Microbial Groundwater Quality Status of Hand-Dug Wells and Boreholes in the Dodowa Area of Ghana
Abstract
1. Introduction
2. Materials and Methods
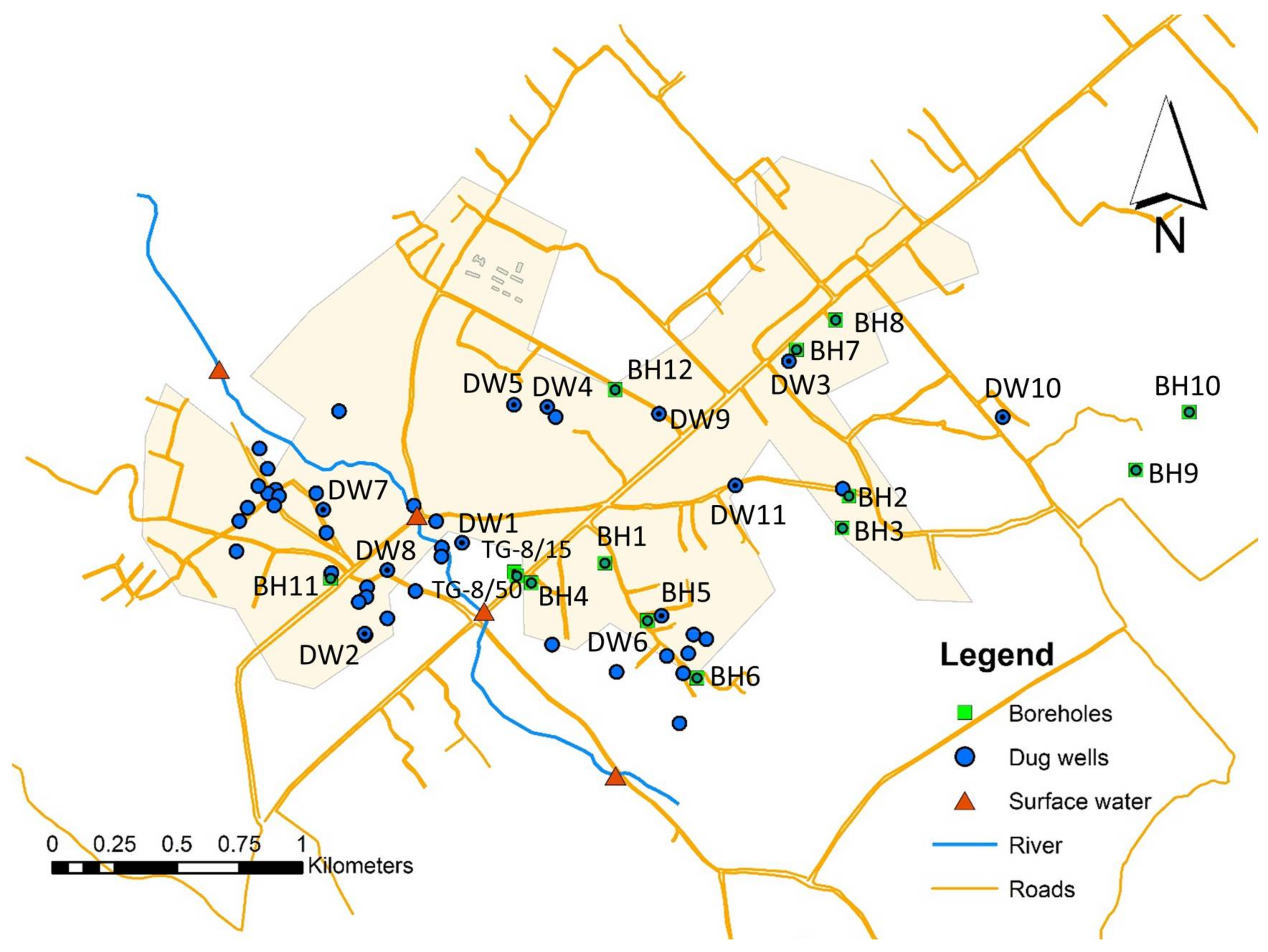
2.1. The Study Area
2.2. Sanitary Risk Inspection
2.3. Bacteriological Quality Assessment
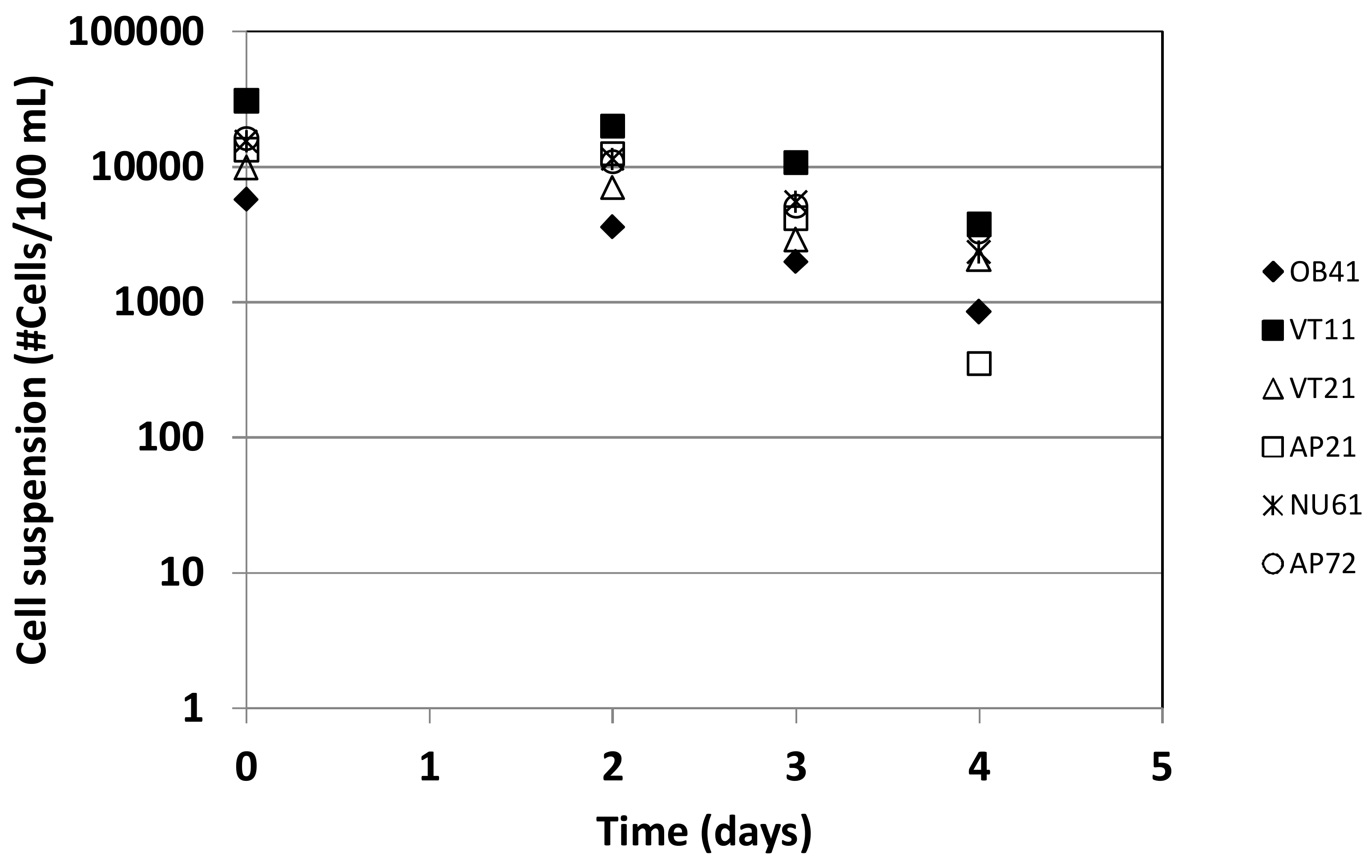
2.4. Bacteria Growth Experiments
2.5. Viruses Concentration
2.6. Nucleic Acids Isolation
2.7. Q-PCR and Nested PCR
3. Results
3.1. Direct Sanitary Risk Inspection and Bacteriological Quality Assessment
3.2. Bacteriological Quality of Hand-Dug Wells and Boreholes
3.3. Bacterial Survival Experiments
3.4. Virus Content in Groundwater
4. Discussion
4.1. Risk Assessment and Bacteriological Quality of Boreholes and Hand-Dug Wells
4.2. Presence of Viruses in Water Samples
5. Conclusions
- There is widespread fecal pollution of groundwater in the area; both hand-dug wells and boreholes are heavily loaded with fecal matter. This situation will not change unless the contaminating risk factors as we found in Table 1 are fixed and sanitary measures are taken.
