among Albanian migrants to Southern Italy. Allergy 2004, 59, 632–636. [Google Scholar] [CrossRef] [PubMed]
- Beggs, P.J. Impacts of climate change on aeroallergens: Past and future. Clin. Exp. Allergy 2004, 34, 1507–1513. [Google Scholar] [CrossRef] [PubMed]
- De Jong, N.W.; Vermeulen, A.M.; Gerth van Wijk, R.; de Groot, H. Occupational allergy caused by flowers. Allergy 1998, 53, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Anguita, J.L.; Palacios, L.; Ruiz-Valenzuela, L.; Bartolome, B.; Lopez-Urbano, M.J.; Saenz de San Pedro, B.; Cano, E.; Quiralte, J. An occupational respiratory allergy caused by Sinapis alba pollen in olive farmers. Allergy 2007, 62, 447–450. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Ortega, P.; Bartolome, B.; Enrique, E.; Gaig, P.; Richart, C. Allergy to Diplotaxis erucoides pollen: Occupational sensitization and cross-reactivity with other common pollens. Allergy 2001, 56, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Hermanides, H.K.; Lahey-de Boer, A.M.; Zuidmeer, L.; Guikers, C.; van Ree, R.; Knulst, A.C. Brassica oleracea pollen, a new source of occupational allergens. Allergy 2006, 61, 498–502. [Google Scholar] [CrossRef] [PubMed]
- Swierczyniska-Machura, D.; Krakowiak, A.; Palczynski, C. Occupational allergy caused by ornamental plants. Med. Pr. 2006, 57, 359–364. [Google Scholar] [PubMed]
- Miesen, W.M.A.J.; Van der Heide, S.; Kerstjens, H.A.M.; Dubois, A.E.J.; de Monchy, J.G.R. Occupational asthma due to IgE mediated allergy to the flower Molucella laevis (Bells of Ireland). Occup. Environ. Med. 2003, 60, 701–703. [Google Scholar] [CrossRef] [PubMed]
- Eire, M.A.; Pineda, F.; Losada, S.V.; de la Cuesta, C.G.; Villalva, M.M. Occupational rhinitis and asthma due to cedroarana (Cedrelinga catenaeformis Ducke) wood dust allergy. J. Investig. Allergol. Clin. Immunol. 2006, 16, 385–387. [Google Scholar] [PubMed]
- Blanco, C.; Ortega, N.; Castillo, R.; Alvarez, M.; Dumpierrez, A.G.; Carrillo, T. Carica papaya pollen allergy. Ann. Allergy Asthma Immunol. 1998, 81, 171–175. [Google Scholar] [CrossRef]
- Barbato, A.; Pisetta, F.; Norbiato, M.; Ragusa, A.; Mesirca, P.; Pesenti, P.; Marcer, G. Influence of aeroallergens on bronchial reactivity in children sensitized to grass pollens. Ann. Allergy 1986, 56, 138–141. [Google Scholar] [PubMed]
- Tovey, E.R.; Almqvist, C.; Li, Q.; Crisafulli, D.; Marks, G.B. Nonlinear relationship of mite allergen exposure to mite sensitization and asthma in a birth cohort. J. Allergy Clin. Immunol. 2008, 122, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Lebel, B.; Bousquet, J.; Morel, A.; Chanal, I.; Godard, P.; Michel, F.B. Correlation between symptoms and the threshold for release of mediators in nasal secretions during nasal challenge with grass-pollen grains. J. Allergy Clin. Immunol. 1988, 82, 869–877. [Google Scholar] [CrossRef]
- Twaroch, T.E.; Curin, M.; Valenta, R.; Swoboda, I. Mold allergens in respiratory allergy: From structure to therapy. Allergy Asthma Immunol. Res. 2015, 7, 205–220. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Spieksma, F.T. Aerobiologic and clinical aspects of mould allergy in Europe. Allergy 1995, 50, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Bernardis, P.; Agnoletto, M.; Puccinelli, P.; Parmiani, S.; Pozzan, M. Injective versus sublingual immunotherapy in Alternaria tenuis allergic patients. J. Investig. Allergol. Clin. Immunol. 1996, 6, 55–62. [Google Scholar] [PubMed]
