Lagged Influence of Fine Particulate Matter and Geographic Disparities on Clinic Visits for Children’s Asthma in Taiwan
Abstract
1. Introduction
2. Materials and Methods
2.1. Asthma Data
2.2. Environmental Data
2.3. Study Area
2.4. Imputation of Missing Environmental Data
2.5. Statistical Modeling Approach
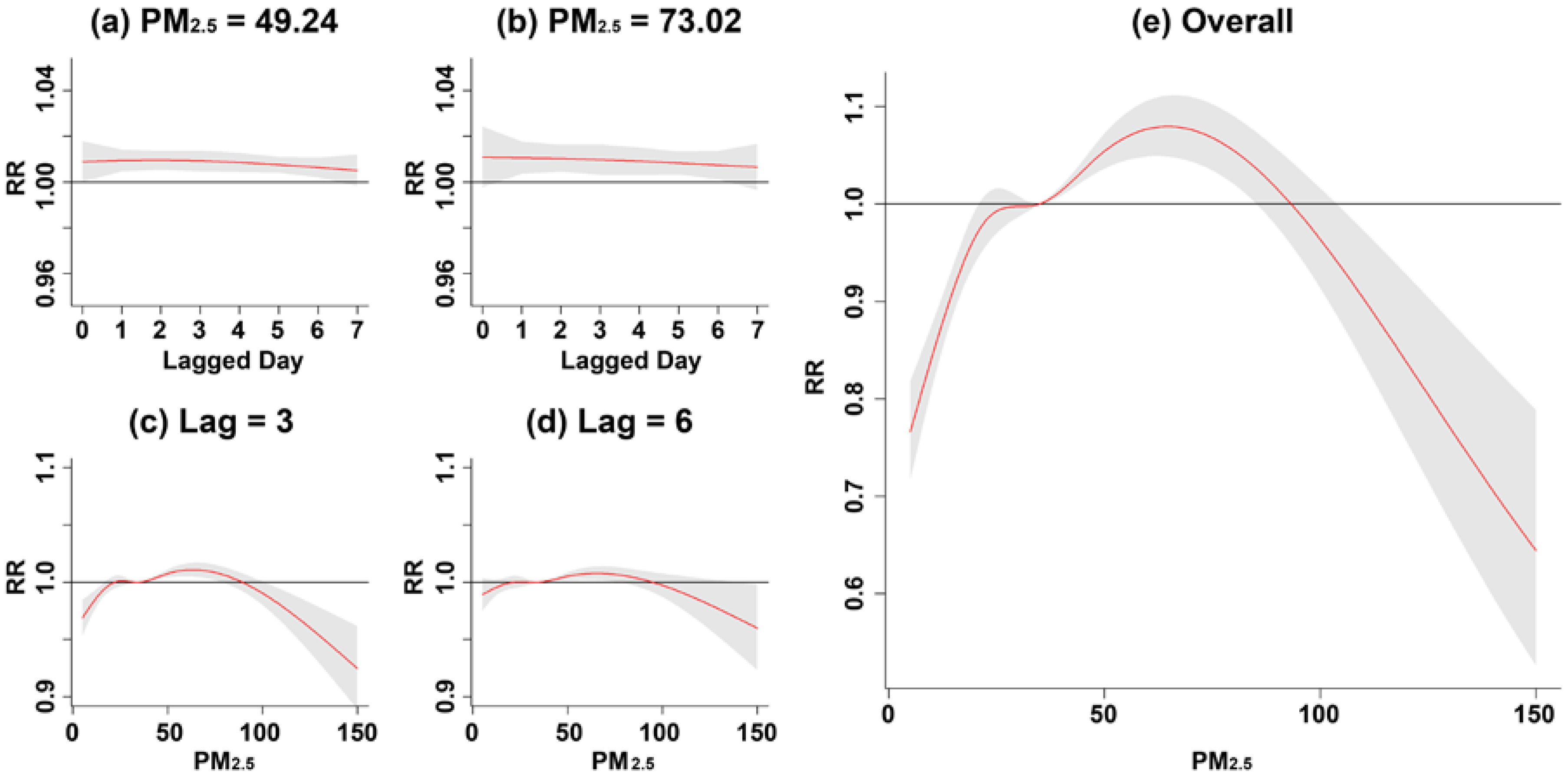
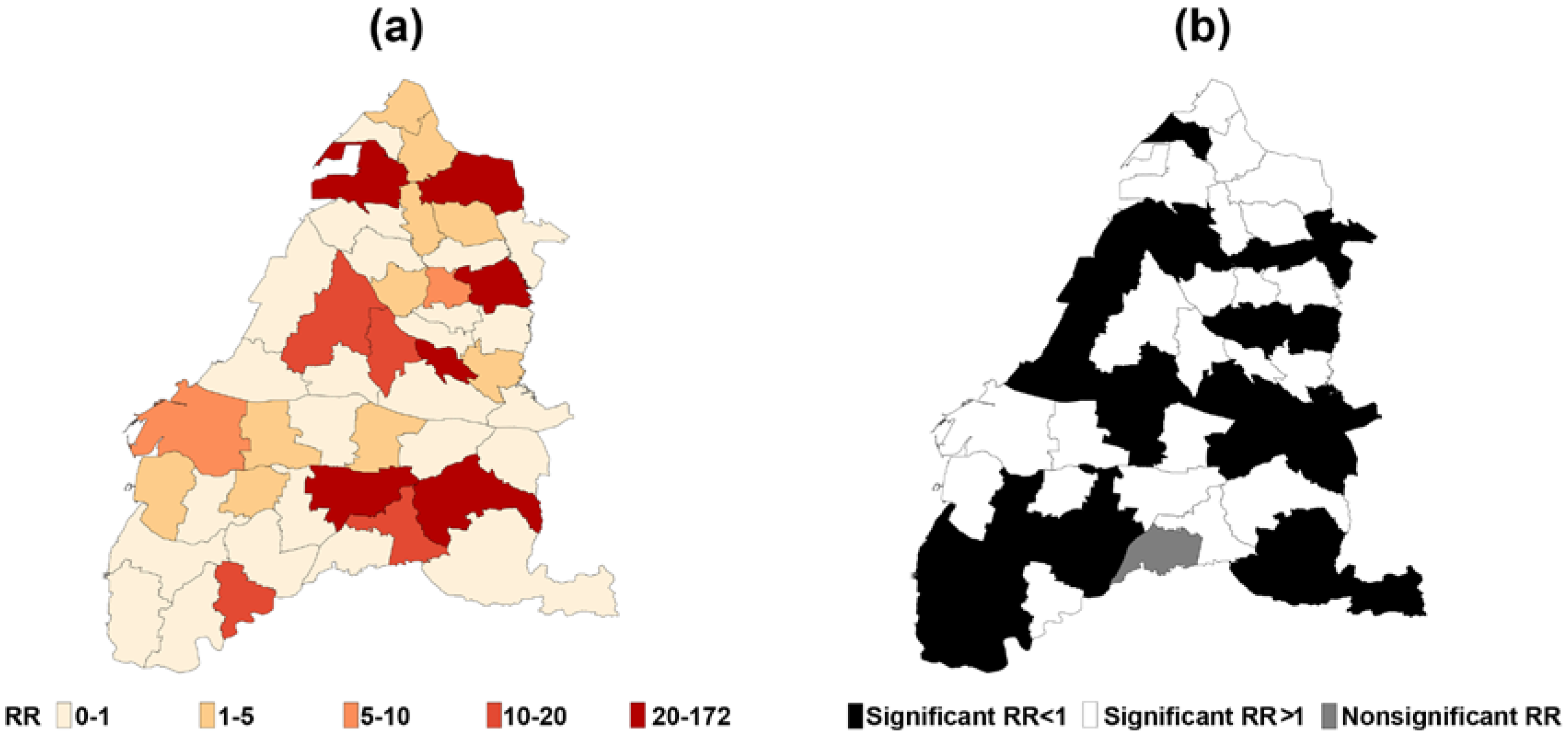
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Akinbami, L.J.; Schoendorf, K.C. Trends in childhood asthma: Prevalence, health care utilization, and mortality. Pediatrics 2002, 110, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Asher, M.I.; Montefort, S.; Bjorksten, B.; Lai, C.K.; Strachan, D.P.; Weiland, S.K.; William, H.; ISAAC Phase Three Study Group. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC Phases One and Three repeat multicountry cross-sectional surveys. Lancet 2006, 368, 733–743. [Google Scholar] [CrossRef]
- Asher, M.I.; Weiland, S.K. The International Study of Asthma and Allergies in Childhood (ISAAC). ISAAC Steering Committee. Clin. Exp. Allergy 1998, 28 (Suppl. 5), 52–66. [Google Scholar] [CrossRef] [PubMed]
- Magnus, P.; Jaakkola, J.J. Secular trend in the occurrence of asthma among children and young adults: Critical appraisal of repeated cross sectional surveys. BMJ 1997, 314, 1795–1799. [Google Scholar] [CrossRef] [PubMed]
- Maziak, W.; Behrens, T.; Brasky, T.M.; Duhme, H.; Rzehak, P.; Weiland, S.K.; Keil, U. Are asthma and allergies in children and adolescents increasing? Results from ISAAC phase I and phase III surveys in Munster, Germany. Allergy 2003, 58, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Anandan, C.; Nurmatov, U.; van Schayck, O.C.; Sheikh, A. Is the prevalence of asthma declining? Systematic review of epidemiological studies. Allergy 2010, 65, 152–167. [Google Scholar] [CrossRef] [PubMed]
- Burney, P. The changing prevalence of asthma? Thorax 2002, 57 (Suppl. 2), II36–II39. [Google Scholar] [PubMed]
