Does Molimina Indicate Ovulation? Prospective Data in a Hormonally Documented Single-Cycle in Spontaneously Menstruating Women
Abstract
1. Introduction
2. Methods
2.1. Participants
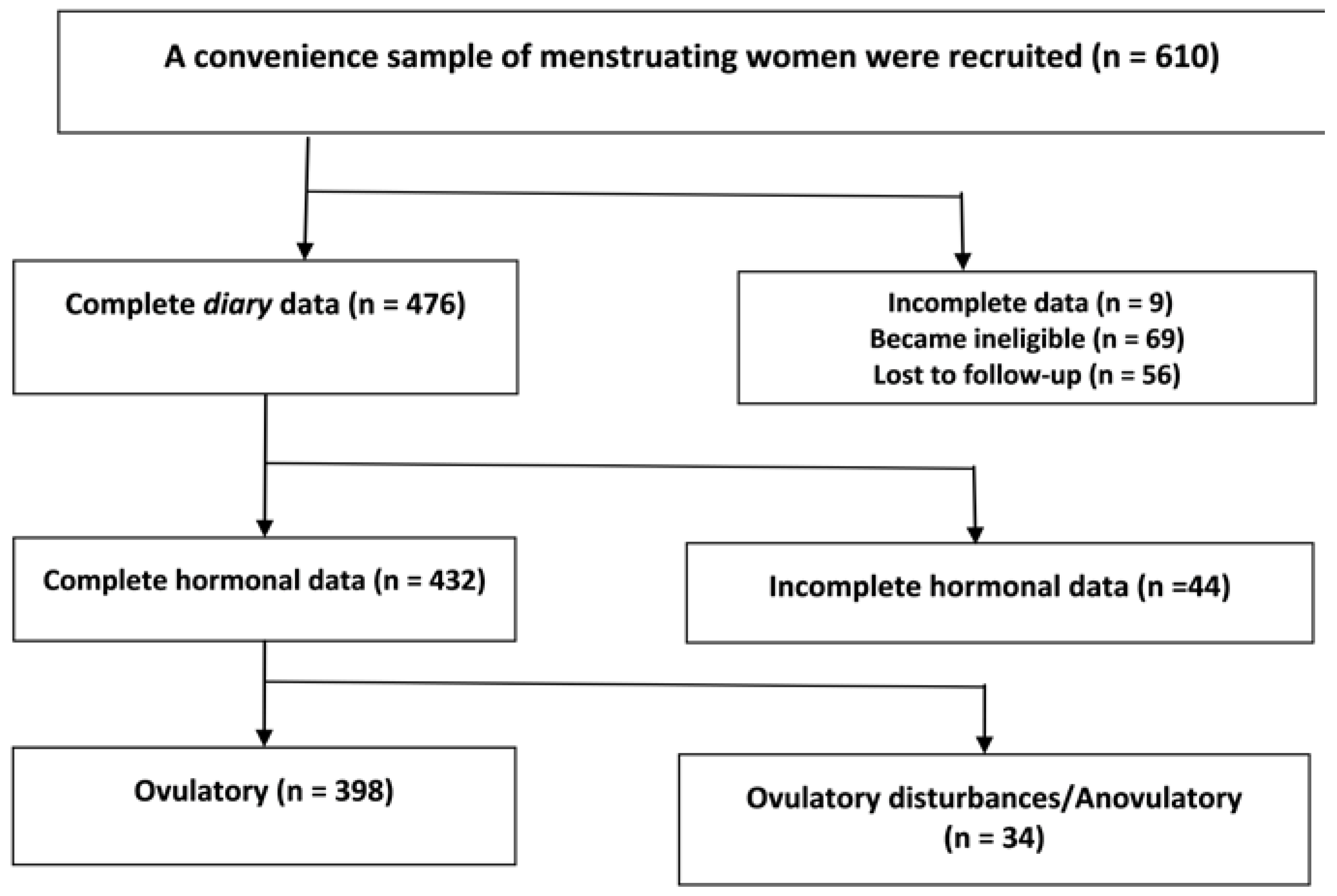
2.2. Flow of Participants through the Study
2.3. Molimina Documentation
2.4. Hormonal Cycle Documentation
2.5. Statistical Analysis
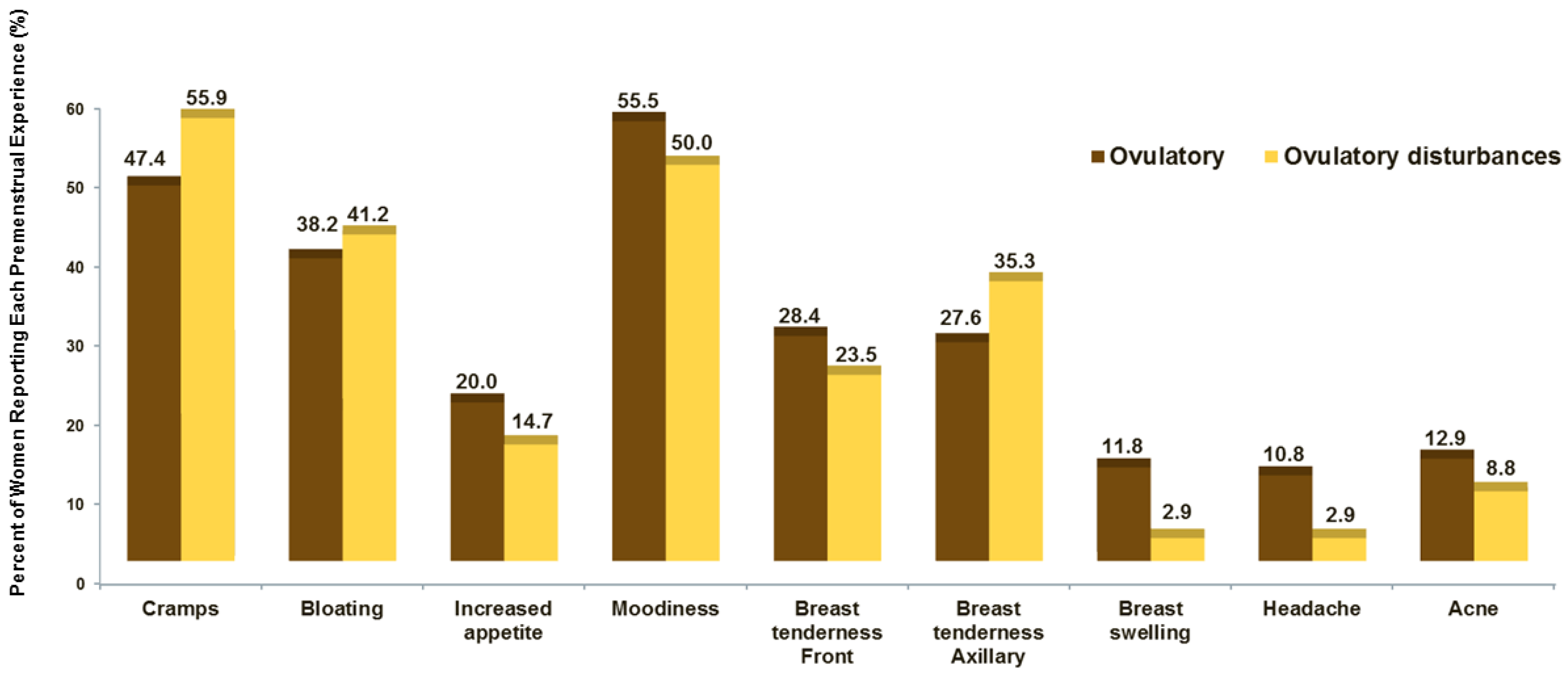
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khosla, S.; Melton, L.J., III; Riggs, B.L. The unitary model for estrogen deficiency and the pathogenesis of osteoporosis: Is a revision needed? J. Bone Miner. Res. 2011, 26, 441–451. [Google Scholar] [CrossRef] [PubMed]
- Nicodemus, K.K.; Folsom, A.R.; Anderson, K.E. Menstrual history and risk of hip fractures in postmenopausal women. The Iowa Women’s Health Study. Am. J. Epidemiol. 2001, 153, 251–255. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, F.; Wang, X.; Arguelles, L.; Rosul, L.L.; Venners, S.A.; Chen, C.; Hsu, Y.H.; Terwedow, H.; Wu, D.; Tang, G.; et al. Menstrual cycle lengths and bone mineral density: A cross-sectional, population-based study in rural Chinese women ages 30–49 years. Osteoporos. Int. 2007, 18, 221–233. [Google Scholar] [CrossRef] [PubMed]
- Clarke, C.L.; Sutherland, R.L. Progestin regulation of cellular proliferation. Endocr. Rev. 1990, 11, 266–301. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Vigna, Y.M.; Schechter, M.T.; Burgess, A.E. Spinal bone loss and ovulatory disturbances. N. Engl. J. Med. 1990, 323, 1221–1227. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Naess, M.; Langhammer, A.; Forsmo, S. Ovulation Prevalence in Women with Spontaneous Normal-Length Menstrual Cycles—A Population-Based Cohort from HUNT3, Norway. PLoS ONE 2015, 10, e0134473. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Hitchcock, C.L.; Barr, S.I.; Yu, T.; Prior, J.C. Negative Spinal Bone Mineral Density Changes and Subclinical Ovulatory Disturbances—Prospective Data in Healthy Premenopausal Women With Regular Menstrual Cycles. Epidemiol. Rev. 2014, 36, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.V.; Center, J.R.; Sambrook, P.N.; Eisman, J.A. Risk factors for proximal humerus, forearm, and wrist fractures in elderly men and women: The Dubbo Osteoporosis Epidemiology Study. Am. J. Epidemiol. 