Association Between PM2.5 and Daily Hospital Admissions for Heart Failure: A Time-Series Analysis in Beijing
Abstract
1. Introduction
2. Materials and Methods
2.1. Data on Hospitalization for CHF
2.2. Environmental Data
2.3. Statistical Analysis
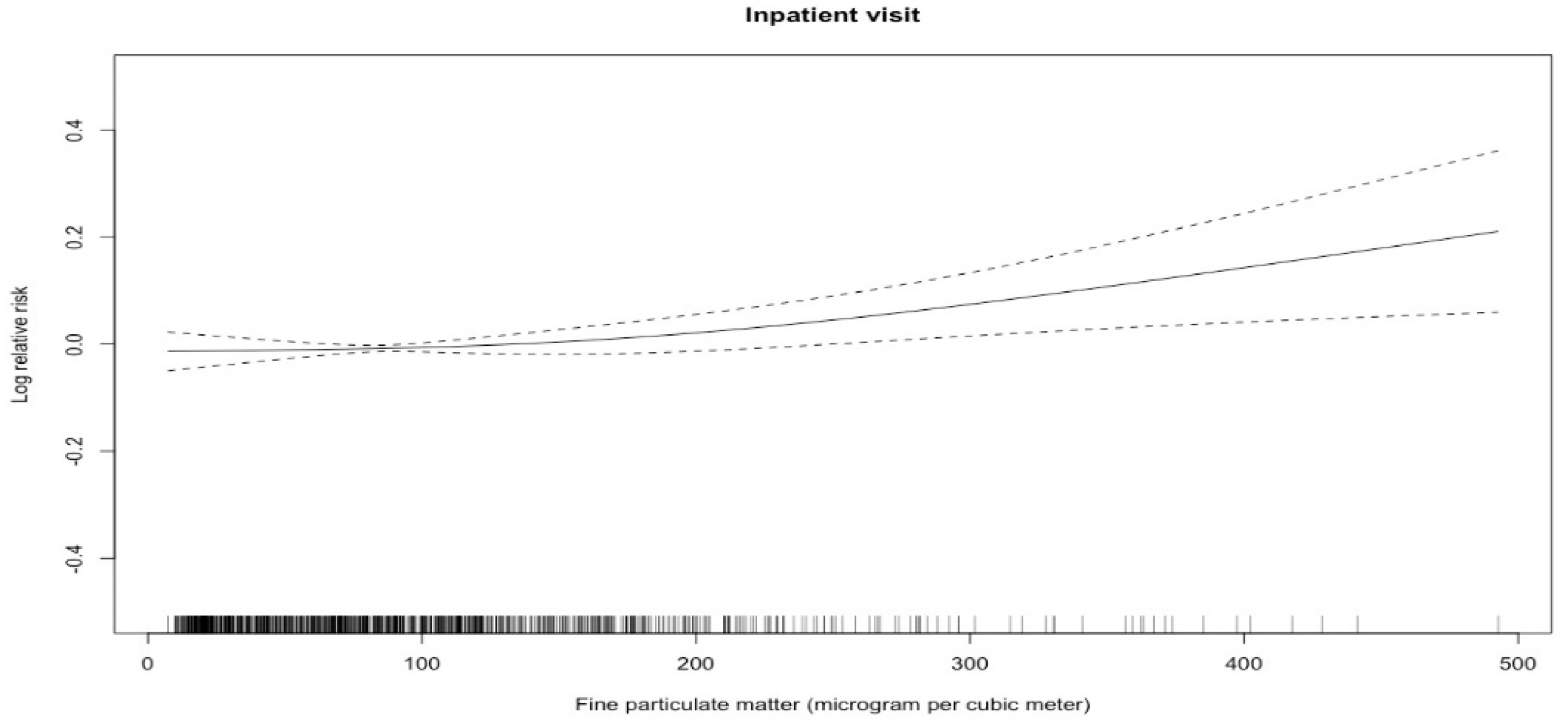
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Statement
References
- Mahmood, S.S.; Wang, T.J. The epidemiology of congestive heart failure: The Framingham Heart Study perspective. Glob. Heart 2013, 8, 77–82. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Allen, C.; Arora, M.; Barber, R.M.; Bhutta, Z.A.; Brown, A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Huynh, Q.L.; Blizzard, C.L.; Marwick, T.H.; Negishi, K. Association of ambient particulate matter with heart failure incidence and all-cause readmissions in Tasmania: An observational study. BMJ Open 2018, 8, e021798. [Google Scholar] [CrossRef] [PubMed]
- Vanasse, A.; Talbot, D.; Chebana, F.; Belanger, D.; Blais, C.; Gamache, P.; Giroux, J.X.; Dault, R.; Gosselin, P. Effects of climate and fine particulate matter on hospitalizations and deaths for heart failure in elderly: A population-based cohort study. Environ. Int. 2017, 106, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.; Langrish, J.P.; Nair, H.; McAllister, D.A.; Hunter, A.L.; Donaldson, K.; Newby, D.E.; Mills, N.L. Global association of air pollution and heart failure: A systematic review and meta-analysis. Lancet 2013, 382, 1039–1048. [Google Scholar] [CrossRef]
- Sacks, J.D.; Stanek, L.W.; Luben, T.J.; Johns, D.O.; Buckley, B.J.; Brown, J.S.; Ross, M. Particulate matter–induced health effects: Who is susceptible? Environ. Health Perspect. 2011, 119, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Peel, J.L.; Metzger, K.B.; Klein, M.; Flanders, W.D.; Mulholland, J.A.; Tolbert, P.E. Ambient air pollution and cardiovascular emergency department visits in potentially sensitive groups. Am. J. Epidemiol. 2007, 165, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, R.W.; Mills, I.C.; Walton, H.A.; Anderson, H.R. Fine particle components and health-a systematic review and meta-analysis of epidemiological time series studies of daily mortality and hospital admissions. J. Expo. Sci. Environ. Epidemiol. 2015, 25, 208–214. [Google Scholar] [CrossRef] [PubMed]
- Briggs, D. Environmental pollution and the global burden of disease. Br. Med. Bull. 2003, 68, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.L.; Tsai, S.S.; Yang, C.Y. Fine particulate air pollution and hospital admissions for congestive heart failure: A case-crossover study in Taipei. Inhal. Toxicol. 2013, 25, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Tian, Y.; Song, J.; Cao, Y.; Xiang, X.; Huang, C.; Li, M.; Hu, Y. Effect of Ambient Air Pollution on Hospitalization for Heart Failure in 26 of China’s Largest Cities. Am. J. Cardiol. 