- Baxter, D.M.; Perkins, J.L.; McGhee, C.R.; Seltzer, J.M. A regional comparison of mold spore concentrations outdoors and inside “clean” and “mold contaminated” Southern California buildings. J. Occup. Environ. Hyg. 2005, 2, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Nolte, H.; Hébert, J.; Berman, G.; Gawchik, S.; White, M.; Kaur, A.; Liu, N.; Lumry, W.; Maloney, J. Randomized controlled trial of ragweed allergy immunotherapy tablet efficacy and safety in North American adults. Ann. Allergy Asthma Immunol. 2013, 110, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Weichenthal, S.; Lavigne, E.; Villeneuve, P.J.; Reeves, F. Airborne Pollen Concentrations and Emergency Room Visits for Myocardial Infarction: A Multicity Case-Crossover Study in Ontario, Canada. Am. J. Epidemiol. 2016, 183, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Durham, S.R.; Emminger, W.; Kapp, A.; de Monchy, J.G.; Rak, S.; Scadding, G.K.; Wurtzen, P.A.; Andersen, J.S.; Tholstrup, B.; Riis, B.; et al. SQ-standardized sublingual grass immunotherapy: Confirmation of disease modification 2 years after 3 years of treatment in a randomized trial. J. Allergy Clin. Immunol. 2012, 129, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Connell, J.T. Quantitative intranasal pollen challenges. 3. The priming effect in allergic rhinitis. J. Allergy 1969, 43, 33–44. [Google Scholar] [CrossRef]
- Jacobs, R.L.; Andrews, C.P.; Ramirez, D.A.; Rather, C.G.; Harper, N.; Jimenez, F.; Martinez, H.; Manoharan, M.; Carrillo, A.; Gerardi, M.; et al. Symptom dynamics during repeated serial allergen challenge chamber exposures to house dust mite. J. Allergy Clin. Immunol. 2015, 135, 1071–1075. [Google Scholar] [CrossRef] [PubMed]
- O’Hehir, R.E.; Varese, N.P.; Deckert, K.; Zubrinich, C.M.; van Zelm, M.C.; Rolland, J.M.; Hew, M. Epidemic Thunderstorm Asthma Protection with Five-grass Pollen Tablet Sublingual Immunotherapy. Am. J. Respir. Crit. Care Med. 2018, 198, 126–128. [Google Scholar] [CrossRef] [PubMed]
- Subbarao, P.; Becker, A.; Brook, J.R.; Daley, D.; Mandhane, P.J.; Miller, G.E.; Turvey, S.E.; Sears, M.R. Epidemiology of asthma: Risk factors for development. Expert Rev. Clin. Immunol. 2009, 5, 77–95. [Google Scholar] [CrossRef] [PubMed]
- Clifford, R.L.; Jones, M.J.; MacIsaac, J.L.; McEwen, L.M.; Goodman, S.J.; Mostafavi, S.; Kobor, M.S.; Carlsten, C. Inhalation of diesel exhaust and allergen alters human bronchial epithelium DNA methylation. J. Allergy Clin. Immunol. 2017, 139, 112–121. [Google Scholar] [CrossRef] [PubMed]
- Gershwin, L.J. Infectious and Environmental Triggers of Asthma. In Current Clinical Practice: Bronchial Asthma: A Guide for Practical Understanding and Treatment, 5th ed.; Humana Press Inc.: Totowa, NJ, USA, 2006. [Google Scholar]
- Parameswaran, K.; Hildreth, A.J.; Taylor, I.K.; Keaney, N.P.; Bansal, S.K. Predictors of asthma severity in the elderly: Results of a community survey in Northeast England. J. Asthma 1999, 36, 613–618. [Google Scholar] [CrossRef] [PubMed]
- Anyo, G.; Brunekreef, B.; de Meer, G.; Aarts, F.; Janssen, N.A.H.; van Vliet, P. Early, current and past pet ownership: Associations with sensitization, bronchial responsiveness and allergic symptoms in school children. Clin. Exp. Allergy 2002, 32, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Galli, L.; Facchetti, S.; Raffetti, E.; Donato, F.; D’Anna, M. Respiratory diseases and allergic sensitization in swine breeders: A population-based cross-sectional study. Ann. Allergy Asthma Immunol. 2015, 115, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Waser, M.; von Mutius, E.; Riedler, J.; Nowak, D.; Maisch, S.; Carr, D.; Eder, W.; Tebow, G.; Schierl, R.; Schreuer, M.; et al. Exposure to pets, and the association with hay fever, asthma, and atopic sensitization in rural children. Allergy 2005, 60, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Gassner-Bachmann, M.; Wuthrich, B. Farmers’ children suffer less from hay fever and asthma. Dtsch. Med. Wochenschr. 2000, 125, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Bjerg, A.; Ekerljung, L.; Eriksson, J.; Naslund, J.; Sjolander, S.; Ronmark, E.; Dahl, Å.; Holmberg, K.; Wennergren, G.; Torén, K.; et al. Increase in pollen sensitization in Swedish adults and protective effect of keeping animals in childhood. Clin. Exp. Allergy 2016, 46, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Leynaert, B.; Neukirch, C.; Jarvis, D.; Chinn, S.; Burney, P.; Neukirch, F. Does living on a farm during childhood protect against asthma, allergic rhinitis, and atopy in adulthood? Am. J. Respir. Crit. Care Med. 2001, 164, 1829–1834. [Google Scholar] [CrossRef] [PubMed]
