Validity of Research-Grade Actigraphy Unit for Measuring Exercise Intensity
Abstract
:1. Introduction
2. Literature Review
2.1. Actigraphy Devices
2.2. Metabolic Heat (MH)
3. Methodology
3.1. Research Design
3.2. Instruments and Data Collection
3.3. Participants
4. Results
4.1. Descriptive Statistics of Sample Distribution
4.2. Analyses of Variance
4.3. Correlation Analysis
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Barton, J.; Hine, R.; Pretty, J. The health benefits of walking in green spaces of high natural and heritage value. J. Integr. Environ. Sci. 2009, 6, 261–278. [Google Scholar] [CrossRef]
- Pretty, J.; Peacock, J.; Sellengs, M.; Griffen, M. The mental and physical health outcomes of green exercise. Int. J. Environ. Health Res. 2005, 15, 319–337. [Google Scholar] [CrossRef] [PubMed]
- Hallman, D.M.; Sato, T.; Kristiansen, J.; Gupta, N.; Skotte, J.; Holtermann, A. Prolonged sitting is associated with attenuated heart rate variability during sleep in blue-collar workers. Int. J. Environ. Res. Public Health 2015, 12, 14811–14827. [Google Scholar] [CrossRef] [PubMed]
- DCMS. Game Plan: A Strategy for Delivering Government’s Sport and Physical Activity Objectives; Department of Culture, Media and Sport and Cabinet Office: London, UK, 2002.
- Sports Affair Council, Executive Yuan. Sport City Survey; Sports Affair Council, Executive Yuan: Taipei, Taiwan, 2010.
- World Health Organization. The World Health Report 2002-Reducing Risks, Promoting Healthy Life; World Health Organization: Geneva, Switzerland, 2011; p. 61. [Google Scholar]
- Lai, C.T.; Su, C.H. The differences of physical and mental health on male collegiate students at the different levels of active life. J. Phys. Educ. Sport Sci. 2009, 9, 43–50. [Google Scholar]
- Walker, R.K.; Hickey, A.M.; Freedson, P.S. Advantages and limitations of wearable activity trackers: Considerations for patients and clinicians. Clin. J. Oncol. Nurs. 2016, 20, 606–610. [Google Scholar] [CrossRef] [PubMed]
- Calkins, M.; Szmerekovsky, J.G.; Biddle, S. Effect of increased time spent outdoors on individuals with dementia residing in nursing homes. J. Hous. Elder. 2007, 21, 211–228. [Google Scholar] [CrossRef]
- Rapport, M.D.; Kofler, M.J.; Himmerich, C. Activity measurement. In Clinician’s Handbook of Child Behavioral Assessment; Hersen, M., Ed.; Elsevier Academic Press: New York, NY, USA, 2006; pp. 125–157. [Google Scholar]
- Diouf, A.; Thiam, M.; Idohou-Dossou, N.; Diongue, O.; Mégné, N.; Diallo, K.; Sembène, P.M.; Wade, S. Physical activity level and sedentary behaviors among public school children in Dakar (Senegal) measured by PAQ-C and accelerometer: Preliminary results. Int. J. Environ. Res. Public Health 2016, 13, 998. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Lee, P.H.; Macfarlane, D. Associations between moderate-to-vigorous physical activity and neighbourhood recreational facilities: The features of the facilities matter. Int. J. Environ. Res. Public Health 2014, 11, 12594–12610. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.J.; Baranowski, T.; Lau, P.W.C.; Chen, T.A.; Zhang, S.G. Psychological correlates of self-reported and objectively measured physical activity among Chinese children—Sychological correlates of PA. Int. J. Environ. Res. Public Health 2016, 13, 1006. [Google Scholar] [CrossRef] [PubMed]