- Hand-dug wells in the area seem at even greater risk to contamination compared to the boreholes; identified dominant risk factors to contamination are on-site sanitation facilities e.g., pit latrines and unlined pit latrines. However, even the boreholes are not sufficiently constructed or sited to prevent contamination.
- Systemic contamination by combined point sources is the main source of fecal contamination of groundwater sources.
- Based on the WHO [23] drinking water standards, groundwater from all points sampled is not suitable for drinking and needs to be treated before consumption.
Recommendations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dalu, T.; Barson, M.; Nhiwatiwa, T. Impact of intestinal microorganisms and protozoan parasites on drinking water quality in Harare, Zimbabwe. J. Water Sanit. Hyg. Dev. 2011, 1, 153. [Google Scholar] [CrossRef]
- Howard, G.; Pedley, S.; Barrett, M.; Nalubega, M.; Johal, K. Risk factors contributing to microbiological contamination of shallow groundwater in Kampala, Uganda. Water Res. 2003, 37, 3421–3429. [Google Scholar] [CrossRef]
- Parker, A.H.; Youlten, R.; Dillon, M.; Nussbaumer, T.; Carter, R.C.; Tyrrel, S.F.; Webster, J. An assessment of microbiological water quality of six water source categories in north-east Uganda. J. Water Health 2010, 8, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Lapworth, D.J.; Nkhuwa, D.C.W.; Okotto-Okotto, J.; Pedley, S.; Stuart, M.E.; Tijani, M.N.; Wright, J. Urban groundwater quality in sub-Saharan Africa: Current status and implications for water security and public health. Hydrogeol. J. 2017, 25, 1093–1116. [Google Scholar] [CrossRef]
- Roy, B.; Vilane, T.; Dlamini, T.L. An Assessment of Groundwater Pollution from On-Site Sanitation in Malkerns, Swaziland. J. Agric. Sci. Eng. 2016, 2, 11–17. [Google Scholar]
- Kanyerere, T.; Levy, J.; Xu, Y.; Saka, J. Assessment of microbial contamination of groundwater in upper Limphasa River catchment, located in a rural area of northern Malawi. Water SA 2012, 38, 581. [Google Scholar] [CrossRef]
- Lutterodt, G.; Foppen, J.W.A.; Uhlenbrook, S. Escherichia coli strains harvested from springs in Kampala, Uganda: Cell characterization and transport in saturated porous media. Hydrol. Process. 2014, 28, 1973–1988. [Google Scholar] [CrossRef]
- Moyo, N.A.G. An analysis of the chemical and microbiological quality of ground water from boreholes and shallow wells in Zimbabwe. Phys. Chem. Earth 2013, 66, 27–32. [Google Scholar] [CrossRef]
- Sorensen, J.P.R.; Lapworth, D.J.; Nkhuwa, D.C.W.; Stuart, M.E.; Gooddy, D.C.; Bell, R.A.; Chirwa, M.; Kabika, J.; Liemisa, M.; Chibesa, M.; et al. Emerging contaminants in urban groundwater sources in Africa. Water Res. 2015, 72, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Wanke, H.; Nakwafila, A.; Hamutoko, J.T.; Lohe, C.; Neumbo, F.; Petrus, I.; David, A.; Beukes, H.; Masule, N.; Quinger, M. Hand dug wells in Namibia: An underestimated water source or a threat to human health? Phys. Chem. Earth 2014, 76–78, 104–113. [Google Scholar] [CrossRef]
