Use of Iodine-131 to Tellurium-132 Ratios for Assessing the Relationships between Human Inhaled Radioactivity and Environmental Monitoring after the Accident in Fukushima
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Ethical Considerations
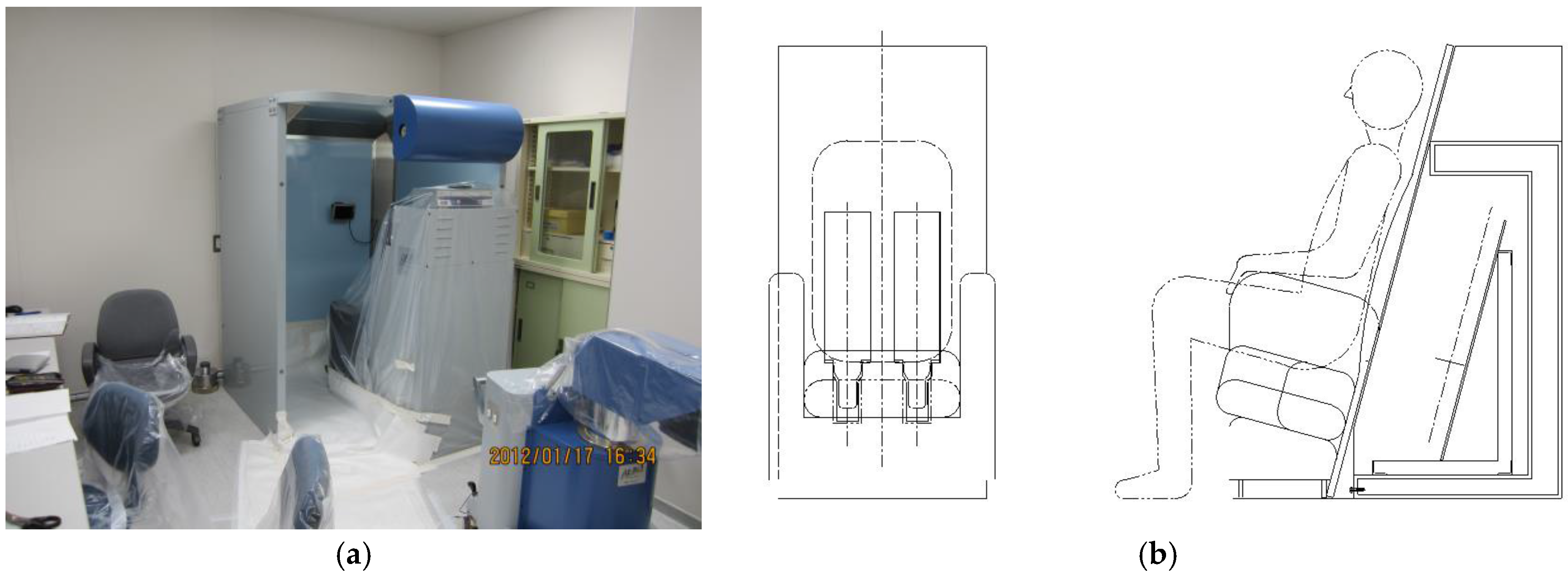
2.2. Whole Body Counter Measurements
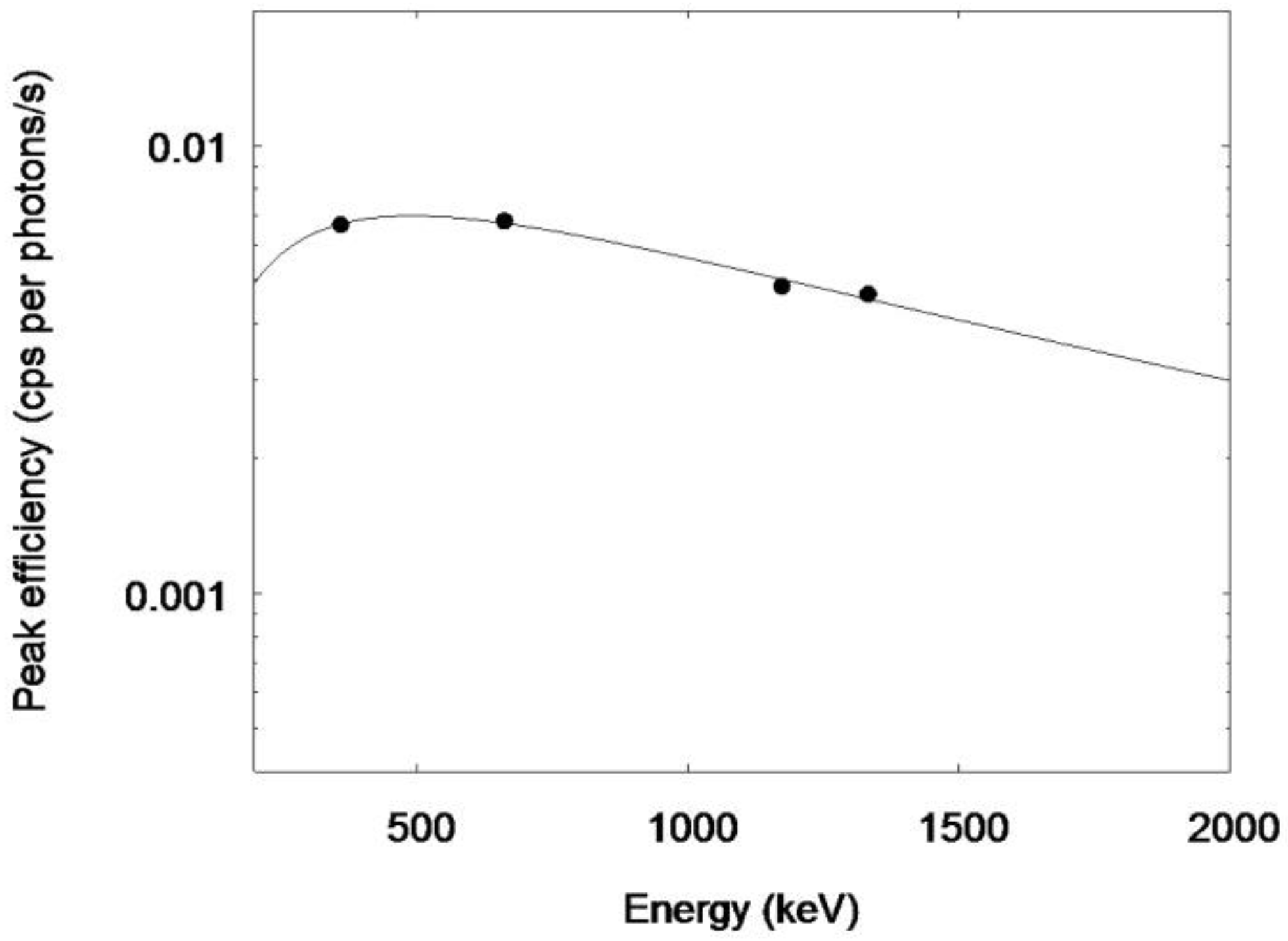
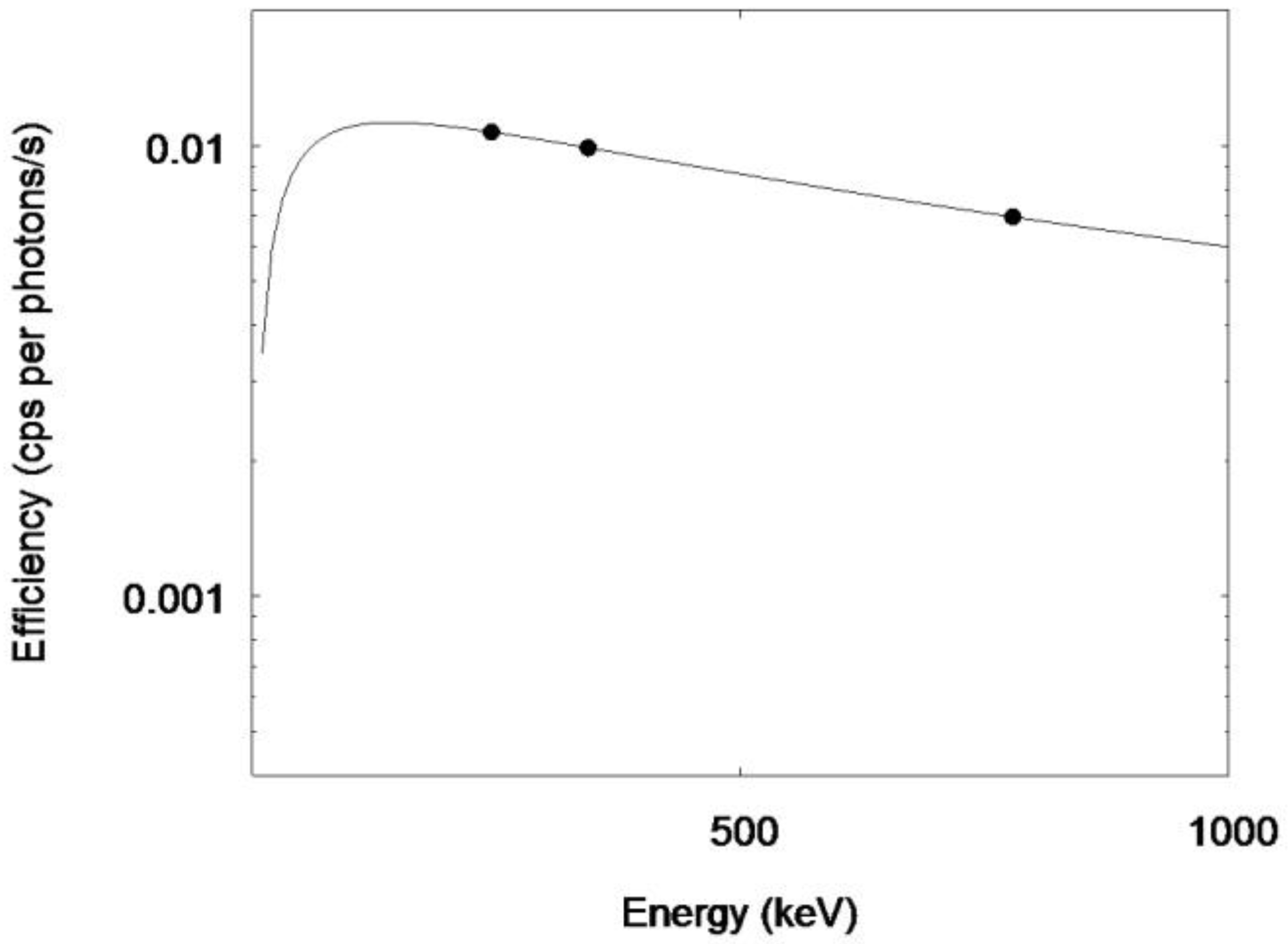
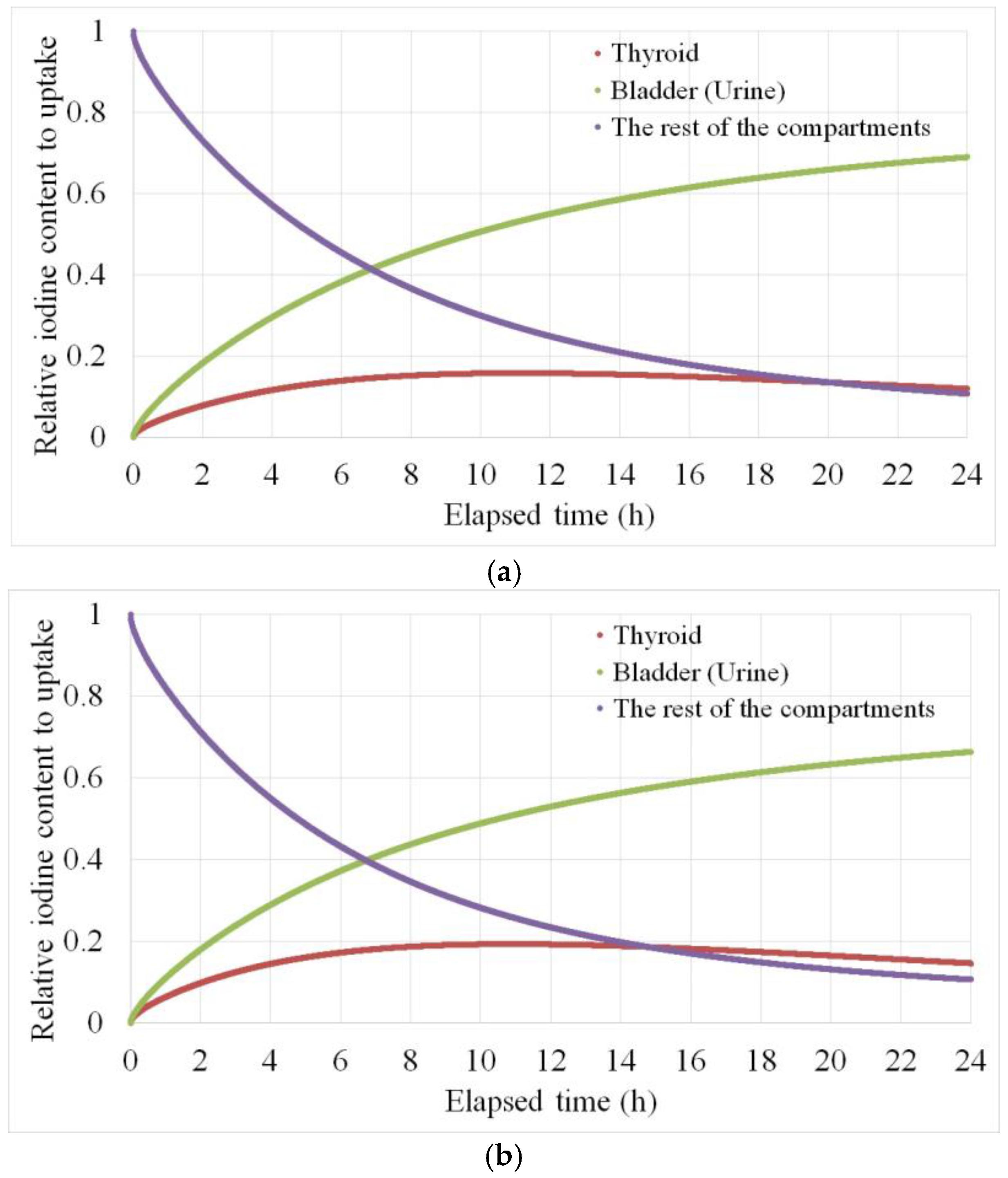
2.3. Biokinetic Calibration
2.4. Nutritional Survey
3. Results
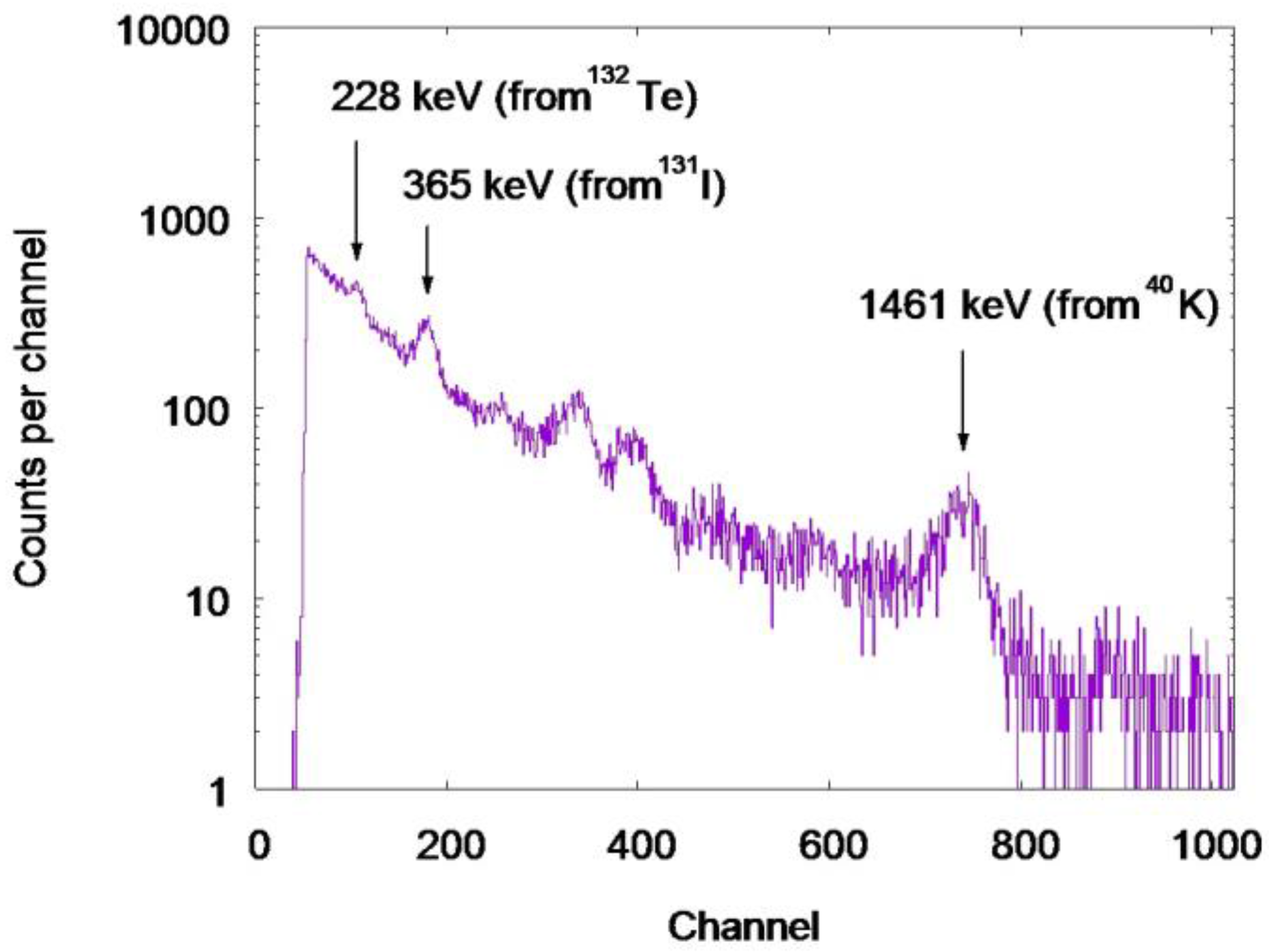
3.1. Whole Body Counter Measurements
3.2. Nutritional Survey
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hänscheid, H.; Reiners, C.; Goulko, G.; Luster, M.; Schneider-Ludorff, M.; Buck, A.K.; Lassmann, M. Facing the nuclear threat: Thyroid blocking revisited. J. Clin. Endocrinol. Metab. 