- Sadeh, A.; Hauri, P.J.; Kripke, D.F.; Lavie, P. An American sleep disorders association review: The role of actigraphy in the evaluation of sleep disorders. Sleep 1995, 18, 288–302. [Google Scholar] [CrossRef] [PubMed]
- Tryon, W.W. Actigraphy Measurement in Psychology and Medicine; Plenum Press: New York, NY, USA, 1991. [Google Scholar]
- Ancoli-Israel, S.; Cole, R.; Alessi, C.; Chambers, M.; Moorcroft, W.; Pollak, C. The role of actigraphy in the study of sleep and circadian rhythms. Sleep 2003, 26, 342–392. [Google Scholar] [CrossRef] [PubMed]
- Stone, K.L.; Ancoli-Israel, S. Actigraphy. In Principles and Practice of Sleep Medicine; Kryger, M., Roth, T., Dement, W., Eds.; Elsevier: New York, NY, USA, 2011; pp. 1668–1675. [Google Scholar]
- Sadeh, A. Actigraphy. In Clinical Neurophysiology of Sleep Disorders Handbook of Clinical Neurophysiology; Guilleminault, C., Ed.; Elsevier: New York, NY, USA, 2005; pp. 67–72. [Google Scholar]
- Sadeh, A. The role and validity of actigraphy in sleep medicine: An update. Sleep Med. Rev. 2011, 15, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Tryon, W.W. Human activity: A review of quantitative findings. In Behavioral Assessment in Behavioral Medicine; Tryon, W.W., Ed.; Springer: New York, NY, USA, 1985; pp. 257–299. [Google Scholar]
- Cook, T.D.; Campbell, D.T. Quasi-Experimentation: Design & Analysis issues for Field Settings; Houghton Mifflin: Boston, MA, USA, 1979. [Google Scholar]
- Evenson, K.R.; Goto, M.M.; Furberg, R.D. Systematic review of the validity and reliability of consumer-wearable activity trackers. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 159. [Google Scholar] [CrossRef] [PubMed]
- Cuberek, R.; Ansari, W.E.; Frömel, K.; Skalik, K.; Sigmund, E.A. Comparison of two motion sensors for the assessment of free-living physical activity of adolescents. Int. J. Environ. Res. Public Health 2010, 7, 1558–1576. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, T.; Rowlands, A.V.; Olds, T.; Maher, C. The validity of consumer-level, activity monitors in healthy adults worn in free-living conditions: A cross-sectional study. Int. J. Behav. Nutr. Phys. Act. 2015, 12, 42. [Google Scholar]
- Kaplan, R.M.; Saccuzzo, D.P. Psychological Testing: Principles, Applications, and Issues; Brooks/Cole Publishing: Pacific Grove, CA, USA, 1989. [Google Scholar]
- Sports Administration, Ministry of Education. The 102 Year Sports Statistics of the Republic of China; Sports Administration, Ministry of Education: Taipei, Taiwan, 2013.
