Interventions to Improve Vaccination Uptake and Cost Effectiveness of Vaccination Strategies in Newly Arrived Migrants in the EU/EEA: A Systematic Review
Abstract
1. Introduction
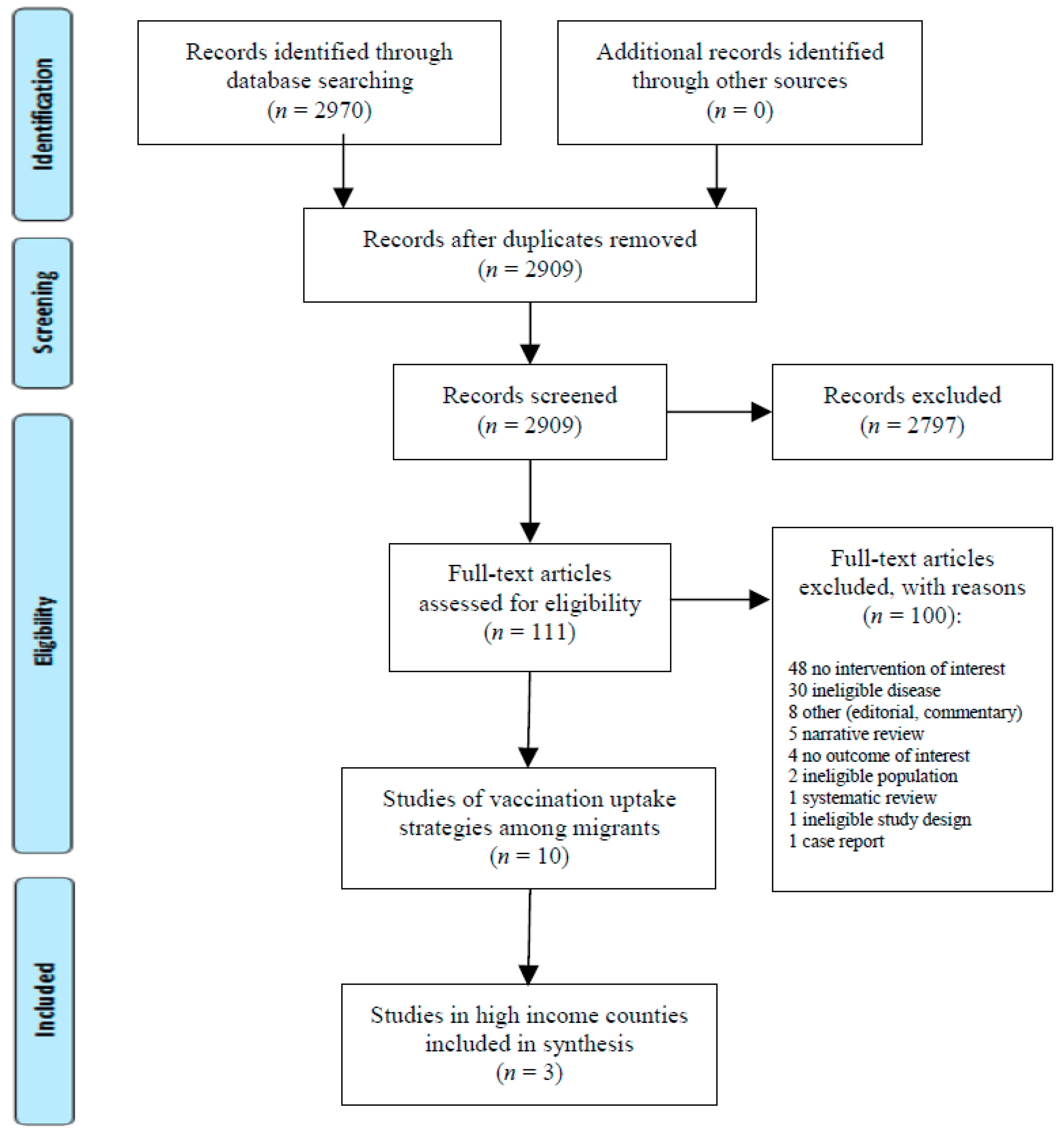
2. Methods
3. Results
3.1. What Interventions Increase Uptake of Vaccinations in Migrants?
3.2. What are Cost-Effective Approaches to Vaccinating Newly Arrived Migrants?
4. Discussion
5. Strengths and Limitations
6. Implementation Considerations and Evidence Gaps
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Migration Report 2017: Highlights; United Nations, Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2017.
