Systematic Review and Meta-Analysis of Human Skin Diseases Due to Particulate Matter
Abstract
1. Introduction
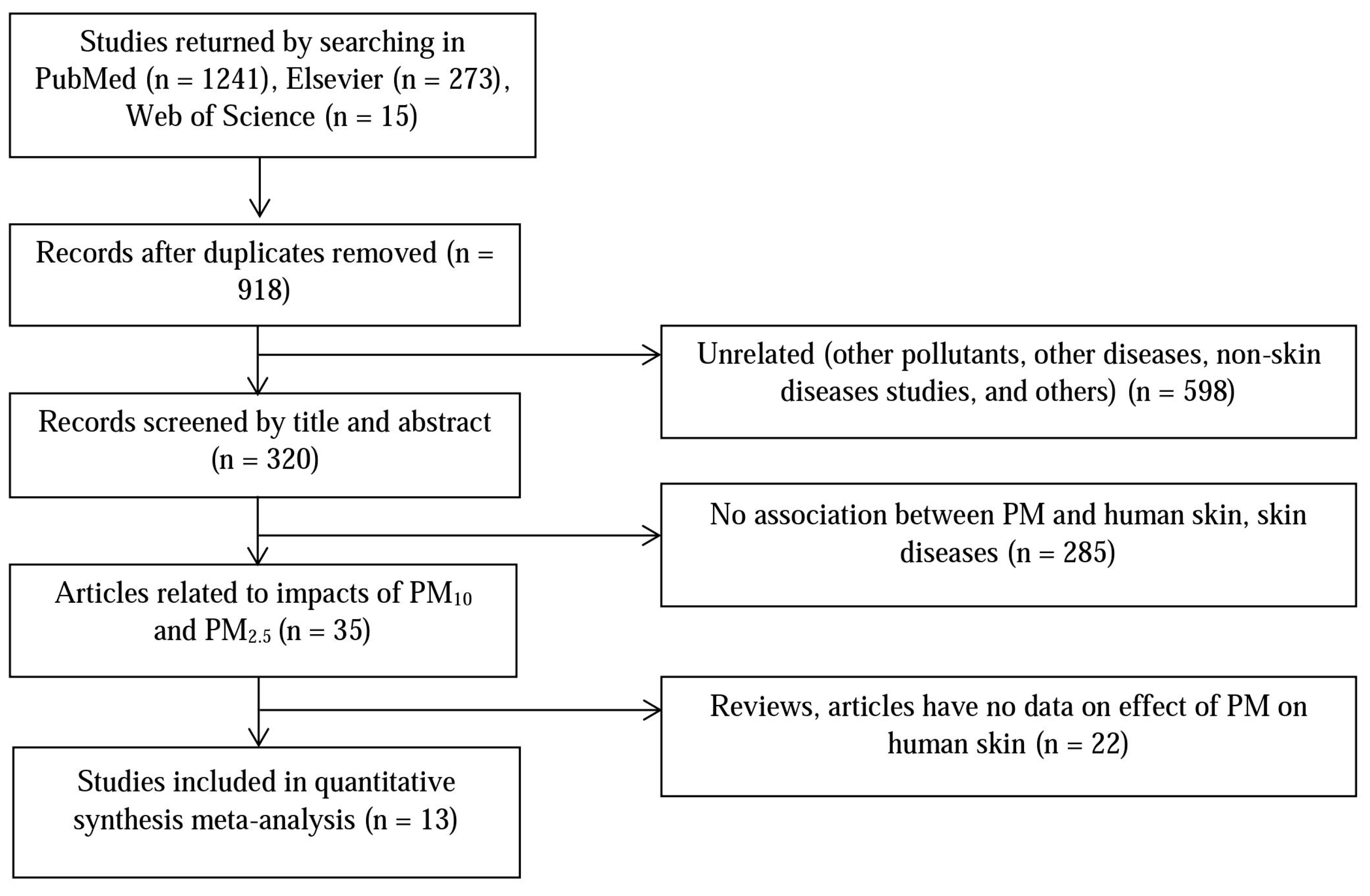
2. Materials and Methods
2.1. Literature Search and Data Extraction
2.2. Meta-Analysis
3. Results
4. Discussion
5. Conclusions
6. Limitations of Study
Acknowledgments
Author Contributions
Conflicts of Interest
References
- SOGA. State of Global Air. A Special Report on Global Exposure to Air Pollution and Its Disease Burden; Health Effects Institute: Cambridge, MA, USA, 2017; pp. 1–15. [Google Scholar]
- WHO. Ambient (Outdoor) Air Quality and Health. Available online: http://www.who.int/mediacentre/factsheets/fs313/en/ (accessed on 5 July 2017).