- Downs, S.H.; Marks, G.B.; Sporik, R.; Belosouva, E.G.; Car, N.G.; Peat, J.K. Continued increase in the prevalence of asthma and atopy. Arch. Dis. Child. 2001, 84, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, K.H.; Shen, J.J. Prevalence of childhood asthma in Taipei, Taiwan, and other Asian Pacific countries. J. Asthma 1988, 25, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Tsuang, H.C.; Su, H.J.; Kao, F.F.; Shih, H.C. Effects of changing risk factors on increasing asthma prevalence in southern Taiwan. Paediatr. Perinat. Epidemiol. 2003, 17, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.Y.; Chen, Y.J.; Lin, M.W.; Chen, T.J.; Chu, S.Y.; Chen, C.C.; Lee, D.D.; Chang, Y.T.; Wang, W.J.; Liu, H.N. Prevalence of atopic dermatitis, allergic rhinitis and asthma in Taiwan: A national study 2000 to 2007. Acta Derm. Venereol. 2010, 90, 589–594. [Google Scholar] [PubMed]
- Wieslander, G.; Norback, D.; Bjornsson, E.; Janson, C.; Boman, G. Asthma and the indoor environment: The significance of emission of formaldehyde and volatile organic compounds from newly painted indoor surfaces. Int. Arch. Occup. Environ. Health 1997, 69, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.S.; Chang, L.T.; Lee, H.C.; Chan, C.C. Effects of personal particulate matter on peak expiratory flow rate of asthmatic children. Sci. Total Environ. 2007, 382, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Chan, T.C.; Chen, M.L.; Lin, I.F.; Lee, C.H.; Chiang, P.H.; Wang, D.W.; Chuang, J.H. Spatiotemporal analysis of air pollution and asthma patient visits in Taipei, Taiwan. Int. J. Health Geogr. 2009, 8, 26. [Google Scholar] [CrossRef] [PubMed]
- Hyrkas, H.; Jaakkola, M.S.; Ikaheimo, T.M.; Hugg, T.T.; Jaakkola, J.J. Asthma and allergic rhinitis increase respiratory symptoms in cold weather among young adults. Respir. Med. 2014, 108, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.L.; Lin, Y.C.; Sung, F.C.; Huang, S.L.; Ko, Y.C.; Lai, J.S.; Dockery, D.W. Climate, traffic-related air pollutants, and asthma prevalence in middle-school children in Taiwan. Environ. Health Perspect. 1999, 107, 1001–1006. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, F.A.; Muller, A.; Busi, L.E.; Cianni, N.; Massolo, L.; Schlink, U.; Porta, A.; Sly, P.D. Increased asthma and respiratory symptoms in children exposed to petrochemical pollution. J. Allergy Clin. Immunol. 2009, 123, 632–638. [Google Scholar] [CrossRef] [PubMed]
- USEPA. Asthma Facts. United States Environmental Protection Agency, 2015. Available online: https://www.epa.gov/sites/production/files/2015-10/documents/asthma_fact_sheet_eng_july_30_2015_v2.pdf (accessed on 17 January 2018).