- Mara, D.; Lane, J.; Scott, B.; Trouba, D. Sanitation and health. PLoS Med. 2010, 7, e1000363. [Google Scholar] [CrossRef] [PubMed]
- Katukiza, A.Y.; Ronteltap, M.; Oleja, A.; Niwagaba, C.B.; Kansiime, F.; Lens, P.N.L. Selection of sustainable sanitation technologies for urban slums—A case of Bwaise III in Kampala, Uganda. Sci. Total Environ. 2010, 409, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Van Geen, A.; Ahmed, K.M.; Akita, Y.; Alam, M.J.; Culligan, P.J.; Emch, M.; Escamilla, V.; Feighery, J.; Ferguson, A.S.; Knappett, P.; et al. Fecal contamination of shallow tubewells in Bangladesh inversely related to arsenic. Environ. Sci. Technol. 2011, 45, 1199–1205. [Google Scholar] [CrossRef] [PubMed]
- Grönwall, J. Self-supply and accountability: To govern or not to govern groundwater for the (peri-) urban poor in Accra, Ghana. Environ. Earth Sci. 2016, 75, 1163. [Google Scholar] [CrossRef]
- Gibson, K.E.; Opryszko, M.C.; Schissler, J.T.; Guo, Y.; Schwab, K.J. Evaluation of human enteric viruses in surface water and drinking water resources in southern Ghana. Am. J. Trop. Med. Hyg. 2011, 84, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Kortatsi, B.K.; Jørgensen, N.O. The origin of high salinity waters in the accra plains groundwaters. In Proceedings of the First International Conference on Saltwater Intrusion and Coastal Aquifersó Monitoring, Modeling, and Management, Essaouira, Morocco, 23–25 April 2001. [Google Scholar]
- Buadee, J.; Yirenkyi, K.R. 2010 Population and Housing Census; Ghana Statistical Service: Accra, Ghana, 2014. [Google Scholar]
- Katukiza, A.Y.; Temanu, H.; Chung, J.W.; Foppen, J.W.A.; Lens, P.N.L. Genomic copy concentrations of selected waterborne viruses in a slum environment in Kampala, Uganda. J. Water Health 2013, 11, 358–370. [Google Scholar] [CrossRef] [PubMed]
- Boom, R.; Sol, C.; Beld, M.; Weel, J.; Goudsmit, J.; Dillen, P.W. Improved silica-guanidiniumthiocyanate DNA isolation procedure based on selective binding of bovine alpha-casein to silica particles. J. Clin. Microbiol. 1999, 37, 615–619. [Google Scholar] [PubMed]
- Heim, A.; Ebnet, C.; Harste, G.; Pring-Akerblom, P. RapN2 and quantitative detection of human adenovirus DNA by real-time PCR. J. Med. Virol. 2003, 70, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Allard, A.; Albinsson, B.; Wadell, G. Rapid typing of human adenoviruses by a general PCR combined with restriction endonuclease analysis. J. Clin. Microbiol. 2001, 39, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Freeman, M.M.; Kerin, T.; Hull, J.; McCaustland, K.; Gentsch, J. Enhancement of detection and quantification of rotavirus in stool using a modified real-time RT-PCR assay. J. Med. Virol. 2008, 80, 1489–1496. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for Drinking-Water Quality, 3rd ed.; World Health Organization: Geneva, Swertzerland, 2004. [Google Scholar]
- Ercumen, A.; Naser, A.M.; Arnold, B.F.; Unicomb, L.; Colford, J.M.; Luby, S.P. Can sanitary inspection surveys predict risk of microbiological contamination of groundwater sources? Evidence from shallow tubewells in rural Bangladesh. Am. J. Trop. Med. Hyg. 2017, 96, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Misati, A.G.; Ogendi, G.; Peletz, R.; Khush, R.; Kumpel, E. Can sanitary surveys replace water quality testing? Evidence from Kisii, Kenya. Int. J. Environ. Res. Public Health 2017, 14, 152. [Google Scholar] [CrossRef] [PubMed]
