Antibiotic Resistance Characterization of Environmental E. coli Isolated from River Mula-Mutha, Pune District, India
Abstract
1. Introduction
2. Materials and Methods
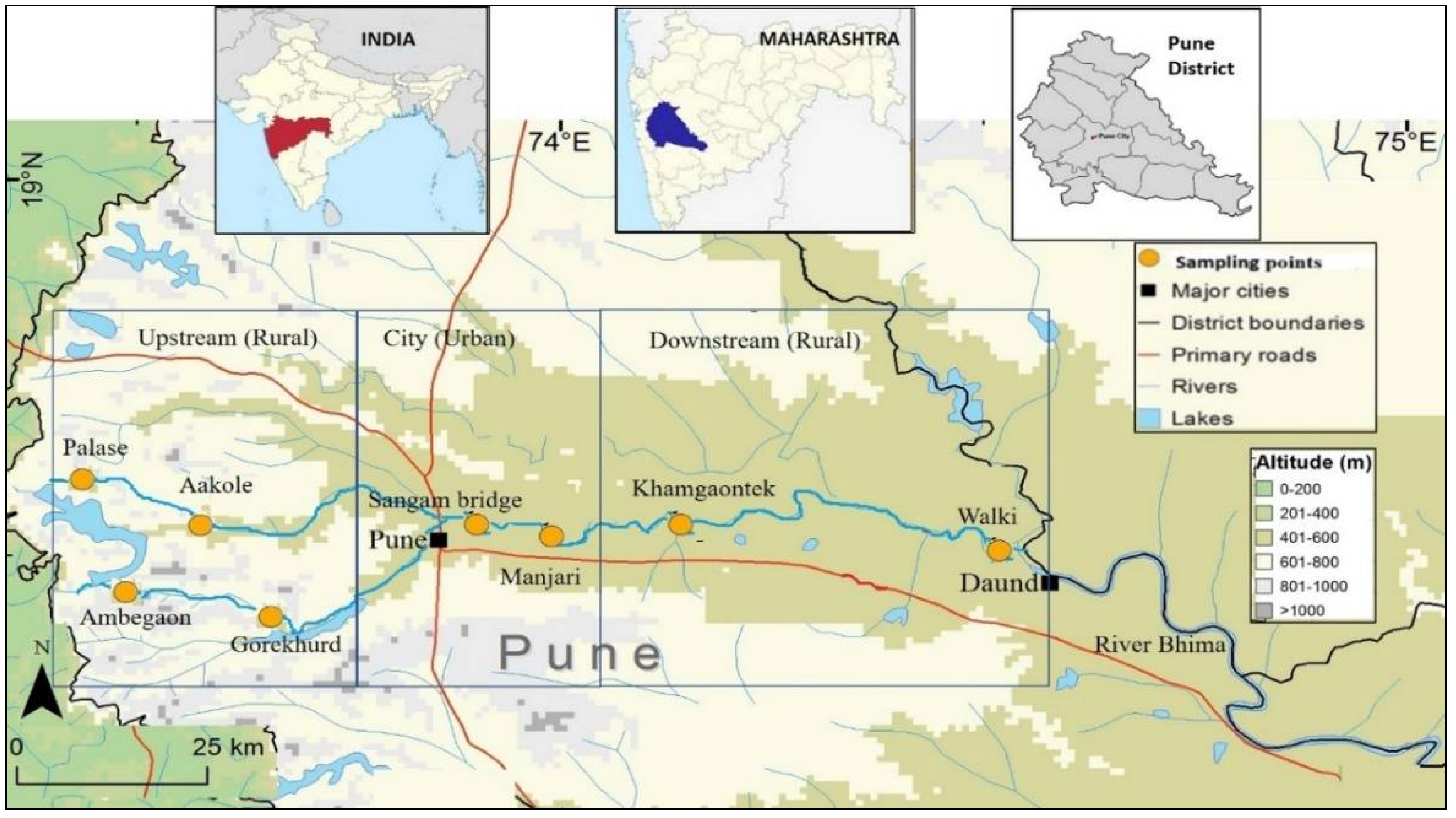
2.1. Study Area and Sample Collection
2.2. Isolation of Dual Drug-Resistant (DDR) E. coli
2.3. Antibiotic Susceptibility Testing
2.4. Genetic Determinants of Antibiotic Resistance and Horizontal Gene Transfer (HGT) in DDR E. coli (HGT)

3. Results
3.1. Antibiotic Resistance Profile of DDR E. coli
3.2. Detection of Genes Responsible for Fluoroquinolone and Cephalosporin Resistance in DDR E. coli
3.3. Horizontal Gene Transfer (HGT) of Antibiotic Resistance through Integron 1 and 2 in DDR E. coli
4. Discussion
4.1. Antibiotic Resistance Profile of DDR E. coli
4.2. Genotypic Antibiotic Resistance in DDR E. coli
4.3. Horizontal Gene Transfer of Antibiotic Resistance in DDR E. coli
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Drexler, M. What You Need to Know about Infectious Disease; National Academies Press: Washington, DC, USA, 2010. [Google Scholar]
- Dowling, P.M. Antimicrobial therapy of urinary tract infections. Can. Vet. J. 1996, 37, 438–441. [Google Scholar] [PubMed]
- Nigam, A.; Gupta, D.; Sharma, A. Treatment of infectious disease: Beyond antibiotics. Microbiol. Res. 2014, 169, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Meyer, C.N.; Skinhøj, P.; Prag, J. Bacteremia in HIV-positive and AIDS patients: Incidence, species distribution, risk-factors, outcome, and influence of long-term prophylactic antibiotic treatment. Scand. J. Infect. Dis. 1994, 26, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Wiström, J.; Norrby, S.R.; Myhre, E.B.; Eriksson, S.; Granström, G.; Lagergren, L.; Englund, G.; Nord, C.E.; Svenungsson, B. Frequency of antibiotic-associated diarrhoea in 2462 antibiotic-treated hospitalized patients: A prospective study. J. Antimicrob. Chemother. 