Efficient Removal of Tetracycline from Aqueous Media with a Fe3O4 Nanoparticles@graphene Oxide Nanosheets Assembly
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Fe3O4@GO
2.1.1. Preparation of Preoxidized Graphite
2.1.2. Preparation of GO
2.1.3. Preparation of Fe3O4@GO
2.2. Characterization
2.3. Adsorption Experiment
2.4. Desorption Experiment
3. Results and Discussion
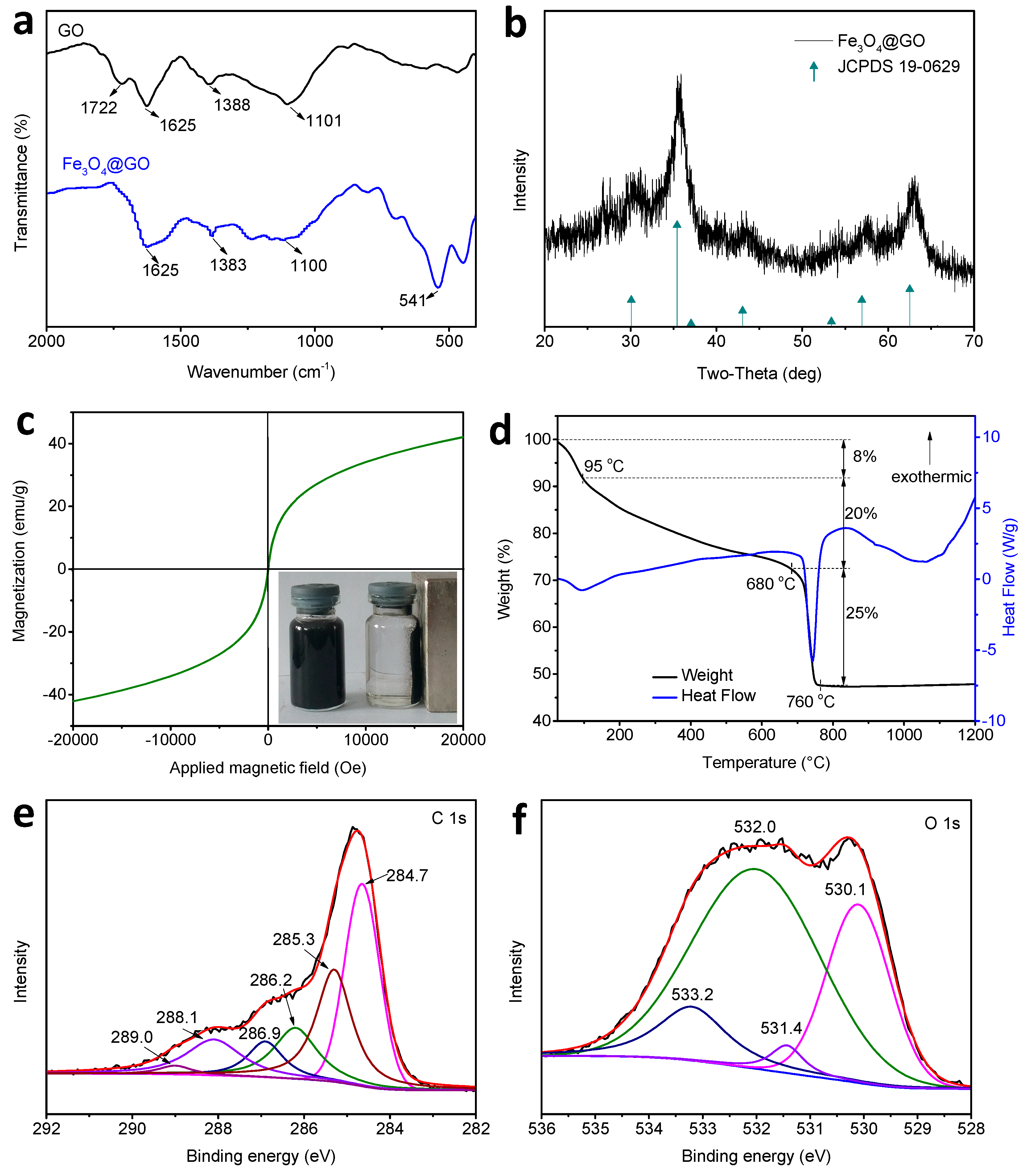
3.1. Characterization
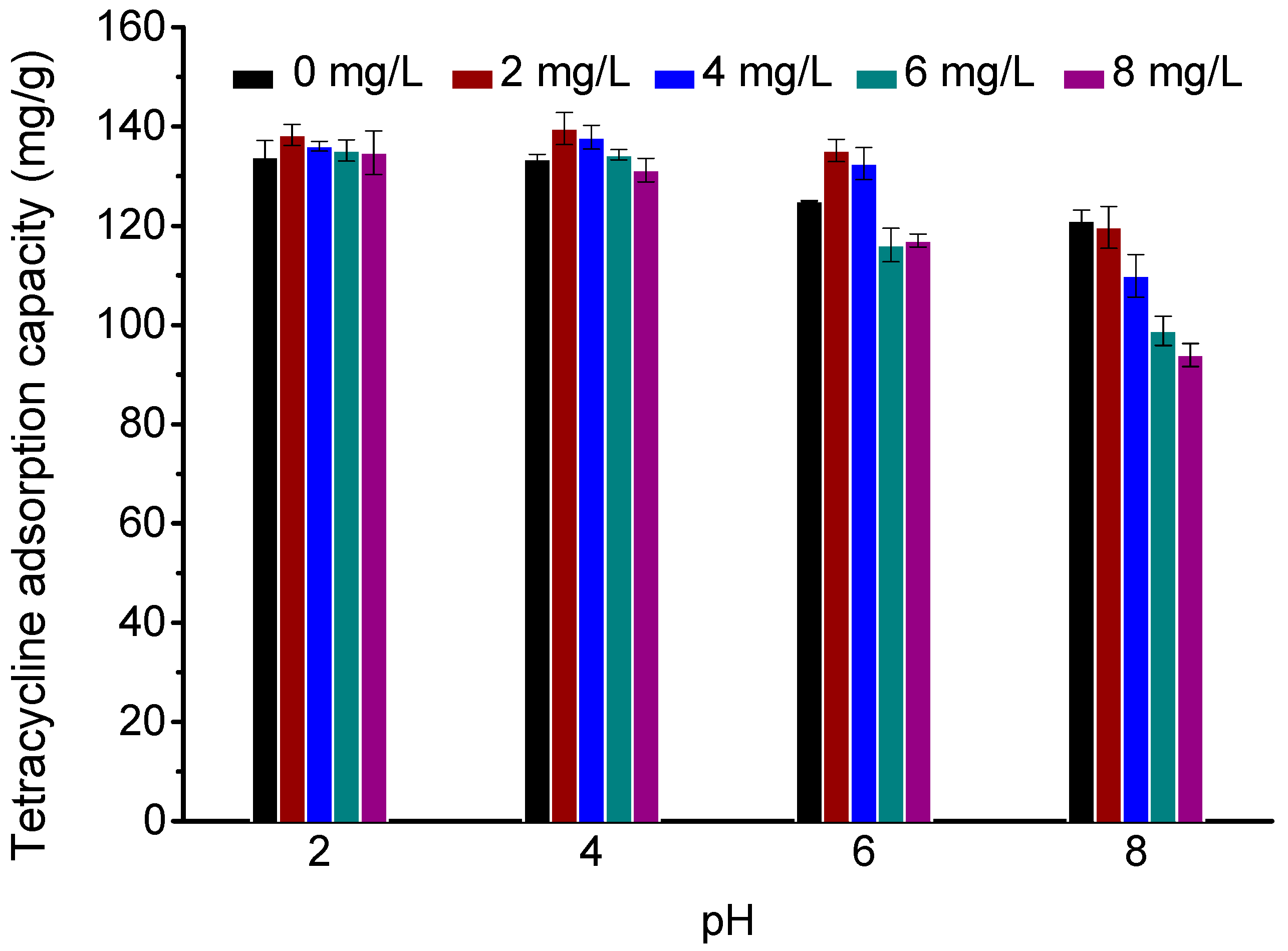
3.2. Effect of pH
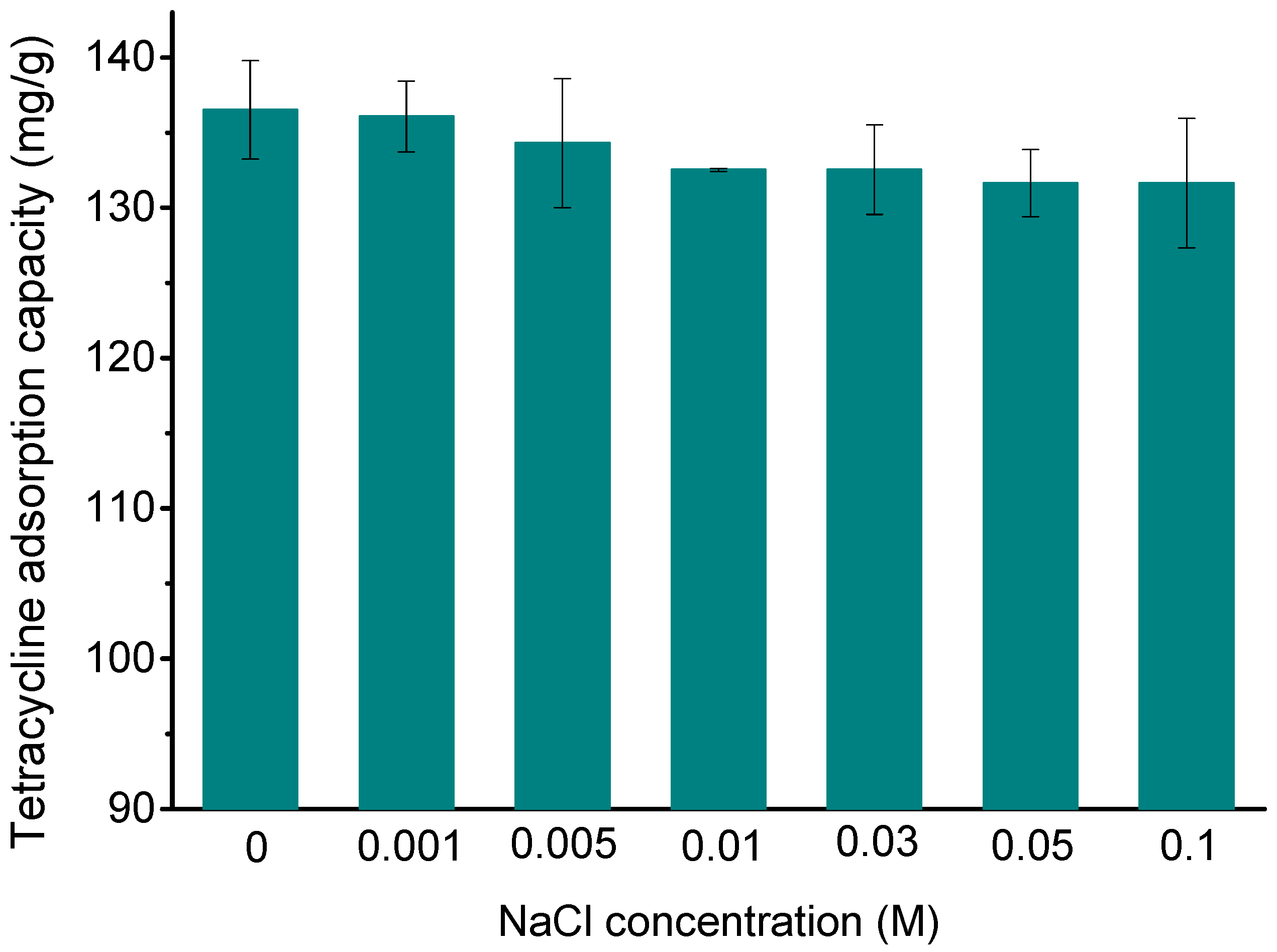
3.3. Effect of Ionic Strength
3.4. Effect of HA Concentration
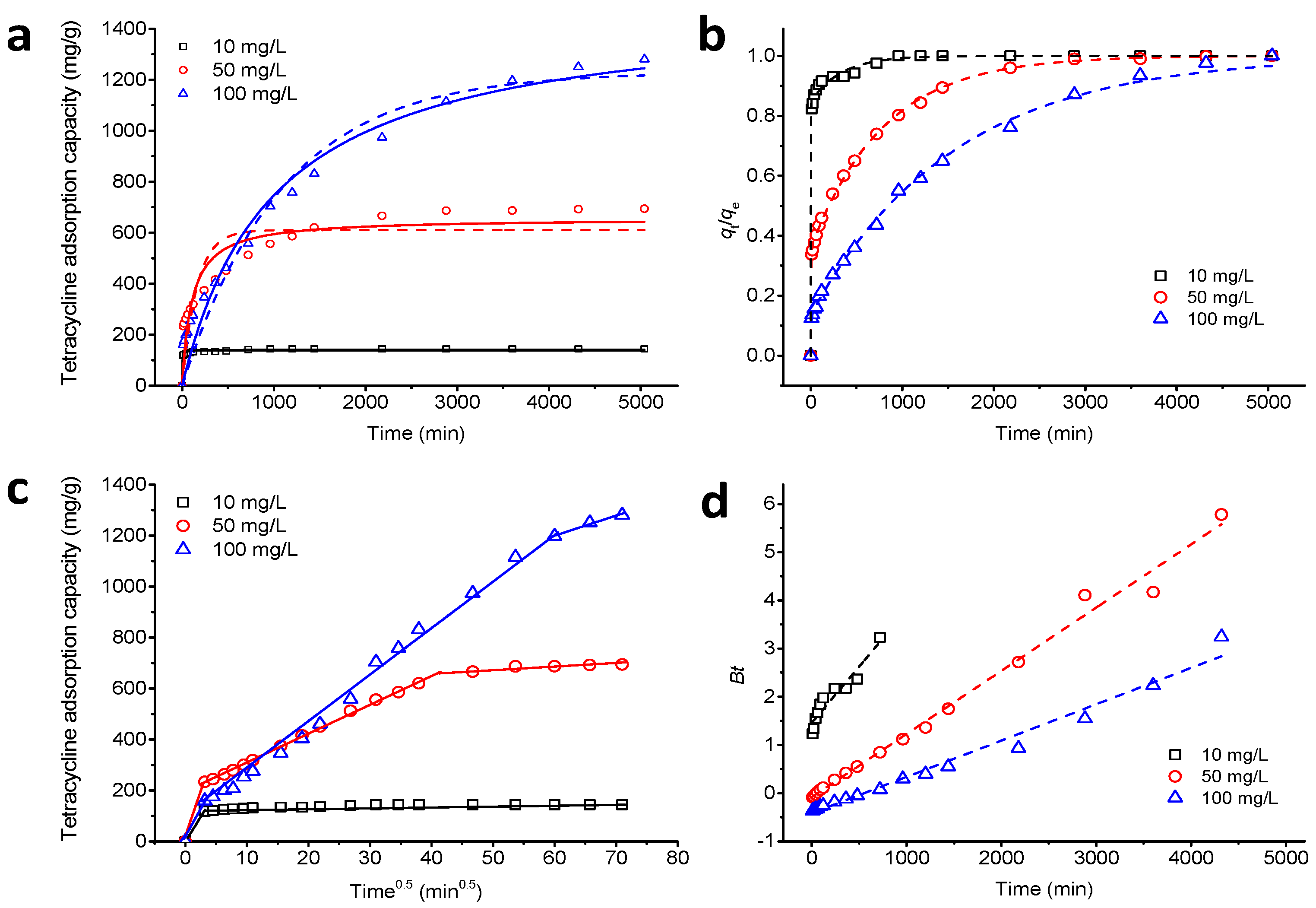
3.5. Adsorption Kinetics
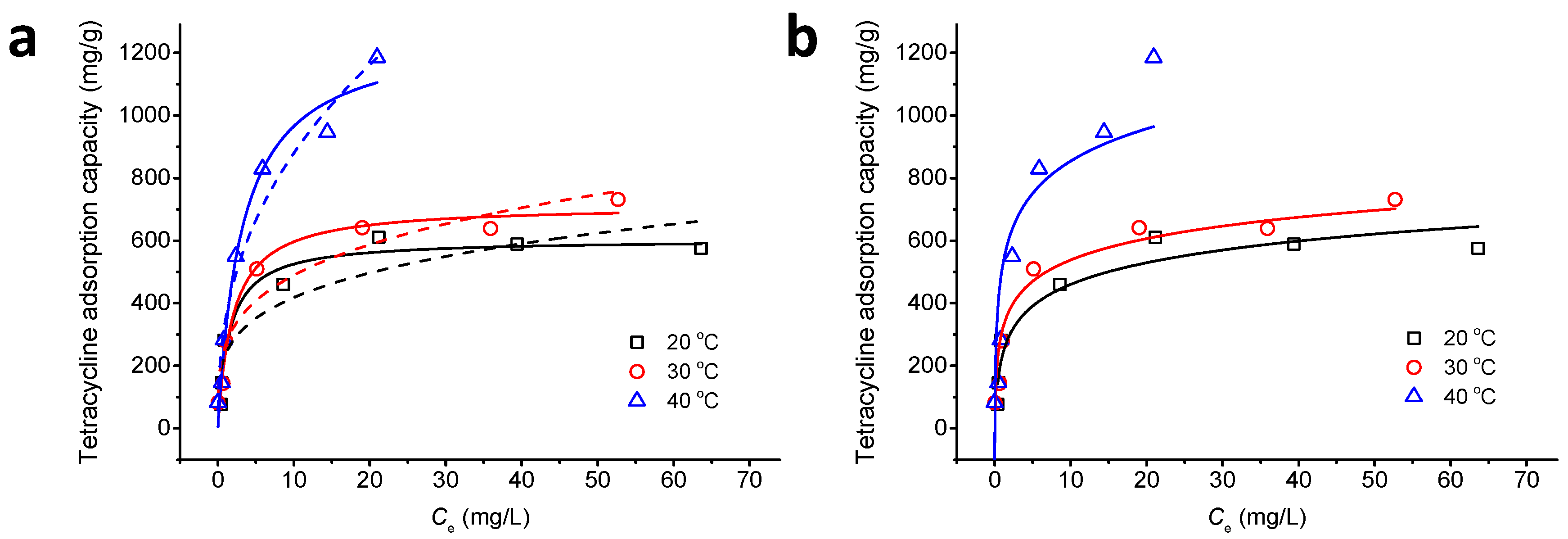
3.6. Adsorption Isotherm
3.7. Desorption Experiment
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Passananti, M.; Lavorgna, M.; Iesce, M.R.; DellaGreca, M.; Brigante, M.; Criscuolo, E.; Cermola, F.; Isidori, M. Photochemical fate and eco-genotoxicity assessment of the drug etodolac. Sci. Total Environ. 2015, 518, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Liu, Y.; Li, B.; Liu, S.; Zeng, G.; Zeng, Z.; Wang, X.; Ning, Q.; Zheng, B.; Yang, C. Effect of Cu(II) ions on the enhancement of tetracycline adsorption by Fe3O4@SiO2-Chitosan/graphene oxide nanocomposite. Carbohydr. Polym. 2017, 157, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Hu, X.; Zhao, Y.; Cui, L.; Huang, Z.; Long, J.; Xu, J.; Deng, J.; Wu, C.; Liao, W. Decontamination of tetracycline by thiourea-dioxide-reduced magnetic graphene oxide: Effects of pH, ionic strength, and humic acid concentration. J. Colloid Interface Sci. 2017, 495, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Yang, T.; Wang, X.; Sun, Y.; Ai, Y.; Sheng, G.; Hayat, T.; Wang, X. Experimental and theoretical evidence for competitive interactions of tetracycline and sulfamethazine with reduced graphene oxides. Environ. Sci. Nano 2016, 3, 1318–1326. [Google Scholar] [CrossRef]
