Spondylodiscitis in Paediatric Patients: The Importance of Early Diagnosis and Prolonged Therapy
Abstract
1. Background
2. Case Presentations
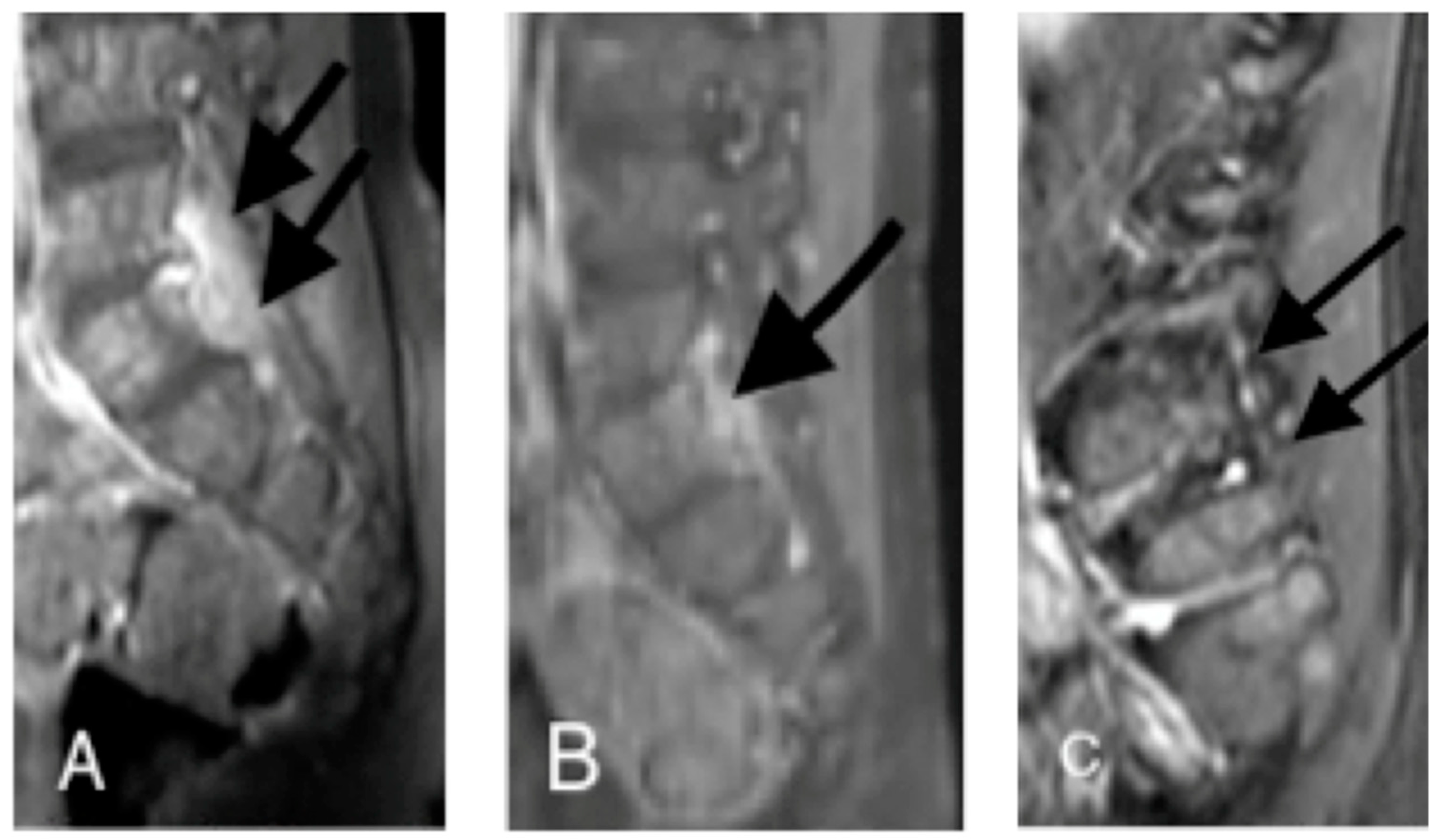
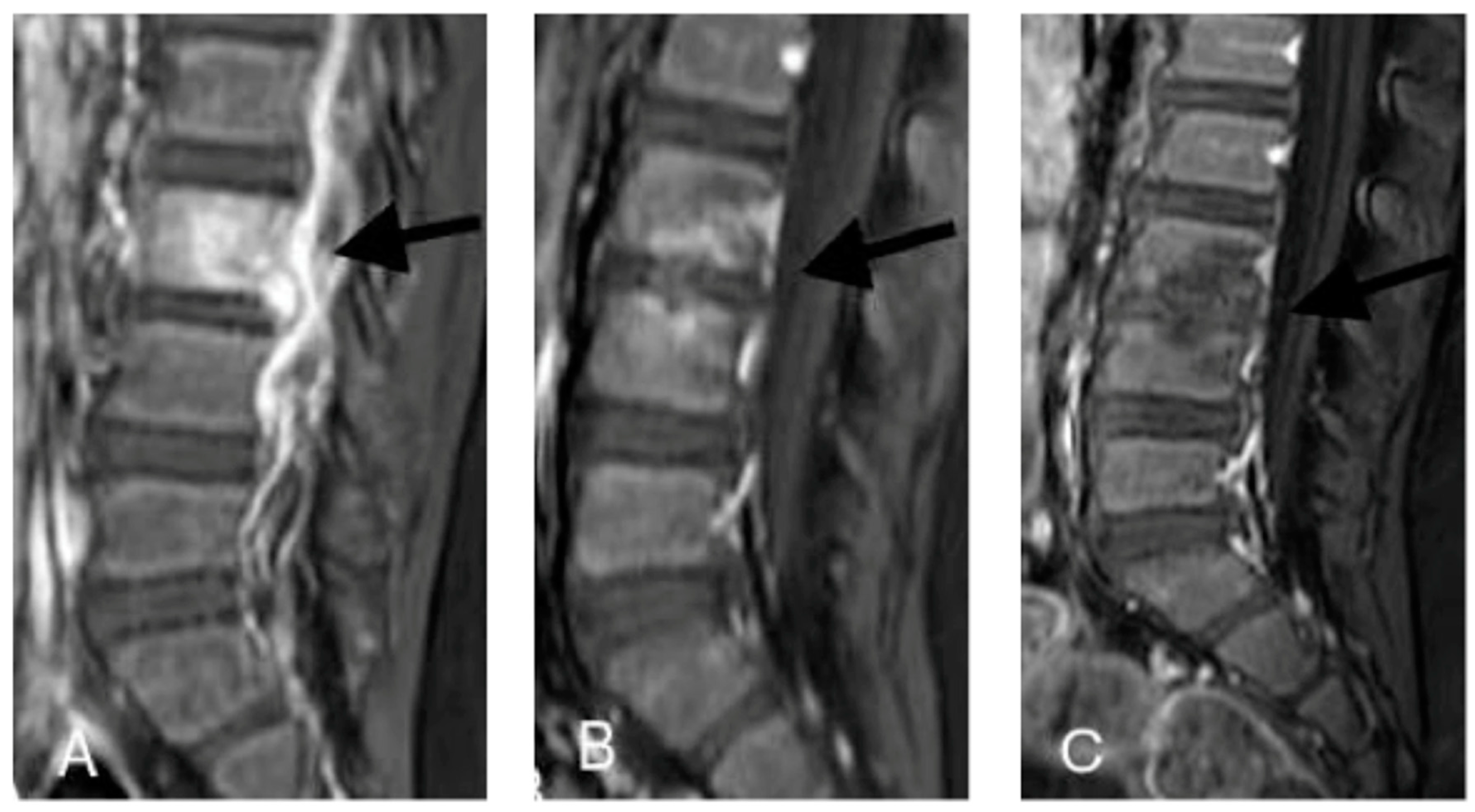
2.1. Case 1
2.2. Case 2
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Ethical Approval and Consent to Participate
Consent to Publish
Conflicts of Interest
References
- Cottle, L.; Riordan, T. Infectious spondylodiscitis. J. Infect. 2008, 56, 401–412. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.M.; Choi, E.H.; Lee, H.J.; Yun, K.W.; Lee, C.K.; Cho, T.J.; Cheon, J.E.; Lee, H. The etiology, clinical presentation and long-term outcome of spondylodiscitis in children. Pediatr. Infect. Dis. J. 2016, 35, e102–e106. [Google Scholar] [CrossRef] [PubMed]
- Principi, N.; Esposito, S. Infectious discitis and spondylodiscitis in children. Int. J. Mol. Sci. 2016, 17, 539. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.; Carrol, C.L.; Baker, C.J. Discitis and vertebral osteomyelitis in children: An 18-year review. Pediatrics 2000, 105, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Van Tuyl, J.S.; Jones, A.N.; Johnson, P.N. Meropenem-Induced Neutropenia in a Neonate. J. Pediatr. Pharmacol. Ther. 2016, 21, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Segarra-Newnham, M.; Tagoff, S.S. Probable vancomycin-induced neutropenia. Ann. Pharmacother. 2004, 38, 1855–1859. [Google Scholar] [CrossRef] [PubMed]