- Grap, M.J.; Hamilton, V.A.; McNallen, A.; Ketchum, J.M.; Best, A.M.; Arief, N.Y.I.; Wetzel, P.A. Actigraphy: Analyzing patient movement. Heart Lung 2011, 40, 52–59. [Google Scholar] [CrossRef] [PubMed]
- Hersen, M. Clinician’s Handbook of Child Behavioral Assessment; Elsevier Academic Press: New York, NY, USA, 2005. [Google Scholar]
- Rapport, M.D.; Bolden, J.; Kofler, M.J.; Sarver, D.E.; Raiker, J.S.; Alderson, R.M. Hyperactivity in boys with attention-deficit/hyperactivity disorder (ADHD): A ubiquitous core symptom or manifestation of working memory deficits? J. Abnorm. Child Psychol. 2009, 37, 521–534. [Google Scholar] [CrossRef] [PubMed]
- De Souza, L.; Benedito-Silva, A.A.; Pires, M.L.; Poyares, D.; Tufik, S.; Calil, H.M. Further validation of actigraphy for sleep studies. Sleep 2003, 26, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Welk, G.J.; Nam, Y.H.; Lee, J.A.; Lee, J.M.; Kim, Y.; Meier, N.F.; Dixon, P.M. Comparison of consumer and research monitors under semistructured settings. Med. Sci. Sports Exerc. 2016, 48, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Kooiman, T.J.M.; Dontje, M.L.; Sprenger, S.R.; Krijnen, W.P.; van der Schans, C.P.; de Groot, M. Reliability and validity of ten consumer activity trackers. Sports Sci. Med. Rehabil. 2015, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.C.; Smolensky, M.H.; D’Alonzo, G.E.; Redman, D.P. Actigraphy: A means of assessing circadian patterns in human activity. Chronobiol. Int. 1990, 7, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.J.; Kripke, D.F.; Gruen, W.; Mullaney, D.J.; Gillin, J.C. Automatic sleep/wake identification from wrist activity. Sleep 1992, 15, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Miyagishi, T.; Sack, R.L.; Hughes, R.J.; Blood, M.L.; Lewy, A.J. Evaluation of the Actillume wrist actigraphy monitor in the detection of sleeping and waking. Psychiatry Clin. Neurosci. 1998, 52, 160–161. [Google Scholar] [CrossRef] [PubMed]
- Grap, M.J.; Borchers, C.T.; Munro, C.L.; Elswick, R.K., Jr.; Sessler, C.N. Actigraphy in the critically ill: Correlation with activity, agitation, and sedation. Am. J. Crit. Care 2005, 14, 52–60. [Google Scholar] [PubMed]
- Janssen, X.; Cliff, D.P.; Reilly, J.J.; Hinkley, T.; Jones, R.A.; Batterham, M.; Ekelund, U.; Brage, S.; Okely, A.D. Predictive validity and classification accuracy of ActiGraph energy expenditure equations and cut-points in young children. PLoS ONE 2013, 8, e79124. [Google Scholar] [CrossRef] [PubMed]
- Abel, M.G.; Hannon, J.C.; Sell, K.; Lillie, T.; Conlin, G.; Anderson, D. Validation of the Kenz Lifecorder EX and ActiGraph GT1M accelerometers for walking and running in adults. Appl. Physiol. Nutr. Metab. 2008, 33, 1155–1164. [Google Scholar] [CrossRef] [PubMed]
- Gusmer, R.J.; Bosch, T.A.; Watkins, A.N.; Ostrem, J.D.; Dengel, D.R. Comparison of FitBit® Ultra to ActiGraph™ GT1M for assessment of physical activity in young adults during treadmill walking. J. Open Sports Med. 2014, 8, 11–15. [Google Scholar] [CrossRef]
- Kreider, J.F.; Curtiss, P.S.; Rabl, A. Heating and Cooling of Buildings: Design for Efficiency, revised 2nd ed.; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- DuBois, D.; DuBois, E.F. A formula to estimate the approximate surface area if height and weight be known. Arch. Intern. Med. 1916, 17, 863–871. [Google Scholar] [CrossRef]
- Lin, C.H. Establishment of a Database and Calculation Formula for Human Body Surface Areas, Hsinchu. Ph.D. Thesis, Department of Industrial Engineering and Engineering Management, National Tsing Hua University, Hsinchu, Taiwan, 2010. [Google Scholar]
- Buchowski, M.S. Doubly labeled water is a validated and verified reference standard in nutrition research. J. Nutr. 2014, 144, 573–574. [Google Scholar] [CrossRef] [PubMed]
- Kaiyala, K.J.; Ramsay, D.S. Direct animal calorimetry, the underused gold standard for quantifying the fire of life. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2011, 158, 252–264. [Google Scholar] [CrossRef] [PubMed]
- Shadish, W.R.; Cook, T.D.; Campbell, D.T. Experimental and Quasi-Experimental Designs for Generalized Causal Inference; Houghton Mifflin: Boston, MA, USA, 2002. [Google Scholar]
- Passmore, R.; Durnin, J.V.G.A. Human energy expenditure. Physiol. Rev. 1955, 35, 801–840. [Google Scholar] [PubMed]
- Torres, N. Accuracy of the RT3, BioTrainer-Pro, and SensWear Armband to Measure Energy Cost of Treadmill Walking and Running. Unpublished Master’s Thesis, The University of Texas at El Paso, El Paso, TX, USA, 2003. [Google Scholar]
- Reece, J.D. Reduce Your Sit and be More Fit: An Examination of Sedentary Behavior. Unpublished Ph.D. Thesis, Middle Tennessee State University, Murfreesboro, TN, USA, 2013. [Google Scholar]
- Bowler, D.E.; Buyung-Ali, L.M.; Knight, T.M.; Pullin, A.S. A systematic review of evidence for the added benefits to health of exposure to natural environments. BMC Public Health 2010, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Xia, H. Quora. Available online: https://www.quora.com/How-accurate-is-the-Nike+-iPhone-app-in-measuring-distance (accessed on 7 March 2017).