2001, 47, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Gabrielle, D. Study on Antibiotic Resistance Finds Concern, but No ‘Epidemic’. Available online: http://search.excite.com/excite.309.01.in/search/web?q=antimicrobial+resistance&cid=333014653&ad.segment=excite.309.01.in&ad.searchtermmatchtype=p&ad.device=c&ad.keyword=antimicrobial%20resistance&ad.ismobile=&ad.creative=75728903473435&ad.querystring=Global%20epidemic%20of%20antimicrobial%20resistance&ad.matchtype=bb (accessed on 22 May 2018).
- WHO & WTO. WTO Agreements and Public Health: A Joint Study by The WHO and The WTO Secretariat; World Health Organization, World Trade Organization Secretariat: Geneva, Switzerland, 2002. [Google Scholar]
- Laxminarayan, R.; Duse, A.; Wattal, C.; Zaidi, A.K.; Wertheim, H.F.; Sumpradit, N.; Vlieghe, E.; Hara, G.L.; Gould, I.M.; Goossens, H. Antibiotic resistance—The need for global solutions. Lancet Infect. Dis. 2013, 13, 1057–1098. [Google Scholar] [CrossRef]
- WHO. Antimicrobial Resistance: Global Report on Surveillance; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- EC. Amr: A Major European and Global Challenge. Available online: https://ec.europa.eu/health/amr/sites/amr/files/amr_factsheet_en.pdf (accessed on 22 May 2018).
- EMA. Baytril 10% Oral Solution. Available online: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/veterinary/referrals/Baytril_10/vet_referral_000065.jsp&mid=WC0b01ac05805c5170 (accessed on 6 June 2018).
- Emmerson, A.; Jones, A. The quinolones: Decades of development and use. J. Antimicrob. Chemother. 2003, 51, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Porter, G.; Grills, N. Medication misuse in India: A major public health issue in India. J. Public Health 2015, 38, e150–e157. [Google Scholar] [CrossRef] [PubMed]
- Travasso, C. India draws a red line under antibiotic misuse. Br. Med. J. 2016, 352. [Google Scholar] [CrossRef] [PubMed]
- Chandy, S.J. Consequences of irrational use of antibiotics. Indian J. Med. Ethics 2008, 5, 174–175. [Google Scholar] [CrossRef] [PubMed]
- FSSAI. Antibiotic Substances in Food Products and FSSAI Regulations. Available online: https://foodsafetyhelpline.com/2018/01/antibiotic-substances-in-food-products-and-fssai-regulations/ (accessed on 20 May 2018).
