The Efficacy of Dog Assisted Therapy in Detained Drug Users: A Pilot Study in an Italian Attenuated Custody Institute
Abstract
:1. Introduction
1.1. Drug Addiction as Health Challenge in Prison Settings
1.2. Animal Assisted Program in Prison Settings
2. Materials and Methods
2.1. Ethical Statements
2.2. Study Design
2.3. Setting
- selection of inmates with the suitable features to be involved in the program by the prison health care unit and the prison administration;
- subscription of the “therapeutic-rehabilitative agreement” by the inmate;
- observation and psycho-diagnostic evaluation of the inmate by the clinical and functional point of view, taking into consideration his personal resources;
- planning of the “individual therapeutic program” (I.T.P.);
- integration of the patient in care and rehabilitative activities;
- evaluation of results reached by the patient compared to the fixed aims.
- ✓
- individual interviews with a psychotherapist once a week (1 h or more according to patients’ needs);
- ✓
- psychotherapy sessions in small group focused on emotion control and assertiveness (1 h, twice a week);
- ✓
- relaxation techniques and stress management course (2 h, once a week);
- ✓
- educational activities: design and painting course, theatre course (3–4 h, once a week);
- ✓
- sport activities (rugby), (2–3 h, twice a week).
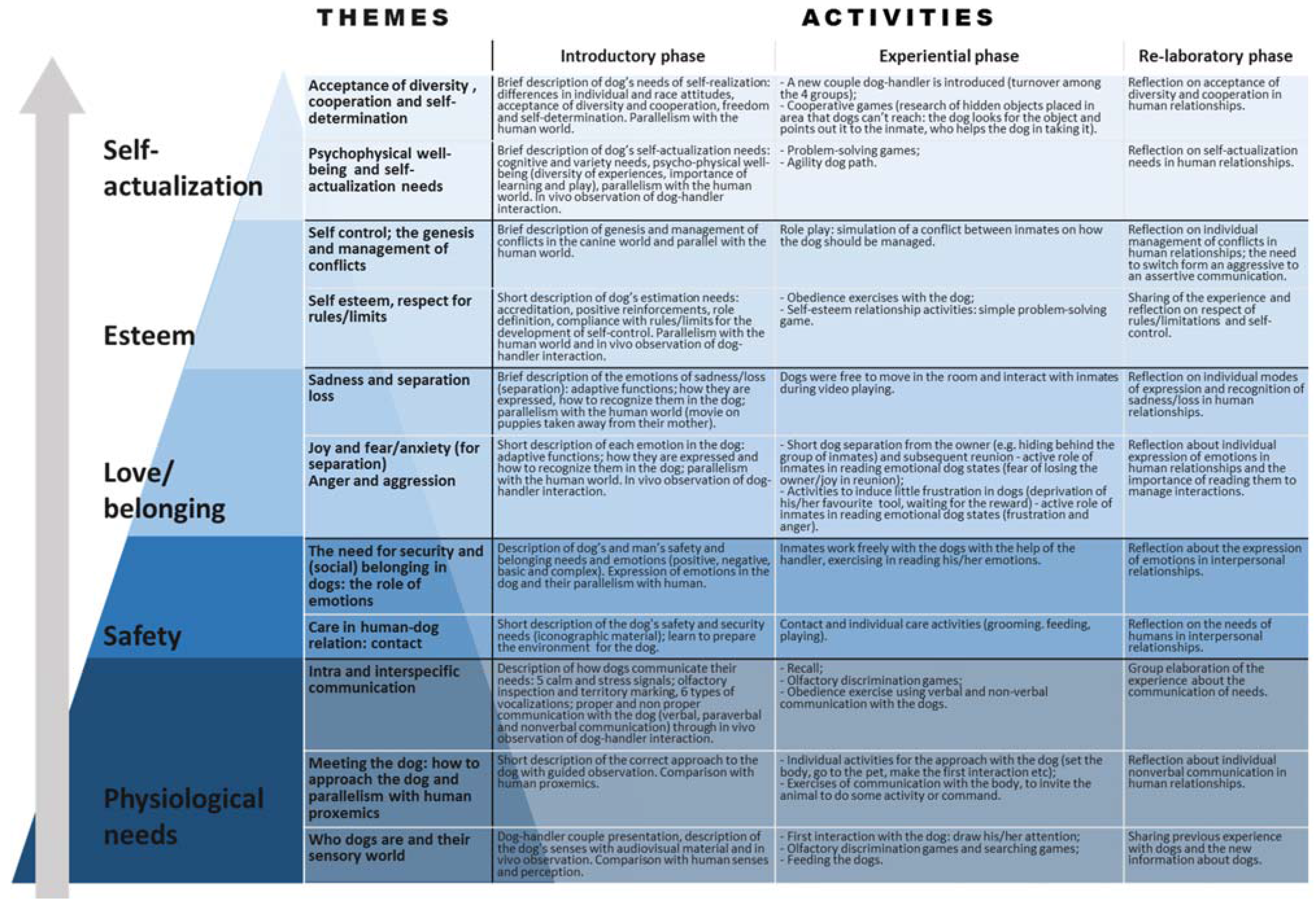
2.4. Dog Assisted Therapy Sessions
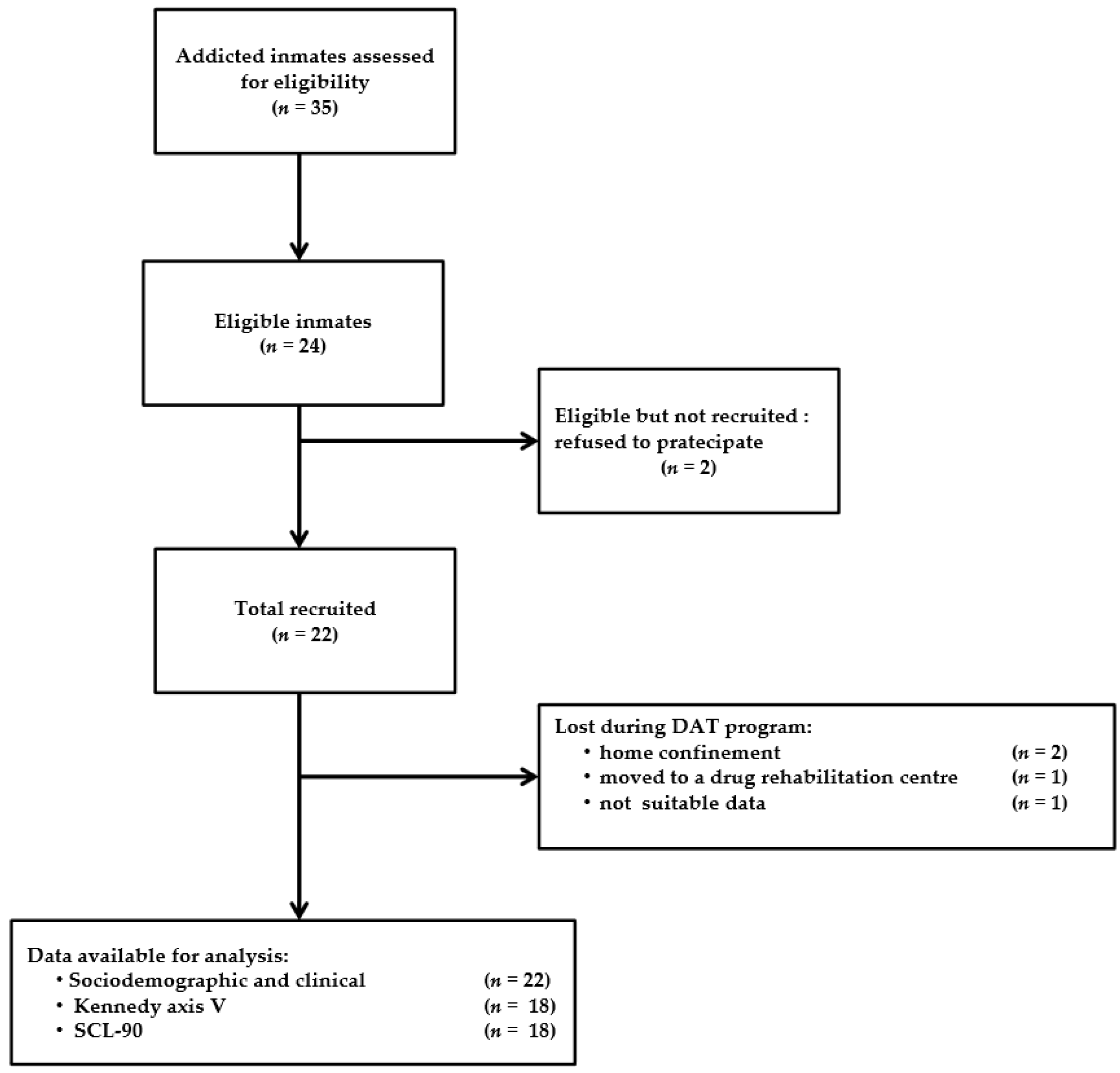
2.5. Study Population and Eligibility Criteria
2.6. Sample Size
2.7. Data Collection
2.8. Data Analysis
3. Results
3.1. Study Population
