Quantitative Microbial Risk Assessment and Opportunist Waterborne Infections–Are There Too Many Gaps to Fill?
Abstract
1. Introduction
2. Clinical Presentations
3. Sources of Contamination
4. Exposure Route
5. Dose Response
5.1. Dose
5.2. Virulence
5.3. Animal Models of Exposure
5.4. Population Susceptibility
6. Exposure Frequency
7. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Buchanan, R. The role of microbiological criteria and risk assessment in haccp. Food Microbiol. 1995, 12, 421–424. [Google Scholar] [CrossRef]
- Haas, C.N.; Rose, J.B.; Gerba, C.P. Quantitative Microbial Risk Assessment; John Wiley & Sons: Hoboken, NJ, USA, 1999. [Google Scholar]
- Westrell, T.; Schonning, C.; Stenstrom, T.; Ashbolt, N. Qmra and haccp for management of pathogens in wastewater and sewage sludge treatment and reuse. Water Sci. Technol. 2004, 50, 23–30. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Quantitative Microbial Risk Assessment: Application for Water Safety Management; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Ashbolt, N. Environmental (saprozoic) pathogens of engineered water systems: Understanding their ecology for risk assessment and management. Pathogens 2015, 4, 390–405. [Google Scholar] [CrossRef] [PubMed]
- Petterson, S.; Ashbolt, N. Qmra and water safety management: Review of application in drinking water systems. J. Water Health 2016, 14, 571–589. [Google Scholar] [CrossRef] [PubMed]
- Whiley, H.; Keegan, A.; Giglio, S.; Bentham, R. Mycobacterium avium complex—The role of potable water in disease transmission. J. Appl. Microbiol. 2012, 113, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, T.W.; Haas, C.N. Quantitative microbial risk assessment model for legionnaires’ disease: Assessment of human exposures for selected spa outbreaks. J. Occup. Environ. Hyg. 2007, 4, 634–646. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, T.W.; Haas, C.N. A quantitative microbial risk assessment model for legionnaires’ disease: Animal model selection and dose-response modeling. Risk Anal. 2007, 27, 1581–1596. [Google Scholar] [CrossRef] [PubMed]
- Buse, H.Y.; Schoen, M.E.; Ashbolt, N.J. Legionellae in engineered systems and use of quantitative microbial risk assessment to predict exposure. Water Res. 2012, 46, 921–933. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, T.; Haas, C. Legionnaires’ disease: Evaluation of a quantitative microbial risk assessment model. J. Water Health 2008, 6, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Whiley, H.; Keegan, A.; Fallowfield, H.; Ross, K. Uncertainties associated with assessing the public health risk from Legionella. Front. Microbiol. 2014, 5, 501. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.; Haas, C. Critical review of mathematical approaches for quantitative microbial risk assessment (qmra) of Legionella in engineered water systems: Research gaps and a new framework. Environ. Sci. Water Res. Technol. 2016, 2, 599–613. [Google Scholar] [CrossRef]