- Braun-Fahrlander, C.; Gassner, M.; Grize, L.; Neu, U.; Sennhauser, F.H.; Varonier, H.S.; Vuille, J.C.; Wüthrich, B. Prevalence of hay fever and allergic sensitization in farmer’s children and their peers living in the same rural community. SCARPOL team. Swiss Study on Childhood Allergy and Respiratory Symptoms with Respect to Air Pollution. Clin. Exp. Allergy 1999, 29, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Stein, M.M.; Hrusch, C.L.; Gozdz, J.; Igartua, C.; Pivniouk, V.; Murray, S.E.; Ledford, J.G.; Marques dos Santos, M.; Anderson, R.L.; Metwali, N.; et al. Innate Immunity and Asthma Risk in Amish and Hutterite Farm Children. N. Engl. J. Med. 2016, 375, 411–421. [Google Scholar] [CrossRef] [PubMed]
- Klopp, A.; Vehling, L.; Becker, A.B.; Subbarao, P.; Mandhane, P.J.; Turvey, S.E.; Lefebvre, D.L.; Sears, M.R.; Azad, M.B.; CHILD Study Investigators. Modes of Infant Feeding and the Risk of Childhood Asthma: A Prospective Birth Cohort Study. J. Pediatr. 2017, 190, 192–199. [Google Scholar] [CrossRef] [PubMed]
- Dogaru, C.M.; Nyffenegger, D.; Pescatore, A.M.; Spycher, B.D.; Kuehni, C.E. Breastfeeding and Childhood Asthma: Systematic Review and Meta-Analysis. Am. J. Epidemiol. 2014, 179, 1153–1167. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.B.; Becker, A.B.; Guttman, D.S.; Sears, M.R.; Scott, J.A.; Kozyrskyj, A.L. Gut microbiota diversity and atopic disease: Does breast-feeding play a role? J. Allergy Clin. Immunol. 2013, 131, 247–248. [Google Scholar] [CrossRef] [PubMed]
- Rosas, I.; McCartney, H.A.; Payne, R.W.; Calderón, C.; Lacey, J.; Chapela, R.; Ruiz-Velazco, S. Analysis of the relationships between environmental factors (aeroallergens, air pollution, and weather) and asthma emergency admissions to a hospital in Mexico City. Allergy 2007, 53, 394–401. [Google Scholar] [CrossRef]
- North, M.L.; Soliman, M.; Walker, T.; Steacy, L.M.; Ellis, A.K. Controlled Allergen Challenge Facilities and Their Unique Contributions to Allergic Rhinitis Research. Curr. Allergy Asthma Rep. 2015, 15, 11. [Google Scholar] [CrossRef] [PubMed]
- Barnes, C.; Pacheco, F.; Landuyt, J.; Hu, F.; Portnoy, J. Hourly variation of airborne ragweed pollen in Kansas City. Ann. Allergy Asthma Immunol. 2001, 86, 166–171. [Google Scholar] [CrossRef]
- Weber, R.W. Meteorologic variables in aerobiology. Aerobiology 2003, 23, 411–422. [Google Scholar] [CrossRef]
- Barnes, C.; Pacheco, F.; Landuyt, J.; Hu, F.; Portnoy, J. The effect of temperature, relative humidity and rainfall on airborne ragweed pollen concentrations. Aerobiologia 2001, 17, 61–68. [Google Scholar] [CrossRef]
- Andrew, E.; Nehme, Z.; Bernard, S.; Smith, K. 6 Characteristics of thunderstorm asthma EMS attendances in Victoria, Australia. BMJ Open 2017, 7 (Suppl. 3), A2–A3. [Google Scholar]