2001, 153, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.I.; Berga, S.L.; Cameron, J.L. Synergism between psychosocial and metabolic stressors: Impact on reproductive function in cynomolgus monkeys. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E270–E276. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Vigna, Y.M.; Barr, S.I.; Rexworthy, C.; Lentle, B.C. Cyclic medroxyprogesterone treatment increases bone density: A controlled trial in active women with menstrual cycle disturbances. Am. J. Med. 1994, 96, 521–530. [Google Scholar] [CrossRef]
- Michopoulos, V.; Mancini, F.; Loucks, T.L.; Berga, S.L. Neuroendocrine recovery initiated by cognitive behavioral therapy in women with functional hypothalamic amenorrhea: A randomized, controlled trial. Fertil. Steril. 2013, 99, 2084–2091. [Google Scholar] [CrossRef] [PubMed]
- Berga, S.L.; Loucks, T.L. Use of cognitive behavior therapy for functional hypothalamic amenorrhea. Ann. N. Y. Acad. Sci. 2006, 1092, 114–129. [Google Scholar] [CrossRef] [PubMed]
- Munster, K.; Helm, P.; Schmidt, L. Secondary amenorrhoea: Prevalence and medical contact—A cross-sectional study from a Danish county. Br. J. Obstet. Gynaecol. 1992, 99, 430–433. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Ho Yeun, B.; Clement, P.; Bowie, L.; Thomas, J. Reversible luteal phase changes and infertility associated with marathon training. Lancet 1982, 1, 269–270. [Google Scholar] [CrossRef]
- Magyar, D.M.; Boyers, S.P.; Marshall, J.R.; Abraham, G.E. Regular menstrual cycles and premenstrual molimina as indicators of ovulation. Obstet. Gynecol. 1979, 53, 441–444. [Google Scholar]
- White, C.P.; Hitchcock, C.L.; Vigna, Y.M.; Prior, J.C. Fluid Retention over the Menstrual Cycle: 1-Year Data from the Prospective Ovulation Cohort. Obstet. Gynecol. Int. 2011, 2011, 138451. [Google Scholar] [CrossRef] [PubMed]
- Harvey, A.; Hitchcock, C.L.; Prior, J.C. Ovulation disturbances and mood across the menstrual cycles of healthy women. J. Psychosom. Obstet. Gynaecol. 2009, 30, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C. Ovulatory disturbances: They do matter. Can. J. Diagn. 1997, 14, 64–80. [Google Scholar]