- Euser, S.M.; Pelgrim, M.; Den Boer, J.W. Legionnaires’ disease and pontiac fever after using a private outdoor whirlpool spa. Scand. J. Infect. Dis. 2010, 42, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, J.; Hampton, L.; Fleming-Dutra, K.; Marten, C.; McClusky, C.; Perry, C.; Clemmons, N.; McCormic, Z.; Peik, S.; Mancuso, J. Large outbreak of legionnaires’ disease and pontiac fever at a military base. Epidemiol. Infect. 2014, 142, 2336–2346. [Google Scholar] [CrossRef] [PubMed]
- Glick, T.H.; Gregg, M.B.; Berman, B.; Mallison, G.; Rhodes, W.W.J.; Kassanoff, I. Pontiac fever. An epidemic of unknown etiology in a health department: I. Clinical and epidemiologic aspects. Am. J. Epidemiol. 1978, 107, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, N.; Marmet, D.; Surgot, M.; Nowicki, M.; Arslan, A.; Esteve, J.; Fleurette, J. Exposure to legionellaceae at a hot spring spa: A prospective clinical and serological study. Epidemiol. Infect. 1989, 102, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Bartram, J.; Chartier, Y.; Lee, J.V.; Pond, K.; Surman-Lee, S. Legionella and the Prevention of Legionellosis; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Van Heijnsbergen, E.; Schalk, J.A.; Euser, S.M.; Brandsema, P.S.; den Boer, J.W.; de Roda Husman, A.M. Confirmed and potential sources of Legionella reviewed. Environ. Sci. Technol. 2015, 49, 4797–4815. [Google Scholar] [CrossRef] [PubMed]
- Fisman, D.N. Of Time and the River: How Our Understanding of Legionellosis Has Changed Since 1976; Oxford University Press: New York, NY, USA, 2017. [Google Scholar]
- Rhoads, W.J.; Garner, E.; Ji, P.; Zhu, N.; Parks, J.; Schwake, D.O.; Pruden, A.; Edwards, M.A. Distribution system operational deficiencies coincide with reported legionnaires’ disease clusters in flint, michigan. Environ. Sci. Technol. 2017, 51, 11986–11995. [Google Scholar] [CrossRef] [PubMed]
- Lesnik, R.; Brettar, I.; Höfle, M.G. Legionella species diversity and dynamics from surface reservoir to tap water: From cold adaptation to thermophily. ISME J. 2016, 10, 1064–1080. [Google Scholar] [CrossRef] [PubMed]
- Berkelman, R.L. Consideration of prophylactic antibiotic therapy during an outbreak of legionnaires’ disease. Clin. Infect. Dis. 2014, 58, 144–145. [Google Scholar] [CrossRef] [PubMed]
- Whiley, H. Legionella risk management and control in potable water systems: Argument for the abolishment of routine testing. Int. J. Environ. Res. Public Health 2016, 14, 12. [Google Scholar] [CrossRef] [PubMed]
- Crimi, P.; Macrina, G.; Grieco, A.; Tinteri, C.; Copello, L.; Rebora, D.; Galli, A.; Rizzetto, R. Correlation between Legionella contamination in water and surrounding air. Infect. Control Hosp. Epidemiol. 2006, 27, 771–773. [Google Scholar] [CrossRef] [PubMed]
- Pourchez, J.; Leclerc, L.; Girardot, F.; Riffard, S.; Prevot, N.; Allegra, S. Experimental human-like model to assess the part of viable Legionella reaching the thoracic region after nebulization. PLoS ONE 2017, 12, e0186042. [Google Scholar] [CrossRef] [PubMed]
- Berk, S.G.; Ting, R.S.; Turner, G.W.; Ashburn, R.J. Production of respirable vesicles containing live Legionella pneumophila cells by two Acanthamoeba spp. Appl. Environ. Microbiol. 1998, 64, 279–286. [Google Scholar] [PubMed]