- Johansson, M.; Hartig, T.; Staats, H. Psychological benefits of walking: Moderation by company and outdoor environment. Appl. Psychol. Health Well Being 2011, 3, 261–280. [Google Scholar] [CrossRef]
- Canadian Society for Exercise Physiology. The Physical Activity Readiness Questionnaire, PAR-Q; Canadian Society for Exercise Physiology: Ottawa, ON, CA, 2002. [Google Scholar]
- Wu, M.L.; Chang, Y.R. SPSS and Statistical Analysis Application; Wunan Press: Taipei, Taiwan, 2011. [Google Scholar]
- Huck, S.W. Reading Statistics and Research; Pearson Education, Inc.: Boston, MA, USA, 2012. [Google Scholar]
- Fjortoft, I.; Kristofersen, B.; Sageie, J. Children in schoolyards: Tracking movement patterns and physical activity in schoolyards using global positioning system and heart rate monitoring. Landsc. Urban Plan. 2009, 93, 210–217. [Google Scholar] [CrossRef]
- Tang, J.W.; Brown, R.D. The effect of viewing a landscape on physiological health of elderly women. J. Hous. Elder. 2005, 19, 187–202. [Google Scholar] [CrossRef]
- Bushman, B.A. How can I use METS to quantify the amount of aerobic exercise? ACSMS Health Fit. J. 2012, 16, 5–7. [Google Scholar] [CrossRef]
- Zhang, K.; Pi-Sunyer, F.X.; Boozer, C.N. Improving energy expenditure estimation for physical activity. Med. Sci. Sports Exerc. 2004, 36, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Bouchard, C.; Shephard, R.J. Physical activity, fitness, and health: The model and key concepts. In Physical Activity, Fitness, and Health-International Proceedings and Consensus Statement; Bouchard, C., Shephard, R.J., Stephens, T., Eds.; Human Kinetics Publishers: Champaign, IL, USA, 1994; pp. 77–88. [Google Scholar]

| Movement Speed (m/s) | Metabolic Rate (W/m2) | Note |
|---|---|---|
| 0.89 | 115 | 115 W/m2 for <0.89 m/s |
| 1.34 | 150 | 150 W/m2 for 0.90–1.34 m/s |
| 1.79 | 220 | 220 W/m2 for 1.35–1.79 m/s |
| 1.80 | 255 | 255 W/m2 for >1.80 m/s |
| Background | Classification | Number of Participants (n = 116) | Percentage (%) |
