Drinking Water Uranium and Potential Health Effects in the German Federal State of Bavaria
Abstract
:1. Introduction
2. Materials and Methods
2.1. Hydrochemical Data
2.2. Diseases Incidence Rates Data
2.3. Data Processing and Statistical Approaches
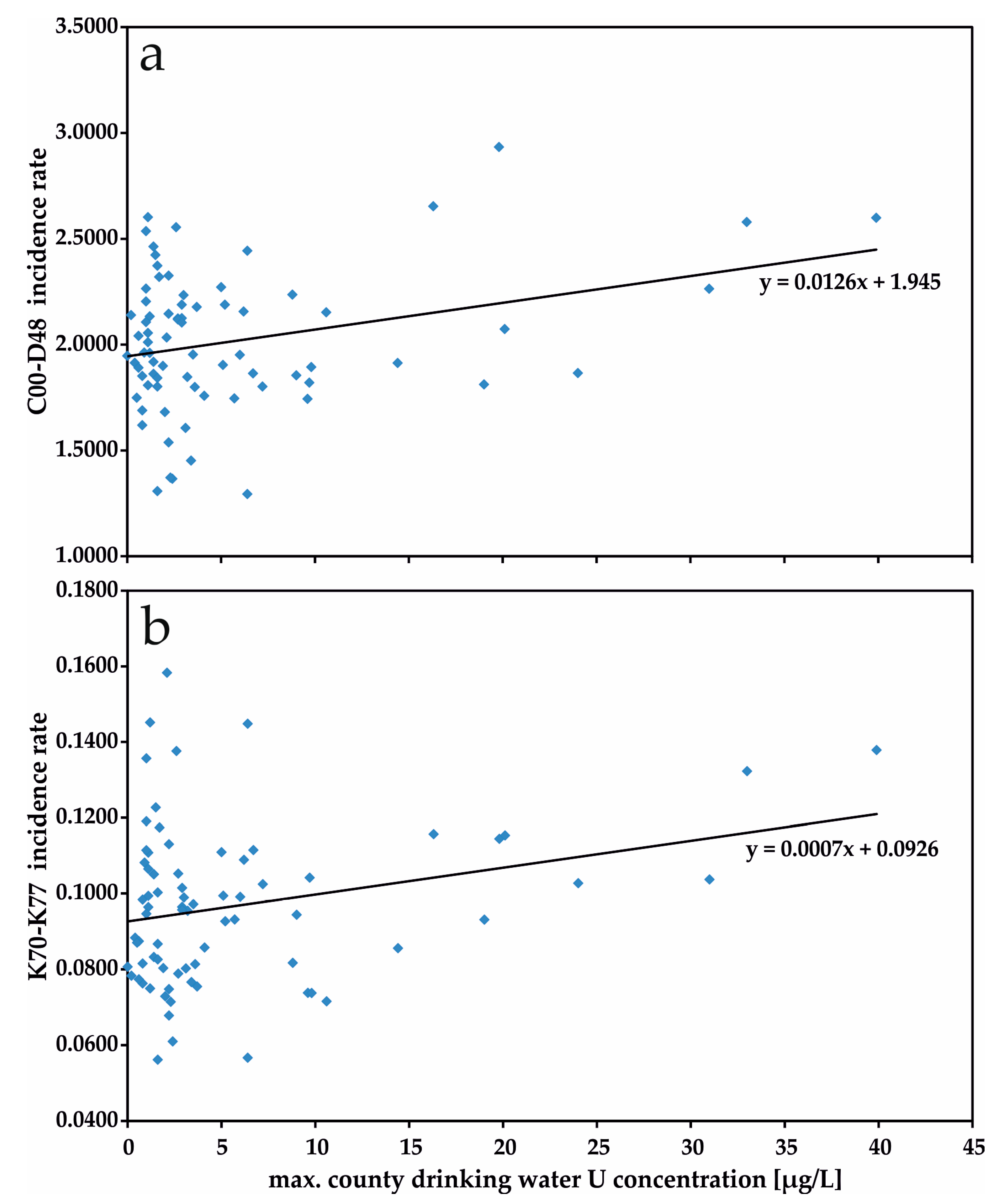
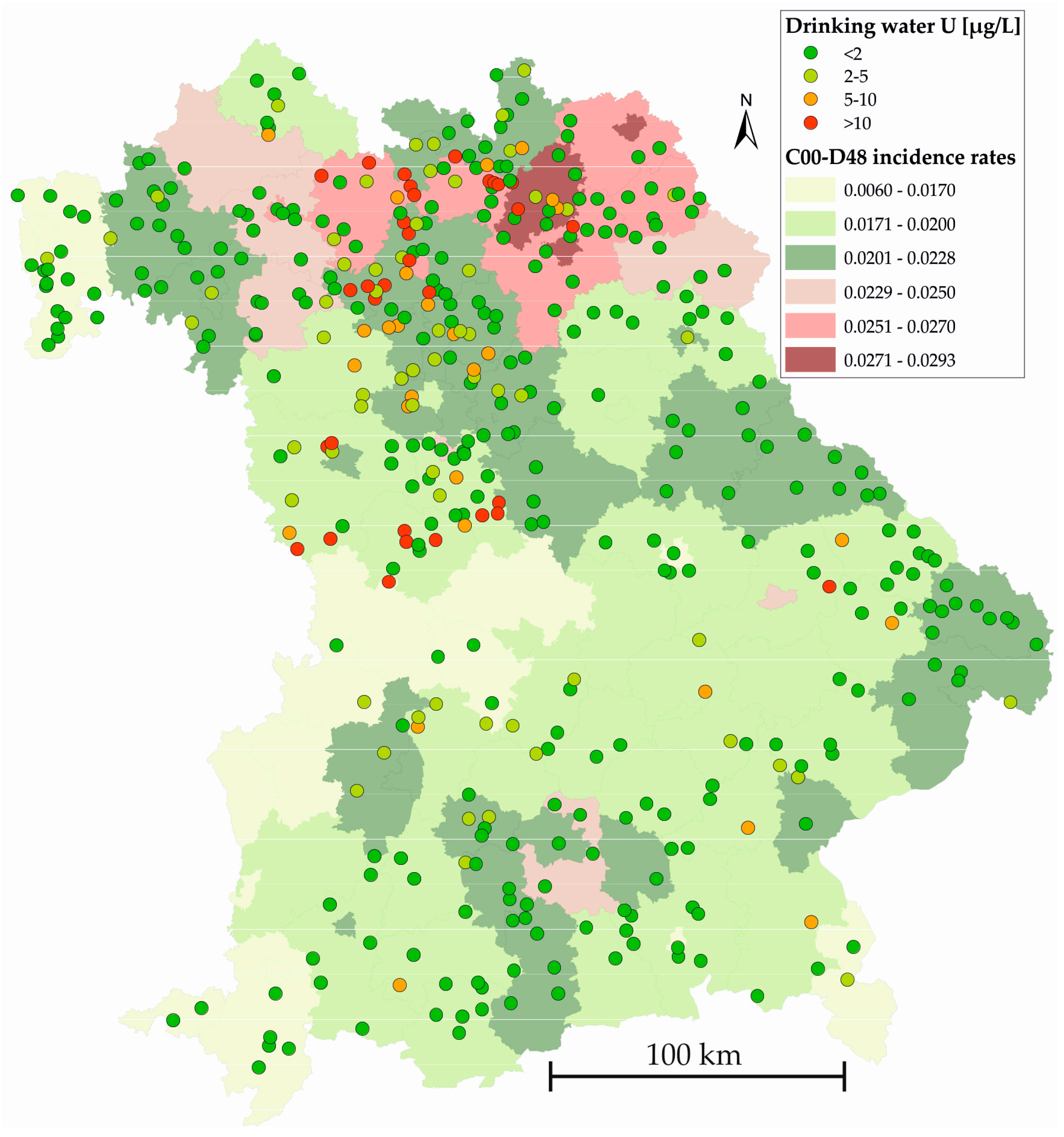
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bots, P.; Behrends, T. Uranium mobility in subsurface aqueous systems: The influence of redox conditions. Mineral. Mag. 2008, 72, 381–384. [Google Scholar] [CrossRef]
- Katsoyiannis, I.A.; Hug, S.J.; Ammann, A.; Zikoudi, A.; Hatziliontos, C. Arsenic speciation and uranium concentrations in drinking water supply wells in Northern Greece: Correlations with redox indicative parameters and implications for groundwater treatment. Sci. Total Environ. 2007, 383, 128–140. [Google Scholar] [CrossRef] [PubMed]
- Curtis, P.C.; Kohler, M.; Davis, J.A. Comparing approaches for simulating the reactive transport of U(VI) in ground water. Mine Water Environ. 2009, 28, 84–93. [Google Scholar] [CrossRef]
- Idiz, E.F.; Carlisle, D.; Kaplan, I.R. Interaction between organic matter and trace metals in a uranium rich bog, Kern County, California, U.S.A. Appl. Geochem. 1986, 1, 573–590. [Google Scholar] [CrossRef]
- Read, D.; Bennett, D.G.; Hooker, P.J.; Ivanovich, M.; Longworth, G.; Milodowski, A.E.; Noy, D.J. The migration of uranium into peat-rich soils at Broubster, Caithness, Scotland, U.K. J. Contam. Hydrol. 1993, 13, 291–308. [Google Scholar] [CrossRef]
- Regenspurg, S.; Margot-Roquier, C.; Harfouche, M.; Froidevaux, P.; Steinmann, P.; Junier, P.; Bernier-Latmani, R. Speciation of naturally-accumulated uranium in an organic-rich soil of an alpine region (Switzerland). Geochim. Cosmochim. Acta 2010, 74, 2082–2098. [Google Scholar] [CrossRef]
- Banning, A.; Demmel, T.; Rüde, T.R.; Wrobel, M. Groundwater uranium origin and fate control in a river valley aquifer. Environ. Sci. Technol. 2013, 47, 13941–13948. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, I.G.; Cidu, R.; Fanfani, L.; Pitsch, H.; Beaucaire, C.; Zuddas, P. Environmental impact of uranium mining and ore processing in the Lagoa Real district, Bahia, Brazil. Environ. Sci. Technol. 2005, 39, 8646–8652. [Google Scholar] [CrossRef] [PubMed]