- Buse, H.Y.; Ashbolt, N.J. Counting Legionella cells within single amoeba host cells. Appl. Environ. Microbiol. 2012, 78, 2070–2072. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.S.; Benson, R.F.; Besser, R.E. Legionella and legionnaires’ disease: 25 years of investigation. Clin. Microbiol. Rev. 2002, 15, 506–526. [Google Scholar] [CrossRef] [PubMed]
- Greig, J.E.; Carnie, J.A.; Tallis, G.F.; Ryan, N.J.; Tan, A.G.; Gordon, I.R.; Zwolak, B.; Leydon, J.A.; Guest, C.S.; Hart, W.G. An outbreak of legionnaires’ disease at the melbourne aquarium, April 2000: Investigation and case-control studies. Med. J. Aust. 2004, 180, 566–572. [Google Scholar] [PubMed]
- Blanchard, D.C.; Syzdek, L.D. Water-to-air transfer and enrichment of bacteria in drops from bursting bubbles. Appl. Environ. Microbiol. 1982, 43, 1001–1005. [Google Scholar] [PubMed]
- Strydom, D.; Küsel, R.R.; Craig, I.K. When is it appropriate to model transmission of tuberculosis using a dose response model? IFAC-PapersOnLine 2017, 50, 31–36. [Google Scholar] [CrossRef]
- Edelstein, P.H.; Metlay, J.P. Legionella pneumophila Goes Clonal—Paris and Lorraine Strain-Specific Risk Factors; The University of Chicago Press: Chicago, IL, USA, 2009. [Google Scholar]
- Robertson, P.; Abdelhady, H.; Garduño, R.A. The many forms of a pleomorphic bacterial pathogen—The developmental network of Legionella pneumophila. Front. Microbiol. 2014, 5, 670. [Google Scholar] [CrossRef] [PubMed]
- Cirillo, J.D.; Cirillo, S.L.; Yan, L.; Bermudez, L.E.; Falkow, S.; Tompkins, L.S. Intracellular growth in Acanthamoeba castellanii affects monocyte entry mechanisms and enhances virulence of Legionella pneumophila. Infect. Immun. 1999, 67, 4427–4434. [Google Scholar] [PubMed]
- Cirillo, J.D. Interaction of Mycobacterium avium with environmental amoebae enhances virulence. Infect. Immun. 1997, 65, 3759. [Google Scholar] [PubMed]
- Bouwknegt, M.; Schijven, J.F.; Schalk, J.A.; Husman, R.; Maria, A. Quantitative risk estimation for a Legionella pneumophila infection due to whirlpool use. Risk Anal. 2013, 33, 1228–1236. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Klein, T.W.; Newton, C.A.; Widen, R.; Friedman, H. Differential growth of Legionella pneumophila in guinea pig versus mouse macrophage cultures. Infect. Immun. 1987, 55, 1369–1374. [Google Scholar] [PubMed]
- Misch, E.A. Legionella: Virulence factors and host response. Curr. Opin. Infect. Dis. 2016, 29, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Berendt, R.F.; Young, H.W.; Allen, R.G.; Knutsen, G.L. Dose-response of guinea pigs experimentally infected with aerosols of Legionella pneumophila. J. Infect. Dis. 1980, 141, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Fitzgeorge, R.; Baskerville, A.; Broster, M.; Hambleton, P.; Dennis, P. Aerosol infection of animals with strains of Legionella pneumophila of different virulence: Comparison with intraperitoneal and intranasal routes of infection. Epidemiol. Infect. 1983, 90, 81–89. [Google Scholar] [CrossRef]
- Beauté, J. Legionnaires’ disease in europe, 2011 to 2015. Eurosurveillance 2017, 22, 30566. [Google Scholar] [CrossRef] [PubMed]
