Insecticidal Effects of Plasma Treated Water
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Plasma Treated Liquids
2.2. Water Analysis
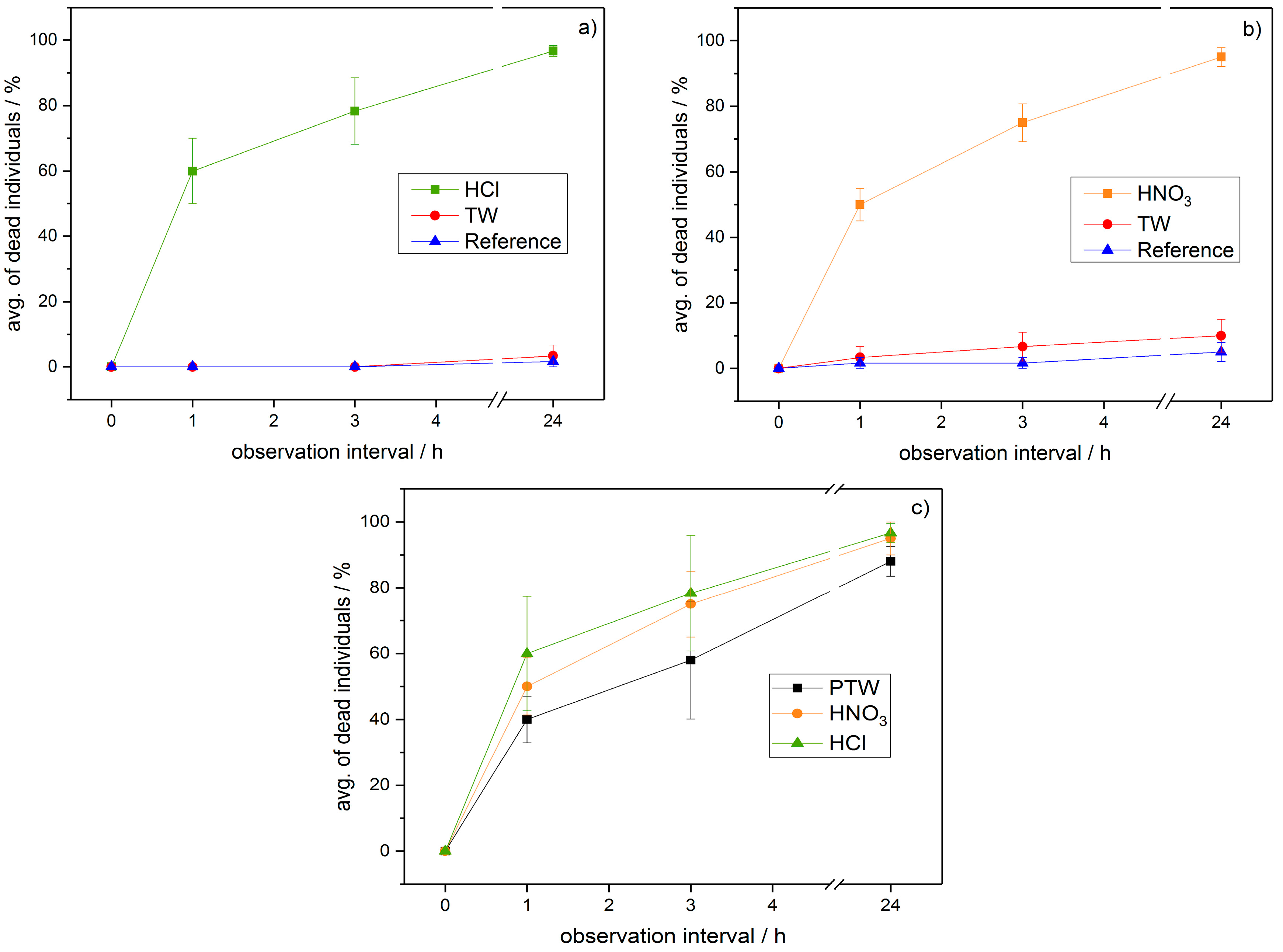
2.3. Classically Acidified Water
2.4. Test Organism Planococcus citri
3. Results
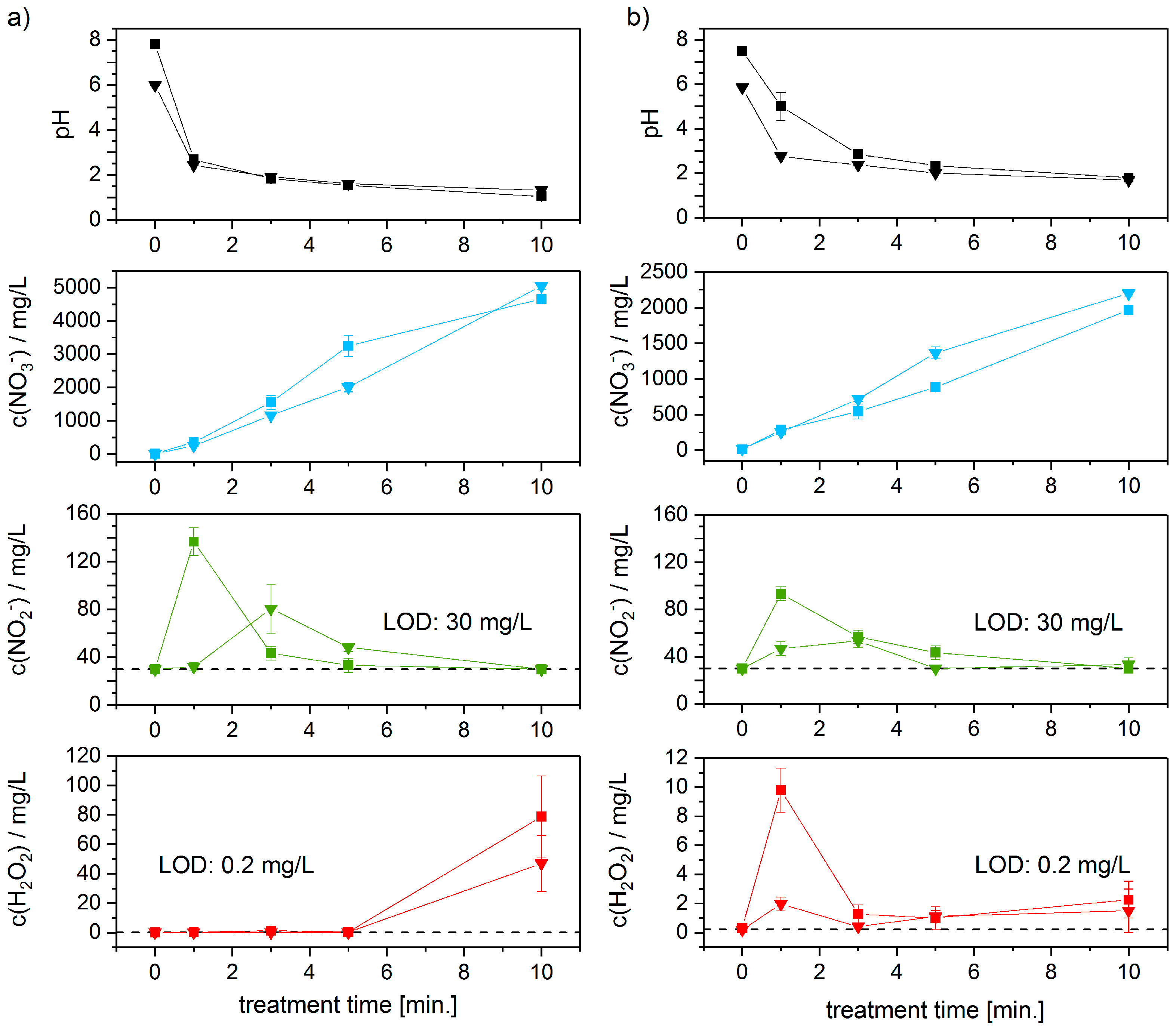
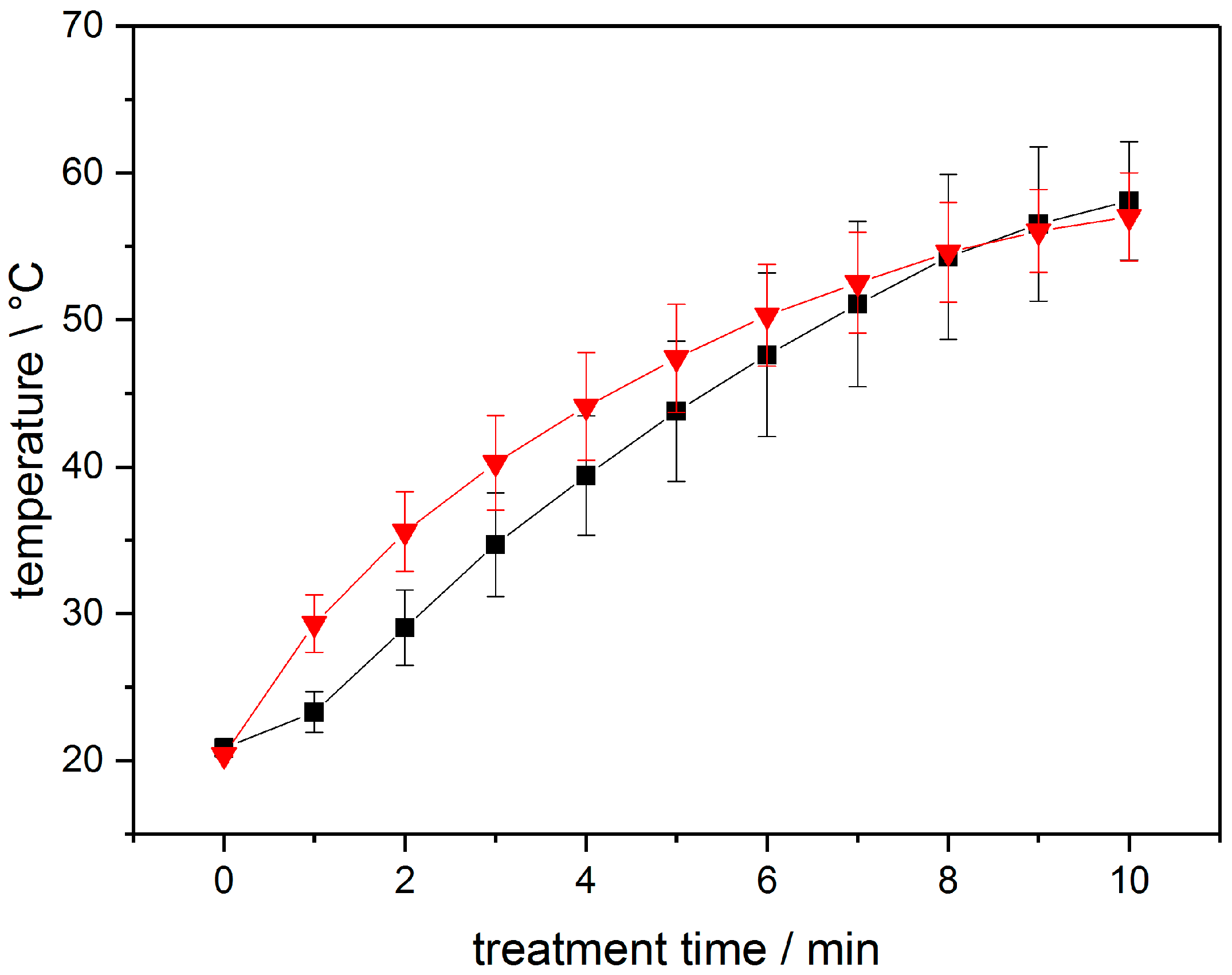
3.1. Analysis of PTW
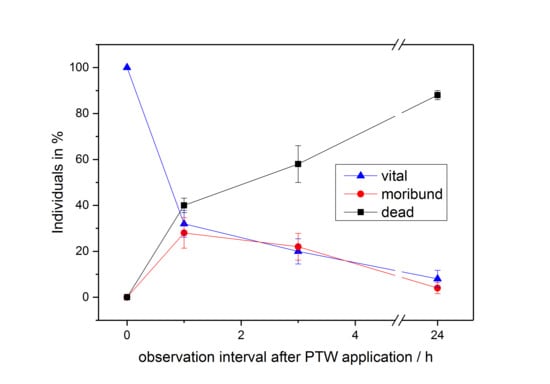
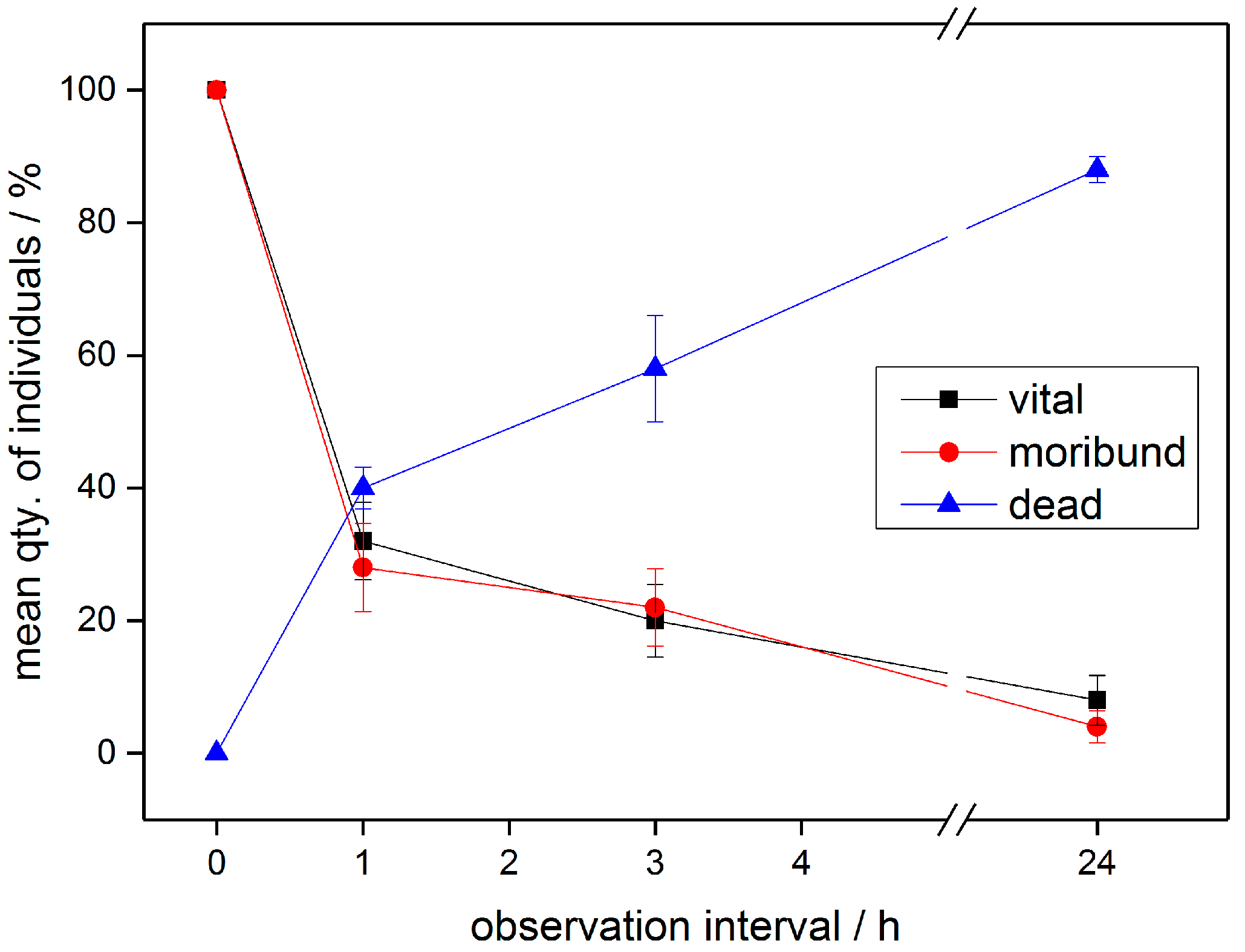
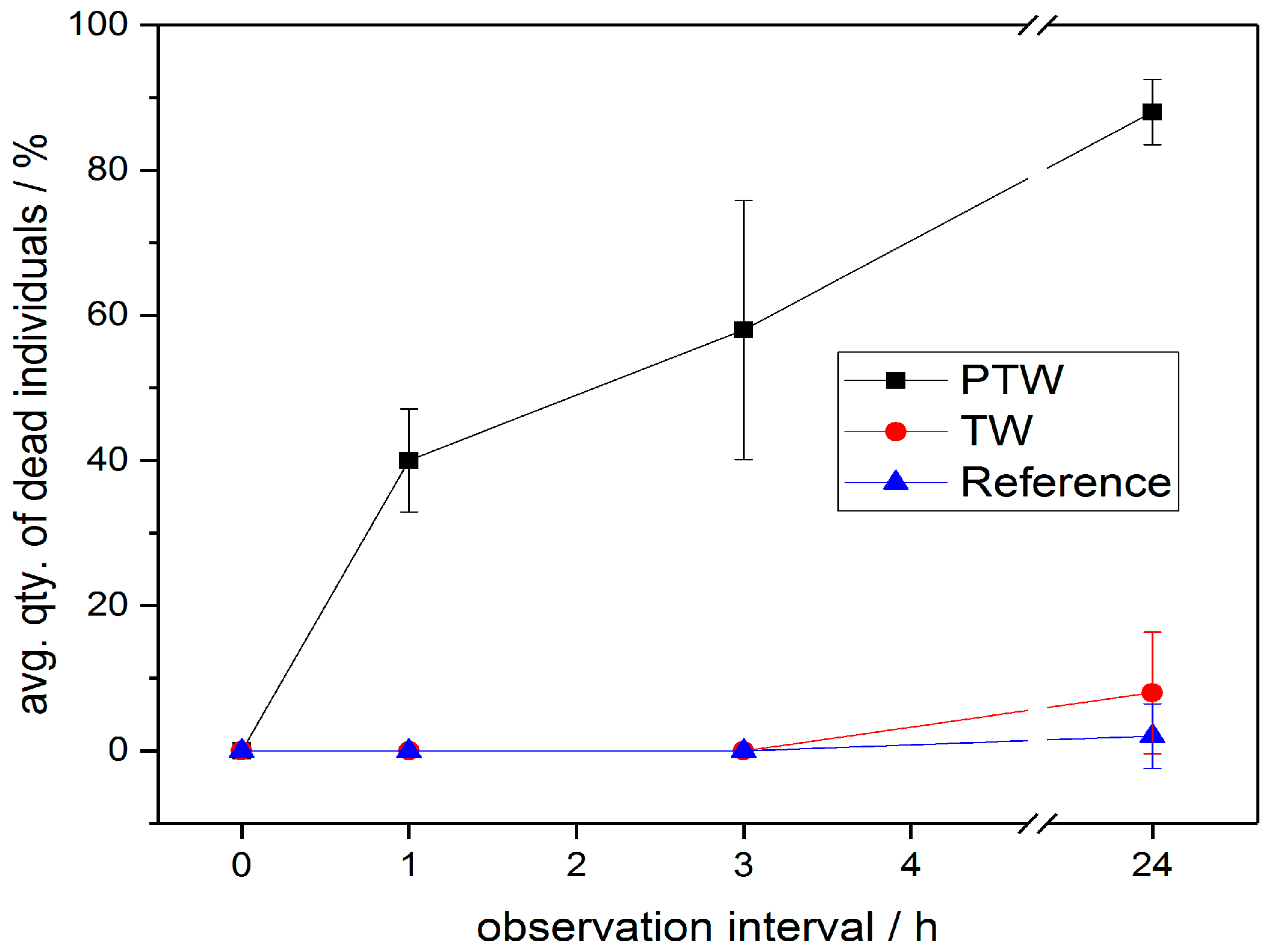
3.2. PTW/CAW Treatment of Mealybugs
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bach, C.E. Direct and indirect interactions between ants (Pheidole megacephala), scales (Coccus viridis) and plants (Pluchea indica). Oecologia 1991, 87, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Cale, J.A.; Ashby, A.W.; West, J.L.; Teale, S.A.; Johnston, M.T.; Castello, J.D.; Vannini, A. Scale insects, decay and canker fungi in American beech. For. Pathol. 2015, 45, 71–75. [Google Scholar] [CrossRef]
- Selvarajan, R.; Balasubramanian, V.; Padmanaban, B. Mealybugs as Vectors. Mealybugs and Their Management in Agricultural and Horticultural Crops; Mani, M., Shivaraju, C., Eds.; Springer: New Delhi, India, 2016; pp. 123–130. [Google Scholar]
- Martelli, G.P.; Agranovsky, A.A.; Bar-Joseph, M.; Boscia, D.; Candresse, T.; Coutts, R.H.A.; Dolja, V.V.; Falk, B.W.; Gonsalves, D.; Jelkmann, W.; et al. The family Closteroviridae revised. Arch. Virol. 2002, 147, 2039–2044. [Google Scholar] [CrossRef] [PubMed]