|---|---|---|---|
| Sex | Male | 52 | 44.8 |
| Female | 64 | 55.2 | |
| Age | 18–20 years | 45 | 38.8 |
| 21–24 years | 70 | 60.3 | |
| 25–30 years | 1 | 0.9 | |
| >31 years | 0 | 0 | |
| Height | 141–150 cm | 6 | 5.2 |
| 151–160 cm | 29 | 24.9 | |
| 161–170 cm | 48 | 42.3 | |
| 171–180 cm | 27 | 23.2 | |
| >181 cm | 5 | 4.4 | |
| Weight | 40–50 kg | 26 | 22.4 |
| 51–60 kg | 56 | 48.1 | |
| 61–70 kg | 24 | 20.7 | |
| 71–80 kg | 7 | 6.1 | |
| 81–90 kg | 2 | 1.8 | |
| >91 kg | 1 | 0.9 | |
| College | Engineering | 0 | 0 |
| Management | 5 | 4.3 | |
| Electrical engineering & computer science | 27 | 23.3 | |
| Humanities and creativity | 84 | 72.4 | |
| Year | Freshman | 17 | 14.6 |
| Sophomore | 43 | 37.1 | |
| Junior | 50 | 43.1 | |
| Senior | 5 | 4.3 | |
| Graduate | 1 | 0.9 |
| Exercise Type | Number of Participants | Minimum (m/s) | Maximum (m/s) | Mean (m/s) | Standard Deviation (m/s) | Skewness | Kurtosis |
|---|---|---|---|---|---|---|---|
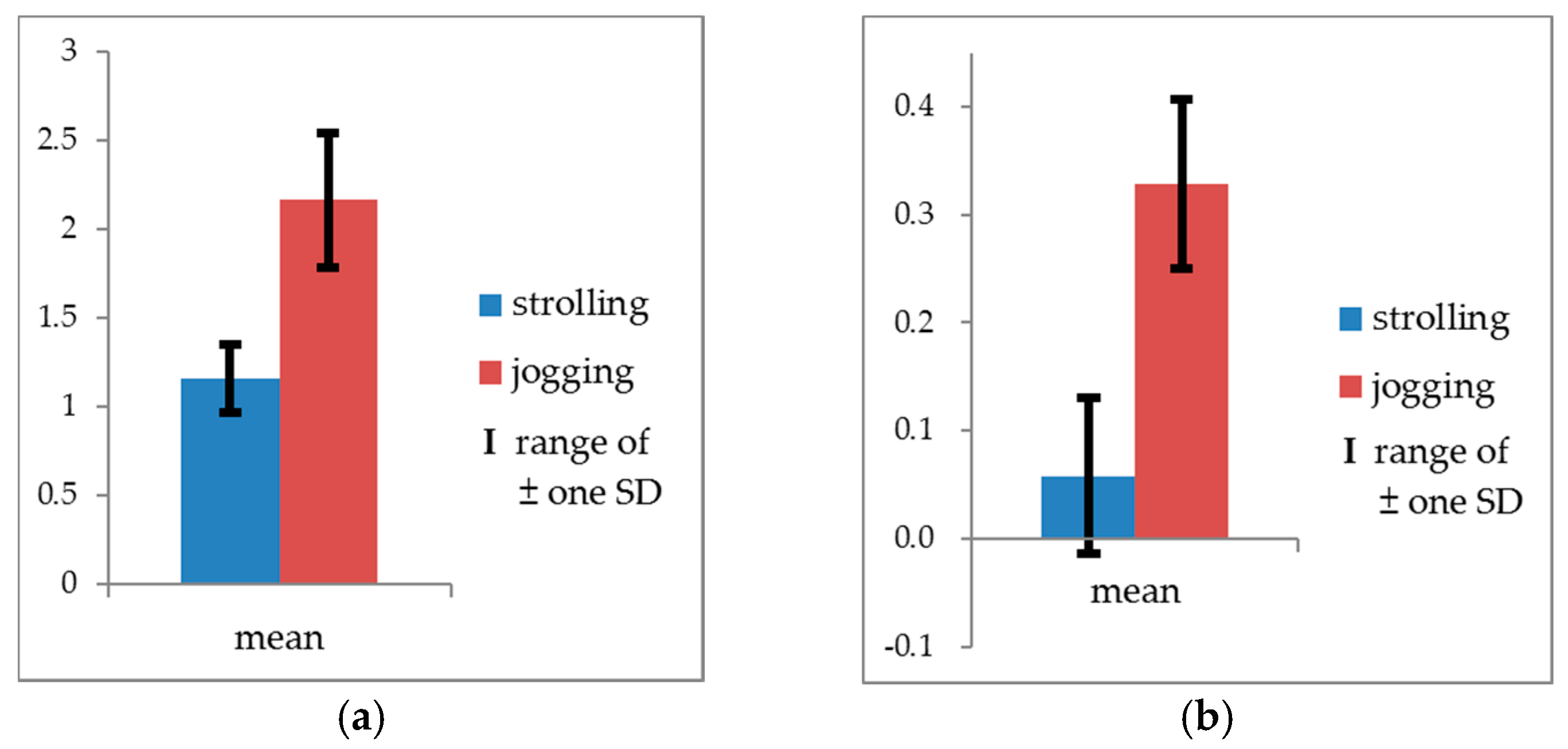