- Cross-National Comparisons of Internal Migration: An Update on Global Patterns and Trends; United Nations, Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2013.
- The EU and the Crisis in Syria, Factsheet—EEAS—European External Action Service—European Commission. Available online: https://eeas.europa.eu/headquarters/headquarters-homepage_en/22664/ (accessed on 17 September 2018).
- World Migration Report 2018; International Organization for Migration: Geneva, Switzerland, 2017.
- European Vaccine Action Plan 2015–2020 (2014). 2017. Available online: http://www.euro.who.int/en/health-topics/disease-prevention/vaccines-and-immunization/publications/2014/european-vaccine-action-plan-20152020-2014 (accessed on 18 March 2017).
- Filia, A.; Amendola, A.; Faccini, M.; Del Manso, M.; Senatore, S.; Bianchi, S.; Borrini, B.M.; Ciampelli, A.; Tanzi, E.; Filipponi, M.T.; et al. Outbreak of a new measles B3 variant in the Roma/Sinti population with transmission in the nosocomial setting, Italy, November 2015 to April 2016. Eurosurveillance 2016, 21, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.A.; Bacci, S.; Shadwick, R.; Tillmann, T.; Rechel, B.; Noori, T.; Suk, J.E.; Odone, A.; Ingleby, J.D.; Mladovsky, P.; et al. Measles among migrants in the European Union and the European Economic Area. Scand. J. Public Health 2016, 44, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.; Haeghebaert, S.; Merlin, B.; Antona, D.; Simon, N.; Elmouden, M.; Battist, F.; Janssens, M.; Wyndels, K.; Chaud, P. Measles outbreak in a refugee settlement in Calais, France: January to February 2016. Eurosurveillance 2016, 21, 30167. [Google Scholar] [CrossRef] [PubMed]
- Khetsuriani, N.; Perehinets, I.; Nitzan, D.; Popovic, D.; Moran, T.; Allahverdiyeva, V.; Huseynov, S.; Gavrilin, E.; Slobodianyk, L.; Izhyk, O.; et al. Responding to a cVDPV1 outbreak in Ukraine: Implications, challenges and opportunities. Vaccine 2017, 35, 4769–4776. [Google Scholar] [CrossRef] [PubMed]
- Werber, D.; Hoffmann, A.; Santibanez, S.; Mankertz, A.; Sagebiel, D. Large measles outbreak introduced by asylum seekers and spread among the insufficiently vaccinated resident population, Berlin, October 2014 to August 2015. Eurosurveillance 2017, 22, 30599. [Google Scholar] [CrossRef] [PubMed]
- Derrough, T.; Salekeen, A. Lessons learnt to keep Europe polio-free: A review of outbreaks in the European Union, European Economic Area, and candidate countries, 1973 to 2013. Eurosurveillance 2016, 21, 30210. [Google Scholar] [CrossRef] [PubMed]
- Grammens, T.; Maes, V.; Hutse, V.; Laisnez, V.; Schirvel, C.; Trémérie, J.M.; Sabbe, M. Different measles outbreaks in Belgium, January to June 2016—A challenge for public health. Eurosurveillance 2016, 21, 30313. [Google Scholar] [CrossRef] [PubMed]
- Woudenberg, T.; van Binnendijk, R.S.; Sanders, E.A.M.; Wallinga, J.; de Melker, H.E.; Ruijs, W.L.M.; Hahné, S.J.M. Large measles epidemic in the Netherlands, May 2013 to March 2014: Changing epidemiology. Eurosurveillance 2017, 22, 30443. [Google Scholar] [CrossRef] [PubMed]
- Antona, D.; Lévy-Bruhl, D.; Baudon, C.; Freymuth, F.; Lamy, M.; Maine, C.; Floret, D.; Parent du Chatelet, I. Measles Elimination Efforts and 2008–2011 Outbreak, France. Emerg. Infect. Dis. 2013, 19, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Hargreaves, S.; Nellums, L.B.; Ramsay, M.; Saliba, V.; Majeed, A.; Mounier-Jack, S.; Friedland, J.S. Who is responsible for the vaccination of migrants in Europe? Lancet 2018, 391, 1752–1754. [Google Scholar] [CrossRef]