- Sforza, R.; Boudon-Padieu, E.; Greif, C. New Mealybug Species Vectoring Grapevine Leafroll-Associated Viruses-1 and -3 (GLRaV-1 and -3). Eur. J. Plant Pathol. 2003, 109, 975–981. [Google Scholar] [CrossRef]
- Bertin, S.; Cavalieri, V.; Graziano, C.; Bosco, D. Survey of mealybug (Hemiptera: Pseudococcidae) vectors of Ampelovirus and Vitivirus in vineyards of northwestern Italy. Phytoparasitica 2010, 38, 401–409. [Google Scholar] [CrossRef]
- Charles, J.G. Economic damage and preliminary economic thresholds for mealybugs (Pseudococcus longispinus T-T.) in Auckland vineyards. N. Z. J. Agric. Res. 2011, 25, 415–420. [Google Scholar] [CrossRef]
- Cox, J.M.; Freeston, A.C. Identification of mealybugs of the genus Planococcus (Homoptera: Pseudococcidae) occurring on cacao throughout the world. J. Nat. Hist. 2007, 19, 719–728. [Google Scholar] [CrossRef]
- Nagrare, V.S.; Kranthi, S.; Biradar, V.K.; Zade, N.N.; Sangode, V.; Kakde, G.; Shukla, R.M.; Shivare, D.; Khadi, B.M.; Kranthi, K.R. Widespread infestation of the exotic mealybug species, Phenacoccus solenopsis (Tinsley) (Hemiptera: Pseudococcidae), on cotton in India. Bull. Entomol. Res. 2009, 99, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Perry, A.S.; Yamamoto, I.; Ishaaya, I.; Perry, R. Insecticides in Agriculture and Environment; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- Ohkawa, H.; Miyagawa, H.; Lee, P.W. Pesticide Chemistry: Crop Protection, Public Health, Environmental Safety; Wiley-VCH: Weinheim, Germany, 2007. [Google Scholar]
- Perveen, F. Insecticides: Advances in Integrated Pest Management; InTech: Rijeka, Croatia, 2014. [Google Scholar]