- USEPA. Ozone and Your Health. United States Environmental Protection Agency, 2009. Available online: https://www3.epa.gov/airnow/ozone-c.pdf (accessed on 17 January 2018).
- Schwartz, J.; Sarnat, J.A.; Coull, B.A.; Wilson, W.E. Effects of exposure measurement error on particle matter epidemiology: A simulation using data from a panel study in Baltimore, MD. J. Expo. Sci. Environ. Epidemiol. 2007, 17 (Suppl. 2), S2. [Google Scholar] [CrossRef] [PubMed]
- Kelly, F.J.; Fussell, J.C. Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos. Environ. 2012, 60, 504–526. [Google Scholar] [CrossRef]
- Villeneuve, P.J.; Goldberg, M.S.; Krewski, D.; Burnett, R.T.; Chen, Y. Fine particulate air pollution and all-cause mortality within the Harvard Six-Cities Study: Variations in risk by period of exposure. Ann. Epidemiol. 2002, 12, 568–576. [Google Scholar] [CrossRef]
- Chen, B.Y.; Chen, C.H.; Chen, P.C.; Wang, G.S.; Guo, Y.L. Air Pollution, Allergic Co-morbidity, and Emergency Department Visit for Pediatric Asthma in Taiwan. Aerosol Air Qual. Res. 2013, 13, 1847–1852. [Google Scholar] [CrossRef]
- Jalaludin, B.; Khalaj, B.; Sheppeard, V.; Morgan, G. Air pollution and ED visits for asthma in Australian children: A case-crossover analysis. Int. Arch. Occup. Environ. Health 2008, 81, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, J.C.; Lumley, T.; Sheppard, L.; Koenig, J.Q.; Shapiro, G.G. Effects of ambient air pollution on symptom severity and medication use in children with asthma. Ann. Allergy Asthma Immunol. 2003, 91, 346–353. [Google Scholar] [CrossRef]
- Tecer, L.H.; Alagha, O.; Karaca, F.; Tuncel, G.; Eldes, N. Particulate matter (PM(2.5), PM(10-2.5), and PM(10)) and children’s hospital admissions for asthma and respiratory diseases: A bidirectional case-crossover study. J. Toxicol. Environ. Health A 2008, 71, 512–520. [Google Scholar] [CrossRef] [PubMed]
- Andersen, Z.J.; Wahlin, P.; Raaschou-Nielsen, O.; Ketzel, M.; Scheike, T.; Loft, S. Size distribution and total number concentration of ultrafine and accumulation mode particles and hospital admissions in children and the elderly in Copenhagen, Denmark. Occup. Environ. Med. 2008, 65, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Iskandar, A.; Andersen, Z.J.; Bonnelykke, K.; Ellermann, T.; Andersen, K.K.; Bisgaard, H. Coarse and fine particles but not ultrafine particles in urban air trigger hospital admission for asthma in children. Thorax 2012, 67, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Chen, Y.; Burnett, R.T.; Villeneuve, P.J.; Krewski, D. The influence of ambient coarse particulate matter on asthma hospitalization in children: Case-crossover and time-series analyses. Environ. Health Perspect. 2002, 110, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, S.; Shima, M.; Yoda, Y.; Oka, K.; Kurosaka, F.; Shimizu, S.; Takahashi, H.; Nikatani, Y.; Nishikawa, J.; Fujiwara, K.; et al. Association between PM2.5 and primary care visits due to asthma attack in Japan: Relation to Beijing’s air pollution episode in January 2013. Environ. Health Prev. Med. 2014, 19, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Malm, W.C.; Schichtel, B.A.; Pitchford, M.L.; Ashbaugh, L.L.; Eldred, R.A. Spatial and monthly trends in speciated fine particle concentration in the United States. J. Geophys. Res. Atmos. 2004, 109. [Google Scholar] [CrossRef]
- Bell, M.L.; HEI Health Review Committee. Assessment of the health impacts of particulate matter characteristics. Res. Rep. Health Eff. Inst. 2012, 161, 5–38. [Google Scholar]
- Englert, N. Fine particles and human health—A review of epidemiological studies. Toxicol. Lett. 2004, 149, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Dominici, F.; Peng, R.D.; Bell, M.L.; Pham, L.; McDermott, A.; Zeger, S.L.; Samet, J.M. Fine particulate air pollution and hospital admission for cardiovascular and respiratory diseases. JAMA 2006, 295, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Oyana, T.J.; Rivers, P.A. Geographic variations of childhood asthma hospitalization and outpatient visits and proximity to ambient pollution sources at a U.S.-Canada border crossing. Int. J. Health Geogr. 2005, 4, 14. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.M.; Vichit-Vadakan, N.; Vajanapoom, N.; Ostro, B.; Thach, T.Q.; Chau, P.Y.; Chan, E.K.; Chung, R.Y.; Ou, C.Q.; Yang, L.; et al. Part 5. Public health and air pollution in Asia (PAPA): A combined analysis of four studies of air pollution and mortality. Res. Rep. Health Eff. Inst. 2010, 154, 377–418. [Google Scholar]
- Lee, S.L.; Wong, W.H.; Lau, Y.L. Association between air pollution and asthma admission among children in Hong Kong. Clin. Exp. Allergy 2006, 36, 1138–1146. [Google Scholar] [CrossRef] [PubMed]
