Preventive Effect of Residential Green Space on Infantile Atopic Dermatitis Associated with Prenatal Air Pollution Exposure
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Exposure to TRAP
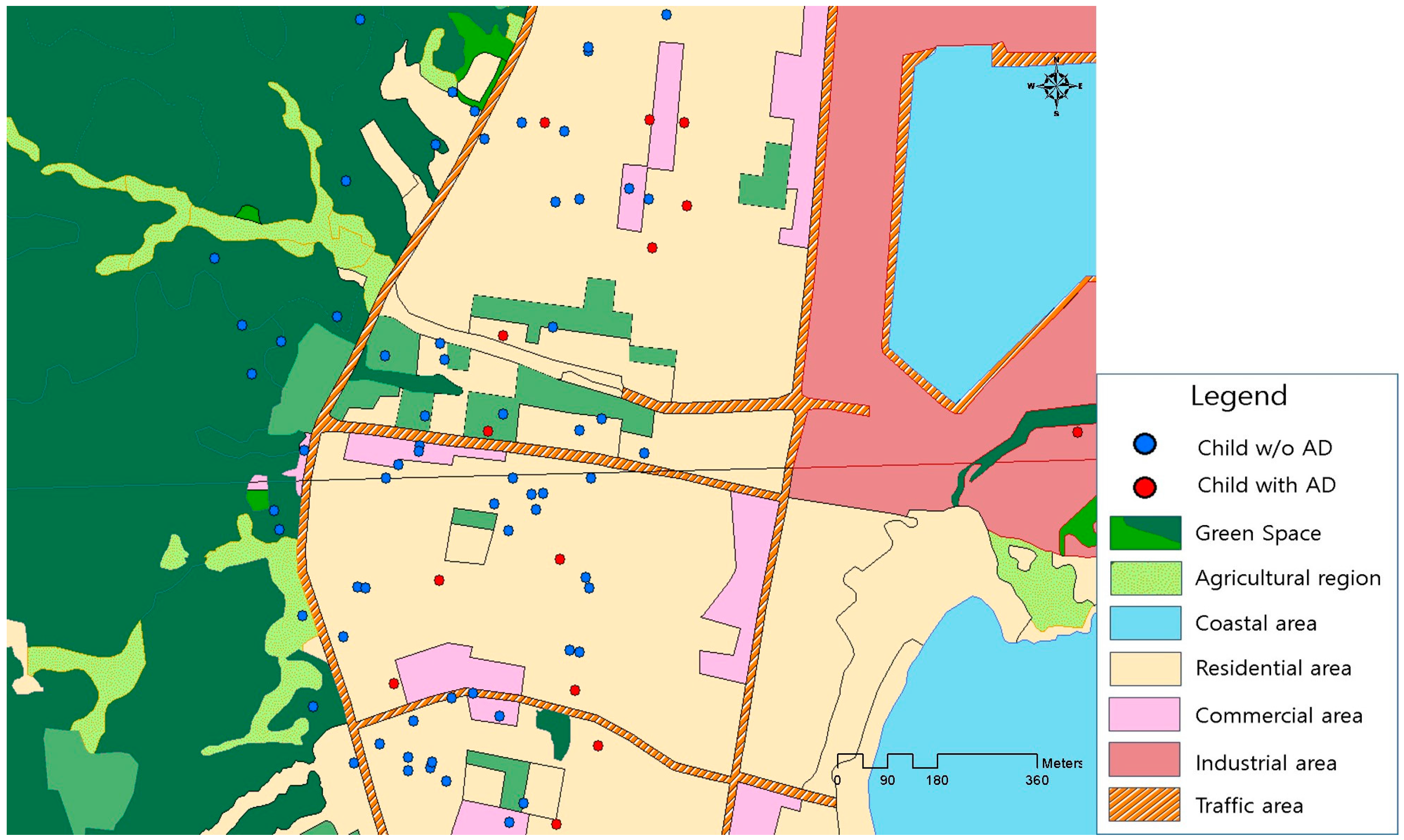
2.3. Assessment of Green Spaces
2.4. Health Outcome
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Weidinger, S.; Novak, N. Atopic dermatitis. Lancet 2016, 387, 1109–1122. [Google Scholar] [CrossRef]
- Williams, H.; Flohr, C. How epidemiology has challenged 3 prevailing concepts about atopic dermatitis. J. Allergy Clin. Immunol. 2006, 118, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Sturgill, S.; Bernard, L.A. Atopic dermatitis update. Curr. Opin. Pediatr. 2004, 16, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.; Kim, J.; Kwon, H.; Chae, Y.; Hahm, M.L.; Lee, K.J.; Park, Y.M.; Lee, S.M.; Han, M.; Kim, W.K. The prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in Korean children: Nationwide cross-sectional survey using complex sampling design. J. Korean Med. Assoc. 2011, 54, 769–778. [Google Scholar] [CrossRef]
- Ricci, G.; Patrizi, A.; Baldi, E.; Menna, G.; Tabanelli, M.; Masi, M. Long term follow-up of atopic dermatitis: Retrospective analysis of related risk factors and association with concomitant allergic diseases. J. Am. Acad. Dermatol. 2006, 55, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.J.; Guo, Y.L.; Weng, H.J.; Hsieh, W.S.; Chuang, Y.L.; Lin, S.J.; Chen, P.C. Environmental risk factors for early infantile atopic dermatitis. Pediatr. Allergy Immunol. 2007, 18, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Hasunuma, H.; Ishimaru, Y.; Yoda, Y.; Shima, M. Decline of ambient air pollution levels due to measures to control automobile emissions and effects on the prevalence of respiratory and allergic disorders among children in Japan. Environ. Res. 2014, 131, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Lee, K.; Lee, Y.M.; Lee, J.H.; Lee, S.I.; Yu, S.D.; Peak, D. Acute health effects of urban fine and ultrafine particles on children with atopic dermatitis. Environ. Res. 2011, 111, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Deng, Q.; Lu, C.; Ou, C.; Chen, L.; Yuan, H. Preconceptional, prenatal and postnatal exposure to outdoor and indoor environmental factors on allergic diseases/symptoms in preschool children. Chemosphere 2016, 152, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Deng, Q.; Lu, C.; Li, Y.; Sundell, J.; Norbäck, D. Exposure to outdoor air pollution during trimesters of pregnancy and childhood asthma, allergic rhinitis, and eczema. Environ. Res. 2016, 150, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.C.; Wen, H.J.; Chen, P.C.; Chiang, T.L.; Lin, S.J.; Guo, Y.L. Prenatal air pollutant exposure and occurrence of atopic dermatitis. Br. J. Dermatol. 