- Carlsten, C.; Melén, E. Air pollution, genetics, and allergy: An update. Curr. Opin. Allergy Clin. Immunol. 2012, 12, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Noh, J.; Sohn, J.; Cho, J.; Cho, S.K.; Choi, Y.J.; Kim, C.; Shin, D.C. Short-term effects of ambient air pollution on emergency department visits for asthma: An assessment of effect modification by prior allergic disease history. J. Prev. Med. Public Health 2016, 49, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Zeng, J.; Liu, K.; Bao, L.; Li, Y. Characterization and cytotoxicity of PM<0.2, PM0.2–2.5 and PM2.5–10 around mswi in Shanghai, China. Int. J. Environ. Res. Public Health 2015, 12, 5076–5089. [Google Scholar] [CrossRef]
- Vu, V.T.; Lee, B.K.; Kim, J.T.; Lee, C.H.; Kim, I.H. Assessment of carcinogenic risk due to inhalation of polycyclic aromatic hydrocarbons in PM10 from an industrial city: A Korean case-study. J. Hazard. Mater. 2011, 189, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, M.; Daneshvar, B.; Hansen, M.; Dragsted, L.O.; Hertel, O.; Knudsen, L.; Loft, S. Personal PM2.5 exposure and markers of oxidative stress in blood. Environ. Health Perspect. 2003, 111, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.E.; Choi, D.; Park, H.J. Air pollution and skin diseases: Adverse effects of airborne PM on various skin diseases. Life Sci. 2016, 152, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.C.; Kim, D.; Min, S.; Paul, C.; Yoo, Y.; Choung, J.T. Gis-based association between PM10 and allergic diseases in seoul: Implications for health and environmental policy. Ann. Allergy Asthma Immunol. 2016, 8, 32–40. [Google Scholar] [CrossRef]
- Achilleos, S.; Kioumourtzoglou, M.A.; Wu, C.D.; Schwartz, J.D.; Koutrakis, P.; Papatheodorou, S.I. Acute effects offine particulate matter constituents on mortality: A systematic review and meta-regression analysis. Environ. Int. 2017, 109, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J.; Liu, W.; Li, L.; Pan, X.; Crawford, M.; Sore, G.; Seite, S. Pollution and skin: From epidemiological and mechanistic studies to clinical implications. J. Dermatol. Sci. 2014, 76, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K. The role of air pollutants in atopic dermatitis. J. Allergy Clin. Immunol. 2014, 134, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.H.; Kim, S.; Lee, J.H.; Kim, J.; Han, Y.; Kim, Y.M.; Kim, G.B.; Jung, K.; Cheong, H.K.; Ahn, K. Indoor air pollution aggravates symptoms of atopic dermatitis in children. PLoS ONE 2015, 10, e0119501. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Lee, H.S.; Park, M.R.; Lee, S.W.; Kim, E.H.; Cho, J.B.; Kim, J.; Han, Y.; Jung, K.; Cheong, H.K.; et al. Relationship between indoor air pollutant levels and residential environment in children with atopic dermatitis. Ann. Allergy Asthma Immunol. 2014, 6, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Annesi-Maesano, I.; Moreau, D.; Caillaudc, D.; Lavaudd, F.O.; Moullece, Y.L.; Taytar, A.; Paulig, G.; Charpinh, D. Residential proximity fine particles related to allergic sensitisation and asthma in primary school children. Respir. Med. 2007, 101, 1721–1729. [Google Scholar] [CrossRef] [PubMed]
- Brauer, M.; Hoek, G.; Smit, H.A.; Jongste, J.C.D.; Gerritsen, J.; Postmae, D.S.; Kerkhof, M.; Brunekreef, B. Air pollution and development of asthma, allergy and infections in a birth cohort. Eur. Respir. J. 2007, 29, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Szyszkowicz, M.; Kousha, T.; Valacchi, G. Ambient air pollution and emergency department visits for skin conditions. Glob. Dermatol. 2016, 3, 323–329. [Google Scholar] [CrossRef]
- Weinmayr, G.; Romeo, E.; Sario, M.D.; Weiland, S.K.; Forastiere, F. Short-term effects of PM10 and NO2 on respiratory health among children with asthma or asthma-like symptoms: A systematic review and meta-analysis. Environ. Health Perspect. 2010, 118, 449–457. [Google Scholar] [CrossRef] [PubMed]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials revisited. Contemp. Clin. Trials 2015, 45, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Vierko, A.; Schikowski, T.; Ranft, U.; Sugiri, D.; Matsui, M.; Kramer, U.; Krutmann, J. Airborne particle exposure and extrinsic skin aging. J. Investig. Dermatol. 2010, 130, 2719–2726. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.J.; Tung, T.H.; Tangd, C.S.; Zhao, Z.H. Allergens, air pollutants, and childhood allergic diseases. Int. J. Hyg. Environ. Health 2015, 219, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Tang, K.T.; Ku, K.C.; Chen, D.Y.; Lin, C.H.; Tsuang, B.J.; Chen, Y.H. Adult atopic dermatitis and exposure to air pollutants—A nationwide population-based study. Ann. Allergy Asthma Immunol. 2017, 118, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Song, S.; Lee, K.; Lee, Y.M.; Lee, J.H.; Lee, S.; Yu, Y.D.; Paek, D. Acute health effects of urban fine and ultrafine particles on children with atopic dermatitis. Environ. Res. 2011, 111, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Peng, F.; Xue, C.H.; Hwang, S.K.; Li, W.H.; Chen, Z.; Zhang, J.Z. Exposure to fine PM associated with senile lentigo in chinese women: A cross-sectional study. J. Eur. Acad. Dermatol. Venereol. 2016, 31, 355–360. [Google Scholar] [CrossRef] [PubMed]