- Crançon, P.; Pili, E.; Charlet, L. Uranium facilitated transport by water-dispersible colloids in field and soil columns. Sci. Total Environ. 2010, 408, 2118–2128. [Google Scholar] [CrossRef] [PubMed]
- Zielinski, R.A.; Orem, W.H.; Simmons, K.R.; Bohlen, P.J. Fertilizer-derived uranium and sulphur in rangeland soil and runoff: A case study in central Florida. Water Air Soil Pollut. 2006, 176, 163–183. [Google Scholar] [CrossRef]
- World Health Organization. Uranium in Drinking-Water—Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO Press: Geneva, Switzerland, 2012. [Google Scholar]
- Schnug, E.; Lottermoser, B.G. Fertilizer-derived uranium and its threat to human health. Environ. Sci. Technol. 2013, 47, 2433–2434. [Google Scholar] [CrossRef] [PubMed]
- Rafsanjani, M.H.; Kratz, S.; Fleckenstein, J.; Schnug, E. Solubility of uranium in fertilizers. Agric. For. Res. 2008, 58, 231–234. [Google Scholar]
- Carrière, M.; Avoscan, L.; Collins, R.; Carrot, F.; Khodja, H.; Ansoborlo, E.; Gouget, B. Influence of uranium speciation on normal rat kidney (NRK-52E) proximal cell cytotoxicity. Chem. Res. Toxicol. 2004, 17, 446–452. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.A.; Geipel, G.; Bernhard, G.; Worch, E. Investigation of uranium binding forms in selected German mineral waters. Environ. Sci. Pollut. Res. 2013, 20, 8629–8635. [Google Scholar] [CrossRef] [PubMed]
- Winde, F.; Erasmus, E.; Geipel, G. Uranium contaminated drinking water linked to leukaemia—Revisiting a case study from South Africa taking alternative exposure pathways into account. Sci. Total Environ. 2017, 574, 400–421. [Google Scholar] [CrossRef] [PubMed]
- Konietzka, R. Gastrointestinal absorption of uranium compounds—A review. Regul. Toxicol. Pharmacol. 2015, 71, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Anke, M.; Seeber, O.; Müller, R.; Schäfer, U.; Zerull, J. Uranium transfer in the food chain from soil to plants, animals and man. Chem. Erde-Geochem. 2009, 69, 75–90. [Google Scholar] [CrossRef]
