Environmental Risk Factors for Developing Type 2 Diabetes Mellitus: A Systematic Review
Abstract
1. Introduction
2. Biology of T2DM
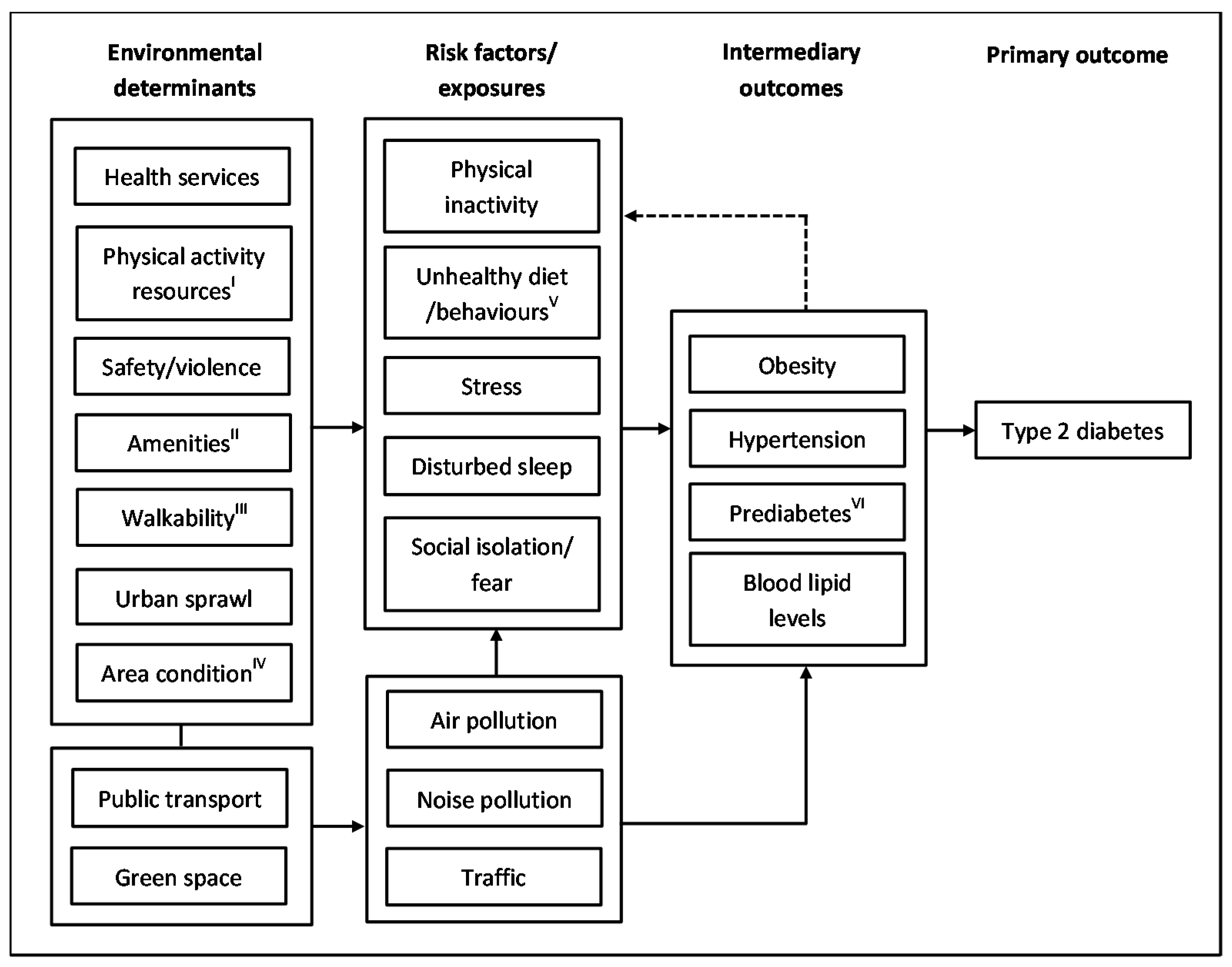
3. Mechanisms Linking Environment and T2DM
4. Methodological Issues in Studying Environmental Characteristics
4.1. Measurement and Misclassification of Exposures
4.2. Confounding and Health-Selective Migration
4.3. Sampling and Secondary Data
5. Identification Strategies
6. Methods
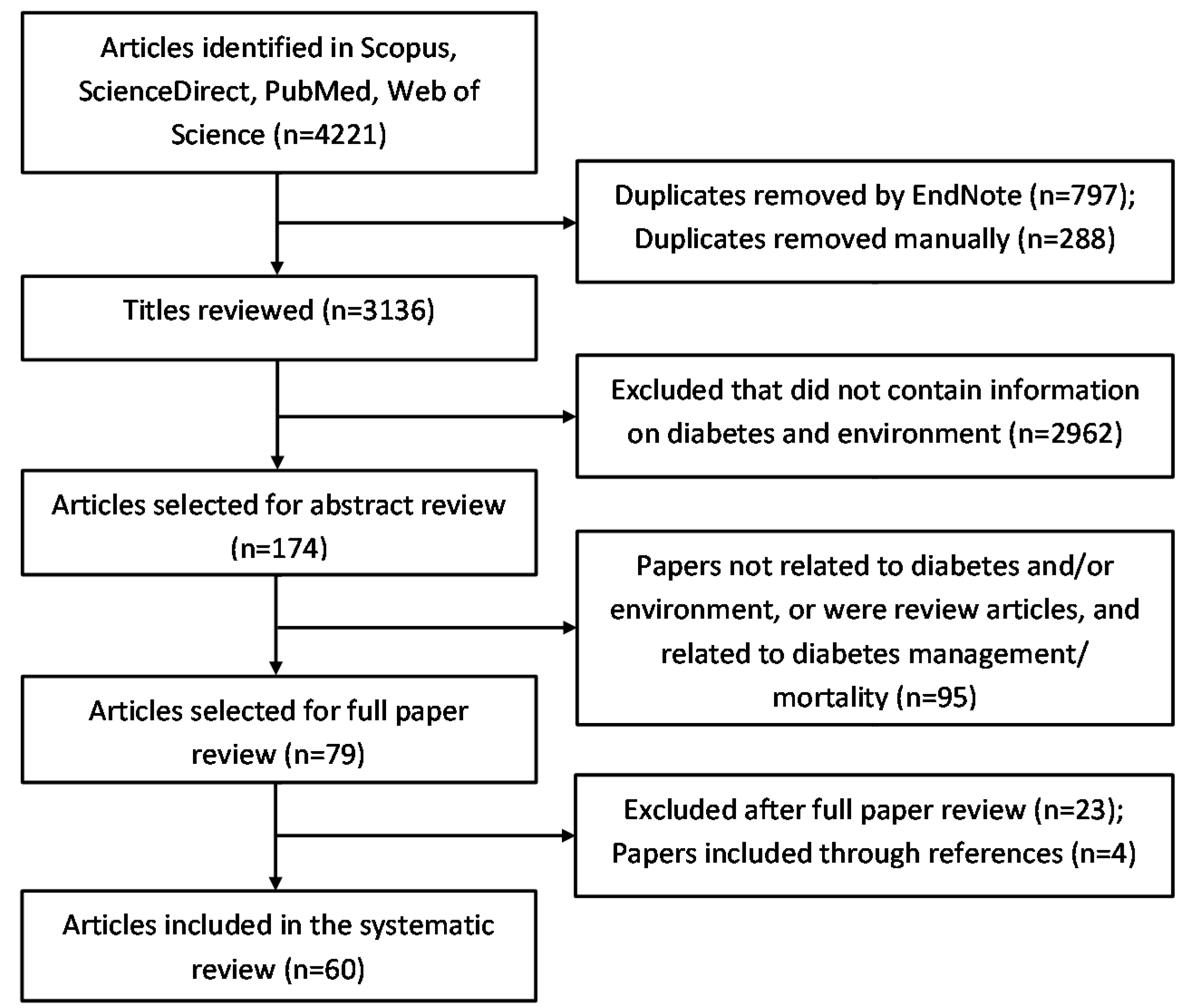
6.1. Search Strategy
6.2. Eligibility Criteria
6.3. Selection Strategy and Data Collection
6.4. Data Analysis
7. Results
7.1. Walkability
7.2. Physical Activity Resources
7.3. Food Environment
7.4. Green Space
7.5. Residential Noise, Traffic, and Proximity to Roads
7.6. Air Pollution
7.7. Neighbourhood Conditions, Safety, and Other Environmental Characteristics
8. Discussion
8.1. Walkability
8.2. Physical Activity Resources
8.3. Food Environment
8.4. Green Space
8.5. Residential Noise, Traffic, and Proximity to Roads
8.6. Air Pollution
8.7. Neighbourhood Conditions, Safety, and Other Environmental Characteristics
8.8. Strengths and Limitations
8.9. Future Research Directions
9. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bommer, C.; Heesemann, E.; Sagalova, V.; Manne-Goehler, J.; Atun, R.; Bärnighausen, T.; Vollmer, S. The global economic burden of diabetes in adults aged 20–79 years: A cost-of-illness study. Lancet Diabetes Endocrinol. 2017, 5, 423–430. [Google Scholar] [CrossRef]
- International Diabetes Federation. IDF Diabetes Atlas Seventh Edition. 2015. Available online: http://www.diabetesatlas.org/resources/2017-atlas.html (accessed on 15 November 2017).