- Tiittanen, P.; Timonen, K.L.; Ruuskanen, J.; Mirme, A.; Pekkanen, J. Fine particulate air pollution, resuspended road dust and respiratory health among symptomatic children. Eur. Respir. J. 1999, 13, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Ostro, B.; Lipsett, M.; Mann, J.; Braxton-Owens, H.; White, M. Air pollution and exacerbation of asthma in African-American children in Los Angeles. Epidemiology 2001, 12, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Halonen, J.I.; Lanki, T.; Yli-Tuomi, T.; Kulmala, M.; Tiittanen, P.; Pekkanen, J. Urban air pollution, and asthma and COPD hospital emergency room visits. Thorax 2008, 63, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Ostro, B.; Roth, L.; Malig, B.; Marty, M. The effects of fine particle components on respiratory hospital admissions in children. Environ Health Perspect. 2009, 117, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Peel, J.L.; Tolbert, P.E.; Klein, M.; Metzger, K.B.; Flanders, W.D.; Todd, K.; Mulholland, J.A.; Ryan, P.B.; Frumkin, H. Ambient air pollution and respiratory emergency department visits. Epidemiology 2005, 16, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Zanobetti, A.; Schwartz, J.; Samoli, E.; Gryparis, A.; Touloumi, G.; Peacock, J.; Anderson, R.H.; Le Tertre, A.; Bobros, J.; Celko, M.; et al. The temporal pattern of respiratory and heart disease mortality in response to air pollution. Environ. Health Perspect. 2003, 111, 1188–1193. [Google Scholar] [CrossRef] [PubMed]
- de Hartog, J.J.; Lanki, T.; Timonen, K.L.; Hoek, G.; Janssen, N.A.; Ibald-Mulli, A.; Peters, A.; Heinrich, J.; Tarkiainen, T.H.; van Grieken, R.; et al. Associations between PM2.5 and heart rate variability are modified by particle composition and beta-blocker use in patients with coronary heart disease. Environ. Health Perspect. 2009, 117, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Slaughter, J.C.; Kim, E.; Sheppard, L.; Sullivan, J.H.; Larson, T.V.; Claiborn, C. Association between particulate matter and emergency room visits, hospital admissions and mortality in Spokane, Washington. J. Expo. Anal. Environ. Epidemiol. 2005, 15, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Batterman, S.; Wasilevich, E.; Wahl, R.; Wirth, J.; Su, F.C.; Mukherjee, B. Association of daily asthma emergency department visits and hospital admissions with ambient air pollutants among the pediatric Medicaid population in Detroit: Time-series and time-stratified case-crossover analyses with threshold effects. Environ. Res. 2011, 111, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Huang, C.; Su, H.; Turner, L.R.; Qiao, Z.; Tong, S. Diurnal temperature range and childhood asthma: A time-series study. Environ. Health 2013, 12, 12. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Huang, C.; Hu, W.; Turner, L.R.; Su, H.; Tong, S. Extreme temperatures and emergency department admissions for childhood asthma in Brisbane, Australia. Occup. Environ. Med. 2013, 70, 730–735. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.F.R.; Hsiao, W.C. Does universal health insurance make health care unaffordable? Lessons from Taiwan. Health Aff. 2003, 22, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.L.; Wang, C.H. Retrospective prediction of intraurban spatiotemporal distribution of PM2.5 in Taipei. Atmos. Environ. 2010, 44, 3053–3065. [Google Scholar]
- Kolovos, A.; Skupin, A.; Jerrett, M.; Christakos, G. Multi-Perspective Analysis and Spatiotemporal Mapping of Air Pollution Monitoring Data. Environ. Sci. Technol. 2010, 44, 6738–6744. [Google Scholar] [CrossRef] [PubMed]
- Zagouras, A.; Kolovos, A.; Coimbra, C.F.M. Objective framework for optimal distribution of solar irradiance monitoring networks. Renew. Energy 2015, 80, 153–165. [Google Scholar] [CrossRef]
- Christakos, G. A Bayesian Maximum-Entropy View to the Spatial Estimation Problem. Math. Geol. 1990, 22, 763–777. [Google Scholar] [CrossRef]
- Christakos, G.; Serre, M.L.; Kovitz, J.L. BME representation of particulate matter distributions in the state of California on the basis of uncertain measurements. J. Geophys. Res. Atmos. 2001, 106, 9717–9731. [Google Scholar] [CrossRef]
- De Nazelle, A.; Arunachalam, S.; Serre, M.L. Bayesian Maximum Entropy Integration of Ozone Observations and Model Predictions: An Application for Attainment Demonstration in North Carolina. Environ. Sci. Technol. 2010, 44, 5707–5713. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.L.; Chen, J.C.; Christakos, G.; Jerrett, M. BME Estimation of Residential Exposure to Ambient PM10 and Ozone at Multiple Time Scales. Environ. Health Perspect. 2009, 117, 537–544. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Christakos, G.; Serre, M.L. BME analysis of spatiotemporal particulate matter distributions in North Carolina. Atmos. Environ. 2000, 34, 3393–3406. [Google Scholar] [CrossRef]
- Kindermann, R.P.; Snell, J.L. Markov random fields and their applications. In Contemporary Mathematics; American Mathematical Society: Providence, RI, USA, 1980. [Google Scholar]
- Yu, H.L.; Kolovos, A.; Christakos, G.; Chen, J.C.; Warmerdam, S.; Dev, B. Interactive spatiotemporal modelling of health systems: The SEKS-GUI framework. Stoch. Environ. Res. Risk A 2007, 21, 555–572. [Google Scholar] [CrossRef]
- WHO. WHO Air Quality Guidelines for Particulate Matter, Ozone, Nitrogen Dioxide and Sulfur Dioxide: Global Update 2005. Summary of Risk Assessment. 2006. Available online: http://apps.who.int/iris/bitstream/10665/69477/1/WHO_SDE_PHE_OEH_06.02_eng.pdf (accessed on 17 January 2018).