2011, 96, 3511–3516. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Uchiyama, K.; Miyashita, M.; Sato, H.; Tanishima, Y.; Maeda, S.; Yoshikawa, J.; Kimura, S. A study of thyroid 131I activity of five human subjects exposed to a radioactive plume at Tamura City in Fukushima. Health Phys. 2015, 109, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, O.; Nakagawa, T.; Takeda, C.; Tani, K.; Kim, E.; Momose, T. Internal doses of three persons staying 110 km south of the Fukushima Daiichi nuclear power station during the arrival of radioactive plumes based on direct measurements. Radiat. Prot. Dosimetry 2016, 170, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Kurihara, O.; Tani, K.; Ohmachi, Y.; Fukutsu, K.; Sakai, K.; Akashi, M. Intake ratio of 131I to 137Cs derived from thyroid and whole-body doses to Fukushima residents. Radiat. Prot. Dosimetry 2016, 168, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Hosoda, M.; Tokonami, S.; Akiba, S.; Kurihara, O.; Sorimachi, A.; Ishikawa, T.; Momose, T.; Nakano, T.; Kashiwakura, I. Estimation of internal exposure of the thyroid to 131I on the basis 134Cs accumulated in the body among evacuees of the Fukushima Daiichi nuclear power station accident. Environ. Int. 2013, 61, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Morita, N.; Miura, M.; Yoshida, M.; Kumagai, A.; Ohtsuru, A.; Usa, T.; Kudo, T.; Takamura, N.; Yamashita, S.; Matsuda, N. Spatiotemporal