- Jonsson, M.; Wratten, S.D.; Landis, D.A.; Gurr, G.M. Recent advances in conservation biological control of arthropods by arthropods. Biol. Control 2008, 45, 172–175. [Google Scholar] [CrossRef]
- Landis, D.A.; Wratten, S.D.; Gurr, G.M. Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu. Rev. Entomol. 2000, 45, 175–201. [Google Scholar] [CrossRef] [PubMed]
- Epino, P.; Yang, H.-C.; Goddard, D.P.; Ahmed, S.; Grainge, M. Handbook of Plants with Pest-Control Properties; Wiley: New York, NY, USA, 1988. [Google Scholar]
- Roy, S.; Handique, G.; Muraleedharan, N.; Dashora, K.; Roy, S.M.; Mukhopadhyay, A.; Babu, A. Use of plant extracts for tea pest management in India. Appl. Microbiol. Biotechnol. 2016, 100, 4831–4844. [Google Scholar] [CrossRef] [PubMed]
- Bickerton, M.W.; Hamilton, G.C. Effects of intercropping with flowering plants on predation of Ostrinia nubilalis (Lepidoptera: Crambidae) eggs by generalist predators in bell peppers. Environ. Entomol. 2012, 41, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Stephens, M.J.; France, C.M.; Wratten, S.D.; Frampton, C. Enhancing Biological Control of Leafrollers (Lepidoptera: Tortricidae) by Sowing Buckwheat (Fagopyrum esculentum) in an Orchard. Biocontrol Sci. Technol. 1998, 8, 547–558. [Google Scholar] [CrossRef]
- Horne, P.A.; Page, J. Integrated Pest Management for Crops and Pastures; Landlinks Press: Melbourne, Australia, 2008. [Google Scholar]
- Barbosa, P. Conservation Biological Control; Academic Press: San Diego, CA, USA, 1998. [Google Scholar]