2015, 173, 981–988. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Cai, J.; Huang, C.; Hu, Y.; Fu, Q.; Zou, Z.; Sun, C.; Shen, L.; Wang, X.; Pan, J.; et al. Associations of gestational and early life exposures to ambient air pollution with childhood atopic eczema in Shanghai, China. Sci. Total Environ. 2016, 572, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Deng, L.; Ou, C.; Yuan, H.; Chen, X.; Deng, Q. Preconceptional and perinatal exposure to traffic-related air pollution and eczema in preschool children. J. Dermatol. Sci. 2017, 85, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Tanaka, K.; Fujiwara, H.; Mitani, Y.; Ikemi, H.; Sasaki, S.; Ohya, Y.; Hirota, Y. Residential proximity to main roads during pregnancy and the risk of allergic disorders in Japanese infants: The Osaka Maternal and Child Health Study. Pediatr. Allergy Immunol. 2010, 21, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Sbihi, H.; Allen, R.W.; Becker, A.; Brook, J.R.; Mandhane, P.; Scott, J.A.; Sears, M.R.; Subbarao, P.; Takaro, T.K.; Turverym, S.W.; et al. Perinatal exposure to traffic-related air pollution and atopy at 1 year of age in a multi-center Canadian birth cohort study. Environ. Health Perspect. 2015, 123, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.J.; Chen, P.C.; Chiang, T.L.; Lin, S.J.; Chuang, Y.L.; Guo, Y.L. Predicting risk for early infantile atopic dermatitis by hereditary and environmental factors. Br. J. Dermatol. 2009, 161, 1166–1172. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, I.; Pedersen, M.; Garcia-Esteban, R.; Ballester, F.; Basterrechea, M.; Esplugues, A.; Fernadez-Somoano, A.; Lertxundi, A.; Tardon, A.; Sunyer, J. Early-life exposure to outdoor air pollution and respiratory health, ear infections, and eczema in infants from the INMA study. Environ. Health Perspect. 2013, 121, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Jedrychowski, W.; Perera, F.; Maugeri, U.; Mrozek-Budzyn, D.; Miller, R.L.; Flak, E.; Morz, E.; Jacek, R.; Spenler, J.D. Effects of prenatal and perinatal exposure to fine air pollutants and maternal fish consumption on the occurrence of infantile eczema. Int. Arch. Allergy Immunol. 2011, 155, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Ruokolainen, L.; von Hertzen, L.; Fyhrquist, N. Green areas around homes reduces atopic sensitization in children. Allergy 2015, 70, 195–202. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.C.; Park, S.J.; Park, C.W.; Yoon, W.S.; Choung, J.T.; Yoo, Y. Clinical and immunological effects of a forest trip in children with asthma and atopic dermatitis. Iran. J. Allergy Asthma Immunol. 2015, 14, 28–36. [Google Scholar] [PubMed]