- World Health Organization. Global Report on Diabetes. 2016. Available online: http://www.who.int/diabetes/global-report/en/ (accessed on 15 November 2017).
- Diez Roux, A.V.; Mair, C. Neighborhoods and health. Ann. N. Y. Acad. Sci. 2010, 1186, 125–145. [Google Scholar] [CrossRef] [PubMed]
- Black, J.L.; Macinko, J. Neighborhoods and obesity. Nutr. Rev. 2008, 66, 2–20. [Google Scholar] [CrossRef] [PubMed]
- Schulz, M.; Romppel, M.; Grande, G. Built environment and health: A systematic review of studies in Germany. J. Public Health 2016, 1–8. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37 (Suppl. 1), S81–S90. [Google Scholar]
- Cnop, M.; Vidal, J.; Hull, R.L.; Utzschneider, K.M.; Carr, D.B.; Schraw, T.; Scherer, P.E.; Boyko, E.J.; Fujimoto, W.Y.; Kahn, S.E. Progressive loss of β-cell function leads to worsening glucose tolerance in first-degree relatives of subjects with type 2 diabetes. Diabetes Care 2007, 30, 677–682. [Google Scholar] [CrossRef] [PubMed]
- Nolan, C.J.; Damm, P.; Prentki, M. Type 2 diabetes across generations: From pathophysiology to prevention and management. Lancet 2011, 378, 169–181. [Google Scholar] [CrossRef]
- Kahn, S.E.; Hull, R.L.; Utzschneider, K.M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006, 444, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, V.A. Defining and characterizing the progression of type 2 diabetes. Diabetes Care 2009, 32 (Suppl. 2), S151–S156. [Google Scholar] [CrossRef] [PubMed]
- Zaccardi, F.; Webb, D.R.; Yates, T.; Davies, M.J. Pathophysiology of type 1 and type 2 diabetes mellitus: A 90-year perspective. Postgrad. Med. J. 2016, 92, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Kahn, S.E.; Cooper, M.E.; Del Prato, S. Pathophysiology and treatment of type 2 diabetes: Perspectives on the past, present, and future. Lancet 2014, 383, 1068–1083. [Google Scholar] [CrossRef]
- Phillips, L.S.; Ratner, R.E.; Buse, J.B.; Kahn, S.E. We can change the natural history of type 2 diabetes. Diabetes Care 2014, 37, 2668–2676. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Khunti, K.; Davies, M.J. Type 2 diabetes. Lancet 2017, 389, 2239–2251. [Google Scholar] [CrossRef]
- Gary-Webb, T.L.; Suglia, S.F.; Tehranifar, P. Social epidemiology of diabetes and associated conditions. Curr. Diabetes Rep. 2013, 13, 850–859. [Google Scholar] [CrossRef] [PubMed]
- Agardh, E.; Allebeck, P.; Hallqvist, J.; Moradi, T.; Sidorchuk, A. Type 2 diabetes incidence and socio-economic position: A systematic review and meta-analysis. Int. J. Epidemiol. 2011, 40, 804–818. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Glass, T.A.; Curriero, F.C.; Stewart, W.F.; Schwartz, B.S. The built environment and obesity: A systematic review of the epidemiologic evidence. Health Place 2010, 16, 175–190. [Google Scholar] [CrossRef] [PubMed]
- Booth, K.M.; Pinkston, M.M.; Poston, W.S.C. Obesity and the Built Environment. J. Am. Diet. Assoc. 2005, 105, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Malambo, P.; Kengne, A.P.; De Villiers, A.; Lambert, E.V.; Puoane, T. Built Environment, Selected Risk Factors and Major Cardiovascular Disease Outcomes: A Systematic Review. PLoS ONE 2016, 11, e0166846. [Google Scholar] [CrossRef] [PubMed]
- Renalds, A.; Smith, T.H.; Hale, P.J. A systematic review of built environment and health. Fam. Community Health 2010, 33, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Wendel-Vos, W.; Droomers, M.; Kremers, S.; Brug, J.; Van Lenthe, F. Potential environmental determinants of physical activity in adults: A systematic review. Obes. Rev. 2007, 8, 425–440. [Google Scholar] [CrossRef] [PubMed]
- Saelens, B.E.; Sallis, J.F.; Frank, L.D. Environmental correlates of walking and cycling: Findings from the transportation, urban design, and planning literatures. Ann. Behav. Med. 2003, 25, 80–91. [Google Scholar] [CrossRef] [PubMed]
- Sallis, J.F.; Owen, N.; Fisher, E.B. Ecological models of health behavior. Health Behav. Health Educ. Theory Res. Pract. 2008, 4, 465–486. [Google Scholar]
- Sallis, J.F.; Floyd, M.F.; Rodríguez, D.A.; Saelens, B.E. Role of built environments in physical activity, obesity, and cardiovascular disease. Circulation 2012, 125, 729–737. [Google Scholar] [CrossRef] [PubMed]
- McNeill, L.H.; Kreuter, M.W.; Subramanian, S.V. Social Environment and Physical activity: A review of concepts and evidence. Soc. Sci. Med. 2006, 63, 1011–1022. [Google Scholar] [CrossRef] [PubMed]
- Coombes, E.; Jones, A.P.; Hillsdon, M. The relationship of physical activity and overweight to objectively measured green space accessibility and use. Soc. Sci. Med. 2010, 70, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Richardson, E.A.; Pearce, J.; Mitchell, R.; Kingham, S. Role of physical activity in the relationship between urban green space and health. Public Health 2013, 127, 318–324. [Google Scholar] [CrossRef] [PubMed]
- Berke, E.M.; Koepsell, T.D.; Moudon, A.V.; Hoskins, R.E.; Larson, E.B. Association of the Built Environment with Physical Activity and Obesity in Older Persons. Am. J. Public Health 2007, 97, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Brownson, R.C.; Baker, E.A.; Housemann, R.A.; Brennan, L.K.; Bacak, S.J. Environmental and Policy Determinants of Physical Activity in the United States. Am. J. Public Health 2001, 91, 1995–2003. [Google Scholar] [CrossRef] [PubMed]
- Cervero, R.; Duncan, M. Walking, bicycling, and urban landscapes: Evidence from the San Francisco Bay Area. Am. J. Public Health 2003, 93, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Gordon-Larsen, P.; Nelson, M.C.; Page, P.; Popkin, B.M. Inequality in the built environment underlies key health disparities in physical activity and obesity. Pediatrics 2006, 117, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Giles-Corti, B.; Broomhall, M.H.; Knuiman, M.; Collins, C.; Douglas, K.; Ng, K.; Lange, A.; Donovan, R.J. Increasing walking: How important is distance to, attractiveness, and size of public open space? Am. J. Prev. Med. 2005, 28, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Poortinga, W. Perceptions of the environment, physical activity, and obesity. Soc. Sci. Med. 2006, 63, 2835–2846. [Google Scholar] [CrossRef] [PubMed]
- Saelens, B.E.; Sallis, J.F.; Black, J.B.; Chen, D. Neighborhood-based differences in physical activity: An environment scale evaluation. Am. J. Public Health 2003, 93, 1552–1558. [Google Scholar] [CrossRef] [PubMed]
- Sallis, J.F.; Glanz, K. Physical activity and food environments: Solutions to the obesity epidemic. Milbank Q. 2009, 87, 123–154. [Google Scholar] [CrossRef] [PubMed]