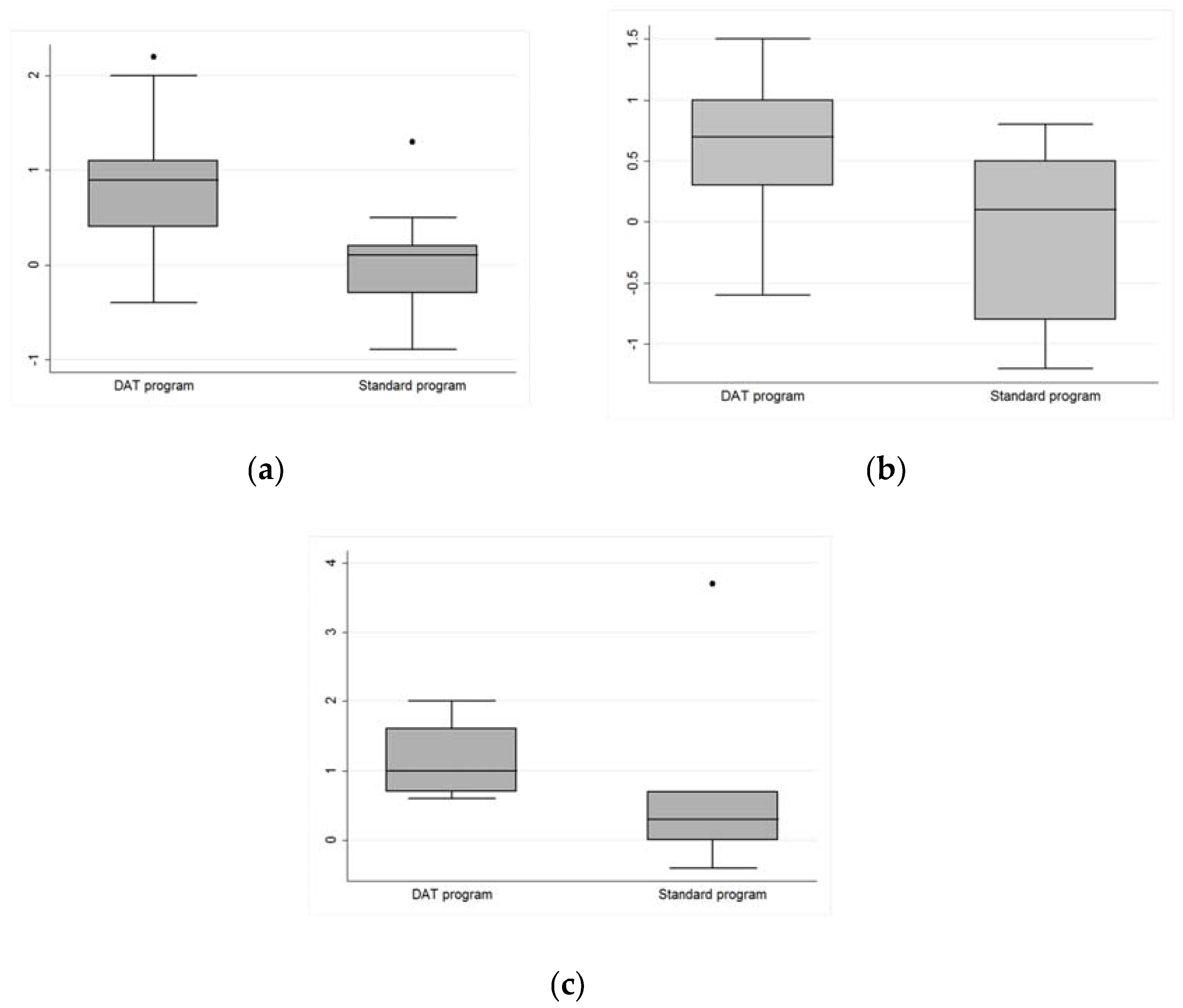
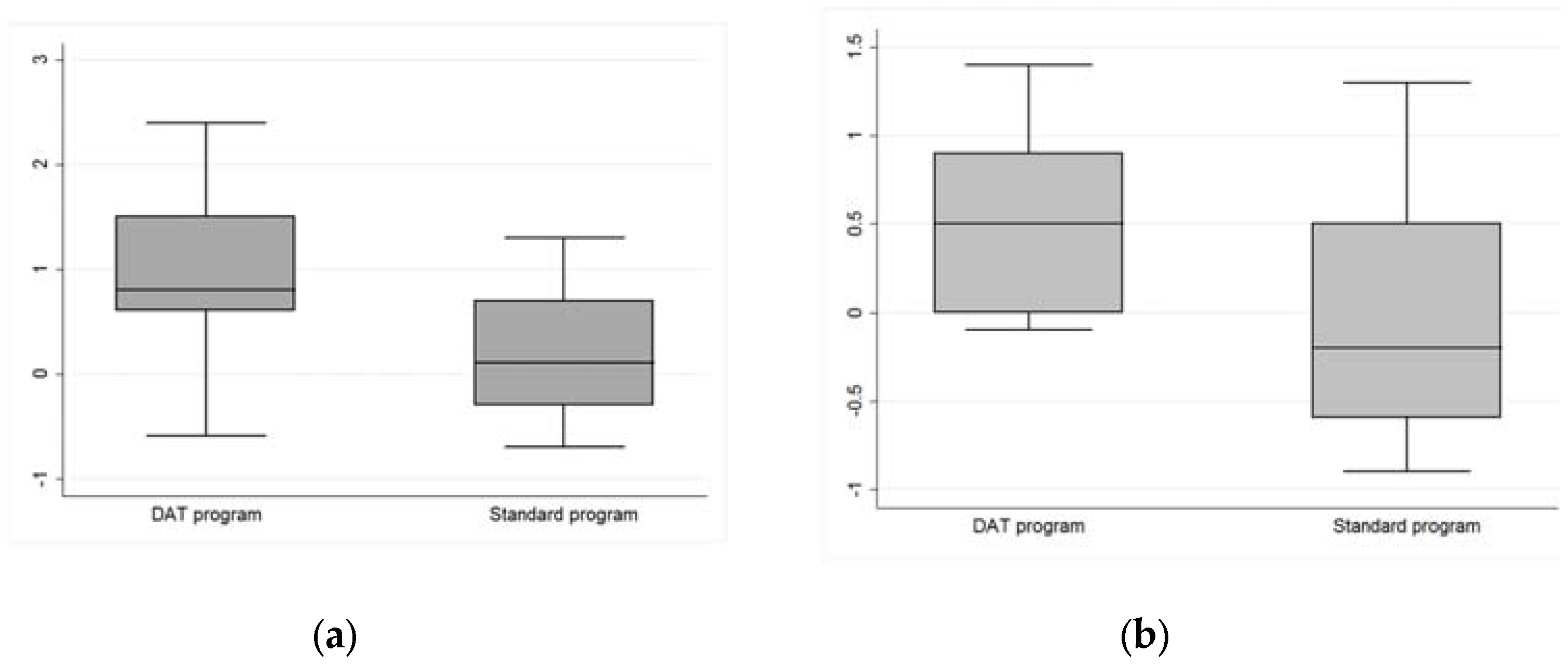
3.2. Test Analysis
4. Discussion
4.1. Functioning and Social Skills
4.2. Craving
4.3. Psychological Dysfunction Symptoms
4.4. Limitations of the Study
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mumola, C.; Karberg, J.C. Drug Use and Dependence, State and Federal Prisoners, 2004; U.S. Department of Justice, Office of Justice Programs, Bureau of Justice Statistics: Washington, DC, USA, 2007.
- Beaudette, J.N.; Power, J.; Stewart, L.A. National Prevalence of Mental Disorders among Incoming Federally Sentenced Men Offenders; Research Report, R-357; Correctional Service Canada: Ottawa, ON, Canada, 2015.
- WHO Regional Office for Europe. Available online: http://www.euro.who.int/prisons (accessed on 20 October 2016).