- Barnett, E.D.; Christiansen, D.; Figueira, M. Seroprevalence of measles, rubella, and varicella in refugees. Clin. Infect. Dis. 2002, 35, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Greenaway, C.; Dongier, P.; Boivin, J.F.; Tapiero, B.; Miller, M.; Schwartzman, K. Susceptibility to measles, mumps, and rubella in newly arrived adult immigrants and refugees. Ann. Intern. Med. 2007, 146, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Mipatrini, D.; Stefanelli, P.; Severoni, S.; Rezza, G. Vaccinations in migrants and refugees: A challenge for European health systems. A systematic review of current scientific evidence. Pathog. Glob. Health 2017, 111, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Toikkanen, S.E.; Baillot, A.; Dreesman, J.; Mertens, E. Seroprevalence of Antibodies against Measles, Rubella and Varicella among Asylum Seekers Arriving in Lower Saxony, Germany, November 2014–October 2015. Int. J. Environ. Res. Public Health 2016, 13, E650. [Google Scholar] [CrossRef] [PubMed]
- Freidl, G.S.; Tostmann, A.; Curvers, M.; Ruijs, W.L.M.; Smits, G.; Schepp, R.; Duizer, E.; Boland, G.; de Melker, H.; van der Klis, F.R.M.; et al. Immunity against measles, mumps, rubella, varicella, diphtheria, tetanus, polio, hepatitis A and hepatitis B among adult asylum seekers in The Netherlands, 2016. Vaccine 2018, 36, 1664–1672. [Google Scholar] [CrossRef] [PubMed]
- Ceccarelli, G.; Vita, S.; Riva, E.; Cella, E.; Lopalco, M.; Antonelli, F.; De Cesaris, M.; Fogolari, M.; Dicuonzo, G.; Ciccozzi, M.; et al. Susceptibility to measles in migrant population: Implication for policy makers. J. Travel Med. 2018, 25, tax080. [Google Scholar] [CrossRef] [PubMed]
- Nakken, C.S.; Skovdal, M.; Nellums, L.B.; Friedland, J.S.; Hargreaves, S.; Norredam, M. Vaccination status and needs of asylum-seeking children in Denmark: A retrospective data analysis. Public Health 2018, 158, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Hubschen, J.M.; Charpentier, E.; Weicherding, P.; Muller, C.P. IgG antibody prevalence suggests high immunization needs in newcomers to Luxembourg, 2012. Vaccine 2018, 36, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Roberton, T.; Weiss, W.; Doocy, S. Challenges in Estimating Vaccine Coverage in Refugee and Displaced Populations: Results From Household Surveys in Jordan and Lebanon. Vaccines 2017, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S.; Orenstein, W.; Offit, P.; Edwards, K.M. Plotkin’s Vaccines, 7th ed.; Elsevier: Philadelphia, PA, USA, 2018; pp. 1645–1691. [Google Scholar]
- GHO|By Category|Measles, 2nd Dose (MCV2)—Immunization Coverage Estimates by WHO Region. Who Minerva Publish Date Minerva Publish Date. Available online: http://apps.who.int/gho/data/view.main.MCV2vREG?lang=en (accessed on 17 September 2018).
- Anderson, R.M. The concept of herd immunity and the design of community-based immunization programmes. Vaccine 1992, 10, 928–935. [Google Scholar] [CrossRef]
- Bica, M.A.; Clemens, R. Vaccination policies of immigrants in the EU/EEA Member States-the measles immunization example. Eur. J. Public Health 2018, 28, 439–444. [Google Scholar] [CrossRef] [PubMed]
- Vito, E.D.; Parente, P.; Waure, C.D.; Poscia, A.; Ricciardi, W. A Review of Evidence on Equitable Delivery, Access and Utilization of Immunization Services for Migrants and Refugees in the WHO European Region; WHO Regional Office for Europe: Copenhagen, Denmark, 2017. [Google Scholar]
- Pottie, K.; Mayhew, A.; Morton, R.; Greenaway, C.; Akl, E.; Rahman, P. Prevention and assessment of infectious diseases among children and adult migrants arriving to the European Union/European Economic Association: A protocol for a suite of systematic reviews for public health and health systems. BMJ Open 2017, 7, e014608. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Wm, B. Improving efficiency and confidence in systematic literature searching. In Proceedings of the EAHIL+ICAHIS + ICLC, Edinburgh, UK, 10–12 June 2015. [Google Scholar]
- Sampson, M.; McGowan, J. Inquisitio validus Index Medicus: A simple method of validating MEDLINE systematic review searches. Res. Synth. Methods 2011, 16, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O’Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 17 September 2018).