- Cockcroft, D.W.; Davis, B.E.; Blais, C.M. Thunderstorm asthma: An allergen-induced early asthmatic response. Ann. Allergy Asthma Immunol. 2018, 120, 120–123. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kronborg, C.; O’Hehir, R.E.; Hew, M. Who’s at risk of thunderstorm asthma? The ryegrass pollen trifecta and lessons learnt from the Melbourne thunderstorm epidemic. Respir. Med. 2017, 132, 146–148. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Annesi Maesano, I.; Molino, A.; Vitale, C.; D’Amato, M. Thunderstorm-related asthma attacks. J. Allergy Clin. Immunol. 2017, 139, 1786–1787. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Holgate, S.T.; Pawankar, R.; Ledford, D.K.; Cecchi, L.; Al-Ahmad, M.; Al-Enezi, F.; Al-Muhsen, S.; Ansotegui, I.; Baena-Cagnani, C.E.; et al. Meteorological conditions, climate change, new emerging factors, and asthma and related allergic disorders. A statement of the World Allergy Organization. World Allergy Organ. J. 2015, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Cecchi, L. Effects of climate change on environmental factors in respiratory allergic diseases. Clin. Exp. Allergy 2008, 38, 1264–1274. [Google Scholar] [CrossRef] [PubMed]
- Valero, A.; Justicia, J.L.; Anton, E.; Dordal, T.; Fernandez-Parra, B.; Lluch, M.; Montoro, J.; Navarro, A.M. Epidemiology of allergic rhinitis caused by grass pollen or house-dust mites in Spain. Am. J. Rhinol. Allergy 2011, 25, e123–e128. [Google Scholar] [CrossRef] [PubMed]
- Canonica, G.W.; Ciprandi, G.; Pesce, G.P.; Buscaglia, S.; Paolieri, F.; Bagnasco, M. ICAM-1 on epithelial cells in allergic subjects: A hallmark of allergic inflammation. Int. Arch. Allergy Immunol. 1995, 107, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Ogi, K.; Takabayashi, T.; Sakashita, M.; Susuki, D.; Yamada, T.; Manabe, Y.; Fujieda, S. Effect of Asian sand dust on Japanese cedar pollinosis. Auris Nasus Larynx 2014, 41, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Weeke, E.R. Epidemiology of allergic diseases in children. Rhinol. Suppl. 1992, 13, 5–12. [Google Scholar] [PubMed]
- D’Amato, G.; Liccardi, G.; D’Amato, M.; Holgate, S. Environmental risk factors and allergic bronchial asthma. Clin. Exp. Allergy 2005, 35, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Carinanos, P.; Sanchez-Mesa, J.A.; Prieto-Baena, J.C.; Lopez, A.; Guerra, F.; Moreno, C.; Domínguez, E.; Galan, C. Pollen allergy related to the area of residence in the city of Cordoba, south-west Spain. J. Environ. Monit. 2002, 4, 734–738. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, T.B.; Thomsen, S.F.; Ulrik, C.S.; Fenger, M.; Nepper-Christensen, S.; Backer, V. Season of birth and risk of atopic disease among children and adolescents. J. Asthma 2007, 44, 257–260. [Google Scholar] [CrossRef] [PubMed]
- Vovolis, V.; Grigoreas, C.; Galatas, I.; Vourdas, D. Is month of birth a risk factor for subsequent development of pollen allergy in adults? Allergy Asthma Proc. 1999, 20, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Lowe, A.J.; Olsson, D.; Bråbäck, L.; Forsberg, B. Pollen exposure in pregnancy and infancy and risk of asthma hospitalization—A register based cohort study. Allergy Asthma Clin. Immunol. 2012, 8, 17. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Bergmann, K.C.; Cecchi, L.; Annesi-Maesano, I.; Sanduzzi, A.; Liccardi, G.; Vitale, C.; Stanziola, A.; D’Amato, M. Climate change and air pollution: Effects on pollen allergy and other allergic respiratory diseases. Allergo J. Int. 2014, 23, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Guinea, J.; Peláez, T.; Alcalá, L.; Bouza, E. Outdoor environmental levels of Aspergillus spp. conidia over a wide geographical area. Med. Mycol. 2006, 44, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Bosch-Cano, F.; Bernard, N.; Sudre, B.; Gillet, F.; Thibaudon, M.; Richard, H.; Badot, P.M.; Ruffaldi, P. Human exposure to allergenic pollens: A comparison between urban and rural areas. Environ. Res. 2011, 111, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Reid, C.E.; Gamble, J.L. Aeroallergens, allergic disease, and climate change: Impacts and adaptation. EcoHealth 2009, 6, 458–470. [Google Scholar] [CrossRef] [PubMed]