- Arzuaga, X.; Gehlhaus, M.; Strong, J. Modes of action associated with uranium induced adverse effects in bone function and development. Toxicol. Lett. 2015, 236, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Brugge, D.; de Lemos, J.L.; Oldmixon, B. Exposure pathways and health effects associated with chemical and radiological toxicity of natural uranium: A review. Rev. Environ. Health 2005, 20, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Wagner, S.E.; Burch, J.B.; Bottai, M.; Puett, R.; Porter, D.; Bolick-Aldrich, S.; Temples, T.; Wilkerson, R.C.; Vena, J.E.; Hébert, J.R. Groundwater uranium and cancer incidences in South Carolina. Cancer Cause Control 2011, 22, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Bean, J.A.; Isacson, P.; Hahne, R.M.; Kohler, J. Drinking water and cancer incidence in Iowa, II. Radioactivity in drinking water. Am. J. Epidemiol. 1982, 116, 924–932. [Google Scholar] [CrossRef] [PubMed]
- Petersen, N.J.; Samuels, L.D.; Lucas, H.F.; Abrahams, S.P. An Epidemiologic Approach to Low-Level Radium 226 Exposure. Public Health Rep. 1966, 81, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Hess, C.T.; Weiffenbach, C.V.; Norton, S.A. Environmental radon and cancer correlations in Maine. Health Phys. 1983, 45, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Collman, G.W.; Loomis, D.P.; Sandler, D.P. Childhood cancer mortality and radon concentration in drinking water in North Carolina. Br. J. Cancer 1991, 63, 626–629. [Google Scholar] [CrossRef] [PubMed]
- Auvinen, A.; Kurttio, P.; Pekkanen, J.; Pukkala, E.; Ilus, T.; Salonen, L. Uranium and other natural radionuclides in drinking water and risk of leukemia: A case-cohort study in Finland. Cancer Cause Control 2002, 13, 825–829. [Google Scholar] [CrossRef]
- Auvinen, A.; Salonen, L.; Pekkanen, J.; Pukkala, E.; Ilus, T.; Kurttio, P. Radon and other natural radionuclides in drinking water and risk of stomach cancer: A case-cohort study in Finland. Int. J. Cancer 2005, 114, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Kurttio, P.; Harmoinen, A.; Saha, H.; Salonen, L.; Karpas, Z.; Komulainen, H.; Auvinen, A. Kidney toxicity of ingested uranium from drinking water. Am. J. Kidney Dis. 2006, 47, 972–982. [Google Scholar] [CrossRef] [PubMed]