- Drummond, M.F.; Sculpher, M.J.; Torrance, G.W.; O’brien, B.J.; Stoddart, G.L. Methods for the Economic Evaluation of Health Care Programmes, 2nd ed.; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Milne, B.; Raman, S.; Thomas, P.; Shah, S. Immunisation of refugee and migrant young people: Can schools do the job? Aust. N. Z. J. Public Health 2006, 30, 526–528. [Google Scholar] [CrossRef] [PubMed]
- Spadea, A.; Semyonov, L.; Unim, B.; Giraldi, G.; Corda, B.; D′Amici, A.M.; Ercole, A.; Boccia, A.; La Torre, G. Action against vaccine-preventable infectious diseases and tuberculosis in Nomad Camps: The experience of a Local Health Unit in Rome. Annali di Igiene Medicina Preventiva e di Comunita 2014, 26, 176–180. [Google Scholar] [PubMed]
- Brockmann, S.O.; Wjst, S.; Zelmer, U.; Carollo, S.; Schmid, M.; Roller, G.; Eichner, M. Public Health initiative for improved vaccination for asylum seekers. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2016, 59, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.L.; Veenstra, D. Economic analysis of prevaccination serotesting compared with presumptive immunization for polio, diphtheria, and tetanus in internationally adopted and immigrant infants. Pediatrics 2006, 117, 1650–1655. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Luo, S.; Tang, X.; Lou, L.; Chen, Y.; Guo, J.; Zhang, B. Does introducing an immunization package of services for migrant children improve the coverage, service quality and understanding? An evidence from an intervention study among 1548 migrant children in eastern China. BMC Public Health 2015, 15, 664. [Google Scholar] [CrossRef] [PubMed]
- Ndiaye, S.M.; Ahmed, M.A.; Denson, M.; Craig, A.S.; Kretsinger, K.; Cherif, B.; Kandolo, P.; Moto, D.D.; Richelot, A.; Tuma, J. Polio outbreak among nomads in Chad: Outbreak response and lessons learned. J. Infect. Dis. 2014, 210 (Suppl. 1), S74–S84. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, P.; Benjamin, A.I.; Myles, P.R.; Babu, B.V. Evaluation of a community-based intervention to improve routine childhood vaccination uptake among migrants in urban slums of Ludhiana, India. J. Public Health Oxf. Engl. 2017, 39, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, M.A.; Makokha, F.; Hussein, A.M.; Mohamed, G.; Mach, O.; Humayun, K.; Okiror, S.; Abrar, L.; Nasibov, O.; Burton, J.; et al. Combined use of inactivated and oral poliovirus vaccines in refugee camps and surrounding communities—Kenya, December 2013. MMWR—Morb. Mortal. Wkly. Rep. 2014, 63, 237–241. [Google Scholar] [PubMed]
- Adam, I.F.; Nakamura, K.; Kizuki, M.; Al Rifai, R.; Vanching, U. Relationship between implementing interpersonal communication and mass education campaigns in emergency settings and use of reproductive healthcare services: Evidence from Darfur, Sudan. BMJ Open 2015, 5, e008285. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.L.; Dang, M.T.; Ly, M.Y.; Diaz, R. Perception of barriers to immunization among parents of Hmong origin in California. Am. J. Public Health 2010, 100, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Harmsen, I.A.; Bos, H.; Ruiter, R.A.C.; Paulussen, T.G.W.; Kok, G.; de Melker, H.E.; Mollema, L. Vaccination decision-making of immigrant parents in the Netherlands; a focus group study. BMC Public Health 2015, 15, 1229. [Google Scholar] [CrossRef] [PubMed]
- Canavati, S.; Plugge, E.; Suwanjatuporn, S.; Sombatrungjaroen, S.; Nosten, F. Barriers to immunization among children of migrant workers from Myanmar living in Tak province, Thailand. Bull. World Health Organ. 2011, 89, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Kowal, S.P.; Jardine, C.G.; Bubela, T.M. “If they tell me to get it, I’ll get it. If they don’t...”: Immunization decision-making processes of immigrant mothers. Can. J. Public Health 2015, 106, e230–e235. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.D.L.; Lam, W.W.T.; Wu, J.T.; Liao, Q. Chinese immigrant parents’ vaccination decision making for children: A qualitative analysis. BMC Public Health 2014, 14, 133. [Google Scholar] [CrossRef] [PubMed]