- Madlen, D.; Rahul, M. A Game of Chicken: How Indian Poultry Farming Is Creating Global Superbugs. Available online: https://www.thebureauinvestigates.com/stories/2018-01-30/a-game-of-chicken-how-indian-poultry-farming-is-creating-global-superbugs (accessed on 22 May 2018).
- Tripathi, K.; Sharma, A. Seasonal variation in bacterial contamination of water sources with antibiotic resistant faecal coliforms in relation to pollution. J. Appl Nat. Sci. 2011, 3, 298–302. [Google Scholar] [CrossRef]
- WHO. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- GOI. National Action Plan for Antimicrobial Resistance (nap_amr); Government of India: New Delhi, India, 2017.
- De Nisco, N.J.; Rivera-Cancel, G.; Orth, K. The biochemistry of sensing: Enteric pathogens regulate type iii secretion in response to environmental and host cues. mBio 2018, 9, e02122-17. [Google Scholar] [CrossRef] [PubMed]
- Roberts, M.C.; Soge, O.O.; No, D. Comparison of multi-drug resistant environmental methicillin-resistant staphylococcus aureus isolated from recreational beaches and high touch surfaces in built environments. Front. Microbiol. 2013, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Yu, T.; Zhang, Y.; Yang, M.; Li, Z.; Liu, M.; Qi, R. Antibiotic resistance characteristics of environmental bacteria from an oxytetracycline production wastewater treatment plant and the receiving river. Appl. Environ. Microbiol. 2010, 76, 3444–3451. [Google Scholar] [CrossRef] [PubMed]
- Akiba, M.; Sekizuka, T.; Yamashita, A.; Kuroda, M.; Fujii, Y.; Murata, M.; Lee, K.-I.; Joshua, D.I.; Balakrishna, K.; Bairy, I. Distribution and relationships of antimicrobial resistance determinants among extended-spectrum-cephalosporin-resistant or carbapenem-resistant escherichia coli isolates from rivers and sewage treatment plants in India. Antimicrob. Agents. Chemother. (Bethesda) 2016, 60, 2972–2980. [Google Scholar] [CrossRef] [PubMed]
- Prado, T.; Bruni, A.d.C.; Barbosa, M.R.F.; Bonanno, V.M.S.; Garcia, S.C.; Sato, M.I.Z. Distribution of human fecal marker gb-124 bacteriophages in urban sewage and reclaimed water of São Paulo city, Brazil. J. Water Health 2017, 16, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Marathe, N.P.; Regina, V.R.; Walujkar, S.A.; Charan, S.S.; Moore, E.R.; Larsson, D.J.; Shouche, Y.S. A treatment plant receiving waste water from multiple bulk drug manufacturers is a reservoir for highly multi-drug resistant integron-bearing bacteria. PLoS ONE 2013, 8, e77310. [Google Scholar] [CrossRef] [PubMed]
- Ram, S.; Vajpayee, P.; Shanker, R. Prevalence of multi-antimicrobial-agent resistant, shiga toxin and enterotoxin producing Escherichia coli in surface waters of River Ganga. Environ. Sci. Technol. 2007, 41, 7383–7388. [Google Scholar] [CrossRef] [PubMed]
- Azam, M.; Jan, A.T.; Haq, Q.M. Blactx-m-152, a novel variant of ctx-m-group-25, identified in a study performed on the prevalence of multidrug resistance among natural inhabitants of River Yamuna, India. Front. Microbiol. 2016, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Skariyachan, S.; Mahajanakatti, A.B.; Grandhi, N.J.; Prasanna, A.; Sen, B.; Sharma, N.; Vasist, K.S.; Narayanappa, R. Environmental monitoring of bacterial contamination and antibiotic resistance patterns of the fecal coliforms isolated from cauvery river, a major drinking water source in Karnataka, India. Environ. Monit. Assess. 2015, 187, 279. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, A. Pune Plans More STPs While Existing Plants under Perform and There Is No Attempt to Fix Them. Available online: https://sandrp.in/2016/08/29/can-thousand-crore-loan-from-jica-save-punes-rivers-understanding-punes-river-pollution-part-i/ (accessed on 6 June 2018).