- Leyden, K.M. Social capital and the built environment: The importance of walkable neighborhoods. Am. J. Public Health 2003, 93, 1546–1551. [Google Scholar] [CrossRef] [PubMed]
- Bramley, G.; Dempsey, N.; Power, S.; Brown, C.; Watkins, D. Social Sustainability and Urban Form: Evidence from Five British Cities. Environ. Plan. A 2009, 41, 2125–2142. [Google Scholar] [CrossRef]
- Li, F.; Fisher, K.J.; Brownson, R.C.; Bosworth, M. Multilevel modelling of built environment characteristics related to neighbourhood walking activity in older adults. J. Epidemiol. Community Health 2005, 59, 558–564. [Google Scholar] [CrossRef] [PubMed]
- McConville, M.E.; Rodriguez, D.A.; Clifton, K.; Cho, G.; Fleischhacker, S. Disaggregate land uses and walking. Am. J. Prev. Med. 2011, 40, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Northridge, M.E.; Sclar, E.D.; Biswas, P. Sorting out the connections between the built environment and health: A conceptual framework for navigating pathways and planning healthy cities. J. Urban Health 2003, 80, 556–568. [Google Scholar] [CrossRef] [PubMed]
- Giles-Corti, B.; Vernez-Moudon, A.; Reis, R.; Turrell, G.; Dannenberg, A.L.; Badland, H.; Foster, S.; Lowe, M.; Sallis, J.F.; Stevenson, M. City planning and population health: A global challenge. Lancet 2016, 388, 2912–2924. [Google Scholar] [CrossRef]
- Quinn, N.; Biggs, H. Creating partnerships to improve community mental health and well-being in an area of high deprivation: Lessons from a study with highrise flat residents in east Glasgow. J. Public Ment. Health 2010, 9, 16–21. [Google Scholar] [CrossRef]
- Bennett, G.G.; McNeill, L.H.; Wolin, K.Y.; Duncan, D.T.; Puleo, E.; Emmons, K.M. Safe to walk? Neighborhood safety and physical activity among public housing residents. PLoS Med. 2007, 4, e306. [Google Scholar] [CrossRef] [PubMed]
- Stockdale, S.E.; Wells, K.B.; Tang, L.; Belin, T.R.; Zhang, L.; Sherbourne, C.D. The importance of social context: Neighborhood stressors, stress-buffering mechanisms, and alcohol, drug, and mental health disorders. Soc. Sci. Med. 2007, 65, 1867–1881. [Google Scholar] [CrossRef] [PubMed]
- Miles, R.; Coutts, C.; Mohamadi, A. Neighborhood urban form, social environment, and depression. J. Urban Health 2012, 89, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Adler, N.E.; Stewart, J. Preface to the Biology of Disadvantage: Socioeconomic Status and Health. Ann. N. Y. Acad. Sci. 2010, 1186, 1–4. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Wingfield, J.C. The concept of allostasis in biology and biomedicine. Horm. Behav. 2003, 43, 2–15. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Brook, R.D. Air pollution and type 2 diabetes: Mechanistic insights. Diabetes 2012, 61, 3037–3045. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, M.; Hjortebjerg, D.; Eriksen, K.T.; Ketzel, M.; Tjønneland, A.; Overvad, K.; Raaschou-Nielsen, O. Exposure to long-term air pollution and road traffic noise in relation to cholesterol: A cross-sectional study. Environ. Int. 2015, 85 (Suppl. C), 238–243. [Google Scholar] [CrossRef] [PubMed]
- Poursafa, P.; Mansourian, M.; Motlagh, M.E.; Ardalan, G.; Kelishadi, R. Is air quality index associated with cardiometabolic risk factors in adolescents? The CASPIAN-III Study. Environ. Res. 2014, 134, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Nowak, D.J.; Crane, D.E.; Stevens, J.C. Air pollution removal by urban trees and shrubs in the United States. Urban For. Urban Green. 2006, 4, 115–123. [Google Scholar] [CrossRef]
- Gordon-Larsen, P. Food availability/convenience and obesity. Adv. Nutr. Int. Rev. J. 2014, 5, 809–817. [Google Scholar] [CrossRef] [PubMed]
- O’campo, P. Invited commentary: Advancing theory and methods for multilevel models of residential neighborhoods and health. Am. J. Epidemiol. 2003, 157, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Diez-Roux, A.V. Neighborhoods and health: Where are we and were do we go from here? Environnement résidentiel et santé: État de la question et perspectives pour le futur. Revue D’epidemiologie et de Sante Publique 2007, 55, 13–21. [Google Scholar] [CrossRef] [PubMed]
- O’Campo, P.; Wheaton, B.; Nisenbaum, R.; Glazier, R.H.; Dunn, J.R.; Chambers, C. The Neighbourhood Effects on Health and Well-being (NEHW) study. Health Place 2015, 31, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Boone-Heinonen, J.; Gordon-Larsen, P.; Guilkey, D.K.; Jacobs, D.R.; Popkin, B.M. Environment and physical activity dynamics: The role of residential self-selection. Psychol. Sports Exerc. 2011, 12, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Schaake, K.; Burgers, J.; Mulder, C.H. Ethnicity, Education and Income, and Residential Mobility between Neighbourhoods. J. Ethn. Migr. Stud. 2014, 40, 512–527. [Google Scholar] [CrossRef]
- Subramanian, S.; Jones, K.; Duncan, C. Multilevel methods for public health research. In Neighbourhoods and Health; Kawachi, I., Berkman, L.F., Eds.; Oxford University Press: New York, NY, USA, 2003; pp. 65–111. [Google Scholar]
- Austin, P.C. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar. Behav. Res. 2011, 46, 399–424. [Google Scholar] [CrossRef] [PubMed]
- Martens, E.P.; Pestman, W.R.; de Boer, A.; Belitser, S.V.; Klungel, O.H. Instrumental variables: Application and limitations. Epidemiology 2006, 17, 260–267. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, J.; Sanbonmatsu, L.; Gennetian, L.; Adam, E.; Duncan, G.J.; Katz, L.F.; Kessler, R.C.; Kling, J.R.; Lindau, S.T.; Whitaker, R.C. Neighborhoods, obesity, and diabetes—A randomized social experiment. N. Engl. J. Med. 2011, 365, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Cummins, S.; Petticrew, M.; Higgins, C.; Findlay, A.; Sparks, L. Large scale food retailing as an intervention for diet and health: Quasi-experimental evaluation of a natural experiment. J. Epidemiol. Community Health 2005, 59, 1035–1040. [Google Scholar] [CrossRef] [PubMed]
- Rutter, M. Proceeding from observed correlation to causal inference: The use of natural experiments. Perspect. Psychol. Sci. 2007, 2, 377–395. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.B. The Environment and Disease: Association or Causation? President’s Address. Proc. R. Soc. Med. 1965, 58, 295–300. [Google Scholar] [PubMed]
- Pasala, S.; Rao, A.; Sridhar, G. Built environment and diabetes. Int. J. Diabetes Dev. Ctries. 2010, 30, 63–68. [Google Scholar] [PubMed]
- Frank, L.; Kavage, S.; Devlin, A. Health and the Built Environment: A Review. Urban Design 4 Health, Ltd., 2012. Available online: http://urbandesign4health.com/projects/health-and-the-built-environment-a-review (accessed on 15 November 2017).