- Kummerer, K. Antibiotics in the aquatic environment—A review-part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.; Son, A.; Her, N.; Zoh, K.-D.; Cho, J.; Yoon, Y. Removal of endocrine disrupting compounds, pharmaceuticals, and personal care products in water using carbon nanotubes: A review. J. Ind. Eng. Chem. 2015, 27, 1–11. [Google Scholar] [CrossRef]
- Yu, F.; Li, Y.; Han, S.; Ma, J. Adsorptive removal of antibiotics from aqueous solution using carbon materials. Chemosphere 2016, 153, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Rattanachueskul, N.; Saning, A.; Kaowphong, S.; Chumha, N.; Chuenchom, L. Magnetic carbon composites with a hierarchical structure for adsorption of tetracycline, prepared from sugarcane bagasse via hydrothermal carbonization coupled with simple heat treatment process. Bioresource Technol. 2017, 226, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Kummerer, K. Antibiotics in the aquatic environment—A review-part II. Chemosphere 2009, 75, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.-Q.; Ying, G.-G.; Pan, C.-G.; Liu, Y.-S.; Zhao, J.-L. Comprehensive Evaluation of Antibiotics Emission and Fate in the River Basins of China: Source Analysis, Multimedia Modeling, and Linkage to Bacterial Resistance. Environ. Sci. Technol. 2015, 49, 6772–6782. [Google Scholar] [CrossRef] [PubMed]
- Homem, V.; Santos, L. Degradation and removal methods of antibiotics from aqueous matrices—A review. J. Environ. Manag. 2011, 92, 2304–2347. [Google Scholar] [CrossRef] [PubMed]
- Rakshit, S.; Sarkar, D.; Elzinga, E.J.; Punamiya, P.; Datta, R. Surface Complexation of Oxytetracycline by Magnetite: Effect of Solution Properties. Vadose Zone J. 2014, 13. [Google Scholar] [CrossRef]