- Tamosiunas, A.; Grazuleviciene, R.; Luksiene, D.; Dedele, A.; Reklaitiene, R.; Baceviciene, M.; Vencloviene, J.; Bernotiene, G.; Radisauskas, R.; Malinauskiene, V.; et al. Accessibility and use of urban green spaces, and cardiovascular health: Findings from a Kaunas cohort study. Environ. Health 2014, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T.; Leslie, E.; Giles-Corti, B.; Owen, N. Associations of neighborhood greenness with physical and mental health: Do walking, social coherence and local social interaction explain the relationships? J. Epidemiol. Community Health 2008, 62, e9. [Google Scholar] [CrossRef] [PubMed]
- Andrusaityte, S.; Grazuleviciene, R.; Kudzyte, J.; Bernotiene, A.; Dedele, A.; Nieuwenhuijsen, M.J. Associations between neighbourhood greenness and asthma in preschool children in Kaunas, Lithuania: A case-control study. BMJ Open 2016, 6, e010341. [Google Scholar] [CrossRef] [PubMed]
- Lovasi, G.S.; O’Neil-Dunne, J.P.; Lu, J.W.; Sheehan, D.; Perzanowski, M.S.; Macfaden, S.W.; King, K.L.; Matte, T.; Miller, R.L.; Hoepner, L.A.; et al. Urban tree canopy and asthma, wheeze, rhinitis, and allergic sensitization to tree pollen in a New York City birth cohort. Environ. Health Perspect. 2013, 121, 494–500. [Google Scholar] [CrossRef] [PubMed]
- Pilat, M.A.; McFarland, A.; Snelgrove, A.; Collins, K.; Waliczek, T.M.; Zajicek, J. The effect of tree cover and vegetation on incidence of childhood asthma in metropolitan statistical areas of Texas. HortTechnology 2012, 22, 631–637. [Google Scholar]
- Dadvand, P.; Villanueva, C.M.; Font-Ribera, L.; Martinez, D.; Basagaña, X.; Belmonte, J.; Vrihheid, M.; Grazulevicieene, R.; Kogevinas, M.; Nieuwenhuijsen, M.J. Risks and benefits of green spaces for children: A cross-sectional study of associations with sedentary behavior, obesity, asthma, and allergy. Environ. Health Perspect. 2014, 122, 1329–1335. [Google Scholar] [CrossRef] [PubMed]