- Van Driesche, R.G.; Simberloff, D.; Blossey, B.; Causton, C.; Hoddle, M.S.; Wagner, D.L.; Marks, C.O.; Heinz, K.M.; Warner, K.D. Integrating Biological Control into Conservation Practice; John Wiley & Sons, Ltd.: Chichester, UK, 2016. [Google Scholar]
- Winkler, K.; Wäckers, F.L.; Termorshuizen, A.J.; van Lenteren, J.C. Assessing risks and benefits of floral supplements in conservation biological control. BioControl 2010, 55, 719–727. [Google Scholar] [CrossRef]
- Abrol, D.P.; Shankar, U. Integrated Pest Management: Principles and Practice; CABI: Wallingford, UK; Boston, MA, USA, 2012. [Google Scholar]
- Pinniger, D. Integrated Pest Management in Cultural Heritage; Archetype Publications: London, UK, 2015. [Google Scholar]
- Bellmann, M.; Avramidis, G.; Wascher, R.; Viöl, W. Accelerated Germination and Altered Surface Characteristics of Pisum Sativum Seeds after Plasma Treatment at Atmospheric Pressure. In Proceedings of the Conference Plasma Surface Engineering, Garmisch-Partenkirchen, Germany, 10–14 September 2012. [Google Scholar]
- Zahoranová, A.; Henselová, M.; Hudecová, D.; Kaliňáková, B.; Kováčik, D.; Medvecká, V.; Černák, M. Effect of Cold Atmospheric Pressure Plasma on the Wheat Seedlings Vigor and on the Inactivation of Microorganisms on the Seeds Surface. Plasma Chem. Plasma Process. 2016, 36, 397–414. [Google Scholar] [CrossRef]
- Misra, N.N.; Tiwari, B.K.; Raghavarao, K.S.M.S.; Cullen, P.J. Nonthermal Plasma Inactivation of Food-Borne Pathogens. Food Eng. Rev. 2011, 3, 159–170. [Google Scholar] [CrossRef]