- Rakshit, S.; Elzinga, E.J.; Datta, R.; Sarkar, D. In situ attenuated total reflectance fourier-transform infrared study of oxytetracycline sorption on magnetite. J. Environ. Qual. 2013, 42, 822–827. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Niu, H.; Zhang, X.; Meng, Z.; Cai, Y. Strong adsorption of chlorotetracycline on magnetite nanoparticles. J. Hazard. Mater. 2011, 192, 1088–1093. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, Y.; Zhang, L.; Huang, H.; Hu, J.; Shah, S.M.; Su, X. Adsorption and removal of tetracycline antibiotics from aqueous solution by graphene oxide. J. Colloid Interface Sci. 2012, 368, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Geng, J.; Wang, X.; Gu, X.; Gao, S. Adsorption of tetracycline onto goethite in the presence of metal cations and humic substances. J. Colloid Interface Sci. 2011, 361, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.T.; Chang, P.H.; Wang, Y.S.; Tsai, Y.; Jean, J.S.; Li, Z. Sorption and desorption of tetracycline on layered manganese dioxide birnessite. Int. J. Environ. Sci. Technol. 2014, 12, 1695–1704. [Google Scholar] [CrossRef]
- Caroni, A.L.P.F.; de Lima, C.R.M.; Pereira, M.R.; Fonseca, J.L.C. Tetracycline adsorption on chitosan: A mechanistic description based on mass uptake and zeta potential measurements. Coll. Surf. Biointerfaces 2012, 100, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Wang, H.; Liu, Y. Statistical Analysis of Main and Interaction Effects on Cu(II) and Cr(VI) Decontamination by Nitrogen-Doped Magnetic Graphene Oxide. Sci. Rep. 2016, 6, 34378. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Chen, J.; Liang, Z.; Xu, W.; Chen, L.; Ye, D. Degradation of phenol using Fe3O4-GO nanocomposite as a heterogeneous photo-Fenton catalyst. Sep. Purif. Technol. 2016, 171, 80–87. [Google Scholar] [CrossRef]
- Wang, D.; Liu, L.; Jiang, X.; Yu, J.; Chen, X. Adsorption and removal of malachite green from aqueous solution using magnetic β-cyclodextrin-graphene oxide nanocomposites as adsorbents. Coll. Surf. Physicochem. Eng. Asp. 2015, 466, 166–173. [Google Scholar] [CrossRef]