| Strolling | 61 | 0.77 | 1.69 | 1.16 | 0.19 | 0.29 | 0.46 |
| Jogging | 55 | 1.54 | 3.03 | 2.17 | 0.38 | 0.04 | −0.85 |
| Dependent Variable | MANOVA (Overall Test) | ANOVA (Follow-Up Tests) | |||||
|---|---|---|---|---|---|---|---|
| F | p | Pillai’s Trace (V) | F | p | Partial Eta Squared () | Post-Hoc Tests | |
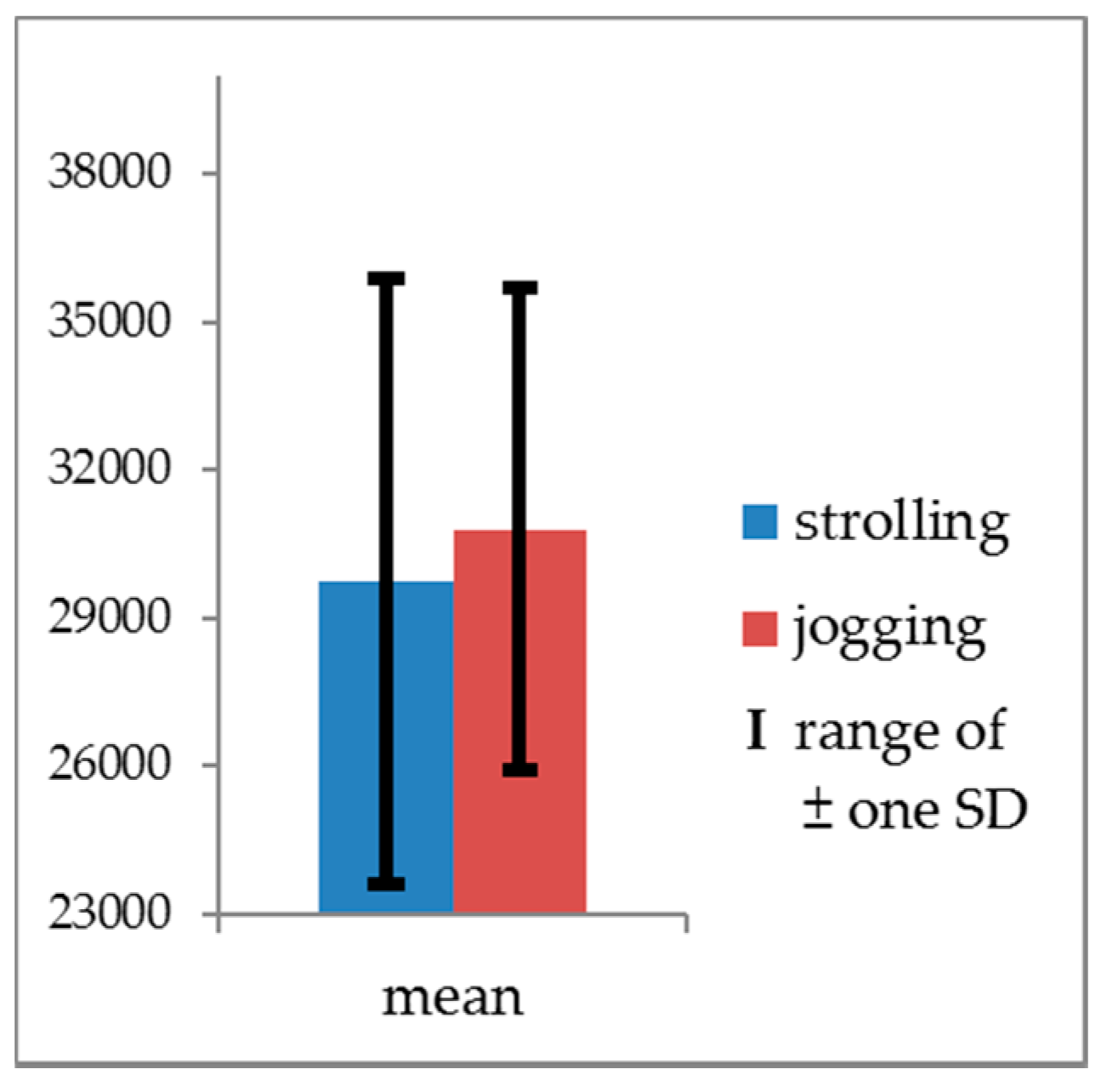
| Actigraph readings | 190.257 *** | <0.001 | 0.771 | 0.995 | 0.321 | 0.009 | N/A |
| Movement speeds | 376.635 *** | <0.001 | 0.768 | Jogging > Strolling | |||
| Variable | Actigraph Readings | Movement Speeds | ||||
|---|---|---|---|---|---|---|
| Strollers | Joggers | Whole | Strollers | Joggers | Whole | |
| (n = 61) | (n = 55) | (n = 116) | (n = 61) | (n = 55) | (n = 116) | |
| Movement speeds | −0.179 | 0.002 | 0.047 | N/A | N/A | N/A |
| (p = 0.167) | (p = 0.990) | (p = 0.614) | ||||
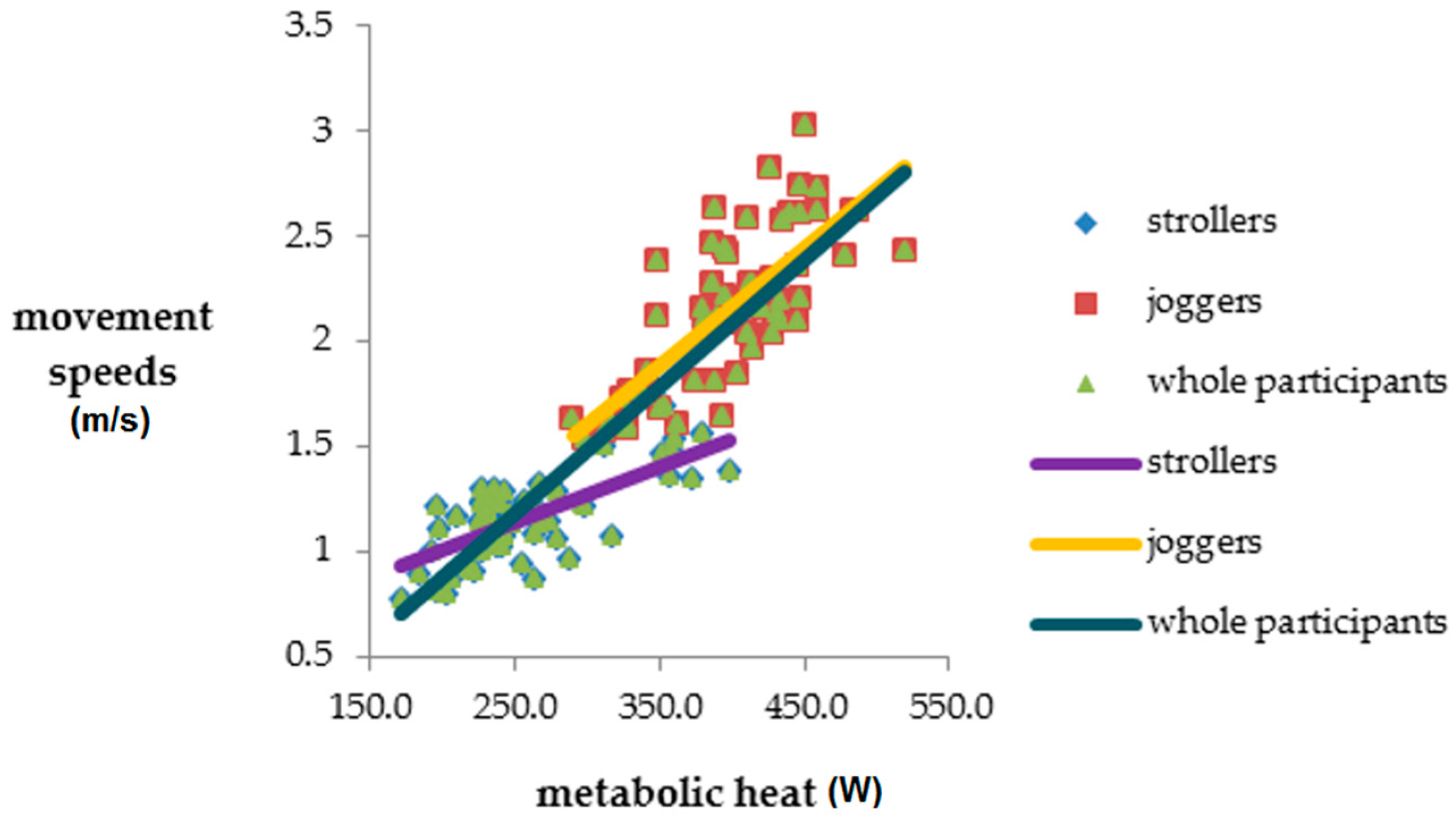
| MH | −0.310 * | −0.009 | −0.032 | 0.714 *** | 0.748 *** | 0.905 *** |
| (p = 0.015) | (p = 0.950) | (p = 0.733) | (p = 0.000) | (p = 0.000) | (p = 0.000) | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, K.-T.; Wang, P.-C. Validity of Research-Grade Actigraphy Unit for Measuring Exercise Intensity. Int. J. Environ. Res. Public Health 2017, 14, 511. https://doi.org/10.3390/ijerph14050511
Han K-T, Wang P-C. Validity of Research-Grade Actigraphy Unit for Measuring Exercise Intensity. International Journal of Environmental Research and Public Health. 2017; 14(5):511. https://doi.org/10.3390/ijerph14050511
Chicago/Turabian StyleHan, Ke-Tsung, and Po-Ching Wang. 2017. "Validity of Research-Grade Actigraphy Unit for Measuring Exercise Intensity" International Journal of Environmental Research and Public Health 14, no. 5: 511. https://doi.org/10.3390/ijerph14050511
APA StyleHan, K.-T., & Wang, P.-C. (2017). Validity of Research-Grade Actigraphy Unit for Measuring Exercise Intensity. International Journal of Environmental Research and Public Health, 14(5), 511. https://doi.org/10.3390/ijerph14050511