- Luby, S.P.; Gupta, S.K.; Sheikh, M.A.; Johnston, R.B.; Ram, P.K.; Islam, M.S. Tubewell water quality and predictors of contamination in three flood-prone areas in Bangladesh. J. Appl. Microbiol. 2008, 105, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Lutterodt, G. Transport of Multiple Escherichia Coli Strains in Saturated Porous Media; TU Delft: Delft, The Netherlands, 2012. [Google Scholar]
- Barker, J.A. A generalized radial flow model for hydraulic tests in fractured rock. Water Resour. Res. 1988, 24, 1796–1804. [Google Scholar] [CrossRef]
- Dershowitz, W.; Miller, I. Dual porosity fracture flow and transport. Geophys. Res. Lett. 1995, 22, 1441–1444. [Google Scholar] [CrossRef]
- Foppen, J.W.A.; Schijven, J.F. Evaluation of data from the literature on the transport and survival of Escherichia coli and thermotolerant coliforms in aquifers under saturated conditions. Water Res. 2006, 40, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Dzwairo, B.; Hoko, Z.; Love, D.; Guzha, E. Assessment of the impacts of pit latrines on groundwater quality in rural areas: A case study from Marondera district, Zimbabwe. Phys. Chem. Earth 2006, 31, 779–788. [Google Scholar] [CrossRef]
- Lutterodt, G.; Foppen, J.W.A.; Maksoud, A.; Uhlenbrook, S. Transport of Escherichia coli in 25 m quartz sand columns. J. Contam. Hydrol. 2011, 119, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Ravenscroft, P.; Mahmud, Z.H.; Islam, M.S.; Hossain, A.K.M.Z.; Zahid, A.; Saha, G.C.; Ali, A.H.M.Z.; Islam, K.; Cairncross, S.; Clemens, J.D.; et al. The public health significance of latrines discharging to groundwater used for drinking. Water Res. 2017, 124, 192–201. [Google Scholar] [CrossRef] [PubMed]
- Costa, D.C.; Madureira, A.P.; Amaral, L.C.; Sanchez, B.A.M.; Gomes, L.T.; Fontes, C.J.F.; Limongi, J.E.; de Brito, C.F.A.; Carvalho, L.H. Submicroscopic malaria parasite carriage: How reproducible are polymerase chain reaction-based methods? Memorias do Instituto Oswaldo Cruz 2014, 109, 21–28. [Google Scholar] [CrossRef] [PubMed]
| Risk Factors to Boreholes | No. of Boreholes | % of Total Number | |
|---|---|---|---|
| Boreholes | Unsanitary/worn-out seal of borehole pump | 6 | 50 |
| Nearest latrine or a pit latrine that percolates to soil, i.e., not sewered | 3 | 25 | |
| Uncapped well within 15–20 m of the borehole | 1 | 8 | |
| Other environmental source of pollution (e.g., animal excreta, rubbish, and surface water discharge) within 10 m radius | 6 | 50 | |
| Dug wells | Latrine or septic tank soak-away within 10 m of the well | 22 | 48 |
| Latrine/septic soak-away at higher ground than well, 10 to 30 m away. | 30 | 65 | |
| Other nearby sources of contamination such as wastewater drain, nearby rubbish dump, animal excreta, etc. within 10 m. | 34 | 74 | |
| Rope/bucket left at potentially contaminated point | 22 | 48 | |