- Salam, M.A.; El-Shishtawy, R.M.; Obaid, A.Y. Synthesis of magnetic multi-walled carbon nanotubes/magnetite/chitin magnetic nanocomposite for the removal of Rose Bengal from real and model solution. J. Ind. Eng. Chem. 2014, 20, 3559–3567. [Google Scholar] [CrossRef]
- Li, X.; Pan, J.; Dai, J.; Dai, X.; Xu, L.; Wei, X.; Hang, H.; Li, C.; Liu, Y. Surface molecular imprinting onto magnetic yeast composites via atom transfer radical polymerization for selective recognition of cefalexin. Chem. Eng. J. 2012, 198–199, 503–511. [Google Scholar] [CrossRef]
- Zhao, G.X.; Li, J.X.; Ren, X.M.; Chen, C.L.; Wang, X.K. Few-layered graphene oxide nanosheets as superior sorbents for heavy metal ion pollution management. Environ. Sci. Technol. 2011, 45, 10454–10462. [Google Scholar] [CrossRef] [PubMed]
- Hummers, W.S.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [Google Scholar] [CrossRef]
- Ma, H.L.; Zhang, Y.W.; Hu, Q.H.; Yan, D.; Yu, Z.Z.; Zhai, M.L. Chemical reduction and removal of Cr(VI) from acidic aqueous solution by ethylenediamine-reduced graphene oxide. J. Mater. Chem. 2012, 22, 5914–5916. [Google Scholar] [CrossRef]
- Jin, Z.; Wang, X.; Sun, Y.; Ai, Y.; Wang, X. Adsorption of 4-n-Nonylphenol and Bisphenol-A on Magnetic Reduced Graphene Oxides: A Combined Experimental and Theoretical Studies. Environ. Sci. Technol. 2015, 49, 9168–9175. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Hu, J.; Zhao, Y.; Shao, D.; Li, J. Efficient removal of cobalt from aqueous solution using β-cyclodextrin modified graphene oxide. RSC Adv. 2013, 3, 9514–9521. [Google Scholar] [CrossRef]
- Bai, L.-Z.; Zhao, D.-L.; Xu, Y.; Zhang, J.-M.; Gao, Y.-L.; Zhao, L.-Y.; Tang, J.-T. Inductive heating property of graphene oxide–Fe3O4 nanoparticles hybrid in an AC magnetic field for localized hyperthermia. Mater. Lett. 2012, 68, 399–401. [Google Scholar] [CrossRef]
- Hou, C.; Zhang, Q.; Zhu, M.; Li, Y.; Wang, H. One-step synthesis of magnetically-functionalized reduced graphite sheets and their use in hydrogels. Carbon 2011, 49, 47–53. [Google Scholar] [CrossRef]