- Grazuleviciene, R.; Danileviciute, A.; Dedele, A.; Vencloviene, J.; Andrusaityte, S.; Uždanaviciute, I.; Nieuwenhuijsen, M.J. Surrounding greenness, proximity to city parks and pregnancy outcomes in Kaunas cohort study. Int. J. Hyg. Environ. Health 2015, 218, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.M.; Ha, M.; Park, H.S.; Lee, B.E.; Kim, Y.J.; Hong, Y.C.; Kim, Y.; Chang, N.; Roh, Y.-M.; Kim, B.-N.; et al. The Mothers and Children’s Environmental Health (MOCEH) study. Eur. J. Epidemiol. 2009, 24, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Leem, J.H.; Kim, H.C.; Hwang, S.S.; Jung, D.Y.; Park, M.S.; Kim, J.-A.; Lee, J.-J.; Park, N.-W.; Kang, S.-C. Land Use Regression Model for Assessing Exposure and Impacts of Air Pollutants in School Children. J. Korean Soc. Atmos. Environ. 2012, 28, 571–580. (In Korean) [Google Scholar] [CrossRef]
- Lamichhane, D.K.; Kim, H.C.; Choi, C.M.; Shin, M.H.; Shim, Y.M.; Leem, J.H.; Ryu, J.S.; Nam, H.S.; Park, S.M. Lung Cancer Risk and Residential Exposure to Air Pollution: A Korean Population-Based Case-Control Study. Yonsei Med. J. 2017, 58, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Schüle, S.A.; Gabriel, K.M.A.; Bolte, G. Relationship between neighbourhood socioeconomic position and neighbourhood public green space availability: An environmental inequality analysis in a large German city applying generalized linear models. Int. J. Hyg. Environ. Health 2017, 220, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Loomis, C.A.; Koss, T.; Chu, D. Embryology. In Dermatology, 2nd ed.; Bolognia, J.L., Jorizzo, J.L., Rapini, R.P., Eds.; Mosby: Maryland Heights, MO, USA, 2008; Volume 1, pp. 7–48. [Google Scholar]
- Bieber, T. Atopic dermatitis. N. Engl. J. Med. 2008, 358, 1483–1494. [Google Scholar] [CrossRef] [PubMed]
- Currie, J.; Neidell, M. Air pollution and infant health: What can we learn from California’s recent experience? Q. J. Econ. 2005, 120, 1003–1030. [Google Scholar]
- Jøhnke, H.; Norberg, L.A.; Vach, W.; Høst, A.; Andersen, K.E. Patterns of sensitization in infants and its relation to atopic dermatitis. Pediatr. Allergy Immunol. 2006, 17, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Kuo, M. How might contact with nature promote human health? Promising mechanisms and a possible central pathway. Front. Psychol. 2015, 6, 1093. [Google Scholar] [CrossRef] [PubMed]
- Rook, G.A. Regulation of the immune system by biodiversity from the natural environment: An ecosystem service essential to health. Proc. Natl. Acad. Sci. USA 2013, 110, 18360–18367. [Google Scholar] [CrossRef] [PubMed]
- Hanski, I.; von Hertzen, L.; Fyhrquist, N.; Koskinen, K.; Torppa, K.; Laatikainen, T.; Karisola, P.; Auvinen, P.; Paulin, l.; Makela, M.J.; et al. Environmental biodiversity, human microbiota, and allergy are interrelated. Proc. Natl. Acad. Sci. USA 2012, 109, 8334–8339. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, T.; Nagao, M.; Hiraguchi, Y.; Katsumata, H.; Nishimori, H.; Iguchi, K.; Kato, Y.; Higasiura, M.; Ogawauchi, I.; Tamaki, K. Serum measurement of thymus and activation-regulated chemokine/CCL17 in children with atopic dermatitis: Elevated normal levels in infancy and age-specific analysis in atopic dermatitis. Pediatr. Allergy Immunol. 2009, 20, 633–641. [Google Scholar] [CrossRef] [PubMed]