- Noriega, E.; Shama, G.; Laca, A.; Diaz, M.; Kong, M.G. Cold atmospheric gas plasma disinfection of chicken meat and chicken skin contaminated with Listeria innocua. Food Microbiol. 2011, 28, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Ten Bosch, L.; Pfohl, K.; Avramidis, G.; Wieneke, S.; Viol, W.; Karlovsky, P. Plasma-Based Degradation of Mycotoxins Produced by Fusarium, Aspergillus and Alternaria Species. Toxins 2017, 9, 97. [Google Scholar] [CrossRef] [PubMed]
- Park, B.J.; Takatori, K.; Sugita-Konishi, Y.; Kim, I.-H.; Lee, M.-H.; Han, D.-W.; Chung, K.-H.; Hyun, S.O.; Park, J.-C. Degradation of mycotoxins using microwave-induced argon plasma at atmospheric pressure. Surf. Coat. Technol. 2007, 201, 5733–5737. [Google Scholar] [CrossRef]
- Ten Bosch, L.; Habedank, B.; Viöl, W. Cold atmospheric pressure plasma as insecticide-free pediculosis treatment approach. In Proceedings of the 7th International Congress of the Society for Vector Ecology, Palma, Spain, 1–6 October 2017. [Google Scholar]
- Donohue, K.V.; Bures, B.L.; Bourham, M.A.; Roe, R.M. Mode of Action of a Novel Nonchemical Method of Insect Control: Atmospheric Pressure Plasma Discharge. J. Econ. Entomol. 2006, 99, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Bures, B.L. Rapid Mortality of Pest Arthropods by Direct Exposure to a Dielectric Barrier Discharge. Ph.D. Thesis, North Carolina State University, Raleigh, NC, USA, 2004. [Google Scholar]
- Kong, M.G.; Kroesen, G.; Morfill, G.; Nosenko, T.; Shimizu, T.; van Dijk, J.; Zimmermann, J.L. Plasma medicine: An introductory review. New J. Phys. 2009, 11, 115012. [Google Scholar] [CrossRef]