- Norris, G.; YoungPong, S.N.; Koenig, J.Q.; Larson, T.V.; Sheppard, L.; Stout, J.W. An association between fine particles and asthma emergency department visits for children in Seattle. Environ. Health Perspect. 1999, 107, 489–493. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.Y.; Chau, T.T. An association between air pollution and daily outpatient visits for respiratory disease in a heavy industry area. PLoS ONE 2013, 8, e75220. [Google Scholar] [CrossRef] [PubMed]
- Taj, T.; Jakobsson, K.; Stroh, E.; Oudin, A. Air pollution is associated with primay health care visits for asthma in Sweden: A case-crossover design with a distributed lag nonlinear model. Spat. Spatio-Tempor. Epidemiol. 2016, 17, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Qiu, H.; Sun, S.; Yang, Y.; Lin, H.; Tian, L. Age-dependent effect of ambient ozone on emergency asthma hospitalizations in Hong Kong. J. Allergy Clin. Immunol. 2018, 141, 1532–1534. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, J. Is there harvesting in the association of airborne particles with daily deaths and hospital admissions? Epidemiology 2001, 12, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Chien, L.-C.; Guo, Y.; Li, X.; Yu, H.-L. Considering spatial heterogeneity in the distributed lag nonlinear model when analyzing spatiotemporal data. J. Expo. Sci. Environ. Epidemol. 2016, 28, 13. [Google Scholar]