- Furusyo, N.; Takeoka, H.; Toyoda, K.; Murata, M.; Maeda, S.; Ohnishi, H.; Fukiwake, N.; Uchi, H.; Furue, M.; Hayashi, J. Thymus and activation regulated chemokines in children with atopic dermatitis: Kyushu University Ishigaki Atopic Dermatitis Study (KIDS). Eur. J. Dermatol. 2007, 17, 397–404. [Google Scholar] [PubMed]
- Baïz, N.; Slama, R.; Béné, M.C.; Charles, M.A.; Kolopp-Sarda, M.N.; Magnan, A.; Thiebaugeorges, O.; Faure, G.; Annesi-Maesano, I. Maternal exposure to air pollution before and during pregnancy related to changes in newborn’s cord blood lymphocyte subpopulations. The EDEN study cohort. BMC Pregnancy Childbirth 2011, 11, 87. [Google Scholar] [CrossRef] [PubMed]
- Dadvand, P.; de Nazelle, A.; Triguero-Mas, M.; Schembari, A.; Cirach, M.; Amoly, E.; Figueras, F.; Basagana, X.; Ostro, B.; Nieuwenhuijsen, M. Surrounding greenness and exposure to air pollution during pregnancy: An analysis of personal monitoring data. Environ. Health Perspect. 2012, 120, 1286–1290. [Google Scholar] [CrossRef] [PubMed]
- Nowak, D.J.; Crane, D.E.; Stevens, J.C. Air pollution removal by urban trees and shrubs in the United States. Urban For. Urban Green. 2006, 4, 115–123. [Google Scholar] [CrossRef]
- Paoletti, E.; Bardelli, T.; Giovannini, G.; Pecchioli, L. Air quality impact of an urban park over time. Procedia Environ. Sci. 2011, 4, 10–16. [Google Scholar] [CrossRef]
- Weir, C.H.; Yeatts, K.B.; Sarnat, J.A.; Vizuete, W.; Salo, P.M.; Jaramillo, R.; Cohn, R.D.; Chu, H.; Zeldin, D.C.; London, S.J. Nitrogen dioxide and allergic sensitization in the 2005–2006 national health and nutrition examination survey. Respir. Med. 2013, 107, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Total (n = 659) | Atopic Dermatitis at Age 6 Months | p-Value 1 | |
|---|---|---|---|---|
| Yes (n = 168) | No (n = 492) | |||
| Maternal | ||||
| Age (years), n (%) | ||||
| <30 | 326 (49.5) | 88 (52.7) | 238 (48.4) | 0.567 |
| 30–34 | 268 (40.7) | 65 (38.9) | 203 (41.3) | |
| ≥35 | 65 (9.8) | 14 (8.4) | 51 (10.3) | |
| Education, n (%) | ||||
| <university | 180 (27.3) | 32 (19.2) | 148 (30.1) | 0.006 |
| ≥university | 479 (72.7) | 135 (80.8) | 344 (69.9) | |
| Income per month, n (%) | ||||
| <$2000 | 177 (26.9) | 48 (28.7) | 129 (26.2) | 0.591 |