- Gregersen, P.; Grunnet, K.; Uldum, S.A.; Andersen, B.H.; Madsen, H. Pontiac fever at a sewage treatment plant in the food industry. Scand. J. Work Environ. Health 1999, 25, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Van Lier, A.; McDonald, S.A.; Bouwknegt, M.; Kretzschmar, M.E.; Havelaar, A.H.; Mangen, M.-J.J.; Wallinga, J.; de Melker, H.E. Disease burden of 32 infectious diseases in the netherlands, 2007–2011. PLoS ONE 2016, 11, e0153106. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.-H.; Chou, P.; Tseng, L.-R.; Lin, H.-C.; Wang, J.-H.; Sheu, J.-N.; Liu, M.-T.; Liu, F.-C.; Wu, H.-H.; Lin, M.-C. Nosocomial neonatal legionellosis associated with water in infant formula, taiwan. Emerg. Infect. Dis. 2014, 20, 1921. [Google Scholar] [CrossRef] [PubMed]
- Yiallouros, P.K.; Papadouri, T.; Karaoli, C.; Papamichael, E.; Zeniou, M.; Pieridou-Bagatzouni, D.; Papageorgiou, G.T.; Pissarides, N.; Harrison, T.G.; Hadjidemetriou, A. First outbreak of nosocomial legionella infection in term neonates caused by a cold mist ultrasonic humidifier. Clin. Infect. Dis. 2013, 57, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Collins, S.; Afshar, B.; Walker, J.; Aird, H.; Naik, F.; Parry-Ford, F.; Phin, N.; Harrison, T.; Chalker, V.; Sorrell, S. Heated birthing pools as a source of legionnaires’ disease. Epidemiol. Infect. 2016, 144, 796–802. [Google Scholar] [CrossRef] [PubMed]
- Leitão Filho, F.S.; Hang Chen, H.; Ngan, D.A.; Tam, A.; Kirby, M.; Sin, D.D. Current methods to diagnose small airway disease in patients with copd. Expert Rev. Respir. Med. 2016, 10, 417–429. [Google Scholar] [CrossRef] [PubMed]
- Den Boer, J.W.; Yzerman, P.; Schellekens, J.; Lettinga, K.D.; Boshuizen, H.C.; Van Steenbergen, J.E.; Bosman, A.; Van den Hof, S.; Van Vliet, H.A.; Peeters, M.F. A large outbreak of legionnaires’ disease at a flower show, the netherlands, 1999. Emerg. Infect. Dis. 2002, 8, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Tobin, J.O.; Dunnill, M.S.; French, M.; Morris, P.J.; Beare, J.; Fisher-Hoch, S.; Mitchell, R.G.; Muers, M.F. Legionnaires’ diseases in a transplant unit: Isolation of the causative agent from shower baths. Lancet 1980, 316, 118–121. [Google Scholar] [CrossRef]
- Gargano, J.; Adam, E.; Collier, S.; Fullerton, K.; Feinman, S.; Beach, M. Mortality from selected diseases that can be transmitted by water–united states, 2003–2009. J. Water Health 2017, 15, 438–450. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, K.A.; Weir, M.H.; Haas, C.N. Dose response models and a quantitative microbial risk assessment framework for the mycobacterium avium complex that account for recent developments in molecular biology, taxonomy, and epidemiology. Water Res. 2017, 109, 310–326. [Google Scholar] [CrossRef] [PubMed]
- Roser, D.; Van den Akker, B.; Boase, S.; Haas, C.; Ashbolt, N.; Rice, S. Pseudomonas aeruginosa dose response and bathing water infection. Epidemiol. Infect. 2014, 142, 449–462. [Google Scholar] [CrossRef] [PubMed]
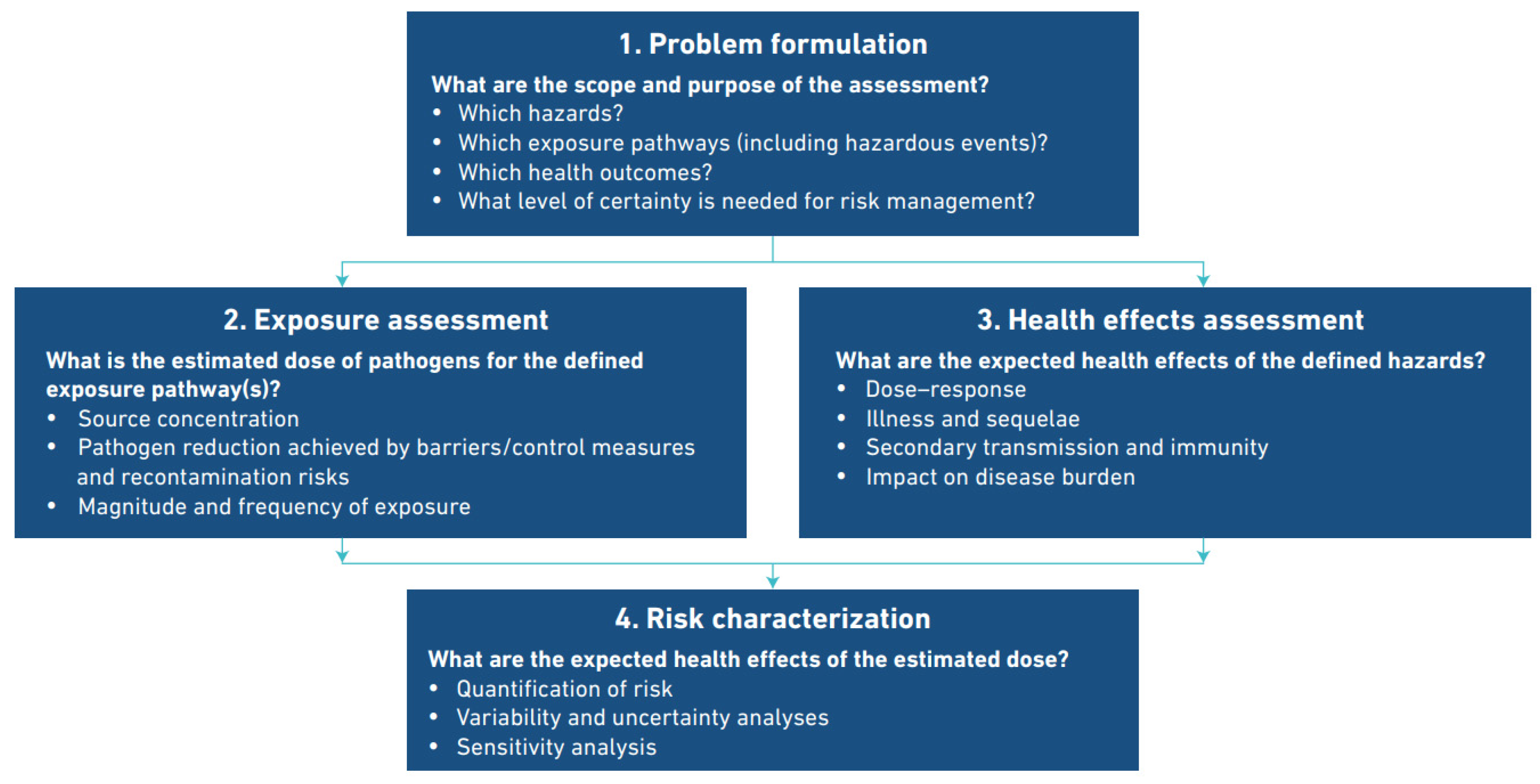
| Problem Formulation What are the scope and purpose of the assessment? | |
| What hazards? | Legionella infections. |
| Which exposure pathways? | Inhalation and/or aspiration of Legionella contaminated water from multiple sources. |