- Tangney, J.P.; Folk, J.B.; Graham, D.M.; Stuewig, J.B.; Blalock, D.V.; Salatino, A.; Blasko, B.L.; Moore, K.E. Changes in inmates’ substance use and dependence from pre-incarceration to one year post-release. J. Crim. Justice 2016, 46, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Kreek, J.; Nielsen, D.A.; Butelman, E.R.; LaForge, K.S. Genetic influences on impulsivity, risk taking, stress responsivity and vulnerability to drug abuse and addiction. Nat. Sci. 2005, 8, 1450–1457. [Google Scholar] [CrossRef] [PubMed]
- Kenny, J.P. Brain reward systems and compulsive drug use. Trend Pharmacol. Sci. 2007, 28, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Perry, J.L.; Carroll, M.E. The role of impulsive behavior in drug abuse. Psychopharmacology 2008, 200, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Conrod, J.P.; Nikolaou, K. Annual research review: On the developmental neuropsychology of substance use disorders. J. Child Psychol. Psychiatry 2016, 57, 371–394. [Google Scholar] [CrossRef] [PubMed]
- Everitt, B.J.; Robbins, T.W. Drug addiction: Updating actions to habit to compulsions ten years on. Annu. Rev. Psychol. 2016, 6, 23–50. [Google Scholar] [CrossRef] [PubMed]
- Gipson, C.D.; Kalivas, P.W. Neural basis of drug addiction. In Drug Abuse in Adolescence. Neurobiological, Cognitive and Psychological; De Micheli, D., Andrade, A.L.M., Da Silva, E.A., Eds.; Springer International Publishing: Berlin, Germany, 2016; pp. 37–56. [Google Scholar]
- Kopak, A.M.; Hoffmann, N.G.; Proctor, S.L. Key risk factors for relapse and rearrest among substance use treatment patients involved in the criminal justice system. Am. J. Crim. Justice 2016, 41, 14–30. [Google Scholar] [CrossRef]
- Kolind, T.; Duke, K. Drugs in prisons: Exploring use, control, treatment and policy. Drugs 2016, 23, 89–92. [Google Scholar] [CrossRef]
- Kanato, M. Drug use and health among prison inmates. Curr. Opin. Psychiatry 2008, 21, 252–254. [Google Scholar] [CrossRef] [PubMed]
- Beijersbergen, K.A.; Dirkzwager, A.J.; van der Laan, P.H.; Nieuwbeerta, P.A. Social building? Prison architecture and staff-prisoner relationships. Crime. Delinq. 2016, 62, 843–874. [Google Scholar] [CrossRef]
- Ireland, J.L.; Ireland, C.A.; Power, C.L. Attitudes towards prisoner-to-prisoner bullying and the association with prison environments: Examining the components. J. Aggress. Confl. Peace Res. 2016, 8, 124–136. [Google Scholar] [CrossRef]
- Chandler, R.K.; Fletcher, B.W.; Volkow, N.D. Treating drug abuse and addiction in the criminal justice system: Improving public health and safety. JAMA 2009, 301, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Hardy, M. Practitioner perspectives on risk: Using governmentality to understand contemporary probation practice. Eur. J. Criminol. 2013, 11, 303–318. [Google Scholar] [CrossRef]
- Galassi, A.; Mpofu, E.; Athanasou, J. Therapeutic community treatment of an inmate population with substance use disorders: Post-release trends in re-arrest, re-incarceration, and drug misuse relapse. Int. J. Environ. Res. Public Health 2015, 12, 7059–7072. [Google Scholar] [CrossRef] [PubMed]
- Baybutt, M.; Chemlal, K. Health-promoting prison: Theory to practice. Glob. Health Promot. 2016, 23, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Fournier, A.K.; Geller, E.S.; Fortney, E.V. Human-animal interaction in a prison setting: Impact on criminal behavior, treatment progress and social skills. Behav. Soc. Issues 2007, 16, 89–105. [Google Scholar] [CrossRef]
- Wesley, M.C.; Minatrea, N.B.; Watson, J.C. Animal-assisted therapy in the treatment of substance dependence. Anthrozoös 2009, 22, 137–148. [Google Scholar] [CrossRef]
- Kern-Godal, A.; Arnevik, E.A.; Walderhaug, E.; Ravndal, E. Substance use disorder treatment retention and completion: A prospective study of horse-assisted therapy (H.A.T.) for young adults. Addict. Sci. Clin. Pract. 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Allison, M.; Ramaswamy, M. Adapting animal-assisted therapy trials to prison-based animal programs. Public Health Nurs. 2016. [Google Scholar] [CrossRef] [PubMed]
- Mercer, J.; Gibson, K.; Clayton, D. The therapeutic potential of a prison-based animal programme in the UK. J. Forensic. Psychol. Pract. 2015, 17, 43–54. [Google Scholar] [CrossRef]
- Bachi, K. Equine-facilitated prison-based programs within the context of prison-based animal programs: State of science review. J. Offender Rehabil. 2013, 52, 46–74. [Google Scholar] [CrossRef]
- Turner, W.G. The experience of offenders in a prison canine program. Fed. Probat. 2007, 71, 38–43. [Google Scholar]
- Harkrader, T.; Burke, T.W.; Owen, S.S. Pound puppies: The rehabilitative uses of dogs in a correctional facility. Correct. Today 2004, 66, 74–79. [Google Scholar]
- Strimple, E.O. A history of prison inmate-animal interaction programs. Am. Behav. Sci. 2003, 47, 70–78. [Google Scholar] [CrossRef]
- Jasperson, R.A. Animal-assisted therapy with female inmates with mental illness: A case example from a pilot program. J. Offender Rehabil. 2010, 49, 417–433. [Google Scholar] [CrossRef]
- Burger, E.; Stetina, B.U.; Turner, K.; McElheney, J.; Handlos, U. Dog-assisted therapy in prison: Emotional competences and emotional status of drug-addicted criminal offenders. J. Vet. Behav. 2011, 6, 79–80. [Google Scholar] [CrossRef]
- Brigham, G.S.; Feaster, D.J.; Wakim, P.G.; Dempsey, C.L. Choosing a control group in effectiveness trials of behavioural drug abuse treatments. J. Subst. Abus. Treat 2009, 37, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Italian National Guidelines for Animal Assisted Interventions (A.A.I.). Agreement between the Italian Government, the Regional Authorities and the Autonomous Provinces of Trento and Bolzano. 2015. Available online: http://www.salute.gov.it/imgs/C_17_opuscoliPoster_276_allegato.pdf (accessed on 25 May 2017).