characteristics of internal radiation exposure in evacuees and first responders after the radiological accident in Fukushima. Radiat. Res. 2013, 180, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, N.; Kumagai, A.; Ohtsuru, A.; Morita, N.; Miura, M.; Yoshida, M.; Kudo, T.; Takamura, N.; Yamashita, S. Assessment of internal exposure doses in Fukushima by a whole body counter within one month after the nuclear power plant accident. Radiat. Res. 2013, 179, 663–668. [Google Scholar] [CrossRef] [PubMed]
- Hargrove, C.K.; Geiger, K.W.; Chatterjee, A. The decay of 131I. Nucl. Phys. 1963, 40, 566–575. [Google Scholar] [CrossRef]
- Khazov, Y.; Mitropolsky, I.; Rodionov, A. Nuclear data sheets for A = 131. Nucl. Data Sheets 2006, 107, 2715–2930. [Google Scholar] [CrossRef]
- American National Standard Institute (ANSI). American National Standard—Specifications for the Bottle Manikin Absorption Phantom. ANSI/HPS N13.35-1999; Health Physics Society: McLean, VA, USA, 1999. [Google Scholar]
- Nakano, T.; Kim, E.; Tani, K.; Kurihara, O.; Sakai, K. A survey on the accuracy of whole-body counters operated in Fukushima after the nuclear disaster. Radiat. Prot. Dosimetry 2016, 170, 100–102. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Agency. Direct Methods for Measuring Radionuclides in the Human Body. Safety Series No. 114; International Atomic Energy Agency: Vienna, Austria, 1996; Available online: https://gnssn.iaea.org/Superseded%20Safety%20Standards/Safety_Series_114_1996.pdf (accessed on 15 December 2017).