- Hu, X.-J.; Liu, Y.-G.; Zeng, G.-M.; Wang, H.; Hu, X.; Chen, A.-W.; Wang, Y.-Q.; Guo, Y.-M.; Li, T.-T.; Zhou, L.; et al. Effect of aniline on cadmium adsorption by sulfanilic acid-grafted magnetic graphene oxide sheets. J. Coll. Interface Sci. 2014, 426, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Togami, M.; Tsushida, M.; Kawai, D. Effect of N-doping of single-walled carbon nanotubes on bioelectrocatalysis of laccase. Anal. Chem. 2014, 86, 5053–5060. [Google Scholar] [CrossRef] [PubMed]
- Li, M.-F.; Liu, Y.-g.; Zeng, G.-M.; Liu, S.-B.; Hu, X.-J.; Shu, D.; Jiang, L.-H.; Tan, X.-F.; Cai, X.-X.; Yan, Z.-L. Tetracycline absorbed onto nitrilotriacetic acid-functionalized magnetic graphene oxide: Influencing factors and uptake mechanism. J. Coll. Interface Sci. 2017, 485, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Lei, Y.; Chen, F.; Luo, Y.; Zhang, L. Three-dimensional magnetic graphene oxide foam/Fe3O4 nanocomposite as an efficient absorbent for Cr(VI) removal. J. Mater. Sci. 2014, 49, 4236–4245. [Google Scholar] [CrossRef]
- Korichi, S.; Bensmaili, A. Sorption of uranium (VI) on homoionic sodium smectite experimental study and surface complexation modeling. J. Hazard. Mater. 2009, 169, 780–793. [Google Scholar] [CrossRef] [PubMed]
- Zong, P.; Wang, S.; Zhao, Y.; Wang, H.; Pan, H.; He, C. Synthesis and application of magnetic graphene/iron oxides composite for the removal of U(VI) from aqueous solutions. Chem. Eng. J. 2013, 220, 45–52. [Google Scholar] [CrossRef]
- Fu, Q.; Hu, B.; Zhou, X.; Hu, Q.; Sheng, J. Impact of key geochemical parameters on the attenuation of Pb(II) from water using a novel magnetic nanocomposite: fulvic acid-coated magnetite nanoparticles. Desalination Water Treat. 2016, 57, 26063–26072. [Google Scholar] [CrossRef]
- Chen, L.-C.; Lei, S.; Wang, M.-Z.; Yang, J.; Ge, X.-W. Fabrication of macroporous polystyrene/graphene oxide composite monolith and its adsorption property for tetracycline. Chin. Chem. Lett. 2016, 27, 511–517. [Google Scholar] [CrossRef]