- D’costa, V.M.; McGrann, K.M.; Hughes, D.W.; Wright, G.D. Sampling the antibiotic resistome. Science 2006, 311, 374–377. [Google Scholar] [CrossRef] [PubMed]
- Gibson, M.K.; Forsberg, K.J.; Dantas, G. Improved annotation of antibiotic resistance determinants reveals microbial resistomes cluster by ecology. ISME J. 2015, 9, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Munck, C.; Albertsen, M.; Telke, A.; Ellabaan, M.; Nielsen, P.H.; Sommer, M.O. Limited dissemination of the wastewater treatment plant core resistome. Nat. Commun. 2015, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- von Wintersdorff, C.J.; Penders, J.; van Niekerk, J.M.; Mills, N.D.; Majumder, S.; van Alphen, L.B.; Savelkoul, P.H.; Wolffs, P.F. Dissemination of antimicrobial resistance in microbial ecosystems through horizontal gene transfer. Front. Microbiol. 2016, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Svara, F.; Rankin, D.J. The evolution of plasmid-carried antibiotic resistance. BMC Evol. Biol. 2011, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Conway, T.; Cohen, P.S. Commensal and pathogenic escherichia coli metabolism in the gut. Microbiol. Spectr. 2015, 3, 1–24. [Google Scholar] [CrossRef]
- Ajayi, A.; Oluyege, A.; Olowe, O.; Famurewa, O. Antibiotic resistance among commensal e. Coli isolated from faeces from cattle Ado-Ekiti Nigeria. J. Anim. Veternary Adv. 2011, 10, 174–179. [Google Scholar] [CrossRef]
- Bush, K.; Jacoby, G.A.; Medeiros, A.A. A functional classification scheme for beta-lactamases and its correlation with molecular structure. Antimicrob. Agents. Chemother. 1995, 39, 1211. [Google Scholar] [CrossRef] [PubMed]
- Bush, K.; Jacoby, G.A. Updated functional classification of β-lactamases. Antimicrob. Agents Chemother. (Bethesda) 2010, 54, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277. [Google Scholar]
- APHA. Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 1999. [Google Scholar]
- BIS. Drinking Water—Specification Is 10500: 2012; Bureau of Indian Standards: New Delhi, India, 2012. [Google Scholar]
- Keche, Y.; Yegnanarayan, R.; Bhoyar, S.; Agrawal, R.; Chavan, R.; Mahendrakar, P. Self medication pattern in rural areas in Pune, India. Int. J. Med. Public Health 2012, 2, 7–11. [Google Scholar] [CrossRef]
- Loharkar, N.; Keche, Y.; Yegnanarayan, R.; Dharma, M.; Bhosale, A.; Makan, A. Self-medication use in urban population of pune, Maharashtra, India. Sch. J. App. Med. Sci. 2013, 1, 732–738. [Google Scholar]
- CLSI. Performance Standards for Antimicrobial Susceptibility Testing; Twenty-First Informational Supplement; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2011; Volume 30. [Google Scholar]
- Bauer, A.; Kirby, W.; Sherris, J.C.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [CrossRef] [PubMed]
- Gales, A.C.; Reis, A.O.; Jones, R.N. Contemporary assessment of antimicrobial susceptibility testing methods for polymyxin b and colistin: Review of available interpretative criteria and quality control guidelines. J. Clin. Microbiol. 2001, 39, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Jacoby, G.A. AmpC beta -lactamases. Clin. Microbiol. Rev. 2009, 22, 161–182. [Google Scholar] [CrossRef] [PubMed]