- Ko, F.W.; Tam, W.; Wong, T.W.; Lai, C.K.; Wong, G.W.; Leung, T.F.; Ng, S.S.; Hui, D.S. Effects of air pollution on asthma hospitalization rates in different age groups in Hong Kong. Clin. Exp. Allergy 2007, 37, 1312–1319. [Google Scholar] [CrossRef] [PubMed]
- Peters, A.; Wichmann, H.E.; Tuch, T.; Heinrich, J.; Heyder, J. Respiratory effects are associated with the number of ultrafine particles. Am. J. Respir. Crit. Care Med. 1997, 155, 1376–1383. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.J.; Wilson, J.D.; Hiller, F.C. Respiratory tract deposition of ultrafine particles in subjects with obstructive or restrictive lung disease. Chest 1990, 97, 1115–1120. [Google Scholar] [CrossRef] [PubMed]
- Parrish, D.D. Critical evaluation of US on-road vehicle emission inventories. Atmos. Environ. 2006, 40, 2288–2300. [Google Scholar] [CrossRef]
- Wang, T.; Cheung, T.F.; Li, Y.S.; Yu, X.M.; Black, D.R. Emission characteristics of CO, NOx, SO2 and indications of biomass burning observed at a rural site in eastern China. J. Geophys. Res. 2002, 107, ACH-9-1–ACH-9-10. [Google Scholar] [CrossRef]
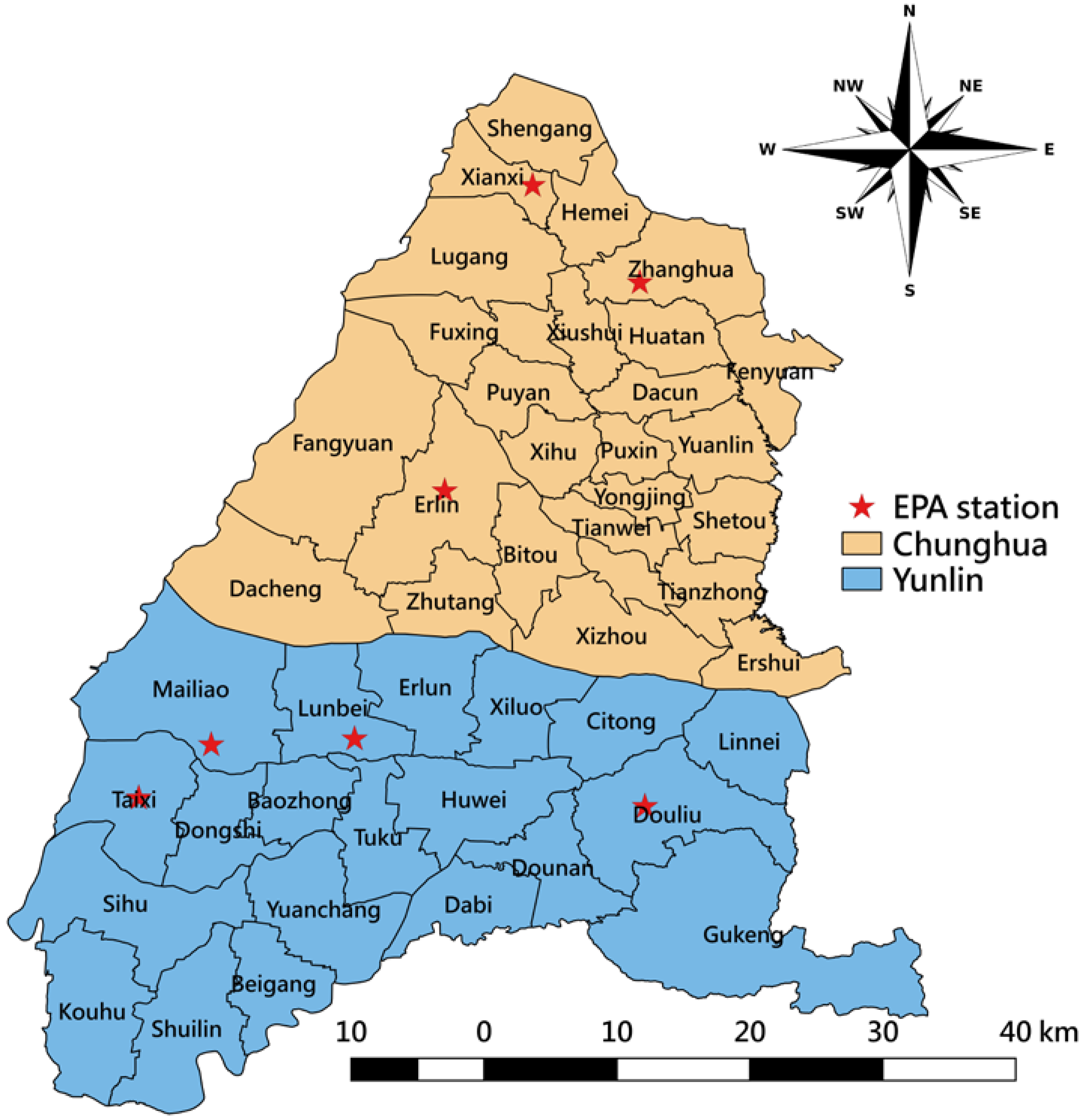
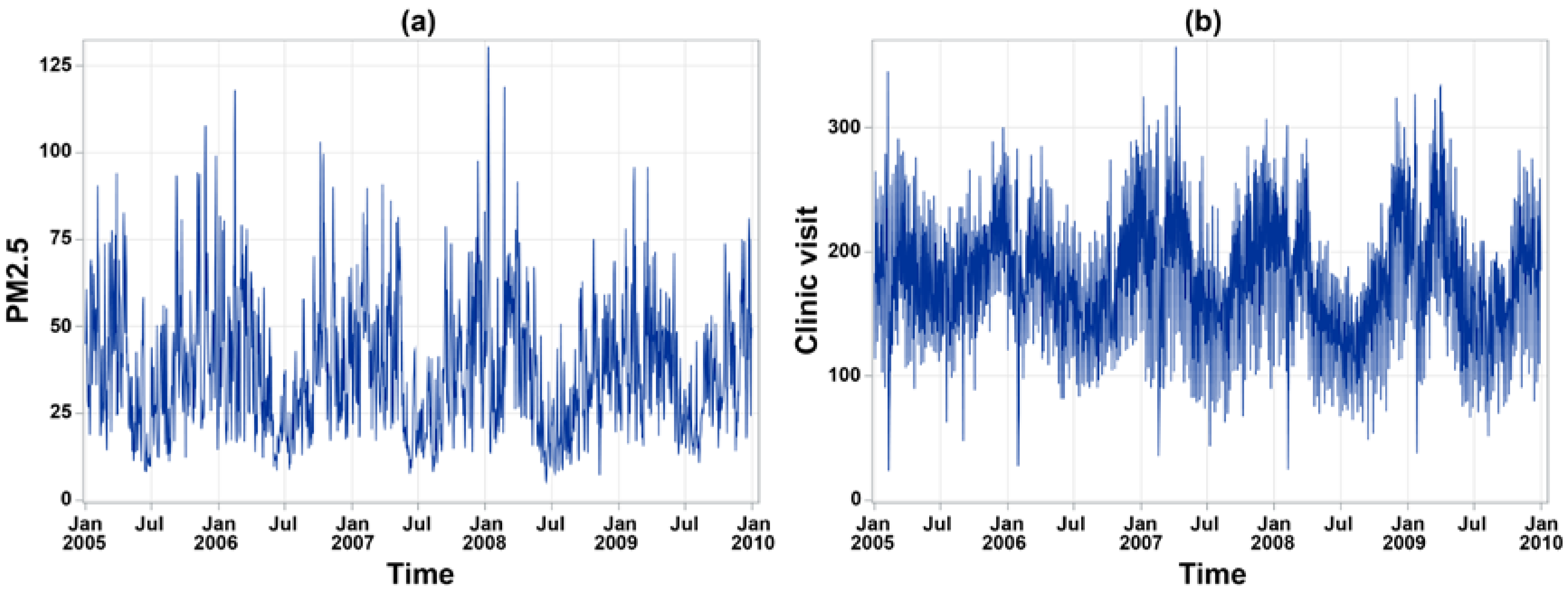
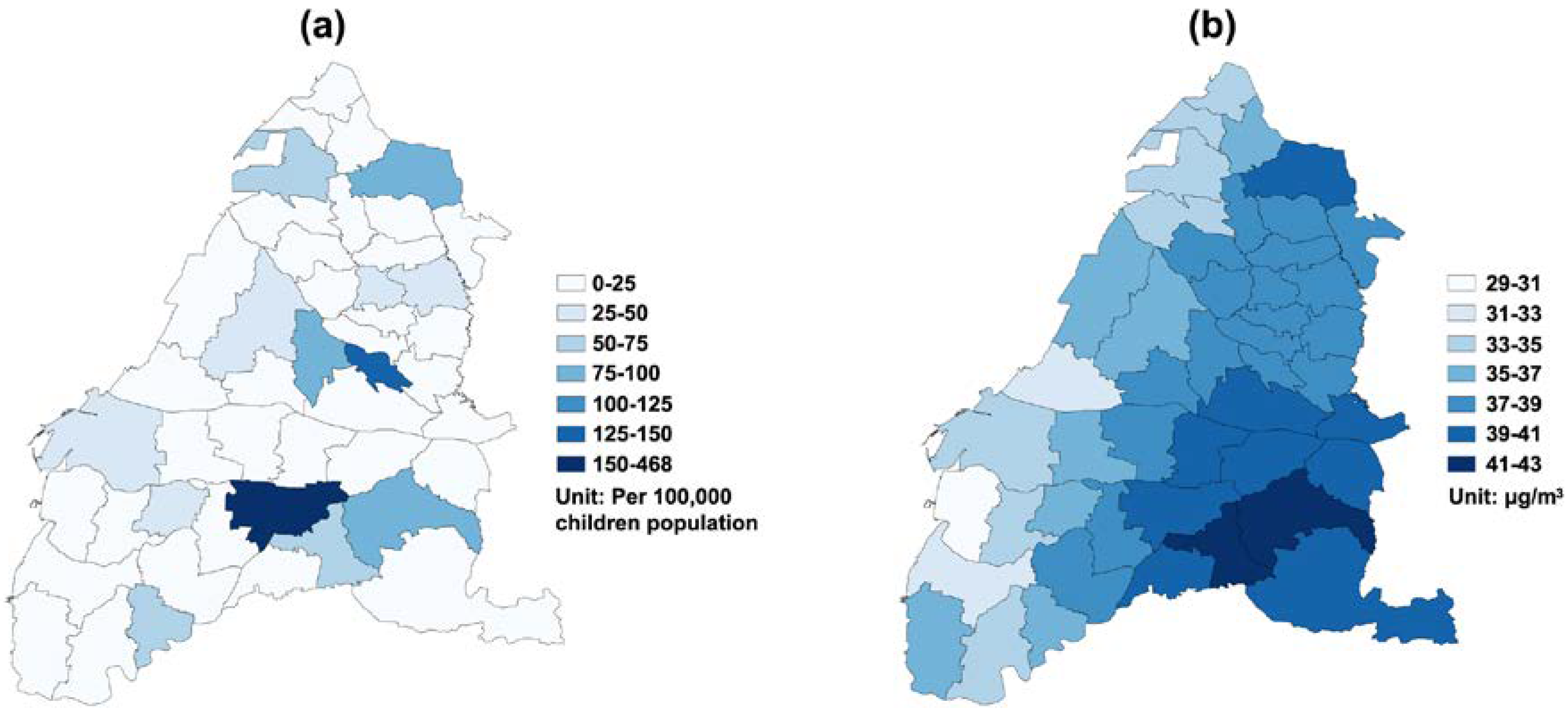
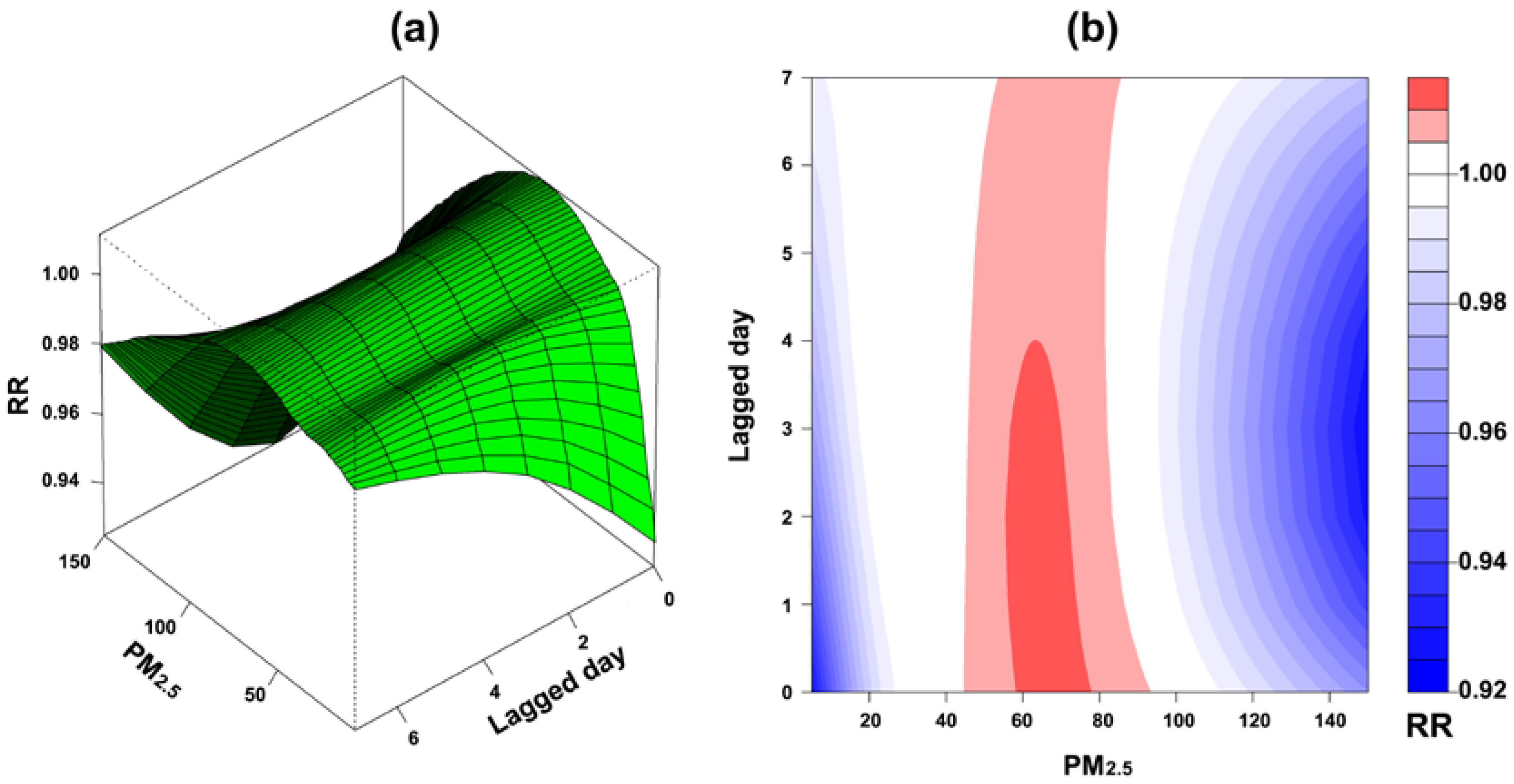
| Study Variables | Changhua | Yunlin | ||
|---|---|---|---|---|
| Mean | Standard deviation | Mean | Standard deviation | |
| Children’s asthma cases | 3.03 | 7.94 | 4.70 | 14.28 |
| PM2.5 (μg/m3) | 37.22 | 18.87 | 37.26 | 19.38 |