- Maslow, A.H. A theory of human motivation. Psychol. Rev. 1943, 50, 370–396. [Google Scholar] [CrossRef]
- Derogatis, L.R. Symptom Check List 90-R: Administration, Scoring and Procedures Manual, 3rd ed.; National Computer Systems: Minneapolis, MN, USA, 1994. [Google Scholar]
- Ignatyev, Y.; Fritsch, R.; Priebe, S.; Mundt, A.P. Psychometric properties of the symptom check-list-90-R in prison inmates. Psychiatry Res. 2016, 239, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, J.A. Mastering the Kennedy Axis V: A New Psychiatric Assessment of Patient Functioning; American Psychiatric Pub: Arlington, MA, USA, 2008. [Google Scholar]
- Moody, L.; Franck, C.; Bickel, W.K. Comorbid depression, antisocial personality, and substance dependence: Relationship with delay discounting. Drug Alcohol. Depend. 2016, 160, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Baltieri, D.A. Predictors of drug use in prison among women convicted of violent crimes. Crim. Behav. Ment. Health 2014, 24, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, G.A.; Rosenheck, R.A. Psychiatric correlates of past incarceration in the national co-morbidity study replication. Crim. Behav. Ment. Health 2014, 24, 18–35. [Google Scholar] [CrossRef] [PubMed]
- Compton, W.M.; Thomas, Y.F.; Stinson, F.S.; Grant, B.F. Prevalence, correlates, disability, and comorbidity of DSM-IV drug abuse and dependence in the United States: Results from the national epidemiologic survey on alcohol and related conditions. Arch. Gen. Psychiatry 2007, 64, 566–576. [Google Scholar] [CrossRef] [PubMed]
- Fazel, S.; Danesh, J. Serious mental disorder in 23,000 prisoners: A systematic review of 62 surveys. Lancet 2002, 359, 545–550. [Google Scholar] [CrossRef]
- Gori, A.; Ponti, L.; Tani, F.; Iraci Sareri, G.; Giannini, M.; Meringolo, P.; Craparo, G.; Bruschi, A.; Caretti, V.; Cacioppo, M.; et al. Psychopathological features in a sample of substance-abusing individuals with criminal history: Towards a definition of a personalita prototype of an “addict with criminal conduct”. Crim. Behav. Ment. Health 2016. [Google Scholar] [CrossRef] [PubMed]
- Boles, M.S.; Miotto, K. Substance abuse and violence: A review of the literature. Aggress. Violent Behav. 2003, 8, 155–174. [Google Scholar] [CrossRef]
- Lino, D.S.; Kelly, M.; Diamond, A. Human-animal interaction and the development of executive functions. In The Social Neuroscience of Human-Animal Interactions; Freund, S.L., McCune, S., Esposito, L., Gee, N.R., McCardle, P., Eds.; American Psychological Association: Washington, DC, 2016. [Google Scholar]
- Hall, S.S.; Wright, H.F.; Mills, D.S. What factors are associated with positive effects of dog ownership in families with children with autism spectrum disorder? The development of the Lincoln autism pet dog impact scale. PLoS ONE 2016, 11, e0149736. [Google Scholar] [CrossRef] [PubMed]
- Koda, N.; Watanabe, G.; Miyaji, Y.; Kuniyoshi, M.; Miyaji, C.; Hirata, T. Effects of dog-assisted intervention assessed by salivary cortisol concentrations in inmates of a Japanese prison. Asian. J. Criminol. 2016. [Google Scholar] [CrossRef]
- Koda, N.; Miyaji, Y.; Kuniyoshi, M.; Adachi, Y.; Watabe, G.; Miyaji, C.; Yamada, K. Effects of dog-assisted program in a Japanese Prison. Asian. J. Criminol. 2015. [Google Scholar] [CrossRef]
- Goldstein, R.Z.; Volkow, N.D. Drug addiction and its underlying neurobiological basis: Neuroimaging evidence for the involvement of the frontal cortex. Am. J. Psychiatry 2002, 159, 1642–1652. [Google Scholar] [CrossRef] [PubMed]
- Dalley, J.W.; Fryer, T.D.; Brichard, L.; Robinson, E.S.; Theobald, D.E.; Laane, K.; Pena, Y.; Murphy, E.R.; Shah, Y.; Probst, K.; et al. Nuclues accumbens D2/3 receptors predict trait impulsivity and cocaine reinforcement. Science 2007, 315, 1267–1270. [Google Scholar] [CrossRef] [PubMed]
- Field, M.; Wiers, R. Automatic and controlled processes in the pathway from drug abuse to addiction. In Drug Abuse and Addiction in Medical Illness. Causes, Consequences and Treatment; Verster, J., Brady, K., Galanter, M., Conrod, P., Eds.; Springer: New York, NY, USA, 2012; pp. 35–45. [Google Scholar]
- Grant, J.E.; Chamberlain, S.R. Impulsive action and impulsive choice across substance and behavioral addictions: Cause or consequence? Addict. Behav. 2014, 39, 1632–1639. [Google Scholar] [CrossRef] [PubMed]