- American National Standard Institute (ANSI). American National Standard Thyroid Radioiodine Uptake Measurements Using a Neck Phantom. ANSI N44.3-1973; American National Standards Institute: New York, NY, USA, 1973. [Google Scholar]
- Gnuplot Homepage. Available online: http://www.gnuplot.info/ (accessed on 15 December 2017).
- Bento, J.; Barros, S.; Teles, P.; Vaz, P.; Zankl, M. Efficiency correction factors of an ACCUSCAN whole-body counter due to the biodistribution of 134Cs, 137Cs and 60Co. Radiat. Prot. Dosimetry 2013, 155, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Leggett, R.W. A physiological systems model for iodine for use in radiation protection. Radiat. Res. 2010, 174, 496–516. [Google Scholar] [CrossRef] [PubMed]
- Giussani, A. A recycling model of the biokinetics of systemic tellurium. Int. J. Radiat. Biol. 2014, 90, 1114–1118. [Google Scholar] [CrossRef] [PubMed]
- Kopp, P. Thyroid hormone synthesis. In Werner & Ingbar’s The Thyroid: A Fundamental and Clinical Text, 10th ed.; Braverman, L.E., Cooper, D.S., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2012; pp. 48–74. ISBN 978-1-4511-2063-9. [Google Scholar]
- Nagataki, S.; Shizume, K.; Nakao, K. Thyroid function in chronic excess iodide ingestion: Comparison of thyroidal absolute iodine uptake and degradation of thyroxine in euthyroid Japanese subjects. J. Clin. Endocrinol. Metab. 1967, 27, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, Y.; Kusama, T. Weight, iodine content and iodine uptake of the thyroid gland of normal Japanese. Hoken-Butsuri 1976, 11, 123–128. (In Japanese) [Google Scholar] [CrossRef]
- Endo, S.; Kimura, S.; Takatsuji, T.; Nanasawa, K.; Imanaka, T.; Shizuma, K. Measurement of soil contamination by radionuclides due to the Fukushima Dai-ichi nuclear power plant accident and associated estimated cumulative external dose estimation. J. Environ. Radioact. 2012, 111, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, H.; Saito, K.; Ishioka, J.; Uwamino, Y. Diffusion of radioactive materials from Fukushima Daiichi nuclear power station obtained by gamma-ray measurements on expressways. Trans. Energy Soc. Jpn. 2011, 10, 152–162. [Google Scholar] [CrossRef]
- Gerhardsson, L. Tellurium. In Handbook on the Toxicology of Metals Volume II: Specific Metals, 4th ed.; Nordberg, G.F., Fowler, B.A., Nordberg, M., Eds.; Academic Press in an imprint of Elsevier: London, UK, 2015; pp. 1217–1228. ISBN 978-0-12-398293-3. [Google Scholar]
- United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR). Sources, Effects and Risks of Ionizing Radiation. UNSCEAR 2013 Report; United Nations Scientific Committee on the Effects of Atomic Radiation: New York, NY, USA, 2014; Available online: http://www.unscear.org/docs/reports/2013/13-85418_Report_2013_Annex_A.pdf (accessed on 15 December 2017).
- International Commission on Radiological Protection. Age-Dependent Doses to Members of the Public from Intake of Radionuclides: Part 4—Inhalation Dose Coefficients. ICRP Publication 71, Ann ICRP; Elsevier: Oxford, UK, 1995; ISBN 0-08-042736-7. [Google Scholar]
- Past Weather Data Search. Available online: http://www.webcitation.org/6vjF1xLmj (accessed on 15 December 2017). (In Japanese).
- Past Weather Data Search. Available online: http://www.webcitation.org/6vjFJTH6p (accessed on 15 December 2017). (In Japanese).