| O3 (ppb) | 28.89 | 10.01 | 31.68 | 10.67 |
| SO2 (ppb) | 3.73 | 1.42 | 3.59 | 1.39 |
| cPM (μg/m3) | 24.20 | 17.82 | 28.76 | 21.00 |
| CO/NOx (ppm/ppb) | 0.02 | 0.01 | 0.02 | 0.01 |
| Temperature (°C) | 22.33 | 4.85 | 21.87 | 4.69 |
| Relative humidity (%) | 68.02 | 8.00 | 69.67 | 7.09 |
| Frequency | Percent | Frequency | Percent | |
| Wind direction | ||||
| South | 15,371 | 26.98% | 11,510 | 26.27% |
| Southwest | 4164 | 7.31% | 2262 | 5.16% |
| West | 307 | 0.54% | 226 | 0.52% |
| Northwest | 136 | 0.24% | 162 | 0.37% |
| North | 1160 | 2.04% | 1714 | 3.91% |
| Northeast | 15,944 | 27.99% | 11,186 | 25.53% |
| East | 8429 | 14.80% | 7073 | 16.14% |
| Southeast | 11,455 | 20.11% | 9687 | 22.21% |
| Variable | RR | 95% CI | p-Value | |
|---|---|---|---|---|
| Day of the Week | ||||
| Sunday | 1.00 | Reference | - | |
| Monday | 2.07 | (2.04, 2.11) | <0.0001 | |
| Tuesday | 1.75 | (1.72, 1.79) | <0.0001 | |
| Wednesday | 1.71 | (1.68, 1.74) | <0.0001 | |
| Thursday | 1.45 | (1.42, 1.48) | <0.0001 | |
| Friday | 1.64 | (1.61, 1.67) | <0.0001 | |
| Saturday | 1.74 | (1.71, 1.77) | <0.0001 | |
| Wind direction | ||||
| South | 1.00 | Reference | - | |
| Southwest | 1.00 | (0.97, 1.02) | 0.6907 | |
| West | 0.82 | (0.76, 0.89) | <0.0001 | |
| Northwest | 0.89 | (0.82, 0.97) | 0.0076 | |
| North | 1.03 | (1.00, 1.06) | 0.0272 | |
| Northeast | 1.00 | (0.98, 1.02) | 0.9091 | |
| East | 1.00 | (0.98, 1.02) | 0.9532 | |
