Short-Term Particulate Air Pollution Exposure is Associated with Increased Severity of Respiratory and Quality of Life Symptoms in Patients with Fibrotic Sarcoidosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Participants
2.2. Exposure Estimation
2.3. Outcomes
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
List of Abbreviations
| O3 | ozone |
| PM2.5 | fine particulate matter with diameter less than 2.5 μm |
| FEV1 | forced expiratory volume in 1 s |
| FVC | forced vital capacity |
| SGRQ | St. George’s Respiratory Questionnaire |
| LCQ | Leicester Cough Questionnaire |
| KSQ | King’s Sarcoidosis Questionnaire |
| IQR | interquartile range |
| APES | acute pulmonary exacerbations of sarcoidosis |
References
- Bonham, C.A.; Strek, M.E.; Patterson, K.C. From granuloma to fibrosis: Sarcoidosis associated pulmonary fibrosis. Curr. Opin. Pulm. Med. 2016, 22, 484–491. [Google Scholar] [CrossRef] [PubMed]
- Hennebicque, A.-S.; Nunes, H.; Brillet, P.-Y.; Moulahi, H.; Valeyre, D.; Brauner, M.W. CT findings in severe thoracic sarcoidosis. Eur. Radiol. 2005, 15, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Panselinas, E.; Judson, M.A. Acute Pulmonary Exacerbations of Sarcoidosis. CHEST 2012, 142, 827–836. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Lower, E.E. Frequency of acute worsening events in fibrotic pulmonary sarcoidosis patients. Respir. Med. 2013, 107, 2009–2013. [Google Scholar] [CrossRef] [PubMed]
- Panselinas, E.; Judson, M.A. Acute Pulmonary Exacerbation of Sarcoidosis. In Pulmonary Sarcoidosis; Springer: New York, NY, USA, 2014; pp. 65–78. [Google Scholar]
- Li, J.; Sun, S.; Tang, R.; Qiu, H.; Huang, Q.; Mason, T.G.; Tian, L. Major air pollutants and risk of COPD exacerbations: A systematic review and meta-analysis. COPD 2016, 11, 3079–3091. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.-Y.; Ding, H.; Jiang, L.-N.; Chen, S.-W.; Zheng, J.-P.; Qiu, M.; Zhou, Y.-X.; Chen, Q.; Guan, W.-J. Association between Air Pollutants and Asthma Emergency Room Visits and Hospital Admissions in Time Series Studies: A Systematic Review and Meta-Analysis. PLoS ONE 2015, 10, e0138146. [Google Scholar] [CrossRef] [PubMed]