- Furuta, S.; Sumiya, S.; Watanabe, H.; Nakano, M.; Imaizumi, K.; Takeyasu, M.; Nakada, A.; Fujita, H.; Mizutani, T.; Morisawa, M.; et al. Results of the Environmental Radiation MONITORING Following the Accident at the Fukushima Daiichi Nuclear Power Plant -Interim Report (Ambient Radiation Dose Rate, Radioactivity Concentration in the Air and Radioactivity Concentration in the Fallout). JAEA-Review 2011-035; Japan Atomic Energy Agency: Ibaraki-ken, Japan, 2011. Available online: http://jolissrch-inter.tokai-sc.jaea.go.jp/pdfdata/JAEA-Review-2011-035.pdf (accessed on 15 December 2017).
- International Commission on Radiological Protection. Human Respiratory Tract Model for Radiological Protection. ICRP Publication 66, Ann ICRP; Elsevier: Oxford, UK, 1994; ISBN 0-08-041154-1. [Google Scholar]
- Koike, T.; Suzuki, Y.; Genyu, S.; Kobayashi, I.; Komori, H.; Otsu, H.; Sakuma, H.; Sarausad, E.M.; Shimada, K.; Shinozuka, T.; et al. Comprehensive data on ionizing radiation from Fukushima Daiichi nuclear power plant in the town of Miharu, Fukushima prefecture: The Misho project. J. Radiol. Prot. 2014, 34, 675–698. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.; Karmisholt, J.; Laurberg, P. Variations in iodine excretion in healthy individuals. In Comprehensive Handbook of Iodine: Nutritional, Biochemical, Pathological and Therapeutic Aspects; Preedy, V.R., Burrow, G.N., Watson, R.R., Eds.; Elsevier Academic Press: Burlington, MA, USA, 2009; pp. 421–428. ISBN 978-0-12-374135-6. [Google Scholar]
- Watanabe, H.; Azuma, Y. Change of bladder capacity during sleep and “intravesical urine vanishment (IVUV)” phenomenon. Yanyosho-Kenkyu 2014, 19, 25–29. (In Japanese) [Google Scholar]
- Leggett, R. An age-specific biokinetic model for iodine. J. Radiol. Prot. 2017, 37, 864–882. [Google Scholar] [CrossRef] [PubMed]


| Age (Year) | 131I Residual Activity in the Thyroid (Bq) |
|---|---|
| 53 | 268 |
| 49 | 249 |
| 35 | 676 |
| 34 | 569 |
| 27 | 1082 |
| Age (Year) | Calibration Model | Total Body Burden (Bq) | Estimated Total Uptake (Bq) | 24-h Thyroid Uptake (%) |
|---|---|---|---|---|
| 53 | Homogenous | 3927 ± 135 | 4243 ± 146 | - |
| Thyroid and others | 3716 ± 128 | 4015 ± 138 | <7.2 ± 1.1 | |
| 49 | Homogenous | 2321 ± 165 | 2508 ± 178 | - |
| Thyroid and others | 2320 ± 165 | 2507 ± 178 | <10.7 ± 3.8 | |
| 35 | Homogenous | 3918 ± 127 | 4233 ± 137 | - |
| Thyroid and others | 4050 ± 131 | 4376 ± 142 | <16.7 ± 2.7 | |
| 34 | Homogenous | 1284 ± 182 | 1387 ± 197 | - |
| Thyroid and others | 1140 ± 162 | 1232 ± 175 | <49.9 ± 11.0 | |
| 27 | Homogenous | 2623 ± 195 | 2834 ± 211 | - |
| Thyroid and others | 2454 ± 182 | 2652 ± 197 | <44.1 ± 5.8 |
| Age (Year) | Blood 1 to Thyroid 1 (λ1) (day−1) | Thyroid 2 to Blood 1 (λ5) (day−1) | Urinary Bladder Contents to Urine (day−1) | Total Body Burden (Bq) | Estimated Total Uptake (Bq) | 24-h Thyroid Uptake (%) |
|---|---|---|---|---|---|---|
| 53 | 7.26 | 1.50 | 0 | 1912 ± 254 | 2065 ± 274 | 13.1 ± 1.7 |
| 49 | 6.65 | 1.50 | 0 | 1913 ± 159 | 2066 ± 172 | 12.1 ± 1.0 |
| 35 | 9.20 | 1.50 | 0 | 3955 ± 131 | 4272 ± 141 | 15.9 ± 0.5 |
| 34 | 9.25 | 1.50 | 12 | 1089 ± 155 | 3589 ± 511 | 16.0 ± 2.3 |
| 27 | 9.25 | 1.50 | 6.2 | 2277 ± 175 | 6829 ± 525 | 16.0 ± 1.2 |
| Age (Year) | Calibration Model | Total Body Burden (Bq) | Estimated Total Uptake (Bq) |