| Southeast | 1.00 | (0.99, 1.02) | 0.7150 | |
| RR% | 95% CI | p-Value | ||
| Air pollutant | ||||
| cPM | 1.42 | (1.01, 1.83) | <0.0001 | |
| CO/NOx | −3.16 | (−3.85, −2.46) | <0.0001 | |
| SO2 | 1.25 | (0.43, 2.08) | 0.0028 | |
| O3 | 0.82 | (−0.07, 1.71) | 0.0704 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chien, L.-C.; Chen, Y.-A.; Yu, H.-L. Lagged Influence of Fine Particulate Matter and Geographic Disparities on Clinic Visits for Children’s Asthma in Taiwan. Int. J. Environ. Res. Public Health 2018, 15, 829. https://doi.org/10.3390/ijerph15040829
Chien L-C, Chen Y-A, Yu H-L. Lagged Influence of Fine Particulate Matter and Geographic Disparities on Clinic Visits for Children’s Asthma in Taiwan. International Journal of Environmental Research and Public Health. 2018; 15(4):829. https://doi.org/10.3390/ijerph15040829
Chicago/Turabian StyleChien, Lung-Chang, Yu-An Chen, and Hwa-Lung Yu. 2018. "Lagged Influence of Fine Particulate Matter and Geographic Disparities on Clinic Visits for Children’s Asthma in Taiwan" International Journal of Environmental Research and Public Health 15, no. 4: 829. https://doi.org/10.3390/ijerph15040829
APA StyleChien, L.-C., Chen, Y.-A., & Yu, H.-L. (2018). Lagged Influence of Fine Particulate Matter and Geographic Disparities on Clinic Visits for Children’s Asthma in Taiwan. International Journal of Environmental Research and Public Health, 15(4), 829. https://doi.org/10.3390/ijerph15040829