- Trinkwv 2001: Trinkwasserverordnung in der Fassung der Bekanntmachung vom 10. März 2016 (BGBl. I S. 459), die Durch Artikel 4 Absatz 21 des Gesetzes vom 18. Juli 2016 (BGBl. I S. 1666) Geändert Worden ist. Available online: https://www.gesetze-im-internet.de/trinkwv_2001/TrinkwV_2001.pdf (accessed on 7 December 2016).
- Banning, A.; Rüde, T.R. Apatite weathering as a geological driver of high uranium concentrations in groundwater. Appl. Geochem. 2015, 59, 139–146. [Google Scholar] [CrossRef]
- Radespiel-Tröger, M.; Meyer, M. Association between drinking water uranium content and cancer risk in Bavaria, Germany. Int. Arch. Occup. Environ. Health 2013, 86, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Statistische Ämter des Bundes und der Länder. Available online: http://www.statistik-portal.de/Statistik-Portal/de_jb10_jahrtabu1.asp (accessed on 3 September 2016).
- Foodwatch: Bayern: Uran Im Trinkwasser, Stand: 03.04.2008. Available online: http://www.foodwatch.org/fileadmin/Themen/Uran/Dokumente/Uranwerte_2008_Bundeslaender/20080403_foodwatch_bayern_uran_wasser_durchsuchbar.pdf (accessed on 14 July 2016).
- Friedmann, L.; Herb, S.; Höbel, W.; Höller, C.; Kaschube, M.; Lessig, U.; Leutner, G.; Lindenthal, W.; Reifenhäuser, C.; Schreff, A. Untersuchungen zum Vorkommen von Uran im Grund- und Trinkwasser in Bayern. Report, Bayerisches Landesamt für Gesundheit und Lebensmittelsicherheit; Bayerisches Landesamt für Umwelt: Hof, Germany, 2007. [Google Scholar]
- Statistisches Bundesamt: Genesis. Available online: https://www-genesis.destatis.de/genesis/online (accessed on 23 August 2016).
- Domingo, J.L. Reproductive and developmental toxicity of natural and depleted uranium: A review. Reprod. Toxicol. 2001, 15, 603–609. [Google Scholar] [CrossRef]
- Canu, I.G.; Laurier, D.; Caër-Lorho, S.; Samson, E.; Timarche, M.; Auriol, B.; Bérard, P.; Collomb, P.; Quesne, B.; Blanchardon, E. Characterisation of protracted low-level exposure to uranium in the workplace: A comparison of two approaches. Int. J. Hyg. Environ. Health 2010, 213, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, O. Estimability and estimation in case-referent studies. Am. J. Epidemiol. 1976, 103, 226–235. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services, Agency for Toxic Substances and Disease Registry. Toxicological Profile for Uranium; Publisher: Atlanta, GA, USA, 2013.