- Goeminne, P.C.; Kiciński, M.; Vermeulen, F.; Fierens, F.; De Boeck, K.; Nemery, B.; Nawrot, T.S.; Dupont, L.J. Impact of air pollution on cystic fibrosis pulmonary exacerbations: A case-crossover analysis. CHEST 2013, 143, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Birring, S.; Judson, M.A.; Culver, D.A.; Parambil, J.G.; Cordova, F.; Lower, E.E. A Double Blind, Placebo Controlled Study of Roflumilast to Prevent Acute Events in Fibrotic Sarcoidosis. Am. J. Respir. Crit. Care Med. 2017, 195, A4752. [Google Scholar]
- Hunninghake, G.W.; Costabel, U.; Ando, M.; Baughman, R.; Cordier, J.F.; du Bois, R.; Eklund, A.; Kitaichi, M.; Lynch, J.; Rizzato, G.; et al. Statement on sarcoidosis. In Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and other Granulomatous Disorders (WASOG) Adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999; American Thoracic Society: New York, NY, USA, 1999; Volume 160, pp. 736–755. [Google Scholar]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.M.; Gustafsson, P.; et al. ATS/ERS Task Force Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Hankinson, J.L.; Odencrantz, J.R.; Fedan, K.B. Spirometric reference values from a sample of the general U.S. population. Am. J. Respir. Crit. Care Med. 1999, 159, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.-C.; Roberts, J.M.; Catov, J.M.; Talbott, E.O.; Ritz, B. First Trimester Exposure to Ambient Air Pollution, Pregnancy Complications and Adverse Birth Outcomes in Allegheny County, PA. Matern. Child Health J. 2012, 17, 545–555. [Google Scholar] [CrossRef] [PubMed]
- American Thoracic Society. ATS statement–Snowbird workshop on standardization of spirometry. Am. Rev. Respir. Dis. 1979, 119, 831–838. [Google Scholar]