- Verdejo-Garcia, A. Cognitive training for substance use disorders: Neuroscientific mechanisms. Neurosci. Biobehav. Rev. 2016, 68, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Rash, C.J.; Weinstock, J.; Van Patten, R. A review of gambling disorder and substance use disorders. Subst. Abus. Rehabil. 2016, 7, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Bernstein, M.H.; McSheffrey, S.N.; van den Berg, J.J.; Vela, E.J.; Stein, L.A.R.; Roberts, B.M.; Martin, R.A.; Clarke, J.G. The association between impulsivity and alcohol/drug use among prison inmates. Addict. Behav. 2015, 42, 140–143. [Google Scholar] [CrossRef] [PubMed]
- Evren, C.; Durkaya, M.; Evren, B.; Dalbudak, E.; Cetin, R. Relationship of relapse with impulsivity, novelty seeking and craving in male alcohol-dependent inpatients. Drug Alcohol. Rev. 2012, 31, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Hulka, L.M.; Vonmoos, M.; Preller, K.H.; Baumgartner, M.R.; Seifritz, E.; Gamma, A.; Quednow, B.B. Changes in cocaine consumption are associated with fluctuations in self-reported impulsivity and gambling decision-making. Psychol. Med. 2015, 45, 3097–3110. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.R.; Dobson, K.S. Cognitive behavioral treatment. In Comprehensive Handbook of Personality and Psychopathology; Amermann, R.T., Ed.; Wiley: Singapore, 2006; Volume 3, p. 487. [Google Scholar]
- Caprilli, S.; Messeri, A. Animal-assisted activity at a Meyer children’s hospital: A pilot study. Evid. Based Complement. Alternat. Med. 2006, 3, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.B.; Knisely, J.S.; Schubert, C.M.; Green, J.D.; Ameringer, S. The effect of an animal-assisted intervention on anxiety and pain in hospitalized children. Anthrozoös 2015, 28, 101–112. [Google Scholar] [CrossRef]
- Marr, C.A.; French, L.; Thompson, D.; Larry, D.; Greening, G.; Mormon, J.; Henderson, I.; Hughes, C.W. Animal-assisted therapy in psychiatric rehabilitation. Anthrozoös 2000, 13, 43–47. [Google Scholar] [CrossRef]
- Berget, B.; Ekeberg, Ø.; Pedersen, I.; Braastad, B.O. Animal-assisted therapy with farm animals for persons with psychiatric disorders: Effects on anxiety and depression, a randomized controlled trial. Occup. Ther. Ment. Health 2011, 27, 50–64. [Google Scholar] [CrossRef]
- Pedersen, I.; Martinsen, E.W.; Berget, B.; Braastad, B.O. Farm animal-assisted intervention for people with clinical depression: A randomized controlled study. Anthrozoös 2012, 25, 149–160. [Google Scholar] [CrossRef]
- Nuremberg, J.R.; Schleifer, S.J.; Shaffer, T.M.; Yellin, M.; Desai, P.J.; Amin, R.; Bouchard, A.; Montalvo, C. Animal-assisted therapy with chronic psychiatric inpatients: Equine-assisted psychotherapy and aggressive behavior. Psychiatr. Serv. Wash. 2015, 66, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Lynch, C.E.; Magann, E.F.; Barringer, S.N.; Ounpraseuth, S.T.; Eastham, D.G.; Lewis, S.D.; Stowe, Z.N. Pet therapy program for antepartum high-risk pregnancies: A pilot study. J. Perinatol. Off. J. Calif. Perinat. Assoc. 2014, 34, 816–818. [Google Scholar] [CrossRef] [PubMed]
- Le Roux, M.; Kemp, R. Effect of a companion dog on depression and anxiety levels of elderly residents in a long-term care facility. Psychogeriatrics 2009, 9, 23–26. [Google Scholar] [CrossRef]
- Moretti, F.; De Ronchi, D.; Bernabei, V.; Marchetti, L.; Ferrari, B.; Forlani, C.; Negretti, F.; Sacchetti, C.; Atti, A.R. Pet therapy in elderly with mental illness. Psychogeriatric 2011, 11, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Majic, T.; Gutzmann, H.; Heinz, A.; Lang, U.E.; Rapp, M.A. Animal-assisted therapy and agitation and depression in nursing home residents with dementia: A matched case-control trial. Am. J. Geriatr. Psychiatry 2013, 21, 1052–1059. [Google Scholar] [CrossRef] [PubMed]
- Serpell, J. the Company of Animals. A Study of Human-Animal Relationships; Cambridge University Press: Cambridge, UK, 1996. [Google Scholar]
- Enders-Slegers, M.J. The meaning of companion animals: Qualitative analysis of the life histories of elderly cat and dog owners. In Companion Animals and U.S.; Podberscek, A.L., Paul, E.S., Serpell, J.A., Eds.; Cambridge University Press: Cambridge, UK, 2000; pp. 237–256. [Google Scholar]
- Wells, D.L. The effects of animals on human health and well-being. J. Soc. Issues 2009, 65, 523–543. [Google Scholar] [CrossRef]
- Pachana, N.A.; Massavelli, B.M.; Robleda-Gomez, S. A developmental psychological perspective on the human-animal bond. In The Psychology of the Human-Animal Bond; Blazina, C., Boyra, G., Shen-Miller, D., Eds.; Springer: New York, NY, USA, 2011; pp. 151–165. [Google Scholar]