- Devroey, D.; Riffi, A.; Balemans, R.; Van De Vijver, E.; Chovanova, H.; Vandevoorde, J. Comparison of knowledge and attitudes about vaccination between Belgian and immigrant adolescents. J. Infect. Public Health 2013, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Infectious Diseases of Specific Relevance to Newly-Arrived Migrants in the EU/EEA—19 November 2015; ECDC: Stockholm, Sweden, 2015. [Google Scholar]
- Blondell, S.J.; Kitter, B.; Griffin, M.P.; Durham, J. Barriers and Facilitators to HIV Testing in Migrants in High-Income Countries: A Systematic Review. AIDS Behav. 2015, 19, 2012–2024. [Google Scholar] [CrossRef] [PubMed]
- Agudelo-Suárez, A.A.; Gil-González, D.; Vives-Cases, C.; Love, J.G.; Wimpenny, P.; Ronda-Pérez, E. A metasynthesis of qualitative studies regarding opinions and perceptions about barriers and determinants of health services’ accessibility in economic migrants. BMC Health Serv. Res. 2012, 12, 461. [Google Scholar] [CrossRef] [PubMed]
- Ingleby, D.; Petrova-Benedict, R. Recommendations on Access to Health Services for Migrants in an Irregular Situation: An Expert Consensus. 2016. Available online: http://equi-health.eea.iom.int/images/Expert_consensus_Recommendations.pdf (accessed on 17 September 2018).
- Coleman, M.S.; Burke, H.M.; Welstead, B.L.; Mitchell, T.; Taylor, E.M.; Shapovalov, D.; Maskery, B.A.; Joo, H.; Weinberg, M. Cost analysis of measles in refugees arriving at Los Angeles International Airport from Malaysia. Hum. Vaccines Immunother. 2017, 13, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Joo, H.; Maskery, B.; Mitchell, T.; Leidner, A.; Klosovsky, A.; Weinberg, M. A comparative cost analysis of the Vaccination Program for US-bound Refugees. Vaccine 2018, 36, 2896–2901. [Google Scholar] [CrossRef] [PubMed]
- Catchpole, M.; Coulombier, D. Refugee crisis demands European Union-wide surveillance! Eurosurveillance 2015, 20, 30063. [Google Scholar] [CrossRef] [PubMed]
- Riccardo, F.; Dente, M.G.; Kärki, T.; Fabiani, M.; Napoli, C.; Chiarenza, A.; Giorgi Rossi, P.; Velasco Munoz, C.; Noori, T.; Declich, S. Towards a European Framework to Monitor Infectious Diseases among Migrant Populations: Design and Applicability. Int. J. Environ. Res. Public Health 2015, 12, 11640–11661. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control. Assessing the Burden of Key Infectious Diseases Affecting Migrant Populations in the EU; European Centre for Disease Prevention and Control: Stolkholm, Sweden, 2014. [Google Scholar]
- Dalla Zuanna, T.; Del Manso, M.; Giambi, C.; Riccardo, F.; Bella, A.; Caporali, M.G.; Dente, M.G.; Declich, S. Immunization Offer Targeting Migrants: Policies and Practices in Italy. Int. J. Environ. Res. Public Health 2018, 15, E968. [Google Scholar] [CrossRef] [PubMed]
- Giambi, C.; Del Manso, M.; Dalla Zuanna, T.; Riccardo, F.; Bella, A.; Caporali, M.G.; Baka, A.; Caks-Jager, N.; Melillo, T.; Mexia, R.; et al. National immunization strategies targeting migrants in six European countries. Vaccine 2018. [Google Scholar] [CrossRef] [PubMed]
- Bell, C.; Atkinson, K.M.; Wilson, K. Modernizing Immunization Practice Through the Use of Cloud Based Platforms. J. Med. Syst. 2017, 41, 57. [Google Scholar] [CrossRef] [PubMed]
- Monitoring of Migrant’s & Refugee’s Health Status—CARE: Common Approach for REfugees and Other Migrants’ Health. Available online: http://careformigrants.eu/monitor-of-migrants-refugees-health-status/ (accessed on 17 September 2018).
