Distribution Characteristics and Risk Assessment of Polycyclic Aromatic Hydrocarbons in the Momoge Wetland, China
Abstract
:1. Introduction
2. Materials and Methods
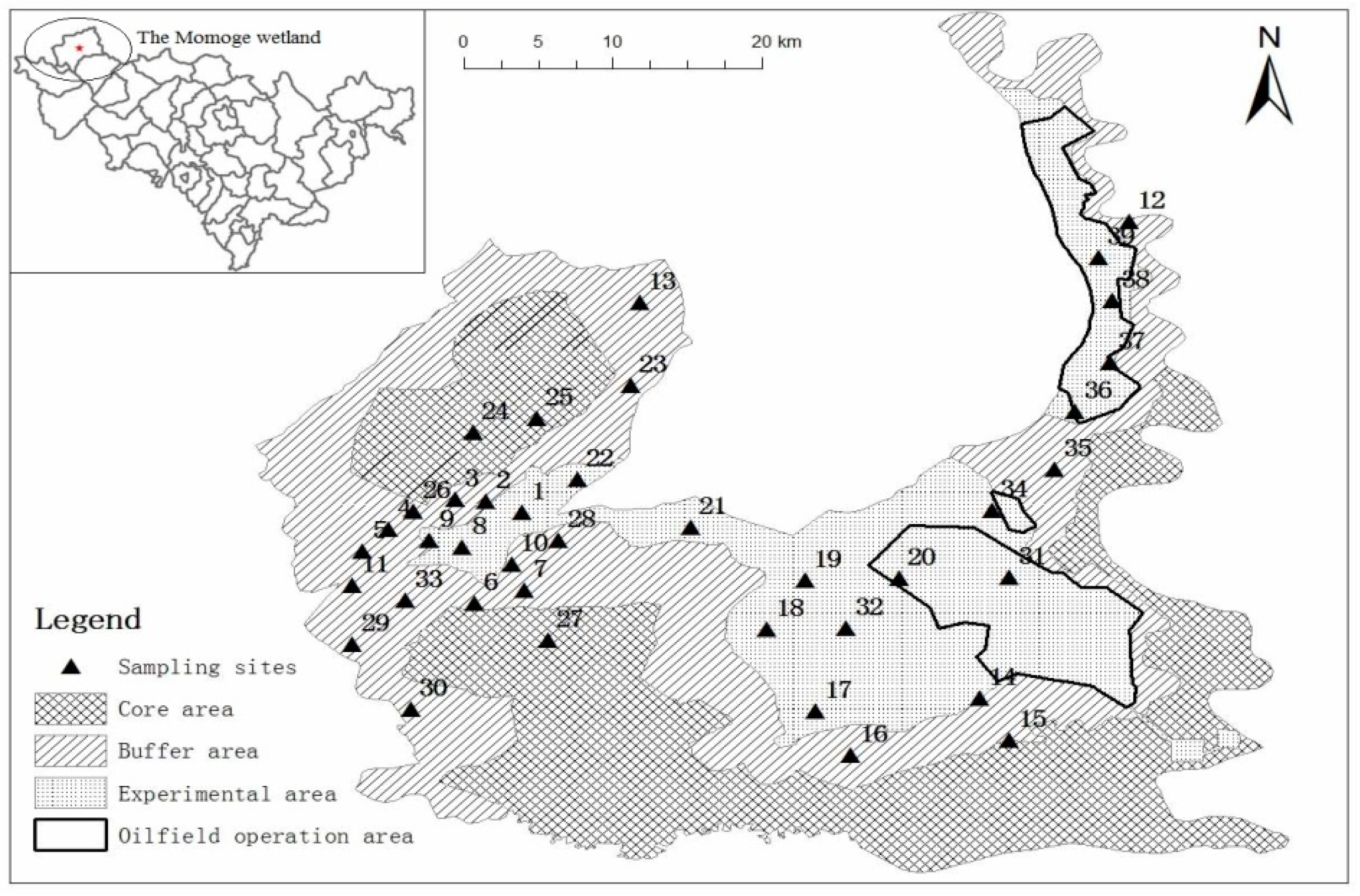
2.1. Study Area