- Kern-Godal, A.; Brenna, I.H.; Kogstad, N.; Arnevik, E.A.; Ravndal, E. Contribution of the patient-horse relationship to substance use disorder treatment: Patients’ experiences. Int. J. Qual. Stud. Health Well-being 2016, 11, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Kern-Godal, A.; Brenna, I.H.; Arnevik, E.A.; Ravndal, E. More than just a break from treatment: How substance use disorder patients experience the stable environment in horse-assisted therapy. Subst. Abus. 2016, 10, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Hauge, H.; Kvalem, I.L.; Berget, B.; Enders-Slegers, M.J.; Braastad, B.O. Equine-assisted activities and the impact on perceived social support, self-esteem and self-efficacy among adolescent—An intervention study. Int. J. Adolesc. Youth 2014, 19, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Maujean, A.; Pepping, A.C.; Kendall, E. A systematic review of randomized controlled trials of animal-assisted therapy on psychosocial outcomes. Anthrozoös 2015, 28, 23–36. [Google Scholar] [CrossRef]
- Maber-Aleksandrowicz, S.; Avent, C.; Hassiotis, A. A systematic review of animal-assisted therapy on psychosocial outcomes in people with intellectual disability. Res. Dev. Disabil. 2016, 49, 322–338. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Wills, T.A. Stress, social support, and the buffering hypothesis. Psychol. Bull. 1985, 98, 310. [Google Scholar] [CrossRef]
- Coyne, J.C.; Downey, G. Social factors and psychopathology: Stress, social support, and coping processes. Annu. Rev. Psychol. 1991, 42, 401–425. [Google Scholar] [CrossRef] [PubMed]
- Bettis, A.H.; Forehand, R.; McKee, L.; Dunbar, J.P.; Watson, K.H.; Compas, B.E. Testing specificity: Associations of stress and coping with symptoms of anxiety and depression in youth. J. Child Fam. Stud. 2016, 25, 949–958. [Google Scholar] [CrossRef]
- Roohafza, H.; Feizi, A.; Afshar, H.; Mazaheri, M.; Behnamfar, O.; Hassanzadeh-Keshteli, A.; Adibi, P. Path analysis of relationship among personality, perceived stress, coping, social support and psychological outcomes. World Psychiatry 2016, 6, 248–256. [Google Scholar] [CrossRef] [PubMed]
- Stern, C.; Chur-Hansen, A. Methodological considerations in designing and evaluating animal-assisted interventions. Animals 2013, 3, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Kazdin, A.E. Establishing the effectiveness of animal-assisted therapies: Methodological standards, issues and strategies. In How Animals Affect Us: Examining the Influence of Human-Animal Interactions on Child Development and Human Health; McCardle, P., McCune, S., Griffin, S.J.A., Maholmes, V., Eds.; American Psychological Association: Washington, DC, 2011; pp. 35–51. [Google Scholar]
- Shean, G. Limitations of randomized control designs in psychotherapy research. Adv. Psychiatry, 2014. [Google Scholar] [CrossRef]
- Shadish, W.R.; Clark, M.H.; Steiner, P.M. Can nonrandomized experiments yield accurate answers? A randomized experiment comparing random and nonrandom assignments. JASA 2008, 103, 1334–1356. [Google Scholar] [CrossRef]
| Demographic Characteristics | Dog Assisted Therapy (D.A.T.) Program (n = 12) | Standard Program (n = 10) |
|---|---|---|
| Age (mean ± SD in years) | 35.5 ± 13.83 | 42.9 ± 9.1 |
| Primary School Education (%) | 22.2 | 0 |
| Secondary School Education (%) | 77.8 | 88.9 |
| High School Education (%) | 0 | 11.1 |
| Degree or PhD (%) | 0 | 0 |
| Prison Stay (median in months) | 12 | 12 |
| Pharmacological treatment (%) | 77.7 | 55.5 |
| Variable | Mean ± SD | t-Student | |
|---|---|---|---|
| T0 | T1 | p-Value | |
| Somatization | 0.98 ± 0.89 | 0.21 ± 0.24 | 0.0227 |
| Obsessive-compulsive symptoms | 1.07 ± 0.61 | 0.46 ± 0.29 | 0.0102 |
| Interpersonal sensitivity | 0.60 ± 0.59 | 0.23 ± 0.24 | 0.1754 * |
| Depression | 1.34 ± 0.84 | 0.45 ± 0.32 | 0.0139 |
| Anxiety | 1.39 ± 0.95 | 0.44 ± 0.35 | 0.0130 |
| Hostility | 0.57 ± 0.58 | 0.43 ± 0.36 | 0.4153 * |
| Phobic anxiety | 0.46 ± 0.55 | 0.06 ± 0.07 | 0.0703 * |
| Paranoid ideation | 1.17 ± 0.72 | 0.54 ± 0.49 | 0.0175 |
| Psychoticism | 0.73 ± 0.62 | 0.19 ± 0.16 | 0.0173 |
| Sleep disorders | 1.78 ± 0.53 | 0.63 ± 0.59 | 0.0002 |
| Global severity index | 1.01 ± 0.54 | 0.35 ± 0.19 | 0.0056 |
| Variable | Mean ± SD | Wilcoxon Signed-Rank Test | |