- Gemmel, F.; Dumarey, N.; Palestro, C.J. Radionuclide imaging of spinal infections. Eur. J. Nucl. Med. Mol. Imaging 2006, 33, 1226–1237. [Google Scholar] [CrossRef] [PubMed]
- Fuster, D.; Tomás, X.; Mayoral, M.; Soriano, A.; Manchón, F.; Cardenal, C.; Monegal, A.; Granados, U.; Garcia, S.; Pons, F. Prospective comparison of whole-body (18)F-FDG PET/CT and MRI of the spine in the diagnosis of haematogenous spondylodiscitis. Eur. J. Nucl. Med. Mol. Imaging 2015, 42, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Modic, M.T.; Feiglin, D.H.; Piraino, D.W.; Boumphrey, F.; Weinstein, M.A.; Duchesneau, P.M. Vertebral osteomyelitis: Assessment using MR. Radiology 1985, 157, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Tsirikos, A.I.; Tome-Bermejo, F. Spondylodiscitis in infancy: A potentially fatal condition that can lead to major spinal complications. J. Bone Jt. Surg. Br. 2012, 94, 1399–1402. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, R. Spinal infections in children: A review. J. Orthop. 2016, 13, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Chandrasenan, J.; Klezl, Z.; Bommireddy, R.; Calthorpe, D. Spondylodiscitis in children: A retrospective series. J. Bone Jt. Surg. Br. 2011, 93, 1122–1125. [Google Scholar] [CrossRef] [PubMed]
- Kapsalaki, E.; Gatselis, N.; Stefos, A.; Makaritsis, K.; Vassiou, A.; Fezoulidis, I.; Dalekos, G.N. Spontaneous spondylodiscitis: Presentation, risk factors, diagnosis, management, and outcome. Int. J. Infect. Dis. 2009, 13, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.J.; Jang, H.C.; Jung, S.I.; Choe, P.G.; Park, W.B.; Kim, C.J.; Song, K.H.; Kim, E.S.; Kim, H.B.; Oh, M.D.; et al. Clinical characteristics and risk factors of pyogenic spondylitis caused by gram-negative bacteria. PLoS ONE 2015, 10, e0127126. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bianchini, S.; Esposito, A.; Principi, N.; Esposito, S. Spondylodiscitis in Paediatric Patients: The Importance of Early Diagnosis and Prolonged Therapy. Int. J. Environ. Res. Public Health 2018, 15, 1195. https://doi.org/10.3390/ijerph15061195
Bianchini S, Esposito A, Principi N, Esposito S. Spondylodiscitis in Paediatric Patients: The Importance of Early Diagnosis and Prolonged Therapy. International Journal of Environmental Research and Public Health. 2018; 15(6):1195. https://doi.org/10.3390/ijerph15061195
Chicago/Turabian StyleBianchini, Sonia, Andrea Esposito, Nicola Principi, and Susanna Esposito. 2018. "Spondylodiscitis in Paediatric Patients: The Importance of Early Diagnosis and Prolonged Therapy" International Journal of Environmental Research and Public Health 15, no. 6: 1195. https://doi.org/10.3390/ijerph15061195
APA StyleBianchini, S., Esposito, A., Principi, N., & Esposito, S. (2018). Spondylodiscitis in Paediatric Patients: The Importance of Early Diagnosis and Prolonged Therapy. International Journal of Environmental Research and Public Health, 15(6), 1195. https://doi.org/10.3390/ijerph15061195