| Height of apron wall less than 1 m and/or lack of top protection covering | 29 | 63 | |
| Depth and effectiveness of internal lining | 28 | 61 |
| Source Type | Source ID | Latitude | Longitude | Depth (m) | E. Coli (cfu/100 mL) | Adenovirus | Rotavirus | Contaminant Risk Level |
|---|---|---|---|---|---|---|---|---|
| Boreholes | TG-8/50 | 5.88014 | −0.099519 | 50 | TNTC | Low | ||
| TG-8/15 | 5.879999 | −0.099432 | 15 | TNTC | Low | |||
| BH1 | 5.88046 | −0.09626 | 10 | + | - | Intermediate | ||
| BH2 | 5.88288 | −0.08747 | 485 | ~ | - | High | ||
| BH3 | 5.88172 | −0.0877 | 425 | - | - | High | ||
| BH4 | 5.879241 | −0.098909 | 825 | ~ | ~ | Intermediate | ||
| BH5 | 5.87838 | −0.09472 | ~ | - | Intermediate | |||
| BH6 | 5.876314 | −0.09294 | 187 | ~ | I | Intermediate | ||
| BH7 | 5.88815 | −0.08936 | TNTC | - | - | Very high | ||
| BH8 | 5.88922 | −0.08795 | 525 | ~ | - | High | ||
| BH9 | 5.88381 | −0.07713 | TNTC | - | - | High | ||
| BH10 | 5.8859 | −0.07521 | 113 | I | - | High | ||
| BH11 | 5.87991 | −0.10614 | 30 | 135 | ~ | - | Very high | |
| BH12 | 5.88671 | −0.09589 | 88 | Intermediate | ||||
| Dug wells | DW1 | 5.8812 | −0.1014 | 3.2 | 300 | ~ | - | High |
| DW2 | 5.877917 | −0.10491 | 0.1 | 475 | ~ | - | High | |
| DW3 | 5.88774 | −0.08963 | 1.3 | 775 | ~ | - | High | |
| DW4 | 5.88609 | −0.09833 | 0.1 | 375 | ~ | ~ | High | |
| DW5 | 5.88617 | −0.09953 | 0.2 | 525 | I | + | High | |
| DW6 | 5.878567 | −0.094222 | 2.5 | TNTC | ~ | - | Very high | |
| DW7 | 5.88239 | −0.1064 | 1.4 | TNTC | + | ~ | Very high | |
| DW8 | 5.880212 | −0.104092 | 0.4 | 201 | ~ | ~ | High | |
| DW9 | 5.88584 | −0.09431 | 0.1 | 300 | I | - | Very high | |
| DW10 | 5.88572 | −0.08193 | 0.3 | 825 | ~ | - | High | |
| DW11 | 5.88326 | −0.09156 | 0.8 | 750 | I | - | Very high |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lutterodt, G.; Van de Vossenberg, J.; Hoiting, Y.; Kamara, A.K.; Oduro-Kwarteng, S.; Foppen, J.W.A. Microbial Groundwater Quality Status of Hand-Dug Wells and Boreholes in the Dodowa Area of Ghana. Int. J. Environ. Res. Public Health 2018, 15, 730. https://doi.org/10.3390/ijerph15040730
Lutterodt G, Van de Vossenberg J, Hoiting Y, Kamara AK, Oduro-Kwarteng S, Foppen JWA. Microbial Groundwater Quality Status of Hand-Dug Wells and Boreholes in the Dodowa Area of Ghana. International Journal of Environmental Research and Public Health. 2018; 15(4):730. https://doi.org/10.3390/ijerph15040730
Chicago/Turabian StyleLutterodt, George, Jack Van de Vossenberg, Yvonne Hoiting, Alimamy K. Kamara, Sampson Oduro-Kwarteng, and Jan Willem A. Foppen. 2018. "Microbial Groundwater Quality Status of Hand-Dug Wells and Boreholes in the Dodowa Area of Ghana" International Journal of Environmental Research and Public Health 15, no. 4: 730. https://doi.org/10.3390/ijerph15040730
APA StyleLutterodt, G., Van de Vossenberg, J., Hoiting, Y., Kamara, A. K., Oduro-Kwarteng, S., & Foppen, J. W. A. (2018). Microbial Groundwater Quality Status of Hand-Dug Wells and Boreholes in the Dodowa Area of Ghana. International Journal of Environmental Research and Public Health, 15(4), 730. https://doi.org/10.3390/ijerph15040730