2.2. Collection and Preservation of Soil Samples
2.3. Laboratory Instruments and Reagents
2.4. Chemical Analysis of Soil Samples
2.5. Data Analysis
2.6. Analysis and Evaluation Method
2.6.1. Sourceidentifications
2.6.2. Evaluation of Wetland Soil Pollution
2.6.3. Ecological Risk Assessment
3. Contents and Characteristics of PAHs in the Momoge Wetland
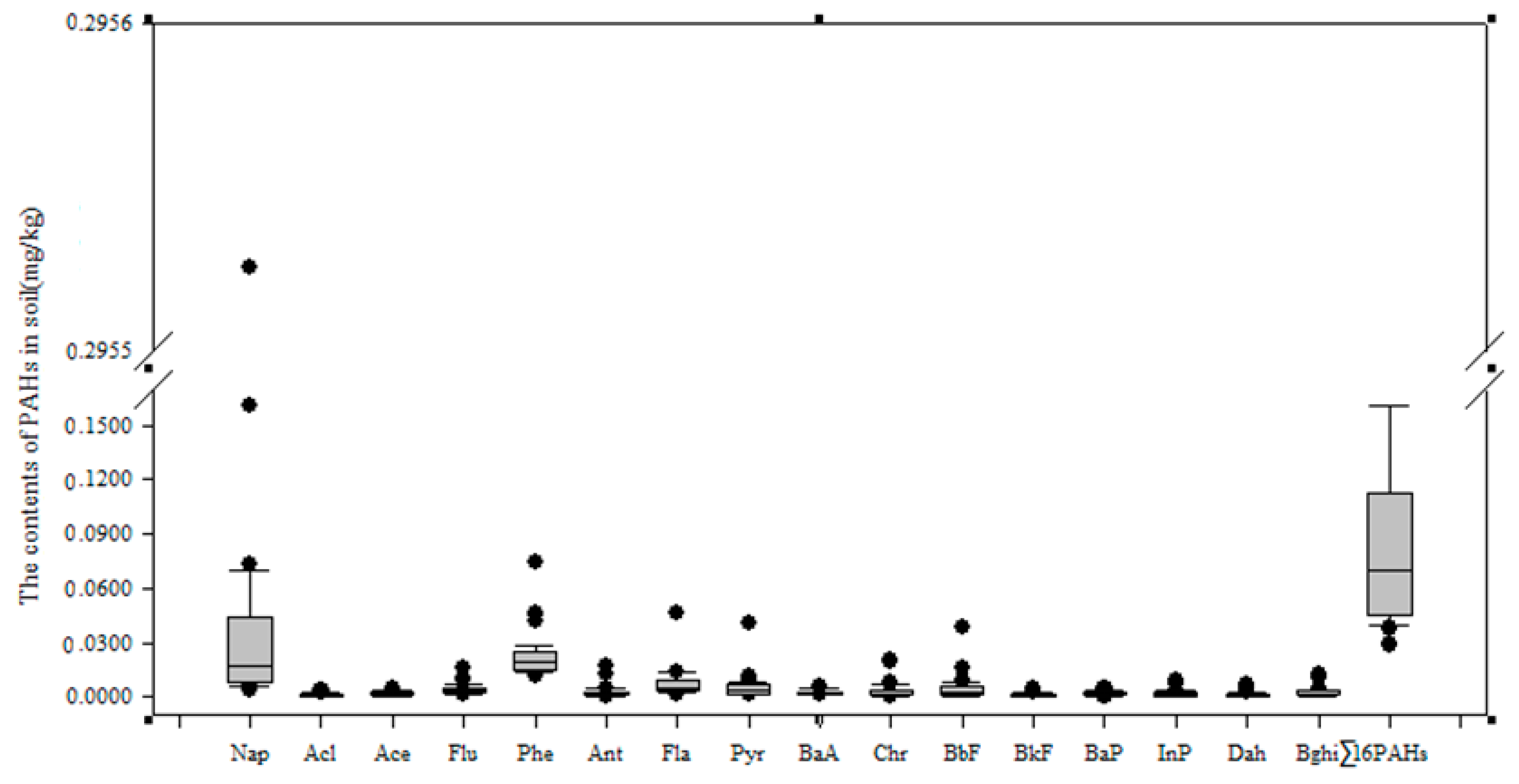
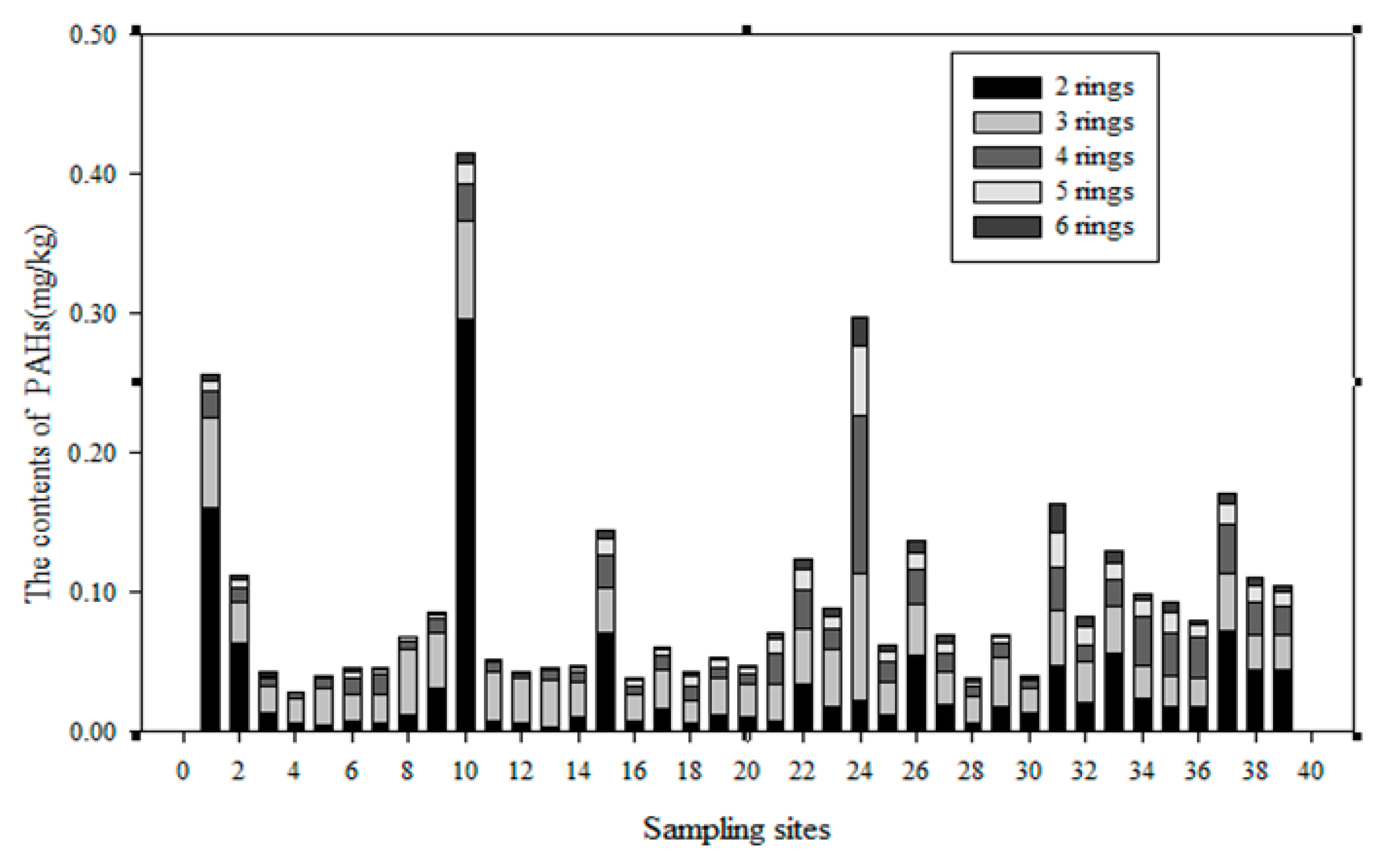
3.1. The PAH Content in the Soils of the Momoge Wetland