- Mackenbach, J.D.; Rutter, H.; Compernolle, S.; Glonti, K.; Oppert, J.-M.; Charreire, H.; De Bourdeaudhuij, I.; Brug, J.; Nijpels, G.; Lakerveld, J. Obesogenic environments: A systematic review of the association between the physical environment and adult weight status, the SPOTLIGHT project. BMC Public Health 2014, 14, 233. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Moudon, A.V. Physical Activity and Environment Research in the Health Field: Implications for Urban and Transportation Planning Practice and Research. J. Plan. Lit. 2004, 19, 147–181. [Google Scholar] [CrossRef]
- National Institutes of Health. Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies. 2017. Available online: https://www.nhlbi.nih.gov/health-pro/guidelines/in-develop/cardiovascular-risk-reduction/tools/cohort (accessed on 23 June 2017).
- Eriksson, C.; Hilding, A.; Pyko, A.; Bluhm, G.; Pershagen, G.; Ostenson, C.G. Long-Term Aircraft Noise Exposure and Body Mass Index, Waist Circumference, and Type 2 Diabetes: A Prospective Study. Environ. Health Perspect. 2014, 122, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Maas, J.; Verheij, R.A.; de Vries, S.; Spreeuwenberg, P.; Schellevis, F.G.; Groenewegen, P.P. Morbidity is related to a green living environment. J. Epidemiol. Community Health 2009, 63, 967–973. [Google Scholar] [CrossRef] [PubMed]
- Gebreab, S.Y.; Hickson, D.A.; Sims, M.; Wyatt, S.B.; Davis, S.K.; Correa, A.; Diez-Roux, A.V. Neighborhood social and physical environments and type 2 diabetes mellitus in African Americans: The Jackson Heart Study. Health Place 2017, 43, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Polsky, J.Y.; Moineddin, R.; Glazier, R.H.; Dunn, J.R.; Booth, G.L. Relative and absolute availability of fast-food restaurants in relation to the development of diabetes: A population-based cohort study. Can. J. Public Health 2016, 107 (Suppl. 1), 5312. [Google Scholar] [CrossRef] [PubMed]
- Stewart, J.E.; Battersby, S.E.; Lopez-De Fede, A.; Remington, K.C.; Hardin, J.W.; Mayfield-Smith, K. Diabetes and the socioeconomic and built environment: Geovisualization of disease prevalence and potential contextual associations using ring maps. Int. J. Health Geogr. 2011, 10, 18. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Rehm, C.D.; Moudon, A.V.; Arterburn, D. The geography of diabetes by census tract in a large sample of insured adults in King County, Washington, 2005–2006. Prev. Chronic Dis. 2014, 11, E125. [Google Scholar] [CrossRef] [PubMed]
- Cunningham-Myrie, C.A.; Theall, K.P.; Younger, N.O.; Mabile, E.A.; Tulloch-Reid, M.K.; Francis, D.K.; McFarlane, S.R.; Gordon-Strachan, G.M.; Wilks, R.J. Associations between neighborhood effects and physical activity, obesity, and diabetes: The Jamaica Health and Lifestyle Survey 2008. J. Clin. Epidemiol. 2015, 68, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Pearson, J.F.; Bachireddy, C.; Shyamprasad, S.; Goldfine, A.B.; Brownstein, J.S. Association between fine particulate matter and diabetes prevalence in the U.S. Diabetes Care 2010, 33, 2196–2201. [Google Scholar] [CrossRef] [PubMed]
- Jerrett, M.; Brook, R.; White, L.F.; Burnett, R.T.; Yu, J.; Su, J.; Seto, E.; Marshall, J.; Palmer, J.R.; Rosenberg, L.; et al. Ambient ozone and incident diabetes: A prospective analysis in a large cohort of African American women. Environ. Int. 2017, 102, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Schootman, M.; Andresen, E.M.; Wolinsky, F.D.; Malmstrom, T.K.; Miller, J.P.; Yan, Y.; Miller, D.K. The Effect of Adverse Housing and Neighborhood Conditions on the Development of Diabetes Mellitus among Middle-aged African Americans. Am. J. Epidemiol. 2007, 166, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.B.; Ravnskjaer, L.; Loft, S.; Andersen, K.K.; Brauner, E.V.; Baastrup, R.; Yao, C.; Ketzel, M.; Becker, T.; Brandt, J.; et al. Long-term exposure to fine particulate matter and incidence of diabetes in the Danish Nurse Cohort. Environ. Int. 2016, 91, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Kramer, U.; Herder, C.; Sugiri, D.; Strassburger, K.; Schikowski, T.; Ranft, U.; Rathmann, W. Traffic-Related Air Pollution and Incident Type 2 Diabetes: Results from the SALIA Cohort Study. Environ. Health Perspect. 2010, 118, 1273–1279. [Google Scholar] [CrossRef] [PubMed]
- Coogan, P.F.; White, L.F.; Jerrett, M.; Brook, R.D.; Su, J.G.; Seto, E.; Burnett, R.; Palmer, J.R.; Rosenberg, L. Air pollution and incidence of hypertension and diabetes mellitus in black women living in Los Angeles. Circulation 2012, 125, 767–772. [Google Scholar] [CrossRef] [PubMed]
- To, T.; Zhu, J.; Villeneuve, P.J.; Simatovic, J.; Feldman, L.; Gao, C.; Williams, D.; Chen, H.; Weichenthal, S.; Wall, C.; et al. Chronic disease prevalence in women and air pollution—A 30-year longitudinal cohort study. Environ. Int. 2015, 80, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Booth, G.L.; Creatore, M.I.; Moineddin, R.; Gozdyra, P.; Weyman, J.T.; Matheson, F.I.; Glazier, R.H. Unwalkable neighborhoods, poverty, and the risk of diabetes among recent immigrants to Canada compared with long-term residents. Diabetes Care 2013, 36, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Lazarevic, N.; Dobson, A.J.; Barnett, A.G.; Knibbs, L.D. Long-term ambient air pollution exposure and self-reported morbidity in the Australian Longitudinal Study on Women’s Health: A cross-sectional study. BMJ Open 2015, 5, e008714. [Google Scholar] [CrossRef] [PubMed]