- O’Connor, K.A.; Brindle, E.; Holman, D.J.; Klein, N.A.; Soules, M.R.; Campbell, K.L.; Kohen, F.; Munro, C.J.; Shofer, J.B.; Lasley, B.L.; et al. Urinary estrone conjugate and pregnanediol 3-glucuronide enzyme immunoassays for population research. Clin. Chem. 2003, 49, 1139–1148. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.A.; Brindle, E.; Miller, R.C.; Shofer, J.B.; Ferrell, R.J.; Klein, N.A.; Soules, M.R.; Holman, D.J.; Mansfield, P.K.; Wood, J.W. Ovulation detection methods for urinary hormones: Precision, daily and intermittent sampling and a combined hierarchical method. Hum. Reprod. 2006, 21, 1442–1452. [Google Scholar] [CrossRef] [PubMed]
- Battistella, E.; Kalyan, S.; Prior, J. Evaluation of Successful Methods and Costs Associated with Recruiting 600 Healthy Women Volunteers to a Study of Ovulation. J. Women’s Health 2010, 19, 1519–1524. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C. Exercise-associated menstrual disturbances. In Reproductive Endocrinology, Surgery and Technology; Adashi, E.Y., Rock, J.A., Rosenwaks, Z., Eds.; Raven Press: New York, NY, USA, 1996; pp. 1077–1091. [Google Scholar]
- Kreiger, N.; Tenenhouse, A.; Joseph, L.; Mackenzie, M.D.; Poliquin, S.; Brown, J.P.; Prior, J.C.; Rittmaster, R.S. The Canadian Multicentre Osteoporosis Study (CaMos): Background, rationale, methods. Can. J. Aging 1999, 18, 376–387. [Google Scholar] [CrossRef]
- Baird, D.D.; Weinberg, C.R.; Wilcox, A.J.; McConnaughey, D.R.; Musey, P.I. Using the ratio of urinary oestrogen and progesterone metabolites to estimate day of ovulation. Stat. Med. 1991, 10, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Israel, R.; Mishell, D.R., Jr.; Stone, S.C.; Thorneycroft, I.H.; Moyer, D.L. Single luteal phase serum progesterone assay as an indicator of ovulation. Am. J. Obstet. Gynecol. 1972, 112, 1043–1046. [Google Scholar] [CrossRef]
- Bedford, J.L.; Prior, J.C.; Barr, S.I. A prospective exploration of cognitive dietary restraint, subclinical ovulatory disturbances, cortisol and change in bone density over two years in healthy young women. J. Clin. Endocrinol. Metab. 2010, 95, 3291–3299. [Google Scholar] [CrossRef] [PubMed]
- Bedford, J.L.; Prior, J.C.; Hitchcock, C.L.; Barr, S.I. Detecting evidence of luteal activity by least-squares quantitative basal temperature analysis against urinary progesterone metabolites and the effect of wake-time variability. Eur. J. Obstet. Gynecol. Reprod. Biol. 2009, 146, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Prior, J.C.; Vigna, Y.M.; Schulzer, M.; Hall, J.E.; Bonen, A. Determination of luteal phase length by quantitative basal temperature methods: Validation against the midcycle LH peak. Clin. Investig. Med. 1990, 13, 123–131. [Google Scholar]