- Ziska, L.H.; Gebhard, D.E.; Frenz, D.A.; Faulkner, S.; Singer, B.D.; Straka, J.G. Cities as harbingers of climate change: Common ragweed, urbanization, and public health. J. Allergy Clin. Immunol. 2003, 111, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Majd, A.; Chehregani, A.; Moin, M.; Gholami, M.; Kohno, S.; Nabe, T.; Shariatzade, M.A. The Effects of Air Pollution on Structures, Proteins and Allergenicity of Pollen Grains. Int. J. Aerobiol. 2004, 20, 111–118. [Google Scholar] [CrossRef]
- Pasqualini, S.; Tedeschini, E.; Frenguelli, G.; Wopfner, N.; Ferreira, F.; D’Amato, G.; Ederli, L. Ozone affects pollen viability and NAD(P)H oxidase release from Ambrosia artemisiifolia pollen. Environ. Pollut. 2011, 159, 2823–2830. [Google Scholar] [CrossRef] [PubMed]
- Raynor, G.S.; Ogden, E.C.; Hayes, J.V. Dispersion and Deposition of Ragweed Pollen from Experimental Sources. J. Appl. Meteorol. 1970, 9, 885–895. [Google Scholar] [CrossRef]
- Ying, Z.; Tie, X.; Li, G. Sensitivity of ozone concentrations to diurnal variations of surface emissions in Mexico City: A WRF/Chem modeling study. Atmos. Environ. 2009, 43, 851–859. [Google Scholar] [CrossRef]
- Lazić, L.; Urošević, M.A.; Mijić, Z.; Vuković, G.; Ilić, L. Traffic contribution to air pollution in urban street canyons: Integrated application of the OSPM, moss biomonitoring and spectral analysis. Atmos. Environ. 2016, 141, 347–360. [Google Scholar] [CrossRef]
- Masiol, M.; Hopke, P.K.; Felton, H.D.; Frank, B.P.; Rattigan, O.V.; Wurth, M.J.; LaDuke, G.H. Analysis of major air pollutants and submicron particles in New York City and Long Island. Atmos. Environ. 2017, 148, 203–214. [Google Scholar] [CrossRef]
- Takaro, T.K.; Knowlton, K.; Balmes, J.R. Climate change and respiratory health: Current evidence and knowledge gaps. Expert Rev. Respir. Med. 2013, 7, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Romieu, I.; Moreno-Macias, H.; London, S.J. Gene by Environment Interaction and Ambient Air Pollution. Proc. Am. Thorac. Soc. 2010, 7, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Peltre, G. Inter-relationship between allergenic pollens and air pollution. Allergie Immunol. 1998, 30, 324–326. [Google Scholar]
- Zhang, X.; Hirota, J.A.; Yang, C.; Carlsten, C. Effect of GST variants on lung function following diesel exhaust and allergen co-exposure in a controlled human crossover study. Free Radic. Biol. Med. 2016, 96, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Alexis, N.E.; Zhou, H.; Lay, J.C.; Harris, B.; Hernandez, M.L.; Lu, T.-S.; Bromberg, P.A.; Diaz-Sanchez, D.; Devlin, R.B.; Kleeberger, S.R.; et al. The glutathione-S-transferase Mu 1 null genotype modulates ozone-induced airway inflammation in human subjects. J. Allergy Clin. Immunol. 2009, 124, 1222–1228. [Google Scholar] [CrossRef] [PubMed]
- Boldogh, I. ROS generated by pollen NADPH oxidase provide a signal that augments antigen-induced allergic airway inflammation. J. Clin. Investig. 2005, 115, 2169–2179. [Google Scholar] [CrossRef] [PubMed]
- Traidl-Hoffmann, C.; Kasche, A.; Menzel, A.; Jakob, T.; Thiel, M.; Ring, J.; Behrendt, H. Impact of pollen on human health: More than allergen carriers? Int. Arch. Allergy Immunol. 2003, 131, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Abou Chakra, O.; Rogerieux, F.; Poncet, P.; Sutra, J.-P.; Peltre, G.; Senechal, H.; Lacroix, G. Ability of pollen cytoplasmic granules to induce biased allergic responses in a rat model. Int. Arch. Allergy Immunol. 2011, 154, 128–136. [Google Scholar] [CrossRef] [PubMed]
- North, M.L.; Jones, M.J.; Macisaac, J.L.; Morin, A.M.; Steacy, L.M.; Gregor, A.; Kobor, M.S.; Ellis, A.K. Blood and nasal epigenetics correlate with allergic rhinitis symptom development in the environmental exposure unit. Allergy 2018, 73, 196–205. [Google Scholar] [CrossRef] [PubMed]
- Holtzman, M.J.; Byers, D.E.; Alexander-Brett, J.; Wang, X. The role of airway epithelial cells and innate immune cells in chronic respiratory disease. Nat. Rev. Immunol. 2014, 14, 686–698. [Google Scholar] [CrossRef] [PubMed]