|---|---|---|---|
| T0 | T1 | p-Value | |
| Somatization | 1.17 ± 1.30 | 0.65 ± 0.74 | 0.2207 |
| Obsessive-compulsive symptoms | 1.37 ± 1.05 | 0.83 ± 0.53 | 0.2026 |
| Interpersonal sensitivity | 0.70 ± 0.51 | 0.52 ± 0.55 | 0.5403 |
| Depression | 1.10 ± 0.77 | 0.83 ± 0.48 | 0.6101 |
| Anxiety | 1.07 ± 0.83 | 0.73 ± 0.41 | 0.3580 |
| Hostility | 0.67 ± 0.75 | 0.53 ± 0.54 | 0.7208 |
| Phobic anxiety | 0.82 ± 1.40 | 0.35 ± 0.51 | 0.2578 |
| Paranoid ideation | 0.86 ± 0.74 | 0.83 ± 0.59 | 0.9188 |
| Psychoticism | 0.84 ± 0.77 | 0.66 ± 0.41 | 0.6831 |
| Sleep disorders | 1.89 ± 1.47 | 1.00 ± 1.02 | 0.0411 * |
| Global severity index | 1.00 ± 0.82 | 0.67 ± 0.43 | 0.5400 |
| Variable | Mean ± SD | Wilcoxon Signed-Rank Test | |
|---|---|---|---|
| D.A.T. Group | Control Group | p-Value | |
| Somatization | 0.78 ± 0.78 | 0.29 ± 0.92 | 0.2152 |
| Obsessive-compulsive symptoms | 0.61 ± 0.55 | 0.26 ± 0.77 | 0.2881 |
| Interpersonal sensitivity | 0.36 ± 0.71 | 0.07 ± 0.75 | 0.5346 |
| Depression | 0.89 ± 0.84 | 0.02 ± 0.65 | 0.0468 * |
| Anxiety | 0.94 ± 0.89 | 0.16 ± 0.73 | 0.0562 ** |
| Hostility | 0.17 ± 0.50 | −0.04 ± 0.66 | 0.4772 |
| Phobic anxiety | 0.37 ± 0.56 | 0.32 ± 1.31 | 0.1133 |
| Paranoid ideation | 0.64 ± 0.62 | −0.13 ± 0.76 | 0.0272 * |
| Psychoticism | 0.54 ± 0.55 | 0.02 ± 0.78 | 0.0926 ** |
| Sleep disorders | 1.13 ± 0.51 | 0.62 ± 1.22 | 0.0201 * |
| Global severity index | 0.76 ± 0.45 | 0.12 ± 0.67 | 0.0627 ** |
| Variable | Mean ± SD | Wilcoxon Signed-Rank Test | |
|---|---|---|---|
| T0 | T1 | p-Value | |
| Psychological impairment | 60.42 ± 7.22 | 69.17 ± 8.48 | 0.0017 |
| Social skills | 58.75 ± 10.25 | 68.33 ± 12.85 | 0.0018 |
| Violence | 62.92 ± 10.10 | 70.42 ± 10.97 | 0.0021 |
| Occupational skills | 64.17 ± 9.96 | 70.83 ± 12.94 | 0.0027 |
| Substance craving | 59.17 ± 13.29 | 67.50 ± 13.57 | 0.0042 |
| Medical impairment | 83.33 ± 10.73 | 82.92 ± 9.16 | 0.6376 * |
| Ancillary impairment | 60.00 ± 8.53 | 65.83 ± 8.75 | 0.0026 |
| Variable | Mean ± SD | Wilcoxon Signed-Rank Test | |
|---|---|---|---|
| T0 | T1 | p-Value | |
| Psychological impairment | 63.75 ± 8.82 | 69.17 ± 10.62 | 0.0058 |
| Social skills | 64.17 ± 9.00 | 68.33 ± 11.15 | 0.0115 |
| Violence | 72.50 ± 14.22 | 76.25 ± 12.64 | 0.0261 |
| Occupational skills | 65.83 ± 13.11 | 69.58 ± 14.37 | 0.0052 |
| Substance craving | 62.50 ± 9.65 | 66.67 ± 10.08 | 0.0056 |
| Medical impairment | 72.08 ± 11.57 | 77.08 ± 12.69 | 0.0092 |
| Ancillary impairment | 62.08 ± 8.65 | 66.67 ± 10.73 | 0.0111 |
| Variable | Mean ± SD | t-Student | |
|---|---|---|---|
| D.A.T. Group | Control Group | p-Value | |
| Psychological impairment | 8.75 ± 3.11 | 5.42 ± 4.50 | 0.0464 * |
| Social skills | 9.58 ± 4.50 | 4.17 ± 4.17 | 0.0058 * |
| Violence | 7.50 ± 5.00 | 3.75 ± 4.83 | 0.0750 |
| Occupational skills | 6.67 ± 5.36 | 3.75 ± 3.11 | 0.1174 |
| Substance craving | 2.08 ± 5.82 | 4.17 ± 3.59 | 0.3028 |
| Medical impairment | −0.42 ± 6.89 | 5.00 ± 4.77 | 0.0356 |
| Ancillary impairment | 5.83 ± 3.59 | 4.58 ± 4.50 | 0.4599 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Contalbrigo, L.; De Santis, M.; Toson, M.; Montanaro, M.; Farina, L.; Costa, A.; Nava, F.A. The Efficacy of Dog Assisted Therapy in Detained Drug Users: A Pilot Study in an Italian Attenuated Custody Institute. Int. J. Environ. Res. Public Health 2017, 14, 683. https://doi.org/10.3390/ijerph14070683
Contalbrigo L, De Santis M, Toson M, Montanaro M, Farina L, Costa A, Nava FA. The Efficacy of Dog Assisted Therapy in Detained Drug Users: A Pilot Study in an Italian Attenuated Custody Institute. International Journal of Environmental Research and Public Health. 2017; 14(7):683. https://doi.org/10.3390/ijerph14070683
Chicago/Turabian StyleContalbrigo, Laura, Marta De Santis, Marica Toson, Maria Montanaro, Luca Farina, Aldo Costa, and Felice Alfonso Nava. 2017. "The Efficacy of Dog Assisted Therapy in Detained Drug Users: A Pilot Study in an Italian Attenuated Custody Institute" International Journal of Environmental Research and Public Health 14, no. 7: 683. https://doi.org/10.3390/ijerph14070683
APA StyleContalbrigo, L., De Santis, M., Toson, M., Montanaro, M., Farina, L., Costa, A., & Nava, F. A. (2017). The Efficacy of Dog Assisted Therapy in Detained Drug Users: A Pilot Study in an Italian Attenuated Custody Institute. International Journal of Environmental Research and Public Health, 14(7), 683. https://doi.org/10.3390/ijerph14070683