| $2000–4000 | 354 (53.7) | 84 (50.3) | 270 (54.9) | |
| >$4000 | 128 (19.4) | 35 (21.0) | 93 (18.9) | |
| Pre-pregnancy BMI 2, mean (SD) | 21.3 (2.9) | 20.9 (3.1) | 21.5 (2.8) | 0.025 |
| Parity, n (%) | ||||
| 0 | 315 (47.8) | 92 (55.1) | 223 (45.3) | 0.029 |
| ≥1 | 344 (52.2) | 75 (44.9) | 269 (54.7) | |
| History of allergy, n (%) | ||||
| No | 470 (71.3) | 113 (67.7) | 357 (72.6) | 0.227 |
| Yes | 189 (28.7) | 54 (32.3) | 135 (27.4) | |
| Exposure to SHS, n (%) | ||||
| No | 400 (60.7) | 106 (63.5) | 294 (59.8) | 0.395 |
| Yes | 259 (39.3) | 61 (36.5) | 198 (40.2) | |
| Residential mobility, n (%) | ||||
| No | 575 (87.2) | 140 (83.8) | 435 (88.4) | 0.125 |
| Yes | 84 (12.8) | 27 (16.2) | 57 (11.6) | |
| Gestational age (weeks), mean (SD) | 39.0 (1.1) | 39.1 (1.1) | 39.0 (1.1) | 0.665 |
| Type of birth, n (%) | ||||
| Vaginal birth | 425 (64.5) | 108 (64.7) | 317 (64.4) | 0.955 |
| Cesarean section | 234 (35.5) | 59 (35.3) | 175 (35.6) | |
| Season of birth, n (%) | ||||
| Winter | 184 (27.9) | 43 (25.8) | 141 (28.7) | 0.469 |
| Others | 475 (72.1) | 124 (74.2) | 351 (71.3) | |
| Presence of pets, n (%) | ||||
| No | 642 (97.4) | 162 (97.0) | 480 (97.6) | 0.696 |
| Yes | 17 (2.6) | 5 (3.0) | 12 (2.4) | |
| Infant | ||||
| Sex, n (%) | ||||
| Male | 358 (54.3) | 89 (53.3) | 269 (54.7) | 0.757 |
| Female | 301 (45.7) | 78 (46.7) | 223 (45.3) | |
| Birth weight (grams), mean (SD) | 3310 (374.4) | 3364 (393.7) | 3292 (366.2) | 0.030 |
| Breastfeeding, n (%) | ||||
| Exclusive breast-feeding | 238 (36.1) | 67 (40.1) | 171 (34.8) | 0.212 |
| Exclusive formula or mixed feeding | 421 (63.9) | 100 (59.9) | 321 (65.2) | |
| Exposure Variables | Mean (SD) | Minimum | Percentiles | Maximum | ||
|---|---|---|---|---|---|---|
| 25 | 50 | 75 | ||||
| PM10 exposure (μg/m3) | ||||||
| First trimester | 54.27 (12.49) | 21.47 | 44.38 | 53.19 | 64.11 | 87.96 |
| Second trimester | 52.53 (9.94) | 26.70 | 45.40 | 51.74 | 59.84 | 81.23 |
| Third trimester | 54.22 (11.08) | 27.31 | 45.68 | 54.12 | 62.50 | 89.22 |
| Entire pregnancy | 53.60 (6.22) | 26.04 | 49.85 | 53.46 | 57.49 | 72.62 |
| NO2 exposure (ppb) | ||||||
| First trimester | 24.13 (9.26) | 7.37 | 18.24 | 22.13 | 26.66 | 62.85 |
| Second trimester | 24.69 (8.51) | 8.40 | 19.02 | 22.77 | 27.70 | 60.92 |
| Third trimester | 25.34 (8.43) | 9.05 | 19.58 | 23.66 | 27.85 | 54.04 |
| Entire pregnancy | 24.69 (8.01) | 9.42 | 20.07 | 22.37 | 26.49 | 55.91 |
| Temperature | ||||||
| First trimester | 11.49 (8.62) | −5.82 | 4.74 | 9.92 | 19.5 | 27.68 |
| Second trimester | 11.82 (8.29) | −2.55 | 4.47 | 9.99 | 19.89 | 25.82 |