- Waller, K.; Reim, J.; Fensler, L.; Swan, S.H.; Brumback, B.; Windham, G.C.; Lasley, B.; Ettinger, B.; Marcus, R. Bone mass and subtle abnormalities in ovulatory function in healthy women. J. Clin. Endocrinol. Metab. 1996, 81, 663–668. [Google Scholar] [PubMed]
- Lynch, K.E.; Mumford, S.L.; Schliep, K.C.; Whitcomb, B.W.; Zarek, S.M.; Pollack, A.Z.; Bertone-Johnson, E.R.; Danaher, M.; Wactawski-Wende, J.; Gaskins, A.J.; et al. Assessment of anovulation in eumenorrheic women: Comparison of ovulation detection algorithms. Fertil. Steril. 2014, 102, 511–518. [Google Scholar] [CrossRef] [PubMed]
- Bujang, M.A.; Adnan, T.H. Requirements for Minimum Sample Size for Sensitivity and Specificity Analysis. J. Clin. Diagn. Res. 2016, 10, YE01–YE06. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, H.K.; Brixen, K.; Bouillon, R.; Mosekilde, L. Changes in biochemical markers of osteoblastic activity during the menstrual cycle. J. Clin. Endocrinol. Metab. 1990, 70, 1431–1437. [Google Scholar] [CrossRef] [PubMed]
- Israel, S.L.; Schnelle, O. The thermogenic property of progesterone. Fertil. Steril. 1950, 1, 53–65. [Google Scholar] [CrossRef]
- Kalyan, S.; Prior, J.C. Bone changes and fracture related to menstrual cycles and ovulation. Crit. Rev. Eukaryot. Gene Expr. 2010, 20, 213–233. [Google Scholar] [CrossRef] [PubMed]
- Barr, S.I.; Janelle, K.C.; Prior, J.C. Energy Intakes Are Higher During the Luteal-Phase of Ovulatory Menstrual Cycles. Am. J. Clin. Nutr. 1995, 61, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Tisdale, J.E.; Jaynes, H.A.; Overholser, B.R.; Sowinski, K.M.; Flockhart, D.A.; Kovacs, R.J. Influence of Oral Progesterone Administration on Drug-Induced QT Interval Lengthening: A Randomized, Double-Blind, Placebo-Controlled Crossover Study. JACC Clin. Electrophysiol. 2016, 2, 765–774. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Total Cohort (N: 610) | Women with Hormonal Data (N: 432) | ||
|---|---|---|---|---|
| Ovulatory N: 398 | Ovulatory Disturbances N: 34 | p Value 1 | ||
| Age (years) Mean (SD) | 31.5 (5.3) | 32.1 (5.3) | 29.4 (5.2) | 0.005 |