- Dallenne, C.; Da Costa, A.; Decré, D.; Favier, C.; Arlet, G. Development of a set of multiplex pcr assays for the detection of genes encoding important β-lactamases in enterobacteriaceae. J. Antimicrob. Chemother. 2010, 65, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Du, X.-X.; Wang, J.-F.; Fu, Y.; Zhao, F.; Chen, Y.; Wang, H.-P.; Yu, Y.-S. Genetic characteristics of blandm-1-positive plasmid in citrobacterfreundii isolate separated from a clinical infectious patient. J. Med. Microbiol. 2013, 62, 1332–1337. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.B.; Wang, M.; Park, C.H.; Kim, E.-C.; Jacoby, G.A.; Hooper, D.C. Oqxab encoding a multidrug efflux pump in human clinical isolates of enterobacteriaceae. Antimicrob. Agents Chemother. (Bethesda) 2009, 53, 3582–3584. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; White, D.G.; Ge, B.; Ayers, S.; Friedman, S.; English, L.; Wagner, D.; Gaines, S.; Meng, J. Identification and characterization of integron-mediated antibiotic resistance among shiga toxin-producing escherichia coli isolates. Appl. Environ. Microbiol. 2001, 67, 1558–1564. [Google Scholar] [CrossRef] [PubMed]
- Byrne-Bailey, K.; Gaze, W.; Kay, P.; Boxall, A.; Hawkey, P.; Wellington, E. Prevalence of sulfonamide resistance genes in bacterial isolates from manured agricultural soils and pig slurry in the United Kingdom. Antimicrob. Agents Chemother. 2009, 53, 696–702. [Google Scholar] [CrossRef] [PubMed]
- DBT. Scoping Report on Antimicrobial Resistance in India; Department of Biotechnology: New Delhi, India, 2017. [Google Scholar]
- Kumar, S.; Tripathi, V.; Garg, S. Antibiotic resistance and genetic diversity in water-borne enterobacteriaceae isolates from recreational and drinking water sources. Int. J. Environ. Sci. Technol. 2013, 10, 789–798. [Google Scholar] [CrossRef]
- Poonia, S.; Singh, T.S.; Tsering, D.C. Antibiotic susceptibility profile of bacteria isolated from natural sources of water from rural areas of east sikkim. Indian J. Community Med. 2014, 39, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Odenholt, I.; Isaksson, B.; Nilsson, L.; Cars, O. Postantibiotic and bactericidal effect of imipenem against Pseudomonas aeruginosa. Eur. J. Clin. Microbiol. Infect. Dis. 1989, 8, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Centres for Disease Control and Prevention (US). Antibiotic Resistance Threats in The United States, 2013; Centres for Disease Control and Prevention, US Department of Health and Human Services: Atlanta, GA, USA, 2013.
- Mulani, M.S.; Azhar, S.; Azharuddin, S.; Tambe, S. Harnessing the power of bacteriophage for pathogen reduction in wastewater. Int. J. Curr. Microbiol. Appl. Sci. 2015, 152–161. [Google Scholar]
- Gonzal, A.C.; Santiago, C.B.; Afuang, W.B. Pasig River Backflow and Its Effect on the Water Quality of Laguna De Bay, Philippines. Available online: https://repository.seafdec.org.ph/bitstream/handle/10862/844/cemplrfa_p162.pdf?sequence=1&isAllowed=y (accessed on 7 June 2018).