| Third trimester | 11.59 (8.55) | −4.34 | 4.54 | 9.74 | 19.18 | 27.01 |
| Entire pregnancy | 11.63 (8.28) | −3.74 | 4.79 | 9.95 | 19.68 | 26.66 |
| Humidity | ||||||
| First trimester | 64.65 (8.64) | 46.14 | 58.24 | 64.29 | 71.27 | 83.38 |
| Second trimester | 64.25 (9.11) | 39.03 | 58.64 | 63.00 | 70.71 | 84.95 |
| Third trimester | 65.05 (8.13) | 49.19 | 58.58 | 65.33 | 71.43 | 83.21 |
| Entire pregnancy | 64.66 (8.31) | 44.79 | 58.76 | 63.88 | 70.94 | 83.26 |
| Green space (m2) | ||||||
| 100-m buffer | 38.44 (49.40) | 0.00 | 5.73 | 19.62 | 47.47 | 205.38 |
| 200-m buffer | 62.00 (63.11) | 0.00 | 13.01 | 38.31 | 98.49 | 292.93 |
| 300-m buffer | 132.52 (89.37) | 2.03 | 52.42 | 123.36 | 184.36 | 440.83 |
| 500-m buffer | 336.67 (232.79) | 2.03 | 111.83 | 247.91 | 538.30 | 903.54 |
| Exposure Variable | Atopic Dermatitis | |
|---|---|---|
| Unadjusted | Adjusted 1 | |
| OR (95% CI) | OR (95% CI) | |
| PM10 (10 μg/m3) | ||
| First trimester | 1.195 (1.038–1.376) * | 1.219 (1.023–1.452) * |
| Second trimester | 1.050 (0.881–1.253) | 0.927 (0.725–1.185) |
| Third trimester | 0.834 (0.710–0.980) * | 0.975 (0.792–1.201) |
| Entire pregnancy | 1.123 (0.844–1.493) | 1.096 (0.795–1.510) |
| NO2 (10 ppb) | ||
| First trimester | 1.202 (1.002–1.444) * | 1.353 (1.027–1.782) * |
| Second trimester | 1.045 (0.852–1.282) | 1.115 (0.839–1.480) |
| Third trimester | 0.999 (0.811–1.230) | 1.254 (0.934–1.684) |
| Entire pregnancy | 1.105 (0.891–1.369) | 1.269 (0.936–1.721) |
| Residential surrounding green space | ||
| 100-m buffer | 0.993 (0.993–1.001) | 0.997 (0.993–1.001) |
| 200-m buffer | 0.997 (0.994–0.999) * | 0.996 (0.993–0.999) * |
| 300-m buffer | 0.999 (0.996–0.999) * | 0.997 (0.995–0.999) * |
| 500-m buffer | 0.999 (0.999–1.000) | 0.999 (0.998–1.000) |
| Green Space in Residential Area (m2) 1 | n | Atopic Dermatitis | |||
|---|---|---|---|---|---|
| PM10 during 1st Trimester (10 μg/m3) | NO2 during 1st Trimester (10 ppb) | ||||
| Unadjusted OR (95% CI) | Adjusted 2 OR (95% CI) | Unadjusted OR (95% CI) | Adjusted 2 OR (95% CI) | ||
| 100-m buffer | |||||
| Tertile 1 (<11.1) | 220 | 1.289 (1.025–1.621) * | 1.454 (1.079–1.959) * | 1.087 (0.828–1.426) | 1.453 (0.944–2.236) |
| Tertile 2 (11.1–28.8) | 221 | 1.055 (0.821–1.356) | 1.047 (0.739–1.482) | 1.189 (0.805–1.756) | 0.838 (0.420–1.675) |
| Tertile 3 (>28.8) | 218 | 1.174 (0.901–1.530) | 0.938 (0.639–1.378) | 1.202 (0.752–1.921) | 1.281 (0.643–2.551) |
| 200-m buffer | |||||
| Tertile 1 (<21.1) | 220 | 1.284 (1.018–1.619) * | 1.581 (1.162–2.152) ** | 1.169 (0.887–1.541) | 1.632 (1.050–2.538) * |