- MSa, C.Z.; Baïz, N.; Banerjee, S.; Charpin, D.A.; Caillaud, D.; Blay, F.D.; Raherison, C.; Lavaud, F.; Annesi-Maesano, I. The relationships between ambient air pollutants and childhood asthma and eczema are modified by emotion and conduct problems. Ann. Epidemiol. 2013, 23, 773–788. [Google Scholar] [CrossRef]
- Morgenstern, V.; Zutavern, A.; Cyrys, J.; Brockow, I.; Koletzko, S.; Krämer, U.; Behrendt, H.; Herbarth, O.; Berg, A.V.; Bauer, C.P.; et al. Atopic diseases, allergic sensitization, and exposure to traffic-related air pollution in children. ATS J. 2008, 135, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Kim, E.H.; Oh, I.; Jung, K.; Han, Y.S.; Cheong, H.K.; Ahn, K. Symptoms of atopic dermatitis are influenced by outdoor air pollution. J. Allergy Clin. Immunol. 2013, 1332, 495–498. [Google Scholar] [CrossRef] [PubMed]
- Shah, L.; Mainelis, G.; Ramagopal, M.; Black, K.; Shalat, S.L. Use of a robotic sampler (piper) for evaluation of PM exposure and eczema in preschoolers. Int. J. Environ. Res. Public Health 2016, 13, 242. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, J.; Lee, S.W.; Suh, J.; Yu, J.S.; Park, E.; Lee, J.; Kim, H.; Lee, K.S.; Chang, E.Y.; et al. The clinical effects of hospitalization in a low pollutant room on atopic dermatitis. Asia Pac. Allergy 2011, 1, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Gehring, U.; Wijga, A.H.; Brauer, M.; Fischer, P.; de Jongste, J.C.; Kerkhof, M.; Oldenwening, M.; Smit, H.A.; Brunekreef, B. Traffic-related air pollution and the development of asthma and allergies during the first 8 years of life. Am. J. Respir. Crit. Care Med. 2009, 181, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Kim, J.; Han, Y.; Jeon, B.H.; Cheong, H.K.; Ahn, K. Short-term effects of weather and air pollution on atopic dermatitis symptoms in children: A panel study in Korea. PLoS ONE 2017, 18, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.K.; Tsang, H.; Wong, C.M. Meta-analysis of adverse health effects due to air pollution in Chinese populations. BMC Public Health 2013, 13, 360–372. [Google Scholar] [CrossRef] [PubMed]
- Magnani, N.D.; Muresan, X.M.; Belmonte, G.; Cervellati, F.; Sticozzi, C.; Pecorelli, A.; Miracco, C.; Marchini, T.; Evelson, P.; Valacchi, G. Skin damage mechanisms related to airborne PM exposure. Toxicol. Sci. 2016, 149, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Pan, T.L.; Wang, P.W.; Aljuffali, I.A.; Huang, C.T.; Lee, C.W.; Fang, J.Y. The impact of urban particulate pollution on skin barrier function and the subsequent drug absorption. J. Dermatol. Sci. 2015, 78, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Wehner, M.R.; Phil, M.; Shive, M.L.; Chren, M.M.; Han, J.; Qureshi, A.A.; Linos, E. Indoor tanning and non-melanoma skin cancer: Systematic review and meta-analysis. BMJ-Br. Med. J. 2012, 345, 590–599. [Google Scholar] [CrossRef]