| BMI (kg/m2) Mean (SD) | 24.4 (4.7) | 24.3 (4.4) | 23.8 (6.2) | 0.558 |
| Education: N (%) | 0.215 | |||
| High-school or less | 36 (5.9) | 18 (4.5) | 4 (11.8) | |
| University certificate or less | 143 (23.4) | 78 (19.6) | 7 (20.6) | |
| University degree | 431 (70.7) | 302 (75.9) | 23 (67.6) | |
| Employment: N (%) | 0.964 | |||
| Full-time | 360 (59) | 237 (59.5) | 20 (58.8) | |
| Part-time | 97 (15.9) | 68 (17.1) | 5 (14.7) | |
| Student | 100 (16.4) | 62 (15.6) | 7 (20.6) | |
| Other | 53 (8.7) | 31 (7.8) | 2 (5.9) | |
| Age at menarche (years) Mean (SD) | 12.7 (1.5) | 12.7 (1.4) | 13.3 (1.5) | 0.012 |
| Cycle length (days) Mean (SD) | 29.3 (6.1) | 29.3 (4.7) | 31.8 (14) | 0.015 |
| Molimina Question (3 = usually and 4 = always) | 545 (89.3) | 353 (88.7) | 33 (97.1) | 0.682 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prior, J.C.; Konishi, C.; Hitchcock, C.L.; Kingwell, E.; Janssen, P.; Cheung, A.P.; Fairbrother, N.; Goshtasebi, A. Does Molimina Indicate Ovulation? Prospective Data in a Hormonally Documented Single-Cycle in Spontaneously Menstruating Women. Int. J. Environ. Res. Public Health 2018, 15, 1016. https://doi.org/10.3390/ijerph15051016
Prior JC, Konishi C, Hitchcock CL, Kingwell E, Janssen P, Cheung AP, Fairbrother N, Goshtasebi A. Does Molimina Indicate Ovulation? Prospective Data in a Hormonally Documented Single-Cycle in Spontaneously Menstruating Women. International Journal of Environmental Research and Public Health. 2018; 15(5):1016. https://doi.org/10.3390/ijerph15051016
Chicago/Turabian StylePrior, Jerilynn C., Chiaki Konishi, Christine L. Hitchcock, Elaine Kingwell, Patti Janssen, Anthony P. Cheung, Nichole Fairbrother, and Azita Goshtasebi. 2018. "Does Molimina Indicate Ovulation? Prospective Data in a Hormonally Documented Single-Cycle in Spontaneously Menstruating Women" International Journal of Environmental Research and Public Health 15, no. 5: 1016. https://doi.org/10.3390/ijerph15051016
APA StylePrior, J. C., Konishi, C., Hitchcock, C. L., Kingwell, E., Janssen, P., Cheung, A. P., Fairbrother, N., & Goshtasebi, A. (2018). Does Molimina Indicate Ovulation? Prospective Data in a Hormonally Documented Single-Cycle in Spontaneously Menstruating Women. International Journal of Environmental Research and Public Health, 15(5), 1016. https://doi.org/10.3390/ijerph15051016