- Liu, D.; Song, N.; Feng, W.; Jia, Q. Synthesis of graphene oxide functionalized surface-imprinted polymer for the preconcentration of tetracycline antibiotics. R. Soc. Chem. Adv. 2016, 6, 11742–11748. [Google Scholar] [CrossRef]
- Figueroa, R.A.; Leonard, A.; Mackay, A.A. Modeling tetracycline antibiotic sorption to clays. Environ. Sci. Technol. 2004, 38, 476–483. [Google Scholar] [CrossRef] [PubMed]
- Hayes, K.F.; Leckie, J.O. Modeling ionic strength effects on cation adsorption at hydrous oxide/solution interfaces. J. Coll. Interface Sci. 1988, 115, 564–572. [Google Scholar] [CrossRef]
- Acheampong, M.A.; Pereira, J.P.C.; Meulepas, R.J.W.; Lens, P.N.L. Kinetics modelling of Cu(II) biosorption on to coconut shell andMoringa oleiferaseeds from tropical regions. Environ. Technol. 2012, 33, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Liu, Y.; Liu, S.; Yin, Y.; Zeng, G.; Tan, X.; Hu, X.; Hu, X.; Jiang, L.; Ding, Y.; et al. Investigation of the adsorption-reduction mechanisms of hexavalent chromium by ramie biochars of different pyrolytic temperatures. Bioresour. Technol. 2016, 218, 351–359. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Ma, W.; Han, M. Biosorption of nickel and copper onto treated alga (Undaria pinnatifida): Application of isotherm and kinetic models. J. Hazard. Mater. 2008, 155, 327–333. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S. Adsorption of Heavy Metals from Waste Streams by Peat. Ph.D. Thesis, University of Birmingham, Birmingham, UK, 5 January 1995. [Google Scholar]
- Ho, Y.S.; Wase, D.A.J.; Forster, C.F. Kinetic Studies of Competitive Heavy Metal Adsorption by Sphagnum Moss Peat. Environ. Technol. 1996, 17, 71–77. [Google Scholar] [CrossRef]