- Birring, S.S.; Prudon, B.; Carr, A.J.; Singh, S.J.; Morgan, M.D.L.; Pavord, I.D. Development of a symptom specific health status measure for patients with chronic cough: Leicester Cough Questionnaire (LCQ). Thorax 2003, 58, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.W.; Quirk, F.H.; Baveystock, C.M.; Littlejohns, P. A self-complete measure of health status for chronic airflow limitation. The St. George’s Respiratory Questionnaire. Am. Rev. Respir. Dis. 1992, 145, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.S.; Siegert, R.J.; Creamer, D.; Larkin, G.; Maher, T.M.; Renzoni, E.A.; Wells, A.U.; Higginson, I.J.; Birring, S.S. The development and validation of the King’s Sarcoidosis Questionnaire for the assessment of health status. Thorax 2013, 68, 57–65. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Newman, L.S.; Rose, C.S.; Bresnitz, E.A.; Rossman, M.D.; Barnard, J.; Frederick, M.; Terrin, M.L.; Weinberger, S.E.; Moller, D.R.; McLennan, G.; et al. ACCESS Research Group A case control etiologic study of sarcoidosis: Environmental and occupational risk factors. Am. J. Respir. Crit. Care Med. 2004, 170, 1324–1330. [Google Scholar] [CrossRef] [PubMed]
- Izbicki, G.; Chavko, R.; Banauch, G.I.; Weiden, M.D.; Berger, K.I.; Aldrich, T.K.; Hall, C.; Kelly, K.J.; Prezant, D.J. World Trade Center “sarcoid-like” granulomatous pulmonary disease in New York City Fire Department rescue workers. CHEST 2007, 131, 1414–1423. [Google Scholar] [CrossRef] [PubMed]
- Jordan, H.T.; Stellman, S.D.; Prezant, D.; Teirstein, A.; Osahan, S.S.; Cone, J.E. Sarcoidosis diagnosed after September 11, 2001, among adults exposed to the World Trade Center disaster. J. Occup. Environ. Med. 2011, 53, 966–974. [Google Scholar] [CrossRef] [PubMed]
- Newman, K.L.; Newman, L.S. Occupational causes of sarcoidosis. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Terčelj, M.; Salobir, B.; Harlander, M.; Rylander, R. Fungal exposure in homes of patients with sarcoidosis-an environmental exposure study. Environ. Health 2011, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Barnard, J.; Rose, C.; Newman, L.; Canner, M.; Martyny, J.; McCammon, C.; Bresnitz, E.; Rossman, M.; Thompson, B.; Rybicki, B.; et