2018, 121, 628–633. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Li, G.; Zhao, D.; Xie, X.; Wei, Z.; Wang, W.; Wang, M.; Li, G.; Liu, W.; Sun, J.; et al. Relationship between fine particulate air pollution and ischaemic heart disease morbidity and mortality. Heart 2015, 101, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.H.; Xiang, X.; Juan, J.; Song, J.; Cao, Y.Y.; Huang, C.; Li, M.; Hu, Y.H. Short-term effects of ambient fine particulate matter pollution on hospital visits for chronic obstructive pulmonary disease in Beijing, China. Environ. Health. 2018, 17, 21. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.F.; Hu, M.G.; Xu, C.D.; Christakos, G.; Zhao, Y. Estimation of citywide air pollution in Beijing. PLoS ONE 2013, 8, e53400. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Xiang, X.; Wu, Y.; Cao, Y.; Song, J.; Sun, K.; Liu, H.; Hu, Y. Fine Particulate Air Pollution and First Hospital Admissions for Ischemic Stroke in Beijing, China. Sci. Rep. 2017, 7, 3897. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Xiang, X.; Juan, J.; Sun, K.; Song, J.; Cao, Y.; Hu, Y. Fine particulate air pollution and hospital visits for asthma in Beijing, China. Environ. Pollut. 2017, 230, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Yin, P.; Meng, X.; Liu, C.; Wang, L.; Xu, X.; Ross, J.A.; Tse, L.A.; Zhao, Z.; Kan, H.; et al. Fine Particulate Air Pollution and Daily Mortality. A Nationwide Analysis in 272 Chinese Cities. Am. J. Respir. Crit. Care Med. 2017, 196, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yin, P.; Chen, R.; Meng, X.; Wang, L.; Niu, Y.; Lin, Z.; Liu, Y.; Liu, J.; Qi, J.; et al. Ambient carbon monoxide and cardiovascular mortality: A nationwide time-series analysis in 272 cities in China. Lancet Planet. Health 2018, 2, e12–e18. [Google Scholar] [CrossRef]
- Yin, P.; Chen, R.; Wang, L.; Meng, X.; Liu, C.; Niu, Y.; Lin, Z.; Liu, Y.; Liu, J.; Qi, J.; et al. Ambient Ozone Pollution and Daily Mortality: A Nationwide Study in 272 Chinese Cities. Environ. Health Perspect. 2017, 125, 117006. [Google Scholar] [CrossRef] [PubMed]
- Wellenius, G.A.; Schwartz, J.; Mittleman, M.A. Particulate air pollution and hospital admissions for congestive heart failure in seven United States cities. Am. J. Cardiol. 2006, 97, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Mann, J.K.; Tager, I.B.; Lurmann, F.; Segal, M.; Quesenberry, C.P., Jr.; Lugg, M.M.; Shan, J.; Van Den Eeden, S.K. Air pollution and hospital admissions for ischemic heart disease in persons with congestive heart failure or arrhythmia. Environ. Health Perspect. 2002, 110, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Belleudi, V.; Faustini, A.; Stafoggia, M.; Cattani, G.; Marconi, A.; Perucci, C.A.; Forastiere, F. Impact of fine and ultrafine particles on emergency hospital admissions for cardiac and respiratory diseases. Epidemiology 2010, 21, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Pope, C.A., 3rd; Renlund, D.G.; Kfoury, A.G.; May, H.T.; Horne, B.D. Relation of heart failure hospitalization to exposure to fine particulate air pollution. Am. J. Cardiol. 2008, 102, 1230–1234. [Google Scholar] [CrossRef] [PubMed]