- Teichert, T.; Vossoughi, M.; Vierkötter, A.; Sugiri, D.; Schikowski, T.; Schulte, T.; Roden, M.; Luckhaus, C.; Herder, C.; Krämer, U. Association between traffic-related air pollution, subclinical inflammation and impaired glucose metabolism: Results from the SALIA study. PLoS ONE 2013, 8, e83042. [Google Scholar] [CrossRef] [PubMed]
- Coogan, P.F.; White, L.F.; Yu, J.; Burnett, R.T.; Marshall, J.D.; Seto, E.; Brook, R.D.; Palmer, J.R.; Rosenberg, L.; Jerrett, M. Long term exposure to NO2 and diabetes incidence in the Black Women’s Health Study. Environ. Res. 2016, 148, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Coogan, P.F.; White, L.F.; Yu, J.; Burnett, R.T.; Seto, E.; Brook, R.D.; Palmer, J.R.; Rosenberg, L.; Jerrett, M. PM2.5 and Diabetes and Hypertension Incidence in the Black Women’s Health Study. Epidemiology 2016, 27, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Leslie, E.; Coffee, N.; Frank, L.; Owen, N.; Bauman, A.; Hugo, G. Walkability of local communities: Using geographic information systems to objectively assess relevant environmental attributes. Health Place 2007, 13, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Paquet, C.; Coffee, N.T.; Haren, M.T.; Howard, N.J.; Adams, R.J.; Taylor, A.W.; Daniel, M. Food environment, walkability, and public open spaces are associated with incident development of cardio-metabolic risk factors in a biomedical cohort. Health Place 2014, 28, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Müller-Riemenschneider, F.; Pereira, G.; Villanueva, K.; Christian, H.; Knuiman, M.; Giles-Corti, B.; Bull, F.C. Neighborhood walkability and cardiometabolic risk factors in australian adults: An observational study. BMC Public Health 2013, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Creatore, M.I.; Glazier, R.H.; Moineddin, R.; Fazli, G.S.; Johns, A.; Gozdyra, P.; Matheson, F.I.; Kaufman-Shriqui, V.; Rosella, L.C.; Manuel, D.G.; et al. Association of neighborhood walkability with change in overweight, obesity, and diabetes. J. Am. Med. Assoc. 2016, 315, 2211–2220. [Google Scholar] [CrossRef] [PubMed]
- Glazier, R.H.; Creatore, M.I.; Weyman, J.T.; Fazli, G.; Matheson, F.I.; Gozdyra, P.; Moineddin, R.; Shriqui, V.K.; Booth, G.L. Density, destinations or both? A comparison of measures of walkability in relation to transportation behaviors, obesity and diabetes in Toronto, Canada. PLoS ONE 2014, 9, e85295. [Google Scholar] [CrossRef] [PubMed]
- Sundquist, K.; Eriksson, U.; Mezuk, B.; Ohlsson, H. Neighborhood walkability, deprivation and incidence of type 2 diabetes: A population-based study on 512,061 Swedish adults. Health Place 2015, 31, 24–30. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Kang, H.M.; Ko, Y.J.; Kim, H.S.; Kim, Y.J.; Bae, W.K.; Park, S.; Cho, B. Influence of urban neighbourhood environment on physical activity and obesity-related diseases. Public Health 2015, 129, 1204–1210. [Google Scholar] [CrossRef] [PubMed]
- Ewing, R.; Meakins, G.; Hamidi, S.; Nelson, A.C. Relationship between urban sprawl and physical activity, obesity, and morbidity—Update and refinement. Health Place 2014, 26, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Auchincloss, A.H.; Diez Roux, A.V.; Brown, D.G.; Erdmann, C.A.; Bertoni, A.G. Neighborhood resources for physical activity and healthy foods and their association with insulin resistance. Epidemiology 2008, 19, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Auchincloss, A.H.; Diez Roux, A.V.; Mujahid, M.S.; Shen, M.; Bertoni, A.G.; Carnethon, M.R. Neighborhood resources for physical activity and healthy foods and incidence of type 2 diabetes mellitus: The Multi-Ethnic study of Atherosclerosis. Arch. Intern. Med. 2009, 169, 1698–1704. [Google Scholar] [CrossRef] [PubMed]
- Christine, P.J.; Auchincloss, A.H.; Bertoni, A.G.; Carnethon, M.R.; Sánchez, B.N.; Moore, K.; Adar, S.D.; Horwich, T.B.; Watson, K.E.; Diez Roux, A.V. Longitudinal associations between neighborhood physical and social environments and incident type 2 diabetes mellitus: The Multi-Ethnic Study of Atherosclerosis (MESA). JAMA Intern. Med. 2015, 175, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Ahern, M.; Brown, C.; Dukas, S. A National Study of the Association between Food Environments and County-Level Health Outcomes. J. Rural Health 2011, 27, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Salois, M.J. Obesity and diabetes, the built environment, and the ‘local’ food economy in the United States, 2007. Econ. Hum. Biol. 2012, 10, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Mezuk, B.; Li, X.; Cederin, K.; Rice, K.; Sundquist, J.; Sundquist, K. Beyond access: Characteristics of the food environment and risk of diabetes. Am. J. Epidemiol. 2016, 183, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Frankenfeld, C.L.; Leslie, T.F.; Makara, M.A. Diabetes, obesity, and recommended fruit and vegetable consumption in relation to food environment sub-types: A cross-sectional analysis of Behavioral Risk Factor Surveillance System, United States Census, and food establishment data. BMC Public Health 2015, 15, 491. [Google Scholar] [CrossRef] [PubMed]
- Piccolo, R.S.; Duncan, D.T.; Pearce, N.; McKinlay, J.B. The role of neighborhood characteristics in racial/ethnic disparities in type 2 diabetes: Results from the Boston Area Community Health (BACH) Survey. Soc. Sci. Med. 2015, 130, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Morland, K.; Diez Roux, A.V.; Wing, S. Supermarkets, other food stores, and obesity: The Atherosclerosis Risk in Communities Study. Am. J. Prev. Med. 2006, 30, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Bodicoat, D.H.; Carter, P.; Comber, A.; Edwardson, C.; Gray, L.J.; Hill, S.; Webb, D.; Yates, T.; Davies, M.J.; Khunti, K. Is the number of fast-food outlets in the neighbourhood related to screen-detected type 2 diabetes mellitus and associated risk factors? Public Health Nutr. 2014, 18, 1698–1705. [Google Scholar] [CrossRef] [PubMed]