al. ACCESS Research Group Job and industry classifications associated with sarcoidosis in A Case-Control Etiologic Study of Sarcoidosis (ACCESS). J. Occup. Environ. Med. 2005, 47, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Deubelbeiss, U.; Gemperli, A.; Schindler, C.; Baty, F.; Brutsche, M.H. Prevalence of sarcoidosis in Switzerland is associated with environmental factors. Eur. Respir. J. 2010, 35, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, O.; Taniguchi, H.; Kondoh, Y.; Kimura, T.; Kato, K.; Kataoka, K.; Ogawa, T.; Watanabe, F.; Arizono, S. A simple assessment of dyspnoea as a prognostic indicator in idiopathic pulmonary fibrosis. Eur. Respir. J. 2010, 36, 1067–1072. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Winget, D.B.; Bowen, E.H.; Lower, E.E. Predicting respiratory failure in sarcoidosis patients. Sarcoidosis Vasc. Diffuse Lung Dis. 1997, 14, 154–158. [Google Scholar] [PubMed]
- Nardi, A.; Brillet, P.-Y.; Letoumelin, P.; Girard, F.; Brauner, M.; Uzunhan, Y.; Naccache, J.-M.; Valeyre, D.; Nunes, H. Stage IV sarcoidosis: Comparison of survival with the general population and causes of death. Eur. Respir. J. 2011, 38, 1368–1373. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Statistic |
|---|---|
| Age, median years (IQR) | 59 (53.25, 62.5) |
| Female N (%) | 12 (75%) |
| African American N (%) | 9 (56%) |
| Current smoker N (%) | 1 (6%) |
| Former smoker N (%) | 10 (62%) |
| Assigned study drug | 8 (50%) |
| FEV1 (L) (mean (SD)) | 1.61 (0.68) |
| FEV1 % predicted (mean (SD)) | 62.38 (21.45) |
| FVC (L) (mean (SD)) | 2.32 (0.86) |
| FVC % predicted (mean (SD)) | 69.56 (19.81) |
| Had episode of FEV1 drop > 10% N (%) | 5 (31%) |
| Pollutant | Time Period | Statistic | 7-Day Average | 10-Day Average | 14-Day Average |
|---|---|---|---|---|---|
| PM2.5 (μg/m3) | All days (N = 69) | Mean (SD) | 11.6 (4.5) | 11.9 (4.3) | 11.9 (3.8) |
| Median (IQR) | 10.6 (4.9) | 10.9 (4.6) | 11.2 (4.4) | ||
| Range | (5.8, 25.3) | (6, 24.7) | (6, 23.5) | ||
| May–October (N = 33) | Mean (SD) | 10.7 (3.1) | 11.3 (2.8) | 11.6 (2.7) | |