- Hu, X.-J.; Wang, J.-S.; Liu, Y.-G.; Li, X.; Zeng, G.-M.; Bao, Z.-L.; Zeng, X.-X.; Chen, A.-W.; Long, F. Adsorption of chromium(VI) by ethylenediamine-modified cross-linked magnetic chitosan resin: Isotherms, kinetics and thermodynamics. J. Hazard. Mater. 2011, 185, 306–314. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Liu, T.; Wang, X.; Hu, X.; Zhang, L. Efficient removal of heavy metal ions from aqueous systems with the assembly of anisotropic layered double hydroxide nanocrystals@carbon nanosphere. Environ. Sci. Technol. 2011, 45, 6181–6187. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.-T.; Shi, L.-Q.; Shen, H.; Chen, X.; Xie, K.-P. Equilibrium, isotherm, kinetic and thermodynamic studies for removal of tetracycline antibiotics by adsorption onto hazelnut shell derived activated carbons from aqueous media. RSC Adv. 2016, 6, 109983–109991. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Textural porosity, surface chemistry and adsorptive properties of durian shell derived activated carbon prepared by microwave assisted NaOH activation. Chem. Eng. J. 2012, 187, 53–62. [Google Scholar] [CrossRef]
- Sarkar, M.; Acharya, P.K.; Bhattacharya, B. Modeling the adsorption kinetics of some priority organic pollutants in water from diffusion and activation energy parameters. J. Colloid Interface Sci. 2003, 266, 28–32. [Google Scholar] [CrossRef]
- Cui, L.M.; Wang, Y.G.; Gao, L.; Hu, L.H.; Yan, L.G.; Wei, Q.; Du, B. EDTA functionalized magnetic graphene oxide for removal of Pb(II), Hg(II) and Cu(II) in water treatment: Adsorption mechanism and separation property. Chem. Eng. J. 2015, 281, 1–10. [Google Scholar] [CrossRef]


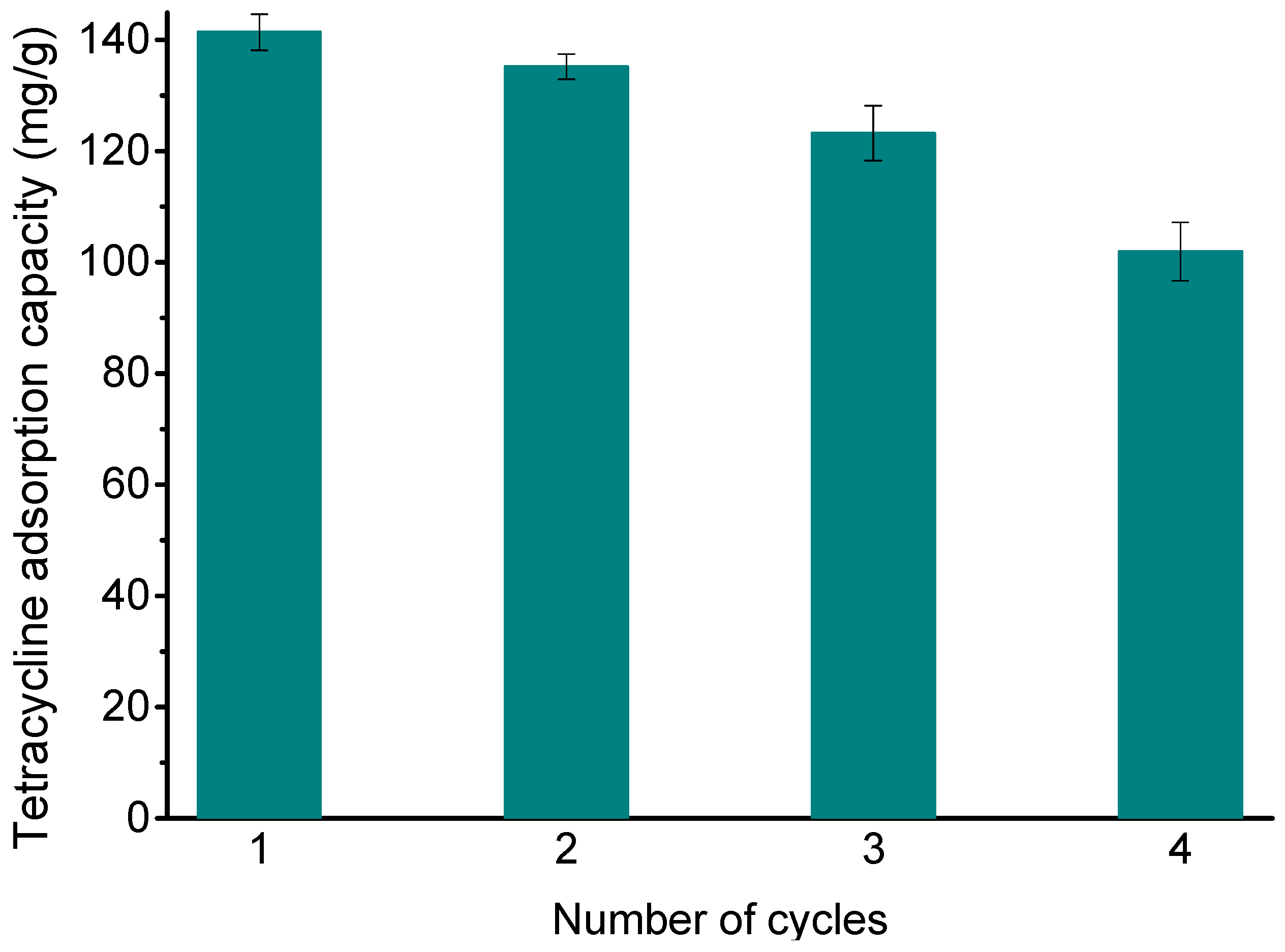
| Models | Parameters | Temperature | ||
|---|---|---|---|---|
| 20 °C | 30 °C | 40 °C | ||
| Pseudo-first-order | k1 × 102 (1/min) | 1.76 × 10−1 | 6.29 × 10−3 | 9.14 × 10−4 |
| qe,1 (mg/g) | 138.27 | 610.88 | 1229.09 | |
| R2 | 0.951 | 0.746 | 0.941 | |
| Pseudo-second-order | k2 (g/mg min) | 2.69 × 10−3 | 1.45 × 10−5 | 6.71 × 10−7 |
| qe,2 (mg/g) | 140.76 | 657.08 | 1492.21 | |
| R2 | 0.977 | 0.857 | 0.956 | |