- Metelmann, H.-R. Plasmamedizin, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Brehmer, F.; Haenssle, H.A.; Daeschlein, G.; Ahmed, R.; Pfeiffer, S.; Gorlitz, A.; Simon, D.; Schon, M.P.; Wandke, D.; Emmert, S. Alleviation of chronic venous leg ulcers with a hand-held dielectric barrier discharge plasma generator (PlasmaDerm((R)) VU-2010): Results of a monocentric, two-armed, open, prospective, randomized and controlled trial (NCT01415622). J. Eur. Dermatol. Venereol. 2015, 29, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Emmert, S.; Brehmer, F.; Hänßle, H.; Helmke, A.; Mertens, N.; Ahmed, R.; Simon, D.; Wandke, D.; Maus-Friedrichs, W.; Däschlein, G.; et al. Atmospheric pressure plasma in dermatology: Ulcus treatment and much more. Clin. Plasma Med. 2013, 1, 24–29. [Google Scholar] [CrossRef]
- Kisch, T.; Schleusser, S.; Helmke, A.; Mauss, K.L.; Wenzel, E.T.; Hasemann, B.; Mailaender, P.; Kraemer, R. The repetitive use of non-thermal dielectric barrier discharge plasma boosts cutaneous microcirculatory effects. Microvasc. Res. 2016, 106, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, J.; Köritzer, J.; Boxhammer, V. Plasma in cancer treatment. Clin. Plasma Med. 2013, 1, 2–7. [Google Scholar] [CrossRef]
- Vandamme, M.; Robert, E.; Pesnel, S.; Barbosa, E.; Dozias, S.; Sobilo, J.; Lerondel, S.; Le Pape, A.; Pouvesle, J.-M. Antitumor Effect of Plasma Treatment on U87 Glioma Xenografts: Preliminary Results. Plasma Process. Polym. 2010, 7, 264–273. [Google Scholar] [CrossRef]