- Kim, J.; Bang, Y.; Lee, W.J. Living near nuclear power plants and thyroid cancer risk: A systematic review and meta-analysis. Environ. Int. 2016, 87, 42–48. [Google Scholar] [CrossRef] [PubMed]
| No. of Counties | With Risk Factor 3 | Without Risk Factor 4 |
|---|---|---|
| with high incidence 1 | a | b |
| with low incidence 2 | c | d |
| ICD-10 Code | Explanation | New Cases | Incidence Rate | Incidence Rate/100,000 |
|---|---|---|---|---|
| C00-D48 | growths/tumors | 1,852,202 | 0.0228 | 2281 |
| E00-E07 | thyroid diseases | 98,166 | 0.0012 | 121 |
| K70-K77 | liver diseases | 87,509 | 0.0011 | 108 |
| M80-M99 | musculoskeletal diseases | 132,771 | 0.0016 | 164 |
| N00-N99 | genito-urinary diseases | 1,044,701 | 0.0129 | 1287 |
| Q00-Q99 | malformation, deformities and chromosome anomalies | 104,793 | 0.0013 | 129 |
| C00-D48 | E00-E07 | K70-K77 | M80-M99 | N00-N99 | Q00-Q99 | |
|---|---|---|---|---|---|---|
| mean U | 0.220 | 0.084 | 0.242 | 0.057 | 0.048 | 0.055 |
| p | 0.06 | 0.47 | 0.04 1 | 0.63 | 0.68 | 0.64 |
| max. U | 0.302 | 0.062 | 0.264 | 0.064 | 0.062 | 0.026 |
| p | 0.008 2 | 0.60 | 0.02 1 | 0.58 | 0.60 | 0.83 |
| Bavaria | C00-D48 | E00-E07 | K70-K77 | M80-M99 | N00-N99 | Q00-Q99 |
|---|---|---|---|---|---|---|
| OR | 1.31 | 3.22 | 1.78 | 0.67 | 1.17 | 1.15 |
| CI (OR) | 0.40; 4.22 | 1.18; 8.97 | 0.66; 4.82 | 0.26; 1.69 | 0.43; 3.22 | 0.36; 3.67 |
| RR | 1.24 | 1.56 | 1.51 | 0.80 | 1.12 | 1.12 |
| CI (RR) | 0.48; 3.23 | 1.11; 2.17 | 0.76; 2.99 | 0.47; 1.37 | 0.55; 2.29 | 0.44; 2.85 |
| Bavaria | C00-D48 | E00-E07 | K70-K77 | M80-M99 | N00-N99 | Q00-Q99 |
|---|---|---|---|---|---|---|
| OR | 4.22 | 2.07 | 3.40 | 0.43 | 1.14 | 0.45 |
| CI (OR) | 1.15; 15.5 | 0.57; 7.51 | 1.00; 11.6 | 0.12; 1.56 | 0.31; 4.23 | 0.07; 3.13 |
| RR | 2.93 | 1.89 | 2.20 | 0.60 | 1.10 | 0.51 |
| CI (RR) | 1.23; 7.02 | 0.60; 5.93 | 1.11; 4.36 | 0.25; 1.44 | 0.43; 2.76 | 0.09; 2.85 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Banning, A.; Benfer, M. Drinking Water Uranium and Potential Health Effects in the German Federal State of Bavaria. Int. J. Environ. Res. Public Health 2017, 14, 927. https://doi.org/10.3390/ijerph14080927
Banning A, Benfer M. Drinking Water Uranium and Potential Health Effects in the German Federal State of Bavaria. International Journal of Environmental Research and Public Health. 2017; 14(8):927. https://doi.org/10.3390/ijerph14080927
Chicago/Turabian StyleBanning, Andre, and Mira Benfer. 2017. "Drinking Water Uranium and Potential Health Effects in the German Federal State of Bavaria" International Journal of Environmental Research and Public Health 14, no. 8: 927. https://doi.org/10.3390/ijerph14080927
APA StyleBanning, A., & Benfer, M. (2017). Drinking Water Uranium and Potential Health Effects in the German Federal State of Bavaria. International Journal of Environmental Research and Public Health, 14(8), 927. https://doi.org/10.3390/ijerph14080927