- WHO. Health Effects of Particulate Matter. 2013. Available online: http://www.euro.who.int/__data/assets/pdf_file/0006/189051/Health-effects-of-particulate-matter-final-Eng.pdf (accessed on 16 November 2017).
- EPA; Agency, U.S.E.P. Particulate Matter Concentrations. 2014. Available online: https://www.epa.gov/roe (accessed on 17 November 2017).
- Kathuria, P.; Silverberg, J.I. Association of pollution and climate with atopic eczema in US children. Pediatr. Allergy Immunol. 2016, 27, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.H.; Kim, J.S.; Kim, Y.C.; Kim, Y.S.; Chung, N.H.; Cho, M.H. Comparative study of PM2.5—And PM10—Induced oxidative stress in rat lung epithelial cells. J. Vet. Sci. 2004, 5, 11–18. Available online: https://www.ncbi.nlm.nih.gov/pubmed/15028881 (accessed on 15 May 2017).
- Diociaiuti, M.; Balduzzi, M.; Berardis, B.D.; Cattani, G.; Stacchini, G.; Ziemacki, G.; Marconi, A.; Paoletti, L. The two PM2.5 (fine) and PM2.5–10 (coarse) fractions: Evidence of different biological activity. Environ. Res. 2011, 86, 254–262. [Google Scholar] [CrossRef]
- Chen, R.; Hu, B.; Liu, Y.; Xu, J.; Yang, G.; Xu, D.; Chen, C. Beyond PM2.5: The role of ultrafine particles on adverse health effects of air pollution. Biochim. Biophys. Acta 2016, 1860, 2844–2855. [Google Scholar] [CrossRef]



| Reference | Year of Study | Location | Pollutant | Diagnosis | Total No. | Age |
|---|---|---|---|---|---|---|
| Vierko et al., 2010 [20] | 2008–2009 | Europe | PM10 | Pigment spots, wrinkles, skin aging | 400 | >18 |
| Song et al., 2011 [23] | 2009 | South Korea | PM10, PM2.5 | Skin itching | 670 | 8–12 |
| Peng et al., 2016 [24] | 2015 | China | PM2.5 | Skin itching | 611 | 10–15 |
| MSa et al., 2013 [25] | 2011 | France | PM10 | Current eczema | 518 | 10–12 |
| Wang et al., 2015 [21] | 2010 | China | PM10 | AD | 5925 | 8–15 |
| Morgenstern et al., 2008 [26] | 2005 | Germany | PM2.5 | Eczema | 10,750 | 4–6 |
| Kim et al., 2013 [27] | 2009–2010 | South Korea | PM10 | AD | 1880 | 8–12 |
| Tang et al., 2017 [22] | 2011 | Taiwan | PM10, PM2.5 | AD | 5115 | 20–30 |
| Shah et al., 2016 [28] | 2015 | United States | PM10 | Eczema | 128 | 28–30 |
| Brauer et al., 2007 [16] | 2004–2006 | Germany | PM2.5 | Eczema, itchy rash | 6982 | 3–6 |
| Lee et al., 2011 [29] | 2010 | South Korea | PM10 | AD | 51 | 2–3 |