- Dominici, F.; Peng, R.D.; Bell, M.L.; Pham, L.; McDermott, A.; Zeger, S.L.; Samet, J.M. Fine particulate air pollution and hospital admission for cardiovascular and respiratory diseases. JAMA 2006, 295, 1127–1134. [Google Scholar] [CrossRef] [PubMed]
- Symons, J.M.; Wang, L.; Guallar, E.; Howell, E.; Dominici, F.; Schwab, M.; Ange, B.A.; Samet, J.; Ondov, J.; Harrison, D.; et al. A case-crossover study of fine particulate matter air pollution and onset of congestive heart failure symptom exacerbation leading to hospitalization. Am. J. Epidemiol. 2006, 164, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Chen, A.; Chen, R.; Qi, Y.; Ye, J.; Li, S.; Li, W.; Liang, Z.; Liang, Q.; Guo, D.; et al. Acute effect of ambient air pollution on heart failure in Guangzhou, China. Int. J. Cardiol. 2014, 177, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Xu, H.; Xu, Q.; Chen, B.; Kan, H. Fine particulate matter constituents and cardiopulmonary mortality in a heavily polluted Chinese city. Environ. Health Perspect. 2012, 120, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Cao, J.J.; Tao, Y.B.; Dai, L.Z.; Lu, S.E.; Hou, B.; Wang, Z.; Zhu, T. Seasonal Variation of Chemical Species Associated With Short-Term Mortality Effects of PM2.5 in Xi’an, a central city in China. Am. J. Epidemiol. 2012, 175, 556–566. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Deng, F.; Wei, H.; Huang, J.; Wang, X.; Hao, Y.; Zheng, C.; Qin, Y.; Lv, H.; Shima, M.; et al. Association of cardiopulmonary health effects with source-appointed ambient fine particulate in Beijing, China: A combined analysis from the Healthy Volunteer Natural Relocation (HVNR) study. Environ. Sci. Technol. 2014, 48, 3438–3448. [Google Scholar] [CrossRef] [PubMed]
- Goldman, G.T.; Mulholland, J.A.; Russell, A.G.; Strickland, M.J.; Klein, M.; Waller, L.A.; Tolbert, P.E. Impact of exposure measurement error in air pollution epidemiology: Effect of error type in time-series studies. Environ. Health 2011, 10, 61. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Hospital Admissions (n = 15,256) |
|---|---|
| Age (year) | |
| <65 (%) | 3210 (21.0%) |
| ≥65 (%) | 12,046 (79.0%) |
| Gender | |
| Male (%) | 8523 (55.9%) |
| Female (%) | 6733 (44.1%) |
| Variables | Mean ± SD | Minimum | Percentile | Maximum | IQR | ||
|---|---|---|---|---|---|---|---|
| 25th | 50th | 75th | |||||
| Daily hospital admissions | 16.7 ± 12.1 | 0 | 3 | 18.5 | 25 | 59 | 22 |