- Li, Y.; Zhang, Q.; Werner, A.D.; Yao, J.; Ye, X. The influence of river-to-lake backflow on the hydrodynamics of a large floodplain lake system (Poyang Lake, China). Hydrol. Process. 2017, 31, 117–132. [Google Scholar] [CrossRef]
- Carattoli, A. Resistance plasmid families in enterobacteriaceae. Antimicrob. Agents Chemother. 2009, 53, 2227–2238. [Google Scholar] [CrossRef] [PubMed]
- Kotlarska, E.; Łuczkiewicz, A.; Pisowacka, M.; Burzyński, A. Antibiotic resistance and prevalence of class 1 and 2 integrons in Escherichia coli isolated from two wastewater treatment plants, and their receiving waters (gulf of Gdansk, Baltic sea, Poland). Environ. Sci. Pollut. Res. 2015, 22, 2018–2030. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.S.; Virdi, J.S. Assessment of antibiotic resistance genes and integrons in commensal escherichia coli from the Indian urban waste water: Implications and significance for public health. Can. J. Biotechnol. 2017, 1, 116. [Google Scholar] [CrossRef]
- Garnier, F.; Raked, N.; Gassama, A.; Denis, F.; Ploy, M.-C. Genetic environment of quinolone resistance gene qnrb2 in a complex sul1-type integron in the newly described salmonella enterica serovar keurmassar. Antimicrob. Agents Chemother. 2006, 50, 3200–3202. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.B.; Park, C.H.; Kim, C.J.; Kim, E.-C.; Jacoby, G.A.; Hooper, D.C. Prevalence of plasmid-mediated quinolone resistance determinants over a 9-year period. Antimicrob. Agents Chemother. 2009, 53, 639–645. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, K.L.; Davies, R.H.; Threlfall, E.J. Mechanisms of quinolone resistance in Escherichia coli and salmonella: Recent developments. Int. J. Antimicrob. Agents 2005, 25, 358–373. [Google Scholar] [CrossRef] [PubMed]
- White, P.A.; McIver, C.J.; Rawlinson, W.D. Integrons and gene cassettes in theenterobacteriaceae. Antimicrob. Agents Chemother. 2001, 45, 2658–2661. [Google Scholar] [CrossRef] [PubMed]
- Sunde, M. Prevalence and characterization of class 1 and class 2 integrons in escherichia coli isolated from meat and meat products of norwegian origin. J. Antimicrob. Chemother. 2005, 56, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Bao, X.; Ji, L.; Chen, L.; Liu, J.; Miao, J.; Chen, D.; Bian, H.; Li, Y.; Yu, G. Resistance integrons: Class 1, 2 and 3 integrons. Ann. Clin. Microbiol. Antimicrob. 2015, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Marathe, N.P.; Pal, C.; Gaikwad, S.S.; Jonsson, V.; Kristiansson, E.; Larsson, D.J. Untreated urban waste contaminates Indian River sediments with resistance genes to last resort antibiotics. Water Res. 2017, 124, 388–397. [Google Scholar] [CrossRef] [PubMed]
- USGS. A Visit to A Wastewater-Treatment Plant: Primary Treatment of Wastewater. Available online: https://water.usgs.gov/edu/wwvisit.html (accessed on 22 May 2018).
- Ma, L.; Zhang, X.-X.; Zhao, F.; Wu, B.; Cheng, S.; Yang, L. Sewage treatment plant serves as a hot-spot reservoir of integrons and gene cassettes. J. Environ. Biol. 2013, 34, 391–399. [Google Scholar] [PubMed]
- Aubertheau, E.; Stalder, T.; Mondamert, L.; Ploy, M.-C.; Dagot, C.; Labanowski, J. Impact of wastewater treatment plant discharge on the contamination of river biofilms by pharmaceuticals and antibiotic resistance. Sci. Total. Environ. 2017, 579, 1387–1398. [Google Scholar] [CrossRef] [PubMed]
- LaPara, T.M.; Burch, T.R.; McNamara, P.J.; Tan, D.T.; Yan, M.; Eichmiller, J.J. Tertiary-treated municipal wastewater is a significant point source of antibiotic resistance genes into duluth-superior harbor. Environ. Sci. Technol. 2011, 45, 9543–9549. [Google Scholar] [CrossRef] [PubMed]
- Gillings, M.; Boucher, Y.; Labbate, M.; Holmes, A.; Krishnan, S.; Holley, M.; Stokes, H.W. The evolution of class 1 integrons and the rise of antibiotic resistance. J. Bacteriol. 2008, 190, 5095–5100. [Google Scholar] [CrossRef] [PubMed]
- Koczura, R.; Mokracka, J.; Taraszewska, A.; Łopacinska, N. Abundance of class 1 integron-integrase and sulfonamide resistance genes in river water and sediment is affected by anthropogenic pressure and environmental factors. Microb. Ecol. 2016, 72, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Leverstein-van Hall, M.A.; Box, A.T.; Blok, H.E.; Paauw, A.; Fluit, A.C.; Verhoef, J. Evidence of extensive interspecies transfer of integron-mediated antimicrobial resistance genes among multidrug-resistant enterobacteriaceae in a clinical setting. J. Infect. Dis. 2002, 186, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Sara Domingues, G.J.d.S.a.K.M.N. Integrons vehicles and pathways for horizontal dissemination in bacteria. Mob. Genet. Elements 2012, 2, 211–223. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Nishino, K.; Roberts, M.C.; Tolmasky, M.; Aminov, R.I.; Zhang, L. Mechanisms of antibiotic resistance. Front. Microbiol. 2015, 6, 1–3. [Google Scholar] [CrossRef] [PubMed]
- CDC. Core Elements of Hospital Antibiotic Stewardship Programs. Available online: https://www.cdc.gov/antibiotic-use/healthcare/implementation/core-elements.html (accessed on 6 June 2018).