| Which Health outcomes? | Legionnaire’s disease in susceptible populations, Pontiac fever in general populations. |
| What certainty is needed for risk management? | This has not been quantified. |
| Exposure Assessment What is the estimated dose of pathogens for the defined exposure pathway(s)? | |
| Pathogen reduction | Achieved by barriers/control measures and managing re-contamination risks–well established control measures for most sources have been developed and applied. |
| Source concentration | Not defined as there are multiple sources. The relationship between source concentration and exposure has not been quantified due to the diversity of exposure scenarios. A robust and broadly applicable relationship between source concentration and dose has not been quantified. |
| Magnitude and frequency of exposure | Not quantifiable. Intermittent or occasional use of variable sources by different demographic groups. Many and variable exposure scenarios have been identified. |
| Health effects assessment What are the expected health effects of the defined hazards? | |
| Dose-response | Not quantified in humans. Susceptibility is dependent on the host immune status. Two principle routes of dose delivery exist. |
| Illness and sequalae | In many cases none. Disease may be self-limited, profound, or fatal. Sequalae range from minor to severe, prolonged and debilitating. Secondary transmission is extremely rare. |
| Impact on disease burden | DALY * for Years Lived with Disability (YLD) is low (8%). DALY for Years of Life Lost is high (92%). |
| Risk characterization What are the expected health effects of the estimated dose? | |
| Quantification of risk | Reported cases range from 0.5 to 5+: 100,000. Prevalence of disease is much higher in susceptible populations but not quantified. Disease is probably considerably under-reported |
| Variability and uncertainty analysis | Not calculable for the range of infecting organisms, sources and exposure routes on available evidence. |
| Sensitivity analysis | Insufficient uncertainty data |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bentham, R.; Whiley, H. Quantitative Microbial Risk Assessment and Opportunist Waterborne Infections–Are There Too Many Gaps to Fill? Int. J. Environ. Res. Public Health 2018, 15, 1150. https://doi.org/10.3390/ijerph15061150
Bentham R, Whiley H. Quantitative Microbial Risk Assessment and Opportunist Waterborne Infections–Are There Too Many Gaps to Fill? International Journal of Environmental Research and Public Health. 2018; 15(6):1150. https://doi.org/10.3390/ijerph15061150
Chicago/Turabian StyleBentham, Richard, and Harriet Whiley. 2018. "Quantitative Microbial Risk Assessment and Opportunist Waterborne Infections–Are There Too Many Gaps to Fill?" International Journal of Environmental Research and Public Health 15, no. 6: 1150. https://doi.org/10.3390/ijerph15061150
APA StyleBentham, R., & Whiley, H. (2018). Quantitative Microbial Risk Assessment and Opportunist Waterborne Infections–Are There Too Many Gaps to Fill? International Journal of Environmental Research and Public Health, 15(6), 1150. https://doi.org/10.3390/ijerph15061150