| Tertile 2 (21.1–64.2) | 220 | 1.174 (0.911–1.512) | 1.120 (0.813–1.542) | 1.060 (0.726–1.546) | 1.044 (0.568–1.919) |
| Tertile 3 (>64.2) | 219 | 1.058 (0.814–1.375) | 0.852 (0.575–1.261) | 1.284 (0.710–2.323) | 1.063 (0.468–2.411) |
| 300-m buffer | |||||
| Tertile 1 (<63.3) | 220 | 1.268 (1.002–1.604) * | 1.455 (1.063–1.993) * | 1.140 (0.872–1.491) | 1.506 (0.973–2.330) |
| Tertile 2 (63.3–171.5) | 220 | 1.042 (0.815–1.330) | 1.183 (0.858–1.631) | 0.935 (0.618–1.413) | 1.020 (0.538–1.934) |
| Tertile 3 (>171.5) | 219 | 1.196 (0.910–1.572) | 0.995 (0.679–1.458) | 1.411 (0.833–2.391) | 1.599 (0.723–3.537) |
| 500-m buffer | |||||
| Tertile 1 (<158.5) | 220 | 1.356 (1.068–1.721) * | 1.451 (1.076–1.957) * | 1.171 (0.892–1.537) | 1.330 (0.877–2.017) |
| Tertile 2 (158.5–444.8) | 220 | 1.107 (0.847–1.446) | 1.246 (0.961–1.803) | 0.998 (0.643–1.550) | 1.066 (0.563–2.020) |
| Tertile 3 (>444.8) | 219 | 1.076 (0.836–1.385) | 1.005 (0.716–1.409) | 1.434 (0.911–2.225) | 1.882 (0.857–4.133) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, J.-Y.; Lamichhane, D.K.; Lee, M.; Ye, S.; Kwon, J.-H.; Park, M.-S.; Kim, H.-C.; Leem, J.-H.; Hong, Y.-C.; Kim, Y.; et al. Preventive Effect of Residential Green Space on Infantile Atopic Dermatitis Associated with Prenatal Air Pollution Exposure. Int. J. Environ. Res. Public Health 2018, 15, 102. https://doi.org/10.3390/ijerph15010102
Lee J-Y, Lamichhane DK, Lee M, Ye S, Kwon J-H, Park M-S, Kim H-C, Leem J-H, Hong Y-C, Kim Y, et al. Preventive Effect of Residential Green Space on Infantile Atopic Dermatitis Associated with Prenatal Air Pollution Exposure. International Journal of Environmental Research and Public Health. 2018; 15(1):102. https://doi.org/10.3390/ijerph15010102
Chicago/Turabian StyleLee, Ji-Young, Dirga Kumar Lamichhane, Myeongjee Lee, Shinhee Ye, Jung-Hyun Kwon, Myung-Sook Park, Hwan-Cheol Kim, Jong-Han Leem, Yun-Chul Hong, Yangho Kim, and et al. 2018. "Preventive Effect of Residential Green Space on Infantile Atopic Dermatitis Associated with Prenatal Air Pollution Exposure" International Journal of Environmental Research and Public Health 15, no. 1: 102. https://doi.org/10.3390/ijerph15010102
APA StyleLee, J.-Y., Lamichhane, D. K., Lee, M., Ye, S., Kwon, J.-H., Park, M.-S., Kim, H.-C., Leem, J.-H., Hong, Y.-C., Kim, Y., Ha, M., & Ha, E. (2018). Preventive Effect of Residential Green Space on Infantile Atopic Dermatitis Associated with Prenatal Air Pollution Exposure. International Journal of Environmental Research and Public Health, 15(1), 102. https://doi.org/10.3390/ijerph15010102