- Wende, K.; Williams, P.; Dalluge, J.; van Gaens, W.; Aboubakr, H.; Bischof, J.; Woedtke, T.; von Goyal, S.M.; Weltmann, K.-D.; Bogaerts, A.; et al. Identification of the biologically active liquid chemistry induced by a nonthermal atmospheric pressure plasma jet. Biointerphases 2015, 10, 29518. [Google Scholar] [CrossRef] [PubMed]
- Adamovich, I.; Baalrud, S.D.; Bogaerts, A.; Bruggeman, P.J.; Cappelli, M.; Colombo, V.; Czarnetzki, U.; Ebert, U.; Eden, J.G.; Favia, P.; et al. The 2017 Plasma Roadmap: Low temperature plasma science and technology. J. Phys. D: Appl. Phys. 2017, 50, 323001. [Google Scholar] [CrossRef]
- Stadtwerke Göttingen AG. Wasseranalyse der Stadtwerke Göttingen AG. 2016. Available online: https://www.stadtwerke-goettingen.de/produkte/wasser/wasseranalyse/ (accessed on 30 March 2017).
- Porter, D.; Poplin, M.D.; Holzer, F.; Finney, W.C.; Locke, B.R. Formation of Hydrogen Peroxide, Hydrogen, and Oxygen in Gliding Arc Electrical Discharge Reactors with Water Spray. IEEE Trans. Ind. Appl. 2009, 45, 623–629. [Google Scholar] [CrossRef]
- Zhao, Y.Y.; Wang, T.; Wilson, M.P.; MacGregor, S.J.; Timoshkin, I.V.; Ren, Q.C. Hydroxyl Radicals and Hydrogen Peroxide Formation at Nonthermal Plasma–Water Interface. IEEE Trans. Plasma Sci. 2016, 44, 2084–2091. [Google Scholar] [CrossRef]
- Oehmigen, K.; Hähnel, M.; Brandenburg, R.; Wilke, C.; Weltmann, K.-D.; von Woedtke, T. The Role of Acidification for Antimicrobial Activity of Atmospheric Pressure Plasma in Liquids. Plasma Process. Polym. 2010, 7, 250–257. [Google Scholar] [CrossRef]