|---|---|---|---|
| 53 | Homogenous | 1856 ± 166 | 2253 ± 202 |
| Biokinetic | 1249 ± 112 | 1516 ± 136 | |
| 49 | Homogenous | 1331 ± 202 | 1616 ± 245 |
| Biokinetic | 1223 ± 185 | 1485 ± 225 | |
| 35 | Homogenous | 3697 ± 155 | 4488 ± 188 |
| Biokinetic | 3744 ± 157 | 4545 ± 191 | |
| 34 | Homogenous | 886 ± 233 | 1075 ± 283 |
| Biokinetic | 868 ± 229 | 1743 ± 460 | |
| 27 | Homogenous | 1627 ± 237 | 1975 ± 288 |
| Biokinetic | 1576 ± 230 | 3025 ± 441 |
| Age (Year) | Ratio of 131I to 132Te | |
|---|---|---|
| Biokinetic Calibrations (131I and 132Te) | Biokinetic (131I) and Homogenous (132Te) Calibrations | |
| 53 | 1.38 ± 0.22 | 0.93 ± 0.15 |
| 49 | 1.41 ± 0.24 | 1.30 ± 0.22 |
| 35 | 0.96 ± 0.05 | 0.97 ± 0.05 |
| 34 | 2.09 ± 0.63 | 3.39 ± 1.01 |
| 27 | 2.29 ± 0.38 | 3.51 ± 0.58 |
| Time and Date | Menu |
|---|---|
| Between 15:30 on 13 March and 2:00 on 14 March | Bread, rice ball, and “CalorieMate” |
| 2:30 on 14 March | Stable iodine pills, 100 mg (4 young members) |
| 7:15 on 14 March | Rice, miso soup (with Welsh onion), scrambled egg (with ketchup), sliced pork and salad (bean sprouts and lettuce) |
| Around 13:00 on 14 March | Bread and rice ball |
| 21:15 on 14 March | Curry and rice, and instant noodles |
| 7:00 on 15 March | Rice, miso soup (with sea weed and tofu), Japanese omelette, and Japanese pickles (napa cabbage) |
| 12:30 on 15 March | Cup noodle (Akai kitsune udon), white bread (with mayonnaise), rice ball and Kashi Pan (bread with sweet filling) |
| 19:20 on 15 March | Tempura udon, and rice ball |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uchiyama, K.; Miyashita, M.; Tanishima, Y.; Maeda, S.; Sato, H.; Yoshikawa, J.; Watanabe, S.; Shibata, M.; Ohhira, S.; Kobashi, G. Use of Iodine-131 to Tellurium-132 Ratios for Assessing the Relationships between Human Inhaled Radioactivity and Environmental Monitoring after the Accident in Fukushima. Int. J. Environ. Res. Public Health 2018, 15, 483. https://doi.org/10.3390/ijerph15030483
Uchiyama K, Miyashita M, Tanishima Y, Maeda S, Sato H, Yoshikawa J, Watanabe S, Shibata M, Ohhira S, Kobashi G. Use of Iodine-131 to Tellurium-132 Ratios for Assessing the Relationships between Human Inhaled Radioactivity and Environmental Monitoring after the Accident in Fukushima. International Journal of Environmental Research and Public Health. 2018; 15(3):483. https://doi.org/10.3390/ijerph15030483
Chicago/Turabian StyleUchiyama, Koji, Masami Miyashita, Yoshinobu Tanishima, Shigenobu Maeda, Hitoshi Sato, Jun Yoshikawa, Shuji Watanabe, Masamichi Shibata, Shuji Ohhira, and Gen Kobashi. 2018. "Use of Iodine-131 to Tellurium-132 Ratios for Assessing the Relationships between Human Inhaled Radioactivity and Environmental Monitoring after the Accident in Fukushima" International Journal of Environmental Research and Public Health 15, no. 3: 483. https://doi.org/10.3390/ijerph15030483
APA StyleUchiyama, K., Miyashita, M., Tanishima, Y., Maeda, S., Sato, H., Yoshikawa, J., Watanabe, S., Shibata, M., Ohhira, S., & Kobashi, G. (2018). Use of Iodine-131 to Tellurium-132 Ratios for Assessing the Relationships between Human Inhaled Radioactivity and Environmental Monitoring after the Accident in Fukushima. International Journal of Environmental Research and Public Health, 15(3), 483. https://doi.org/10.3390/ijerph15030483