| Description | Target Genes | Sequence (5′-3′) | Amplicon Size | Annealing Temp (°C) | Reference |
|---|---|---|---|---|---|
| β-Lactamase | SHV | FP: AGCCGCTTGAGCAAATTAAAC RP: ATCCCGCAGATAAATCACCAC | 713 | 64 | [49] |
| TEM | FP: CATTTCCGTGTCGCCCTTATTC RP: CGTTCATCCATAGTTGCCTGAC | 800 | 62 | ||
| CTX-M-15 | FP: TTAGGAARTGTGCCGCTGYA RP: CGATATCGTTGGTGGTRCCAT | 688 | |||
| CTX-M-27 | FP: TCAAGCCTGCCGATCTGGT RP: TGATTCTCGCCGCTGAAG | 561 | |||
| Plasmid-mediated quinolone resistance | qnrA | FP: AAGGAAGCCGTATGGATATT RP: AGCTAATCCGGCAGCACTAT | 670 | 54 | [50] |
| qnrB | FP: CGACCTGAGCGGCACTGAAT RP: TGAGCAACGATGCCTGGTAG | 515 | |||
| qnrS | FP: ACCTTCACCGCTTGCACATT RP: CCAGTGCTTCGAGAATCAGT | 571 | |||
| oqxA | FP: CTCGGCGCGATGATGCT RP: CCACTCTTCACGGGAGACGA | 280 | 64 | [51] | |
| Horizontal gene transfer | intI1 | FP: CCTCCCGCACGATGATC RP: TCCACGCATCGTCAGGC3 | 280 | 55 | [52] |
| intI2 | FP: CACGGATATGCGACAAAAAGGT RP: GTAGCAAACGAGTGACGAAATG | 788 | 55 | [53] |
| DDR E. coli—CP and CZ Resistant (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Site | CFU (Log10)/100 mL [%] | Resistance to | ||||||||
| Fluro-Quinolone | Cephalosporin | Penicillin | Carbapenem | Amino Glycoside | β-Lactam and Inhibitor | ESBL Production | ||||
| CP | CZ | CTX | CPM | AMP | IMP | GN | PIT | CZ + CZL | ||
| (3rd gen) | (3rd gen) | (4th gen) | ||||||||
| KT | 1.99 [2.2] | 94 | 94 | 94 (100) | 75 (79) | 94 (100) | 44 (46) | 16 (17) | 71 (75) | 6 (6) |
| MJ | 3.61 [1.2] | 60 | 60 | 57 (92) | 41 (68) | 60 (100) | 26 (43) | 5 (8) | 33 (55) | 6 (10) |
| SB | 5.09 [1.8] | 65 | 65 | 64 (98) | 49 (75) | 65 (100) | 29 (44) | 12 (18) | 56 (86) | 10 (15) |
| Total | 219 | 215 (98) | 165 (75) | 219 (100) | 99 (45) | 33 (15) | 160 (73) | 22 (10) | ||
| Resistance to Number of Antibiotics | Ciprofloxacin and Ceftazidime (CZ) + Additional Antibiotics | Resistant Isolates (%) |
|---|---|---|
| 3 | AMP/CPM/CTX/PIT | 4 (1.8) |
| 4 | CTX and AMP/IMP/GN/PIT/CPM CPM and AMP/IMP/GN/PIM GN and IMP/PIT IMP and PIT | 22 (10) |
| 5 | CTX + CPM and AMP/IMP/GN/PIT CTX + AMP and IMP/GN/PIT CTX + IMP and GN/PIT CTX + IMP and GN/PIT CPM + AMP and IMP/GN/PIT AMP + GN and PIT IMP + GN and PIT | 39 (17.8) |
| 6 | CTX + CPM + AMP and IMP/PIT/GN CTX + AMP + IMP and GN/PIT/CPM AMP + IMP + GN and PIT/CPM/CTX AMP + GN + PIT and CTX AMP + PIT+ CTX and CPM IMP + GN + PIT and CTX IMP + PIT+ CTX and CPM IMP + CTX + CPM and AMP GN + PIT + CTX and CPM GN + CTX + CPM and AMP GN + CPM + AMP and IMP | 63 (28.7) |
| 7 | CTX + CPM + AMP + IMP and GN/PIT CTX + CPM + GN + PIT and AMP/IMP | 63 (28.7) |