- Broms, K.; Norback, D.; Eriksson, M.; Sundelin, C.; Svardsudd, K. Prevalence and co-occurrence of parentally reported possible asthma and allergic manifestations in pre-school children. BMC Public Health 2013, 13, 764. [Google Scholar] [CrossRef] [PubMed]
- Bachert, C.; Vignola, A.M.; Gevaert, P.; Leynaert, B.; Van Cauwenberge, P.; Bousquet, J. Allergic rhinitis, rhinosinusitis, and asthma: One airway disease. Immunol. Allergy Clin. N. Am. 2004, 24, 19–43. [Google Scholar] [CrossRef]
- Bousquet, J.; Van Cauwenberge, P.; Khaltaev, N.; Aria Workshop Group; World Health Organization. Allergic rhinitis and its impact on asthma. J. Allergy Clin. Immunol. 2001, 108, S147–S334. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Bouchard, J.; Renzix, P. The link between allergic rhinitis and asthma: A role for antileukotrienes? Can. Respir. J. 2008, 15, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Egan, M.; Bunyavanich, S. Allergic rhinitis: The “Ghost Diagnosis” in patients with asthma. Asthma Res. Pract. 2015, 1, 8. [Google Scholar] [CrossRef] [PubMed]
- Ciprandi, G.; Buscaglia, S.; Scordamaglia, A.; Canonica, G.W. Allergen-specific conjunctival challenge in asthma. An additional diagnostic tool to define sensitization. Int. Arch. Allergy Immunol. 1997, 112, 247–250. [Google Scholar] [CrossRef] [PubMed]
- Boulay, M.-E.; Boulet, L.-P. Influence of natural exposure to pollens and domestic animals on airway responsiveness and inflammation in sensitized non-asthmatic subjects. Int. Arch. Allergy Immunol. 2002, 128, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Cantani, A. Asthma. In Pediatric Allergy, Asthma and Immunology; Springer: Berlin/Heidelberg, Germany, 2008; pp. 725–873. Available online: http://link.springer.com.proxy.lib.sfu.ca/chapter/10.1007/978-3-540-33395-1_11 (accessed on 18 July 2016).
- Hesselmar, B.; Bergin, A.-M.; Park, H.; Hahn-Zoric, M.; Eriksson, B.; Hanson, L.-A.; Padyukov, L. Interleukin-4 receptor polymorphisms in asthma and allergy: Relation to different disease phenotypes. Acta Paediatr. 2010, 99, 399–403. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Asthma. World Health Organization: Geneva, Switzerland, 2017. Available online: http://www.who.int/news-room/fact-sheets/detail/asthma (accessed on 31 May 2018).
- Hertzen, L.; Haahtela, T. Signs of reversing trends in prevalence of asthma. Allergy 2005, 60, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Keith, P.K.; Desrosiers, M.; Laister, T.; Schellenberg, R.R.; Waserman, S. The burden of allergic rhinitis (AR) in Canada: Perspectives of physicians and patients. Allergy Asthma Clin. Immunol. 2012, 8, 7. [Google Scholar] [CrossRef] [PubMed]
- Government of Canada, S.C. Asthma, by Age Group and Sex (Percent). 2016. Available online: https://www.statcan.gc.ca/tables-tableaux/sum-som/l01/cst01/health49b-eng.htm (accessed on 31 May 2018).
- Gershon, A.S.; Guan, J.; Wang, C.; To, T. Trends in Asthma Prevalence and Incidence in Ontario, Canada, 1996–2005: A Population Study. Am. J. Epidemiol. 2010, 172, 728–736. [Google Scholar] [CrossRef] [PubMed]
- Asher, M.I.; Montefort, S.; Björkstén, B.; Lai, C.K.; Strachan, D.P.; Weiland, S.K.; Williams, H.; ISAAC Phase Three Study Group. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet 2006, 368, 733–743. [Google Scholar] [CrossRef]
- Ismaila, A.S.; Sayani, A.P.; Marin, M.; Su, Z. Clinical, economic, and humanistic burden of asthma in Canada: A systematic review. BMC Pulm. Med. 2013, 13, 70. [Google Scholar] [CrossRef] [PubMed]
- Statistics Canada. Population by Sex and Age Group, Population as of July 1. Statistics Canada, CANSIM, Table 051-0001. 2016. Available online: http://www.statcan.gc.ca/tables-tableaux/sum-som/l01/cst01/demo10a-eng.htm (accessed on 18 October 2016).