| PM2.5 (μg/m3) | 99.5 ± 75.3 | 7.2 | 42.5 | 82.8 | 133.3 | 492.8 | 90.8 |
| Temperature (°C) | 12.6 ± 11.6 | −12.5 | 1.5 | 14.1 | 23.8 | 34.5 | 22.3 |
| Relative humidity (%) | 48.6 ± 20.3 | 9 | 30 | 48 | 66 | 92 | 36 |
| Lag Days | Percentage Change | 95% CI | p |
|---|---|---|---|
| Lag 0 days | 0.35 | 0.06–0.64 | 0.0191 |
| Lag 1 days | 0.42 | 0.17–0.67 | 0.000899 |
| Lag 2 days | 0.31 | 0.10–0.53 | 0.00426 |
| Lag 3 days | 0.16 | −0.06–0.38 | 0.148 |
| Lag 0–1 day | 0.59 | 0.26–0.91 | 0.000448 |
| Lag 0–2 days | 0.65 | 0.32–0.99 | 0.000115 |
| Lag 0–3 days | 0.61 | 0.27–0.95 | 0.000436 |
| Variables | Percentage Change | 95% CI | p |
|---|---|---|---|
| Gender | |||
| Male | 0.31 | −0.05–0.67 | 0.09 |
| Female | 0.43 | −0.005–0.87 | 0.0527 |
| Age (year) | |||
| <65 | 0.28 | −0.27–0.82 | 0.318 |
| ≥65 | 0.32 | −0.005–0.65 | 0.0536 |
| Variables | df a | Percentage Change | 95% CI | p |
|---|---|---|---|---|
| Calendar time | 4 | 0.33 | 0.07–0.60 | 0.0142 |
| 5 | 0.35 | 0.07–0.62 | 0.0148 | |
| 6 | 0.33 | 0.06–0.61 | 0.0171 | |
| 7 b | 0.35 | 0.06–0.64 | 0.0191 | |
| 8 | 0.40 | 0.10–0.70 | 0.00804 | |
| Temperature | 2 | 0.35 | 0.06–0.64 | 0.0191 |
| 3 b | 0.35 | 0.06–0.64 | 0.0191 | |
| 4 | 0.47 | 0.20–0.74 | 0.000609 | |
| 5 | 0.46 | 0.19–0.73 | 0.000874 | |
| 6 | 0.45 | 0.18–0.72 | 0.00101 | |
| Relative humidity | 2 | 0.35 | 0.06–0.64 | 0.0191 |
| 3 b | 0.35 | 0.06–0.64 | 0.0191 | |
| 4 | 0.33 | 0.04–0.63 | 0.0238 | |
| 5 | 0.33 | 0.04–0.63 | 0.0239 | |
| 6 | 0.34 | 0.05–0.63 | 0.0211 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Wu, Y.; Tian, Y.-H.; Cao, Y.-Y.; Song, J.; Huang, Z.; Wang, X.-W.; Hu, Y.-H. Association Between PM2.5 and Daily Hospital Admissions for Heart Failure: A Time-Series Analysis in Beijing. Int. J. Environ. Res. Public Health 2018, 15, 2217. https://doi.org/10.3390/ijerph15102217
Li M, Wu Y, Tian Y-H, Cao Y-Y, Song J, Huang Z, Wang X-W, Hu Y-H. Association Between PM2.5 and Daily Hospital Admissions for Heart Failure: A Time-Series Analysis in Beijing. International Journal of Environmental Research and Public Health. 2018; 15(10):2217. https://doi.org/10.3390/ijerph15102217
Chicago/Turabian StyleLi, Man, Yao Wu, Yao-Hua Tian, Ya-Ying Cao, Jing Song, Zhe Huang, Xiao-Wen Wang, and Yong-Hua Hu. 2018. "Association Between PM2.5 and Daily Hospital Admissions for Heart Failure: A Time-Series Analysis in Beijing" International Journal of Environmental Research and Public Health 15, no. 10: 2217. https://doi.org/10.3390/ijerph15102217
APA StyleLi, M., Wu, Y., Tian, Y.-H., Cao, Y.-Y., Song, J., Huang, Z., Wang, X.-W., & Hu, Y.-H. (2018). Association Between PM2.5 and Daily Hospital Admissions for Heart Failure: A Time-Series Analysis in Beijing. International Journal of Environmental Research and Public Health, 15(10), 2217. https://doi.org/10.3390/ijerph15102217