| Study | Quality 1 | Type of study | Setting (Country) | Population | Intervention | Results/Outcomes |
|---|---|---|---|---|---|---|
| Brockmann, 2016 [38] | 4/10 | Cohort study | Housing units (Germany) | Children, adolescent, adult asylum seekers | Vaccination “concept” facilitated by local public health office:
| 58% of refugees exposed to concept were vaccinated compared to 6% of refugees vaccinated in facilities without the intervention |
| Milne, 2006 [36] | 4/10 | Cross-sectional: assessing uptake of MMR, HepB | School (Australia) | Refugee adolescents, young adults |
| 74% students received MMR vaccine 30% historical vaccination rate |
| Spadea, 2014 [37] | 2/10 | Cross-sectional: assessing uptake of MMR and hexavalent (DPT-Hib-IPV-HepB) | Nomadic camp (Italy) | Roma children and women of childbearing age | Vaccination day held on monthly basis | 56.4% coverage of hexavalent vaccine (range 44–91%) at three camps 58.4% coverage of MMR vaccine (range 53–83%) at three camps 30% increase in vaccinations compared with previous year |
| Study | Certainty of Economic Evidence (Quality) | Design | Population | Intervention | Cost-Effectiveness | Resource Requirements |
|---|---|---|---|---|---|---|
| Cohen et al. 2006 [39] | Some allowance made for uncertainty in the estimates of costs and consequences. The costs are provided as base case, and 25% upper and lower range No probabilistic sensitivity analyses performed. Sensitivity analysis was undertaken for costs of serotesting, compliance rate and seroprevalence. Cost-effectiveness results were sensitive to changes in seroprevalence, cost of serotesting. | Decision-analytic model; results presented in 2004 US dollars | US | 1. presumptive vaccination with IPV 2. serotesting for poliovirus type 1, 2, and 3 antibodies followed by vaccination in unprotected patients | For IPV, presumptive vaccination is less costly and more effective, For Dtap, ICER is $7148 USD per person protected. | Difference in costs between 2 interventions are small. For IPV, difference in cost is very minimal: Serotesting is slightly more expensive ($5 USD) than presumptive vaccination. For Dtap, serotesting is more expensive than presumptive vaccine ($57 USD) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hui, C.; Dunn, J.; Morton, R.; Staub, L.P.; Tran, A.; Hargreaves, S.; Greenaway, C.; Biggs, B.A.; Christensen, R.; Pottie, K. Interventions to Improve Vaccination Uptake and Cost Effectiveness of Vaccination Strategies in Newly Arrived Migrants in the EU/EEA: A Systematic Review. Int. J. Environ. Res. Public Health 2018, 15, 2065. https://doi.org/10.3390/ijerph15102065
Hui C, Dunn J, Morton R, Staub LP, Tran A, Hargreaves S, Greenaway C, Biggs BA, Christensen R, Pottie K. Interventions to Improve Vaccination Uptake and Cost Effectiveness of Vaccination Strategies in Newly Arrived Migrants in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health. 2018; 15(10):2065. https://doi.org/10.3390/ijerph15102065
Chicago/Turabian StyleHui, Charles, Jessica Dunn, Rachael Morton, Lukas P. Staub, Anh Tran, Sally Hargreaves, Christina Greenaway, Beverly Ann Biggs, Robin Christensen, and Kevin Pottie. 2018. "Interventions to Improve Vaccination Uptake and Cost Effectiveness of Vaccination Strategies in Newly Arrived Migrants in the EU/EEA: A Systematic Review" International Journal of Environmental Research and Public Health 15, no. 10: 2065. https://doi.org/10.3390/ijerph15102065
APA StyleHui, C., Dunn, J., Morton, R., Staub, L. P., Tran, A., Hargreaves, S., Greenaway, C., Biggs, B. A., Christensen, R., & Pottie, K. (2018). Interventions to Improve Vaccination Uptake and Cost Effectiveness of Vaccination Strategies in Newly Arrived Migrants in the EU/EEA: A Systematic Review. International Journal of Environmental Research and Public Health, 15(10), 2065. https://doi.org/10.3390/ijerph15102065