| Gehring et al., 2009 [30] | 1996–2000 | Netherlands | PM2.5 | Allergen | 3863 | 8 |
| Kim et al., 2017 [31] | 2013–2014 | South Korea | PM10 | AD | 35,158 | 4–8 |
| Reference | Odds Ratio (95% (CI)) | Diagnosis | |
|---|---|---|---|
| PM10 | PM2.5 | ||
| Vierko et al., 2010 [20] | 1.08 (0.82–1.44) | Pigment spots | |
| 1.01 (0.68–1.49) | Wrinkles | ||
| 1.32 (0.93–1.90) | Skin aging | ||
| Song et al., 2011 [23] | 1.23 (0.79–1.93) | 0.67 (0.44–1.01) | Skin itching |
| Peng et al., 2016 [24] | 1.08 (0.77–1.52) | Skin itching | |
| MSa et al., 2016 [25] | 0.99 (0.65–1.51) | Current eczema | |
| Wang et al., 2015 [21] | 1.00 (0.83–1.21) | 1.25 (1.00–1.56) | AD |
| Morgenstern et al., 2008 [26] | 0.93 (0.82–1.05) | Eczema | |
| Kim et al., 2013 [27] | 0.76 (0.66–0.88) | AD | |
| Tang et al., 2017 [22] | 0.98 (0.89–1.08) | 1.05 (0.93–1.19) | AD |
| Shah et al., 2016 [28] | 0.87 (0.41–1.86) | Eczema | |
| Brauer et al., 2007 [16] | 1.10 (0.96–1.25) | Eczema | |
| 0.96 (0.82–1.12) | Itchy rash | ||
| Lee et al., 2011 [29] | 1.89 (0.76–4.74) | AD | |
| Gehring et al., 2009 [30] | 1.11 (1.00–1.24) | Allergen | |
| Kim et al., 2017 [31] | 1.02 (0.78–1.31) | AD | |
| Reference | Odds Ratio (95% CI) | |
|---|---|---|
| PM10 | PM2.5 | |
| Song et al., 2011 [23] | 1.23 (0.79–1.93) | 0.67 (0.44–1.01) |
| Wang et al., 2015 [21] | 1.00 (0.83–1.21) | 1.25 (1.00–1.56) |
| Peng et al., 2016 [24] | 1.08 (0.77–1.52) | |
| Kim et al., 2013 [27] | 0.76 (0.66–0.88) | |
| Tang et al., 2017 [22] | 0.98 (0.89–1.08) | 1.05 (0.93–1.19) |
| Brauer et al., 2007 [16] | 0.96 (0.82–1.12) | |
| Lee et al., 2017 [29] | 1.89 (0.76–4.74) | |
| Kim et al., 2017 [31] | 1.02 (0.78–1.31) | |
| Gehring et al., 2009 [30] | 1.11 (1.00–1.24) | |
| Summary relative risk (95% CI) | 0.96 (0.83–1.11) | 1.05 (0.95–1.16) |
| The coefficient heterogeneity I2 (%) | 62.7 | 46.0 |
| Reference | Location | Diagnosis | ER (%) Skin Disease (95% CI) | |
|---|---|---|---|---|
| PM10 | PM2.5 | |||
| Kim et al., 2013 [27] | South Korea | AD | 0.44 (0.12–0.77) | 0.67 (0.03–1.38) |
| Wang et al., 2015 [21] | China | AD | 1.54 (1.03–2.32) | |
| Kim et al., 2017 [31] | South Korea | AD | 3.20 (1.50–4.90) | |
| Morgenstern et al., 2008 [26] | Europe | Eczema | 1.00 (0.97–1.04) | |
| Song et al., 2011 [23] | South Korea | Skin itching | 1.03 (0.02–2.23) | 3.10 (0.20–6.10) |
| Kim et al., 2017 [31] | South Korea | AD | 0.36 (0.05–0.71) | |
| Msa et al., 2013 [25] | France | Eczema | 1.02 (0.84–1.24) | |
| Seo et al., 2015 [9] | South Korea | AD | 0.57 (0.23-0.98) | |
| Gehring et al., 2010 [30] | Netherlands | Allergen | 1.68 (0.41–2.07) | |
| Ahn Kangmo, 2015 [12] | South Korea | AD | 0.44 (0.16–0.74) | |