- Zhang, S.; Rousseau, A.; Dufour, T. Promoting lentil germination and stem growth by plasma activated tap water, demineralized water and liquid fertilizer. RSC Adv. 2017, 7, 31244–31251. [Google Scholar] [CrossRef]
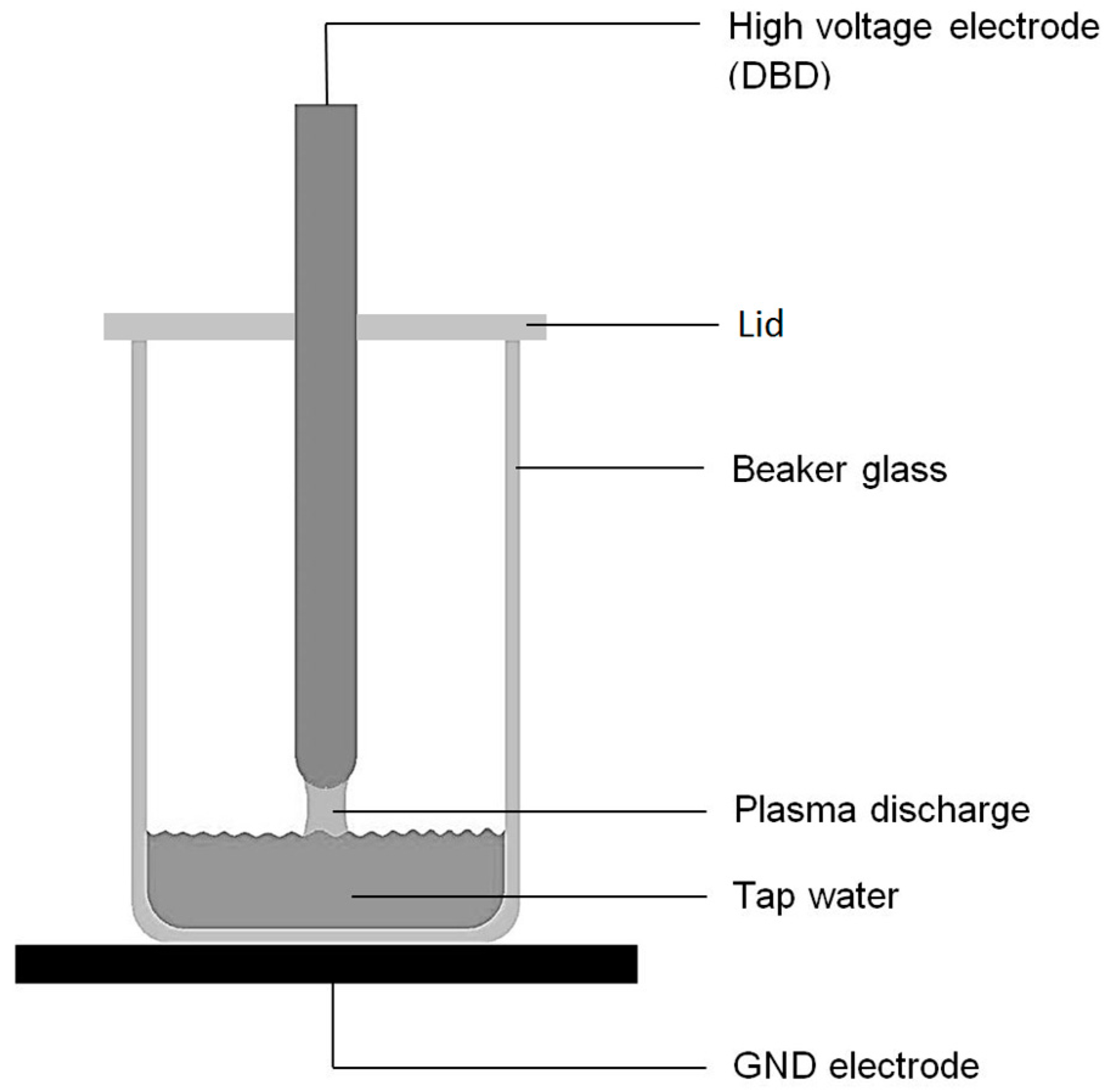

| Input Parameter | Value |
|---|---|
| Electric power | ≈11 W |
| Power density | ≈5 W/cm2 |
| Discharge gap | 3 mm |
| Appl. voltage | ≈30 kV (p-p) |
| Natural frequency | ≈200 kHz |
| Puls rep. Rate | 14 kHz |
| Waveform | decaying sine |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ten Bosch, L.; Köhler, R.; Ortmann, R.; Wieneke, S.; Viöl, W. Insecticidal Effects of Plasma Treated Water. Int. J. Environ. Res. Public Health 2017, 14, 1460. https://doi.org/10.3390/ijerph14121460
Ten Bosch L, Köhler R, Ortmann R, Wieneke S, Viöl W. Insecticidal Effects of Plasma Treated Water. International Journal of Environmental Research and Public Health. 2017; 14(12):1460. https://doi.org/10.3390/ijerph14121460
Chicago/Turabian StyleTen Bosch, Lars, Robert Köhler, Rinat Ortmann, Stephan Wieneke, and Wolfgang Viöl. 2017. "Insecticidal Effects of Plasma Treated Water" International Journal of Environmental Research and Public Health 14, no. 12: 1460. https://doi.org/10.3390/ijerph14121460
APA StyleTen Bosch, L., Köhler, R., Ortmann, R., Wieneke, S., & Viöl, W. (2017). Insecticidal Effects of Plasma Treated Water. International Journal of Environmental Research and Public Health, 14(12), 1460. https://doi.org/10.3390/ijerph14121460