- AlHasan, D.M.; Eberth, J.M. An ecological analysis of food outlet density and prevalence of type II diabetes in South Carolina counties. BMC Public Health 2016, 16, 10. [Google Scholar] [CrossRef] [PubMed]
- Astell-Burt, T.; Feng, X.; Kolt, G.S. Is neighborhood green space associated with a lower risk of type 2 diabetes evidence from 267,072 Australians? Diabetes Care 2014, 37, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Bodicoat, D.H.; O’Donovan, G.; Dalton, A.M.; Gray, L.J.; Yates, T.; Edwardson, C.; Hill, S.; Webb, D.R.; Khunti, K.; Davies, M.J.; et al. The association between neighbourhood greenspace and type 2 diabetes in a large cross-sectional study. BMJ Open 2014, 4, e006076. [Google Scholar] [CrossRef] [PubMed]
- Ulmer, J.M.; Wolf, K.L.; Backman, D.R.; Tretheway, R.L.; Blain, C.J.A.; O’Neil-Dunne, J.P.M.; Frank, L.D. Multiple health benefits of urban tree canopy: The mounting evidence for a green prescription. Health Place 2016, 42, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, M.; Andersen, Z.J.; Nordsborg, R.B.; Becker, T.; Tjonneland, A.; Overvad, K.; Raaschou-Nielsen, O. Long-Term Exposure to Road Traffic Noise and Incident Diabetes: A Cohort Study. Environ. Health Perspect. 2013, 121, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Eze, I.C.; Foraster, M.; Schaffner, E.; Vienneau, D.; Heritier, H.; Rudzik, F.; Thiesse, L.; Pieren, R.; Imboden, M.; von Eckardstein, A.; et al. Long-term exposure to transportation noise and air pollution in relation to incident diabetes in the SAPALDIA study. Int. J. Epidemiol. 2017, 46, 1115–1125. [Google Scholar] [CrossRef] [PubMed]
- Dzhambov, A.M.; Dimitrova, D.D. Exposures to road traffic, noise, and air pollution as risk factors for type 2 diabetes: A feasibility study in Bulgaria. Noise Health 2016, 18, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Heidemann, C.; Niemann, H.; Paprott, R.; Du, Y.; Rathmann, W.; Scheidt-Nave, C. Residential traffic and incidence of Type 2 diabetes: The German Health Interview and Examination Surveys. Diabet. Med. 2014, 31, 1269–1276. [Google Scholar] [CrossRef] [PubMed]
- Andersen, Z.J.; Raaschou-Nielsen, O.; Ketzel, M.; Jensen, S.S.; Hvidberg, M.; Loft, S.; Tjønneland, A.; Overvad, K.; Sørensen, M. Diabetes incidence and long-term exposure to air pollution: A cohort study. Diabetes Care 2012, 35, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Ward-Caviness, C.K.; Kraus, W.E.; Blach, C.; Haynes, C.S.; Dowdy, E.; Miranda, M.L.; Devlin, R.B.; Diaz-Sanchez, D.; Cascio, W.E.; Mukerjee, S.; et al. Association of Roadway Proximity with Fasting Plasma Glucose and Metabolic Risk Factors for Cardiovascular Disease in a Cross-Sectional Study of Cardiac Catheterization Patients. Environ. Health Perspect. 2015, 123, 1007–1014. [Google Scholar] [CrossRef] [PubMed]
- Dijkema, M.B.A.; Mallant, S.F.; Gehring, U.; Van Den Hurk, K.; Alssema, M.; Van Strien, R.T.; Fischer, P.H.; Nijpels, G.; Stehouwer, C.D.A.; Hoek, G.; et al. Long-term exposure to traffic-related air pollution and type 2 diabetes prevalence in a cross-sectional screening-study in the Netherlands. Environ. Health 2011, 10, 76. [Google Scholar] [CrossRef] [PubMed]
- Puett, R.C.; Hart, J.E.; Schwartz, J.; Hu, F.B.; Liese, A.D.; Laden, F. Are particulate matter exposures associated with risk of type 2 diabetes? Environ. Health Perspect. 2011, 119, 384–389. [Google Scholar] [CrossRef] [PubMed]
- Weinmayr, G.; Hennig, F.; Fuks, K.; Nonnemacher, M.; Jakobs, H.; Möhlenkamp, S.; Erbel, R.; Jöckel, K.H.; Hoffmann, B.; Moebus, S. Long-term exposure to fine particulate matter and incidence of type 2 diabetes mellitus in a cohort study: Effects of total and traffic-specific air pollution. Environ. Health 2015, 14, 53. [Google Scholar] [CrossRef] [PubMed]
- Park, S.K.; Adar, S.D.; O’Neill, M.S.; Auchincloss, A.H.; Szpiro, A.; Bertoni, A.G.; Navas-Acien, A.; Kaufman, J.D.; Diez-Roux, A.V. Long-term exposure to air pollution and type 2 diabetes mellitus in a multiethnic cohort. Am. J. Epidemiol. 2015, 181, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Burnett, R.T.; Kwong, J.C.; Villeneuve, P.J.; Goldberg, M.S.; Brook, R.D.; van Donkelaar, A.; Jerrett, M.; Martin, R.V.; Brook, J.R.; et al. Risk of incident diabetes in relation to long-term exposure to fine particulate matter in Ontario, Canada. Environ. Health Perspect. 2013, 121, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Eze, I.C.; Schaffner, E.; Fischer, E.; Schikowski, T.; Adam, M.; Imboden, M.; Tsai, M.; Carballo, D.; von Eckardstein, A.; Künzli, N.; et al. Long-term air pollution exposure and diabetes in a population-based Swiss cohort. Environ. Int. 2014, 70, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Yang, C.; Zhao, Y.; Ma, Z.; Bi, J.; Liu, Y.; Meng, X.; Wang, Y.; Cai, J.; Kan, H.; et al. Associations between long-term exposure to ambient particulate air pollution and type 2 diabetes prevalence, blood glucose and glycosylated hemoglobin levels in China. Environ. Int. 2016, 92–93, 416–421. [Google Scholar] [CrossRef] [PubMed]
- Chien, L.C.; Alamgir, H.; Yu, H.L. Spatial vulnerability of fine particulate matter relative to the prevalence of diabetes in the United States. Sci. Total Environ. 2015, 508, 136–144. [Google Scholar] [CrossRef] [PubMed]
- Sohn, D.; Oh, H. Gender-dependent differences in the relationship between diabetes mellitus and ambient air pollution among adults in South Korean cities. Iran. J. Public Health 2017, 46, 293–300. [Google Scholar] [PubMed]