| Combined estimate | 1.01 (0.08–2.05) | 1.60 (0.45–2.82) | ||
| Reference | Location | Diagnosis | Pollutant | |
|---|---|---|---|---|
| PM10 (μg/m3) | PM2.5 (μg/m3) | |||
| Vierkotter et al., 2010 [20] | Germany | Skin aging | 6.50 | |
| Gehring et al., 2010 [30] | Netherlands | Allergen | 25.20 | |
| Kim et al., 2013 [27] | South Korea | AD | 50.50 | 25.60 |
| Peng et al., 2016 [24] | China | Skin itching | 35.20 | |
| Wang et al., 2015 [21] | China | AD | 48.32 | 29.07 |
| Shah et al., 2016 [28] | United States | Eczema | 56.26 | |
| Kim et al., 2017 [31] | South Korea | AD | 45.20 | |
| Brauer et al., 2007 [16] | Netherlands | Eczema | 25.20 | |
| Morgenstern et al., 2008 [26] | Europe | Eczema | 15.13 | |
| Song et al., 2011 [23] | South Korea | Skin itching | 44.89 | 22.38 |
| Tang et al., 2016 [22] | Taiwan | AD | 56.30 | 33.60 |
| Msa et al., 2013 [25] | France | Eczema | 31.00 | |
| Seo et al., 2015 [9] | South Korea | AD | 46.80 | |
| Szyszkowicz et al., 2016 [17] | New York | Cellulitis | 27.00 | |
| Hamilton | 33.50 | |||
| Halton | 34.20 | |||
| Combined estimate | 47.09 (42.01–52.17) | 26.04 (20.66–31.42) | ||
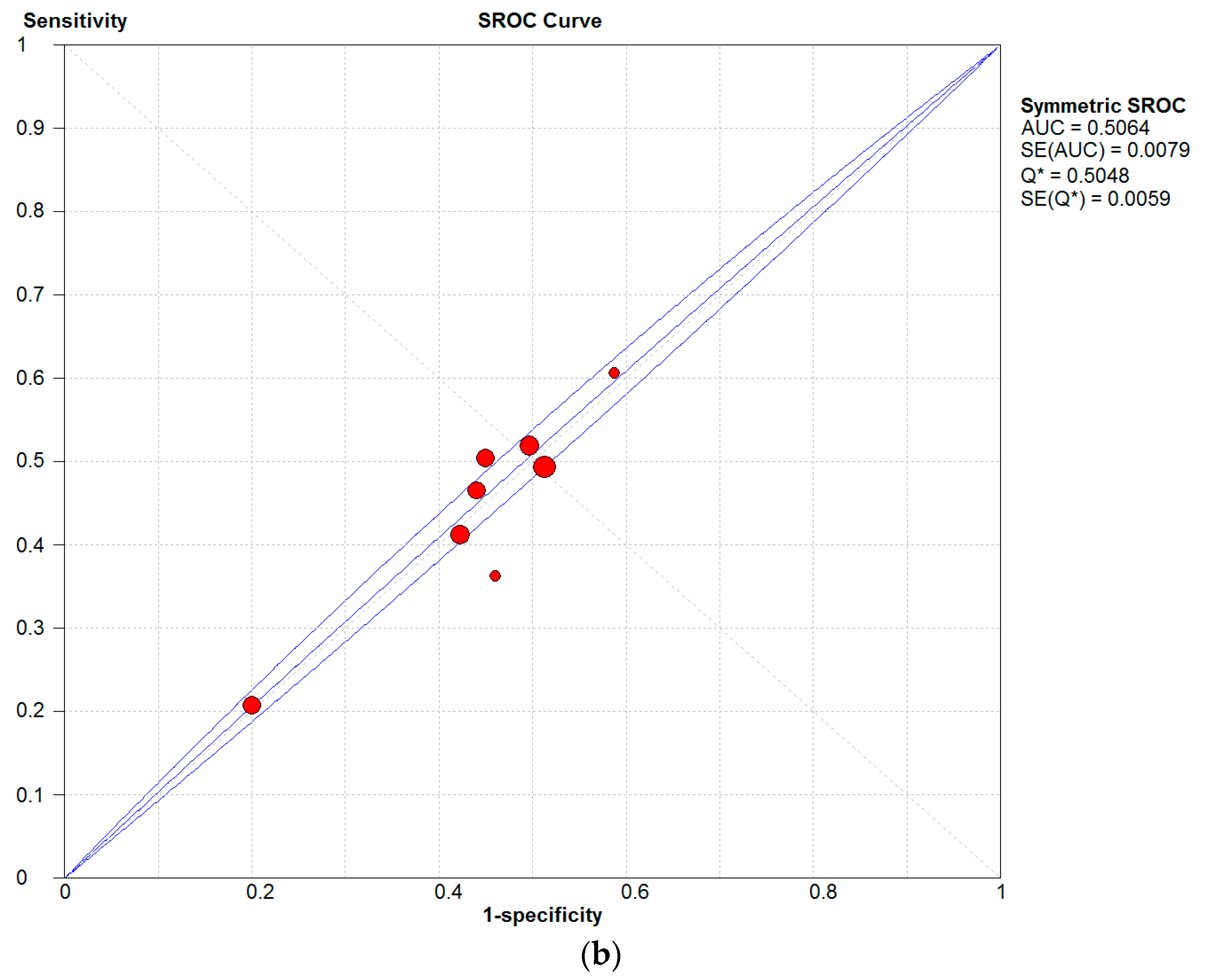
| Category | No. of Studies | Summary Sensitivity, % (95% CI) | Summary Specificity, % (95% CI) | Q * (%) |
|---|---|---|---|---|
| AD | 7 | 26.4 (25.5–27.4) | 46.5 (46.1–46.9) | 50.25 |
| Eczema | 4 | 47.2 (45.2–49.3) | 50.5 (49.8–51.2) | 50.09 |
| Skin allergen | 6 | 54.3 (51.3–57.3) | 49.5 (47.7–51.3) | 49.76 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngoc, L.T.N.; Park, D.; Lee, Y.; Lee, Y.-C. Systematic Review and Meta-Analysis of Human Skin Diseases Due to Particulate Matter. Int. J. Environ. Res. Public Health 2017, 14, 1458. https://doi.org/10.3390/ijerph14121458
Ngoc LTN, Park D, Lee Y, Lee Y-C. Systematic Review and Meta-Analysis of Human Skin Diseases Due to Particulate Matter. International Journal of Environmental Research and Public Health. 2017; 14(12):1458. https://doi.org/10.3390/ijerph14121458
Chicago/Turabian StyleNgoc, Le Thi Nhu, Duckshin Park, Yongil Lee, and Young-Chul Lee. 2017. "Systematic Review and Meta-Analysis of Human Skin Diseases Due to Particulate Matter" International Journal of Environmental Research and Public Health 14, no. 12: 1458. https://doi.org/10.3390/ijerph14121458
APA StyleNgoc, L. T. N., Park, D., Lee, Y., & Lee, Y.-C. (2017). Systematic Review and Meta-Analysis of Human Skin Diseases Due to Particulate Matter. International Journal of Environmental Research and Public Health, 14(12), 1458. https://doi.org/10.3390/ijerph14121458