- Lavigne, E.; Gasparrini, A.; Stieb, D.M.; Chen, H.; Yasseen, A.S.; Crighton, E.; To, T.; Weichenthal, S.; Villeneuve, P.J.; Cakmak, S.; et al. Maternal Exposure to Aeroallergens and the Risk of Early Delivery. Epidemiology 2017, 28, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Dales, R.E.; Cakmak, S.; Judek, S.; Coates, F. Tree pollen and hospitalization for asthma in urban Canada. Int. Arch. Allergy Immunol. 2008, 146, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Dales, R.E.; Cakmak, S.; Burnett, R.T.; Judek, S.; Coates, F.; Brook, J.R. Influence of ambient fungal spores on emergency visits for asthma to a regional children’s hospital. Am. J. Respir. Crit. Care Med. 2000, 162, 2087–2090. [Google Scholar] [CrossRef] [PubMed]
- Héguy, L.; Garneau, M.; Goldberg, M.S.; Raphoz, M.; Guay, F.; Valois, M.-F. Associations between grass and weed pollen and emergency department visits for asthma among children in Montreal. Environ. Res. 2008, 106, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Cakmak, S.; Dales, R.E.; Burnett, R.T.; Judek, S.; Coates, F.; Brook, J.R. Effect of airborne allergens on emergency visits by children for conjunctivitis and rhinitis. Lancet 2002, 359, 947–948. [Google Scholar] [CrossRef]
- Dales, R.E.; Cakmak, S.; Judek, S.; Dann, T.; Coates, F.; Brook, J.R.; Burnett, R.T. Influence of outdoor aeroallergens on hospitalization for asthma in Canada. J. Allergy Clin. Immunol. 2004, 113, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Tavakoli, H.; Fitzgerald, J.M.; Chen, W.; Lynd, L.; Kendzerska, T.; Aaron, S.; Gershon, A.; Marra, C.; Sadatsafavi, M.; Canadian Respiratory Research Network. Ten-year trends in direct costs of asthma: A population-based study. Allergy 2017, 72, 291–299. [Google Scholar] [CrossRef] [PubMed]
- To, T.; Cicutto, L.; Degani, N.; McLimont, S.; Beyene, J. Can a community evidence-based asthma care program improve clinical outcomes? A longitudinal study. Med. Care 2008, 46, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Canadian Institute for Health Information. Asthma Emergency Department Visits: Volume and Median Length of Stay. 2015. Available online: http://indicatorlibrary.cihi.ca/display/HSPIL/Asthma+Emergency+Department+Visits%3A+Volume+and+Median+Length+of+Stay (accessed on 31 May 2018).
- Theriault, L.; Hermus, G.; Goldfarb, D.; Stonebridge, C.; Bounajm, B. Cost Risk Analysis for Chronic Lung Disease in Canada. 2012. Available online: http://www.conferenceboard.ca/e-library/abstract.aspx?did=4585&AspxAutoDetectCookieSupport=1 (accessed on 31 May 2018).
- Ariano, R.; Canonica, G.W.; Passalacqua, G. Possible role of climate changes in variations in pollen seasons and allergic sensitizations during 27 years. Ann. Allergy Asthma Immunol. 2010, 104, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Intergovernmental Panel on Climate Change. Climate Change 2014 Impacts, Adaptation, and Vulnerability Part A: Global and Sectoral Aspects; Cambridge University Press: New York, NY, USA, 2014.
- Ziska, L.H.; Beggs, P.J. Anthropogenic climate change and allergen exposure: The role of plant biology. J. Allergy Clin. Immunol. 2012, 129, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Bonofiglio, T.; Orlandi, F.; Ruga, L.; Romano, B.; Fornaciari, M. Climate change impact on the olive pollen season in Mediterranean areas of Italy: Air quality in late spring from an allergenic point of view. Environ. Monit. Assess. 2013, 185, 877–890. [Google Scholar] [CrossRef] [PubMed]
- Rice, M.B.; Thurston, G.D.; Balmes, J.R.; Pinkerton, K.E. Climate Change. A Global Threat to Cardiopulmonary Health. Am. J. Respir. Crit. Care Med. 2014, 189, 512–519. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Pawankar, R.; Vitale, C.; Lanza, M.; Molino, A.; Stanziola, A.; Vatrella, A.; D’Amato, M. Climate Change and Air Pollution: Effects on Respiratory Allergy. Allergy Asthma Immunol. Res. 2016, 8, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Emberlin, J.; Detandt, M.; Gehrig, R.; Jaeger, S.; Nolard, N.; Rantio-Lehtimaki, A. Responses in the start of Betula (birch) pollen seasons to recent changes in spring temperatures across Europe. Int. J. Biometeorol. 2002, 46, 159–170. [Google Scholar] [PubMed]
- Stach, A.; Garcia-Mozo, H.; Prieto-Baena, J.C.; Czarnecka-Operacz, M.; Jenerowicz, D.; Silny, W.; Galán, C. Prevalence of Artemisia species pollinosis in western Poland: Impact of climate change on aerobiological trends, 1995–2004. J. Investig. Allergol. Clin. Immunol. 2007, 17, 39–47. [Google Scholar] [PubMed]
- Takaro, T.; Henderson, S. Climate change and the new normal for cardiorespiratory disease. Can. Respir. J. 2015, 22, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Ahdoot, S.; Pacheco, S.E. Global Climate Change and Children’s Health. Pediatrics, 26 October 2015. Available online: http://pediatrics.aappublications.org/content/early/2015/10/21/peds.2015-3233.abstract (accessed on 31 March 2017).