- Tahmasebi, A.; Amin, M.M.; Poursafa, P.; Iraj, B.; Sadeghiyan, H.; Kelishadi, R.; Sadeghian, B. Association of geographical distribution of air quality index and type 2 diabetes mellitus in Isfahan, Iran. Pak. J. Med. Sci. 2015, 31, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Tong, L.; Li, K.; Zhou, Q. The association between air pollutants and morbidity for diabetes and liver diseases modified by sexes, ages, and seasons in Tianjin, China. Environ. Sci. Pollut. Res. Int. 2015, 22, 1215–1219. [Google Scholar] [CrossRef] [PubMed]
- Brook, R.D.; Jerrett, M.; Brook, J.R.; Bard, R.L.; Finkelstein, M.M. The relationship between diabetes mellitus and traffic-related air pollution. J. Occup. Environ. Med. 2008, 50, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Villanueva, K.; Knuiman, M.; Koohsari, M.J.; Hickey, S.; Foster, S.; Badland, H.; Nathan, A.; Bull, F.; Giles-Corti, B. People living in hilly residential areas in metropolitan Perth have less diabetes: Spurious association or important environmental determinant? Int. J. Health Geogr. 2013, 12, 59. [Google Scholar] [CrossRef] [PubMed]
- Kauhl, B.; Schweikart, J.; Krafft, T.; Keste, A.; Moskwyn, M. Do the risk factors for type 2 diabetes mellitus vary by location? A spatial analysis of health insurance claims in Northeastern Germany using kernel density estimation and geographically weighted regression. Int. J. Health Geogr. 2016, 15, 38. [Google Scholar] [CrossRef] [PubMed]
- Lewis, L.B.; Sloane, D.C.; Nascimento, L.M.; Diamant, A.L.; Guinyard, J.J.; Yancey, A.K.; Flynn, G. African Americans’ access to healthy food options in South Los Angeles restaurants. Am. J. Public Health 2011, 95, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Morland, K.; Wing, S.; Roux, A.D.; Poole, C. Neighborhood characteristics associated with the location of food stores and food service places. Am. J. Prev. Med. 2002, 22, 23–29. [Google Scholar] [CrossRef]
- Hajna, S.; Ross, N.A.; Brazeau, A.-S.; Bélisle, P.; Joseph, L.; Dasgupta, K. Associations between neighbourhood walkability and daily steps in adults: A systematic review and meta-analysis. BMC Public Health 2015, 15, 768. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wen, M.; Wang, F. Multilevel built environment features and individual odds of overweight and obesity in Utah. Appl. Geogr. 2015, 60, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Cummins, S.; Flint, E.; Matthews, S.A. New neighborhood grocery store increased awareness of food access but did not alter dietary habits or obesity. Health Aff. 2014, 33, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Boehmer, T.; Hoehner, C.; Deshpande, A.; Ramirez, L.B.; Brownson, R.C. Perceived and observed neighborhood indicators of obesity among urban adults. Int. J. Obes. 2007, 31, 968–977. [Google Scholar] [CrossRef] [PubMed]
- McCormack, G.R.; Cerin, E.; Leslie, E.; Du Toit, L.; Owen, N. Objective versus perceived walking distances to destinations: Correspondence and predictive validity. Environ. Behav. 2008, 40, 401–425. [Google Scholar] [CrossRef]
- Wolch, J.R.; Byrne, J.; Newell, J.P. Urban green space, public health, and environmental justice: The challenge of making cities ‘just green enough’. Landsc. Urban Plan. 2014, 125, 234–244. [Google Scholar] [CrossRef]
- Lee, A.C.; Maheswaran, R. The health benefits of urban green spaces: A review of the evidence. J. Public Health 2011, 33, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Lachowycz, K.; Jones, A. Greenspace and obesity: A systematic review of the evidence. Obes. Rev. 2011, 12, e183–e189. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Lin, F.; Wang, B.; Cao, Y.; Hou, X.; Wang, Y. Residential Proximity to Major Roadways and Risk of Type 2 Diabetes Mellitus: A Meta-Analysis. Int. J. Environ. Res. Public Health 2016, 14, 3. [Google Scholar] [CrossRef] [PubMed]
- Dzhambov, A.M. Long-term noise exposure and the risk for type 2 diabetes: A meta-analysis. Noise Health 2015, 17, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Kolb, H.; Martin, S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017, 15, 131. [Google Scholar] [CrossRef] [PubMed]
- Cappuccio, F.P.; D’Elia, L.; Strazzullo, P.; Miller, M.A. Quantity and Quality of Sleep and Incidence of Type 2 Diabetes: A systematic review and meta-analysis. Diabetes Care 2010, 33, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Ising, H.; Kruppa, B. Health effects caused by noise: Evidence in the literature from the past 25 years. Noise Health 2004, 6, 5–13. [Google Scholar] [PubMed]
- Kyrou, I.; Tsigos, C. Stress mechanisms and metabolic complications. Horm. Metab. Res. 2007, 39, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Janghorbani, M.; Momeni, F.; Mansourian, M. Systematic review and metaanalysis of air pollution exposure and risk of diabetes. Eur. J. Epidemiol. 2014, 29, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Balti, E.V.; Echouffo-Tcheugui, J.B.; Yako, Y.Y.; Kengne, A.P. Air pollution and risk of type 2 diabetes mellitus: A systematic review and meta-analysis. Diabetes Res. Clin. Pract. 2014, 106, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Eze, I.C.; Hemkens, L.G.; Bucher, H.C.; Hoffmann, B.; Schindler, C.; Künzli, N.; Schikowski, T.; Probst-Hensch, N.M. Association between ambient air pollution and diabetes mellitus in Europe and North America: Systematic review and meta-analysis. Environ. Health Perspect. 2015, 123, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Brook, R.D.; Rajagopalan, S.; Pope, C.A.; Brook, J.R.; Bhatnagar, A.; Diez-Roux, A.V.; Holguin, F.; Hong, Y.; Luepker, R.V.; Mittleman, M.A.; et al. Particulate Matter Air Pollution and Cardiovascular Disease. Update Sci. Statement Am. Heart Assoc. 2010, 121, 2331–2378. [Google Scholar] [CrossRef]