| Two-compartment | Ffast | 0.15 | 0.36 | 0.86 |
| Fslow | 0.85 | 0.64 | 0.14 | |
| kfast (1/min) | 332.21 | 4.11 | 1554.70 | |
| kslow (1/min) | 7.73 × 10−2 | 793.97 | 5.22 | |
| R2 | 0.996 | 0.999 | 0.997 | |
| Models | Parameters | Temperature | ||
|---|---|---|---|---|
| 20 °C | 30 °C | 40 °C | ||
| Langmuir Isotherm | qmax (mg/g) | 603.74 | 713.88 | 1272.45 |
| KL (L/mg) | 0.67 | 0.51 | 0.31 | |
| R2 | 0.950 | 0.970 | 0.974 | |
| Freundlich Isotherm | n | 4.03 | 3.80 | 2.49 |
| KF | 236.16 | 267.11 | 348.74 | |
| R2 | 0.835 | 0.921 | 0.957 | |
| Temkin Isotherm | aT (L/g) | 9.96 | 23.51 | 30.69 |
| bT × 102 (KJ/mol) | 2.44 | 2.56 | 1.75 | |
| R2 | 0.919 | 0.917 | 0.757 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, X.; Zhao, Y.; Wang, H.; Tan, X.; Yang, Y.; Liu, Y. Efficient Removal of Tetracycline from Aqueous Media with a Fe3O4 Nanoparticles@graphene Oxide Nanosheets Assembly. Int. J. Environ. Res. Public Health 2017, 14, 1495. https://doi.org/10.3390/ijerph14121495
Hu X, Zhao Y, Wang H, Tan X, Yang Y, Liu Y. Efficient Removal of Tetracycline from Aqueous Media with a Fe3O4 Nanoparticles@graphene Oxide Nanosheets Assembly. International Journal of Environmental Research and Public Health. 2017; 14(12):1495. https://doi.org/10.3390/ijerph14121495
Chicago/Turabian StyleHu, Xinjiang, Yunlin Zhao, Hui Wang, Xiaofei Tan, Yuanxiu Yang, and Yunguo Liu. 2017. "Efficient Removal of Tetracycline from Aqueous Media with a Fe3O4 Nanoparticles@graphene Oxide Nanosheets Assembly" International Journal of Environmental Research and Public Health 14, no. 12: 1495. https://doi.org/10.3390/ijerph14121495
APA StyleHu, X., Zhao, Y., Wang, H., Tan, X., Yang, Y., & Liu, Y. (2017). Efficient Removal of Tetracycline from Aqueous Media with a Fe3O4 Nanoparticles@graphene Oxide Nanosheets Assembly. International Journal of Environmental Research and Public Health, 14(12), 1495. https://doi.org/10.3390/ijerph14121495