- Ariano, R.; Berra, D.; Chiodini, E.; Ortolani, V.; Cremonte, L.G.; Mazzarello, M.G.; Galdi, E.; Calosso, C.; Ciprandi, G. Ragweed allergy: Pollen count and sensitization and allergy prevalence in two Italian allergy centers. Allergy Rhinol. 2015, 6, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.E.; Jacobson, K.W.; House, J.M.; Glovsky, M.M. Links between pollen, atopy and the asthma epidemic. Int. Arch. Allergy Immunol. 2007, 144, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Shea, K.M.; Truckner, R.T.; Weber, R.W.; Peden, D.B. Climate change and allergic disease. J. Allergy Clin. Immunol. 2008, 122, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Singer, B.D.; Ziska, L.H.; Frenz, D.A.; Gebhard, D.E.; Straka, J.G. Increasing Amb a 1 content in common ragweed (Ambrosia artemisiifolia) pollen as a function of rising atmospheric CO2 concentration. Funct. Plant Biol. 2005, 32, 667–670. [Google Scholar] [CrossRef]
- Sommer, J.; Plaschke, P.; Poulsen, L.K. Allergic disease—Pollen allergy and climate change. Ugeskr. Laeger 2009, 171, 3184–3187. [Google Scholar] [PubMed]
- Lougheed, M.D.; Lemière, C.; Dell, S.D.; Ducharme, F.M.; FitzGerald, J.M.; Leigh, R.; Licskai, C.; Rowe, B.H.; Bowie, D.; Becker, A.; et al. Canadian Thoracic Society Asthma Management Continuum—2010 Consensus Summary for Children Six Years of Age and Over, and Adults. Can. Respir. J. 2010, 17, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Johnston, F.H.; Wheeler, A.J.; Williamson, G.J.; Campbell, S.L.; Jones, P.J.; Koolhof, I.S.; Lucani, C.; Cooling, N.B.; Bowman, D.M.J.S. Using smartphone technology to reduce health impacts from atmospheric environmental hazards. Environ. Res. Lett. 2018, 13, 044019. [Google Scholar] [CrossRef]
- Magnan, A.; Romanet, S.; Vervloet, D. Asthma and allergy. Rev. Prat. 2001, 51, 511–516. [Google Scholar] [PubMed]
- D’Amato, G.; Liccardi, G.; D’Amato, M.; Cazzola, M. The role of outdoor air pollution and climatic changes on the rising trends in respiratory allergy. Respir. Med. 2001, 95, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Palma-Gomez, S.; Gonzalez-Diaz, S.N.; Arias-Cruz, A.; Macias-Weinmann, A.; Amaro-Vivian, L.E.; Perez-Vanzzini, R.; Gutiérrez-Mujica, J.J.; Yong-Rodríguez, A. Effects of reforestation on tree pollen sensitization in inhabitants of Nuevo Leon, Mexico. Rev. Alerg. Méx. 2014, 61, 162–167. [Google Scholar] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sierra-Heredia, C.; North, M.; Brook, J.; Daly, C.; Ellis, A.K.; Henderson, D.; Henderson, S.B.; Lavigne, É.; Takaro, T.K. Aeroallergens in Canada: Distribution, Public Health Impacts, and Opportunities for Prevention. Int. J. Environ. Res. Public Health 2018, 15, 1577. https://doi.org/10.3390/ijerph15081577
Sierra-Heredia C, North M, Brook J, Daly C, Ellis AK, Henderson D, Henderson SB, Lavigne É, Takaro TK. Aeroallergens in Canada: Distribution, Public Health Impacts, and Opportunities for Prevention. International Journal of Environmental Research and Public Health. 2018; 15(8):1577. https://doi.org/10.3390/ijerph15081577
Chicago/Turabian StyleSierra-Heredia, Cecilia, Michelle North, Jeff Brook, Christina Daly, Anne K. Ellis, Dave Henderson, Sarah B. Henderson, Éric Lavigne, and Tim K. Takaro. 2018. "Aeroallergens in Canada: Distribution, Public Health Impacts, and Opportunities for Prevention" International Journal of Environmental Research and Public Health 15, no. 8: 1577. https://doi.org/10.3390/ijerph15081577
APA StyleSierra-Heredia, C., North, M., Brook, J., Daly, C., Ellis, A. K., Henderson, D., Henderson, S. B., Lavigne, É., & Takaro, T. K. (2018). Aeroallergens in Canada: Distribution, Public Health Impacts, and Opportunities for Prevention. International Journal of Environmental Research and Public Health, 15(8), 1577. https://doi.org/10.3390/ijerph15081577