- World Health Organization. Ambient Air Pollution: A Global Assessment of Exposure and Burden of Disease. 2016. Available online: http://who.int/phe/publications/air-pollution-global-assessment/en/ (accessed on 15 November 2017).
- Jocson, R.M.; McLoyd, V.C. Neighborhood and Housing Disorder, Parenting, and Youth Adjustment in Low-Income Urban Families. Am. J. Community Psychol. 2015, 55, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Jones-Rounds, M.L.; Evans, G.W.; Braubach, M. The interactive effects of housing and neighbourhood quality on psychological well-being. J. Epidemiol. Community Health 2014, 68, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Austin, D.M.; Furr, L.A.; Spine, M. The effects of neighborhood conditions on perceptions of safety. J. Crim. Justice 2002, 30, 417–427. [Google Scholar] [CrossRef]
- Fish, J.S.; Ettner, S.; Ang, A.; Brown, A.F. Association of perceived neighborhood safety on body mass index. Am. J. Public Health 2010, 100, 2296–2303. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Aggarwal, A.; Tang, W.; Hurvitz, P.M.; Scully, J.; Stewart, O.; Moudon, A.V. Obesity, diet quality, physical activity, and the built environment: The need for behavioral pathways. BMC Public Health 2016, 16, 1153. [Google Scholar] [CrossRef] [PubMed]
- Ewing, R.; Schmid, T.; Killingsworth, R.; Zlot, A.; Raudenbush, S. Relationship between Urban Sprawl and Physical Activity, Obesity, and Morbidity. Am. J. Health Promot. 2003, 18, 47–57. [Google Scholar] [CrossRef] [PubMed]
| Sl | Search Terms |
|---|---|
| 1 | type 2 diabetes OR non-insulin-dependent diabetes OR prediabetes OR diabetes mellitus |
| 2 | 1 AND built environment OR neighbo?hood environment |
| 3 | 1 AND walkability OR green space OR greenspace OR parks OR open space OR trees OR land use mix |
| 4 | 1 AND food environment OR supermarket OR fast food outlet OR cafe OR bar OR liquor store OR restaurant |
| 5 | 1 AND public transport OR street connectivity OR road traffic OR train OR bus |
| 6 | 1 AND air pollution OR noise pollution |
| 7 | 1 AND neighbo?hood safety OR neighbo?hood crime |
| 8 | 1 AND amenit * OR recreational facility * |
| 9 | 1 AND access to primary health care OR health care accessibility OR access to health care OR availability of health service OR availability of health care OR health facility density OR proximity to health facility |
| Characteristics | Categories | Number i |
|---|---|---|
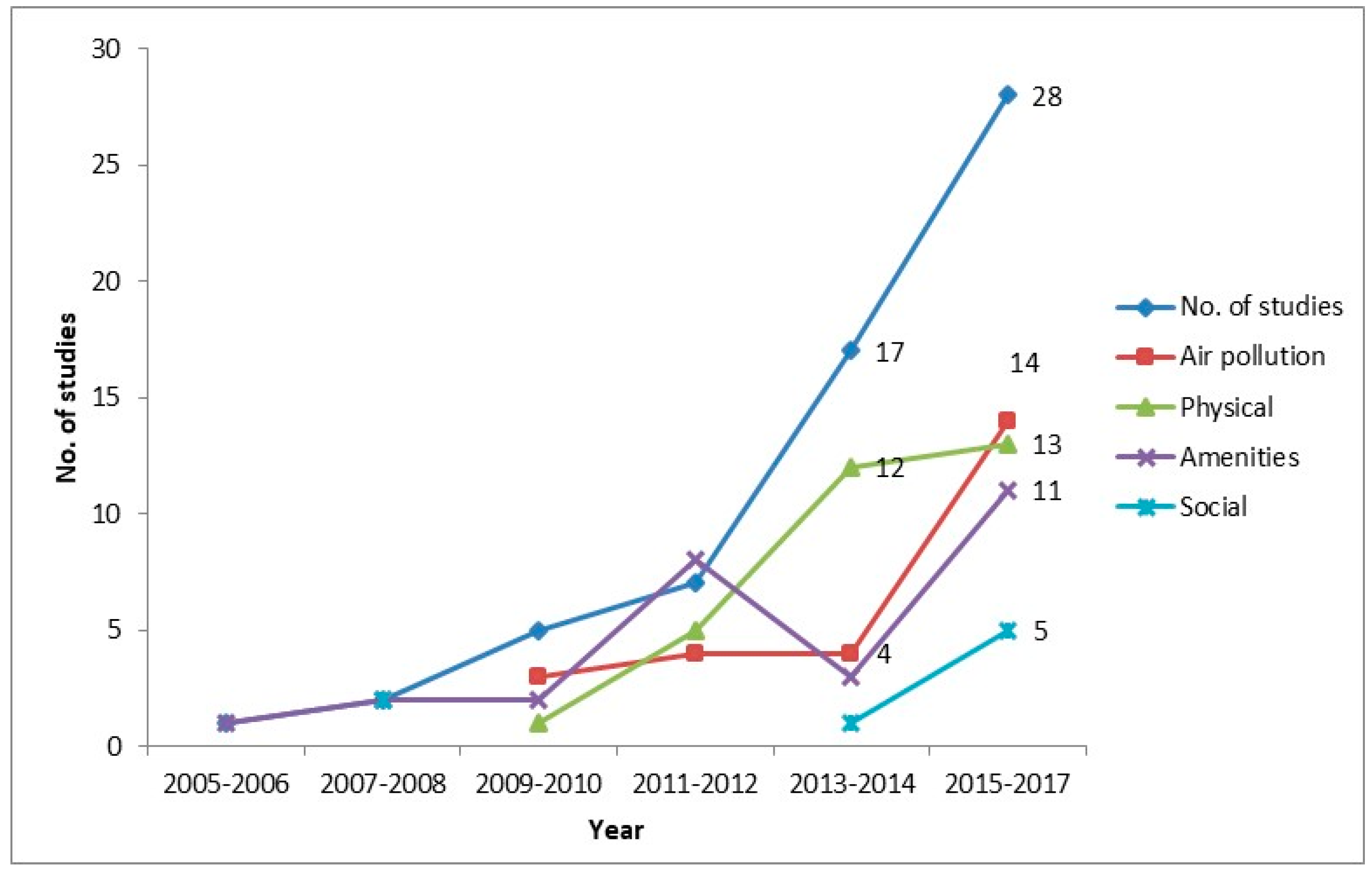
| Study year (publication) | 2005–2006 | 1 |
| 2007–2008 | 2 | |
| 2009–2010 | 5 | |
| 2011–2012 | 7 | |
| 2013–2014 | 17 | |
| 2015–2017 | 28 | |
| Study design | Cohort/longitudinal | 26 |
| Cross-sectional | 24 | |
| Ecological | 10 | |
| Country | USA | 24 |
| Canada | 6 | |
| Germany | 5 | |
| Australia | 5 | |
| Others ii | 20 | |
| Country income level | High income country | 56 |
| Upper middle income country | 4 | |
| Environment focus | Distance to roadways | 7 |
| Food environment | 17 | |
| Physical activity resources | 8 | |
| Walkability iii | 7 | |
| Neighbourhood conditions iv | 4 | |
| Crime/physical disorder/safety | 4 | |
| Green space/tree canopy | 4 | |
| Open space | 2 | |
| Others v | 4 | |
| Air pollution/quality | 25 | |
| Noise pollution | 4 | |
| Air pollutants * | PM2.5 (particulate matter of <2.5 µm) | 14 |
| NO2 (nitrogen dioxide) | 11 | |
| PM10 (particulate matter of <10 µm) | 8 | |
| NOx (nitrogen oxides) | 5 | |
| SO2 (sulphur dioxide) | 2 | |
| PM10–2.5 (particulate matter of 2.5–10 µm) | 2 | |
| BaP (Benzo alpha pyrene) | 1 | |
| Ozone | 1 | |
| Soot | 1 | |
| Air quality | 1 | |
| Noise pollution source * | Traffic noise | 3 |
| Railway noise | 2 | |
| Aircraft noise | 2 | |
| Environment measurement | Objectively measured | 51 |
| Reported by study participants/surveys | 3 | |
| Survey and objective measures combined | 6 | |
| Outcome | Type 2 diabetes mellitus | 25 |
| Diabetes Mellitus | 35 | |
| Prediabetes and diabetes mellitus | 4 | |
| Outcome assessment | Self-reported vi | 25 |
| Blood sugar tests vii | 13 | |
| Database/registers/records | 22 | |
| Association | Significant in expected direction | 82 |
| Non-significant in expected direction | 81 | |
| Non-significant in unexpected direction | 33 | |
| Null association | 6 | |
| Study quality viii | Good | 11 |
| Fair | 32 | |
| Poor | 17 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dendup, T.; Feng, X.; Clingan, S.; Astell-Burt, T. Environmental Risk Factors for Developing Type 2 Diabetes Mellitus: A Systematic Review. Int. J. Environ. Res. Public Health 2018, 15, 78. https://doi.org/10.3390/ijerph15010078
Dendup T, Feng X, Clingan S, Astell-Burt T. Environmental Risk Factors for Developing Type 2 Diabetes Mellitus: A Systematic Review. International Journal of Environmental Research and Public Health. 2018; 15(1):78. https://doi.org/10.3390/ijerph15010078
Chicago/Turabian StyleDendup, Tashi, Xiaoqi Feng, Stephanie Clingan, and Thomas Astell-Burt. 2018. "Environmental Risk Factors for Developing Type 2 Diabetes Mellitus: A Systematic Review" International Journal of Environmental Research and Public Health 15, no. 1: 78. https://doi.org/10.3390/ijerph15010078
APA StyleDendup, T., Feng, X., Clingan, S., & Astell-Burt, T. (2018). Environmental Risk Factors for Developing Type 2 Diabetes Mellitus: A Systematic Review. International Journal of Environmental Research and Public Health, 15(1), 78. https://doi.org/10.3390/ijerph15010078