| Median (IQR) | 10.7 (3.0) | 11.2 (3.4) | 11.4 (4.5) | ||
| Range | (6.1, 19.1) | (6.1, 17.8) | (6, 17) | ||
| November–April (N = 36) | Mean (SD) | 12.5 (5.5) | 12.5 (5.4) | 12.1 (4.6) | |
| Median (IQR) | 10.6 (6.7) | 10.2 (6.1) | 10.3 (5.4) | ||
| Range | (5.8, 25.3) | (6, 24.7) | (6.1, 23.5) | ||
| O3 (ppm) | All days (N = 69) | Mean (SD) | 0.04 (0.009) | 0.041 (0.008) | 0.041 (0.009) |
| Median (IQR) | 0.041 (0.014) | 0.042 (0.014) | 0.043 (0.014) | ||
| Range | (0.023, 0.058) | (0.023, 0.056) | (0.023, 0.055) | ||
| May–October (N = 33) | Mean (SD) | 0.044 (0.007) | 0.045 (0.007) | 0.046 (0.006) | |
| Median (IQR) | 0.044 (0.008) | 0.047 (0.008) | 0.048 (0.007) | ||
| Range | (0.026, 0.058) | (0.027, 0.056) | (0.032, 0.055) | ||
| November–April (N = 36) | Mean (SD) | 0.037 (0.008) | 0.037 (0.008) | 0.037 (0.008) | |
| Median (IQR) | 0.037 (0.012) | 0.036 (0.012) | 0.035 (0.013) | ||
| Range | (0.023, 0.052) | (0.023, 0.052) | (0.023, 0.052) |
| Lung Function Outcome | PM2.5 | Ozone | ||||
|---|---|---|---|---|---|---|
| 7-Day Average | 10-Day Average | 14-Day Average | 7-Day Average | 10-Day Average | 14-Day Average | |
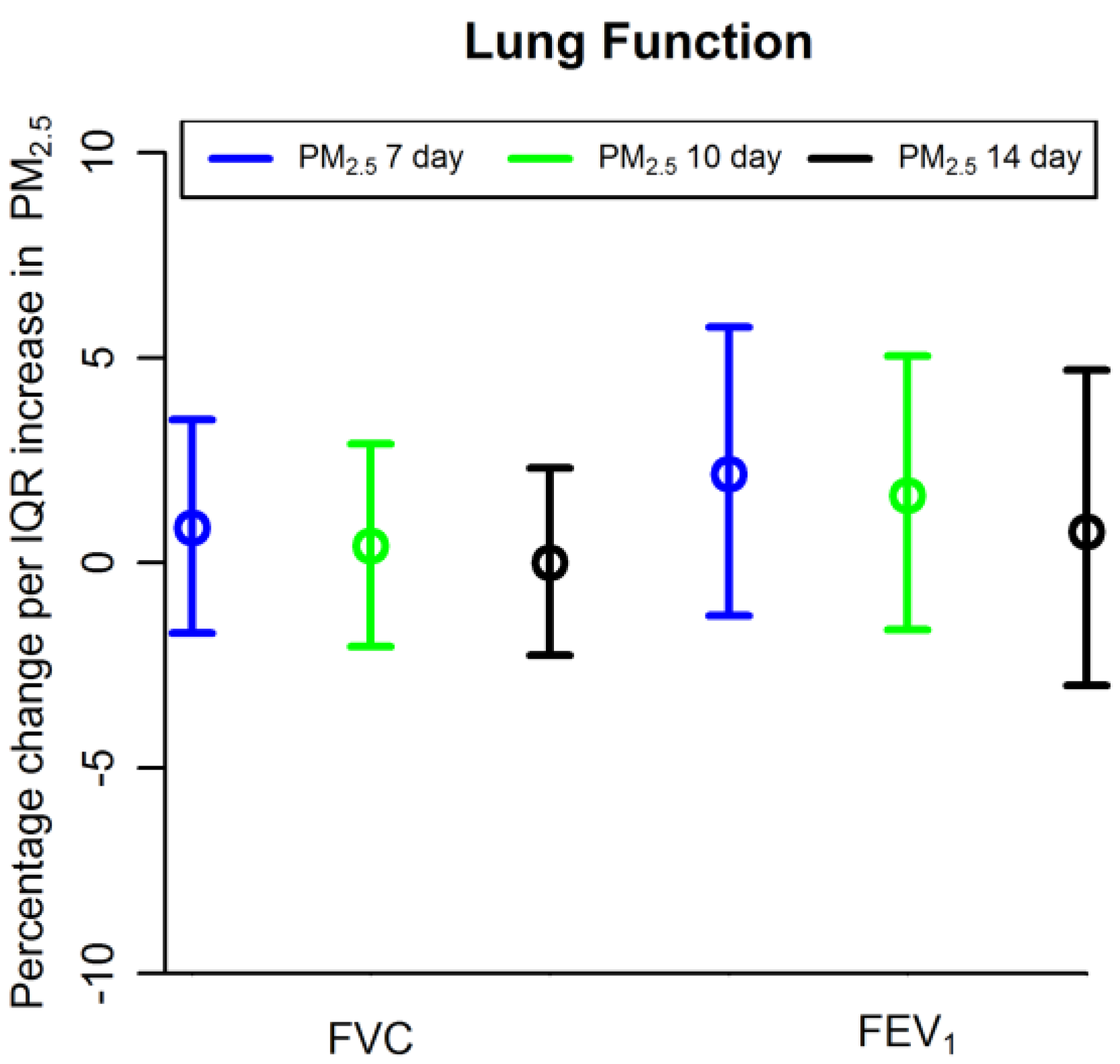
| FVC (L) (% change) 1 | 0.86 (−1.71, 3.50) | 0.41 (−2.03, 2.91) | 0.0 (−2.25, 2.31) | 0.59 (−3.51, 4.86) | 0.46 (−3.58, 4.66) | 0.80 (−3.13, 4.88) |
| FEV1 (L) (% change) 1 | 2.17 (−1.28, 5.75) | 1.65 (−1.63, 5.04) | 0.76 (−2.99, 4.70) | 0.57 (−4.83, 6.29) | 1.11 (−4.25, 6.77) | −0.03 (−5.18, 5.40) |
| Episodes of FEV1 > 10% decline 2 | 0.74 (0.18, 1.19) | 0.76 (0.30, 1.22) | 0.85 (0.32, 1.38) | 0.9 (0.02, 1.79) | 0.93 (0.01, 1.85) | 0.98 (0.04, 1.93) |
| Questionnaire Outcome | PM2.5 | Ozone | ||||
|---|---|---|---|---|---|---|
| 7-Day Average | 10-Day Average | 14-Day Average | 7-Day Average | 10-Day Average | 14-Day Average | |
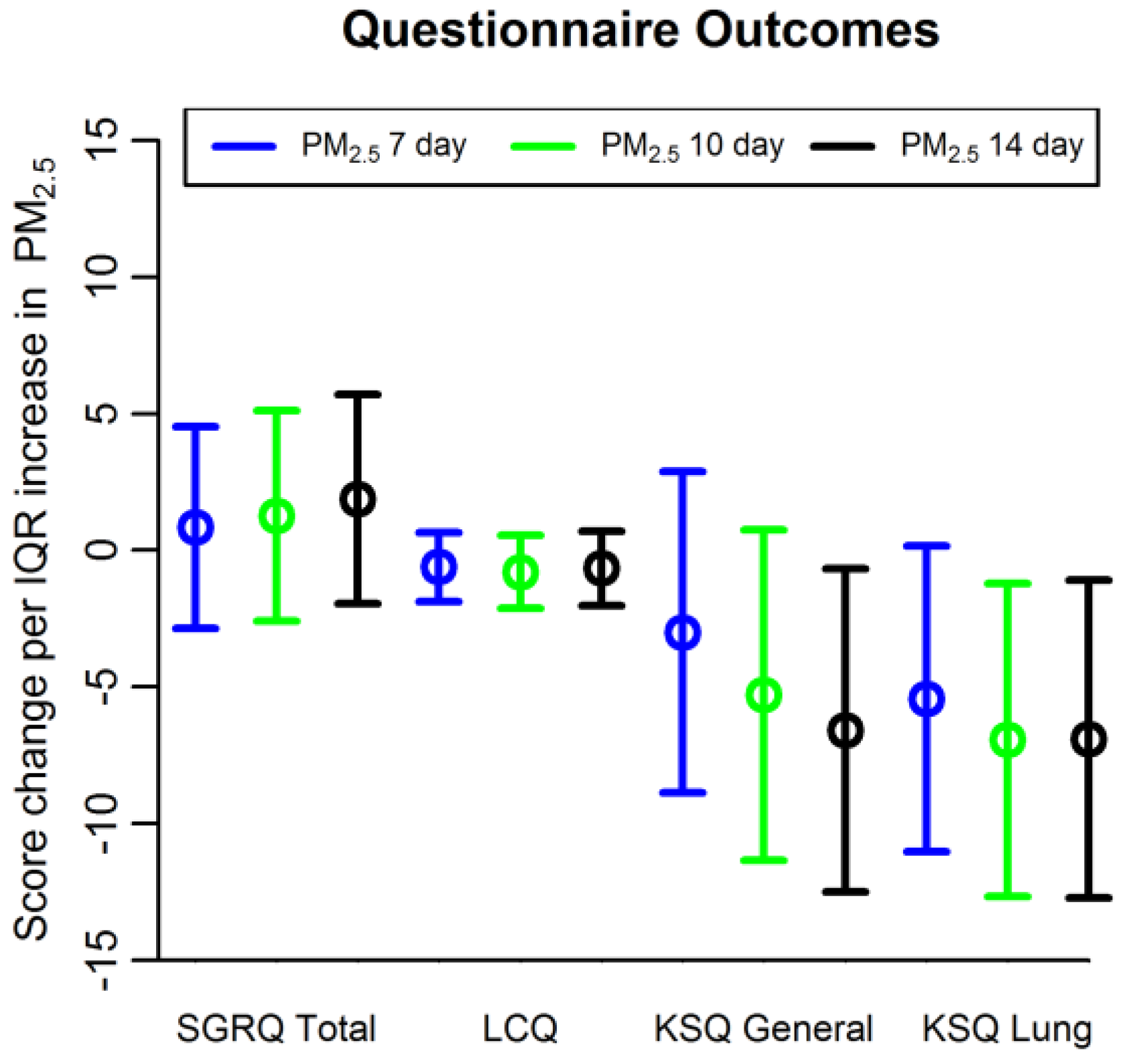
| SGRQ Total Score | 0.84 (−2.85, 4.53) | 1.26 (−2.59, 5.11) | 1.87 (−1.96, 5.70) | −0.33 (−4.73, 4.07) | −1.08 (−5.5, 3.3) | −0.87 (−5.41, 3.66) |