| 8 | CP + CZ+ CTX + CPM + AMP + IMP + GN + PIT | 28 (12.7) |
| Genes | No. of DDR E. coli (%) |
|---|---|
| Associated with fluoroquinolone | |
| qnrB | 1 (0.4) |
| qnrS | 18 (8.2) |
| Associated with β-lactamase | |
| TEM | 70 (32) |
| CTX-M-15 | 100 (46) |
| CTX-M-27 | 2 (0.9) |
| TEM + CTX-M-15 | 22 (10) |
| TEM + CTX-M-27 | 2 (0.9) |
| Associated with fluoroquinolone + β-lactamase | |
| qnrS + TEM | 10 (4.5) |
| Associated with HGT | |
| intI1 | 103 (47) |
| intI2 | 18 (8) |
| intI1 + intI2 | 5 (2) |
| Associated with fluoroquinolone + Class 1 integron | |
| qnrB + intI1 | 1 (0.4) |
| qnrS + intI1 | 10 (4.5) |
| Associated with fluoroquinolone + Class 2 integron | |
| qnrS+int2 | 1 (0.4) |
| Associated with β-lactamase + Class 1 integron | |
| CTX-M-15 + intI1 | 55 (25) |
| CTX-M-27 + intI1 | 1 (0.4) |
| TEM + intI1 | 72 (32) |
| TEM + CTX-M-15 + intI1 | 14 (6) |
| TEM + CTX-M-27 + intI1 | 1 (0.4) |
| Associated with β-lactamase + Class 2 integron | |
| CTX-M-15 + intI2 | 11 (5) |
| TEM + intI2 | 7 (3.1) |
| TEM + CTX-M-15 + intI2 | 3 (1.3) |
| Associated with β-lactamase + Class 1 and Class 2 integron | |
| CTX-M-15 + intI2 + intI1 | 3 (1.3) |
| TEM + intI2 + intI1 | 4 (1.8) |
| TEM + CTX-M-15 + intI2 + intI1 | 2 (0.9) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhawde, R.; Macaden, R.; Saranath, D.; Nilgiriwala, K.; Ghadge, A.; Birdi, T. Antibiotic Resistance Characterization of Environmental E. coli Isolated from River Mula-Mutha, Pune District, India. Int. J. Environ. Res. Public Health 2018, 15, 1247. https://doi.org/10.3390/ijerph15061247
Dhawde R, Macaden R, Saranath D, Nilgiriwala K, Ghadge A, Birdi T. Antibiotic Resistance Characterization of Environmental E. coli Isolated from River Mula-Mutha, Pune District, India. International Journal of Environmental Research and Public Health. 2018; 15(6):1247. https://doi.org/10.3390/ijerph15061247
Chicago/Turabian StyleDhawde, Rutuja, Ragini Macaden, Dhananjaya Saranath, Kayzad Nilgiriwala, Appasaheb Ghadge, and Tannaz Birdi. 2018. "Antibiotic Resistance Characterization of Environmental E. coli Isolated from River Mula-Mutha, Pune District, India" International Journal of Environmental Research and Public Health 15, no. 6: 1247. https://doi.org/10.3390/ijerph15061247
APA StyleDhawde, R., Macaden, R., Saranath, D., Nilgiriwala, K., Ghadge, A., & Birdi, T. (2018). Antibiotic Resistance Characterization of Environmental E. coli Isolated from River Mula-Mutha, Pune District, India. International Journal of Environmental Research and Public Health, 15(6), 1247. https://doi.org/10.3390/ijerph15061247