| LCQ score | −0.61 (−1.87, 0.65) | −0.80 (−2.13, 0.54) | −0.66 (−2.03, 0.70) | −1.00 (−2.57, 0.57) | −0.97 (−2.55, 0.61) | −0.84 (−2.45, 0.77) |
| KSQ General Health Status | −3.00 (−8.87, 2.88) | −5.3 (−11.35, 0.75) | −6.60 (−12.51, −0.68) * | −1.22 (−8.88, 6.44) | −0.69 (−8.5, 7.1) | −0.48 (−8.37, 7.42) |
| KSQ Lung Health Status | −5.44 (−11.03, 0.15) | −6.93 (−12.67, −1.21) * | −6.91 (−12.73, −1.09) * | −3.57 (−10.9, 3.75) | −2.85 (−10.28, 4.58) | −2.34 (−9.98, 5.19) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pirozzi, C.S.; Mendoza, D.L.; Xu, Y.; Zhang, Y.; Scholand, M.B.; Baughman, R.P. Short-Term Particulate Air Pollution Exposure is Associated with Increased Severity of Respiratory and Quality of Life Symptoms in Patients with Fibrotic Sarcoidosis. Int. J. Environ. Res. Public Health 2018, 15, 1077. https://doi.org/10.3390/ijerph15061077
Pirozzi CS, Mendoza DL, Xu Y, Zhang Y, Scholand MB, Baughman RP. Short-Term Particulate Air Pollution Exposure is Associated with Increased Severity of Respiratory and Quality of Life Symptoms in Patients with Fibrotic Sarcoidosis. International Journal of Environmental Research and Public Health. 2018; 15(6):1077. https://doi.org/10.3390/ijerph15061077
Chicago/Turabian StylePirozzi, Cheryl S., Daniel L. Mendoza, Yizhe Xu, Yue Zhang, Mary Beth Scholand, and Robert P. Baughman. 2018. "Short-Term Particulate Air Pollution Exposure is Associated with Increased Severity of Respiratory and Quality of Life Symptoms in Patients with Fibrotic Sarcoidosis" International Journal of Environmental Research and Public Health 15, no. 6: 1077. https://doi.org/10.3390/ijerph15061077
APA StylePirozzi, C. S., Mendoza, D. L., Xu, Y., Zhang, Y., Scholand, M. B., & Baughman, R. P. (2018). Short-Term Particulate Air Pollution Exposure is Associated with Increased Severity of Respiratory and Quality of Life Symptoms in Patients with Fibrotic Sarcoidosis. International Journal of Environmental Research and Public Health, 15(6), 1077. https://doi.org/10.3390/ijerph15061077