3.2. Compounds Analysed in PAHs in Soils of the Momoge Wetland
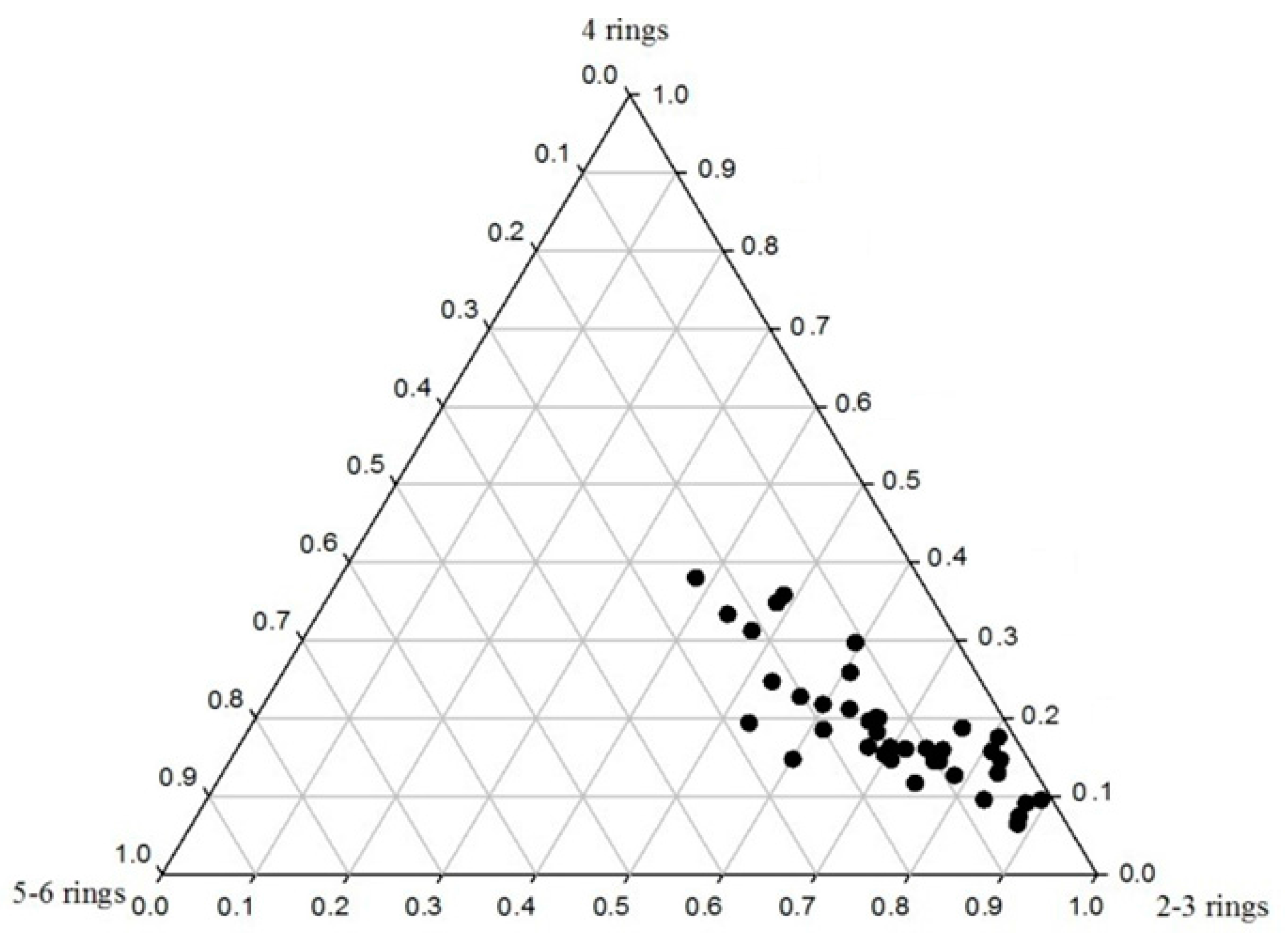
3.3. Distribution of PAHs in the Soils of the Momoge Wetland
4. Source Identification of PAHs in Soils of the Momoge Wetland
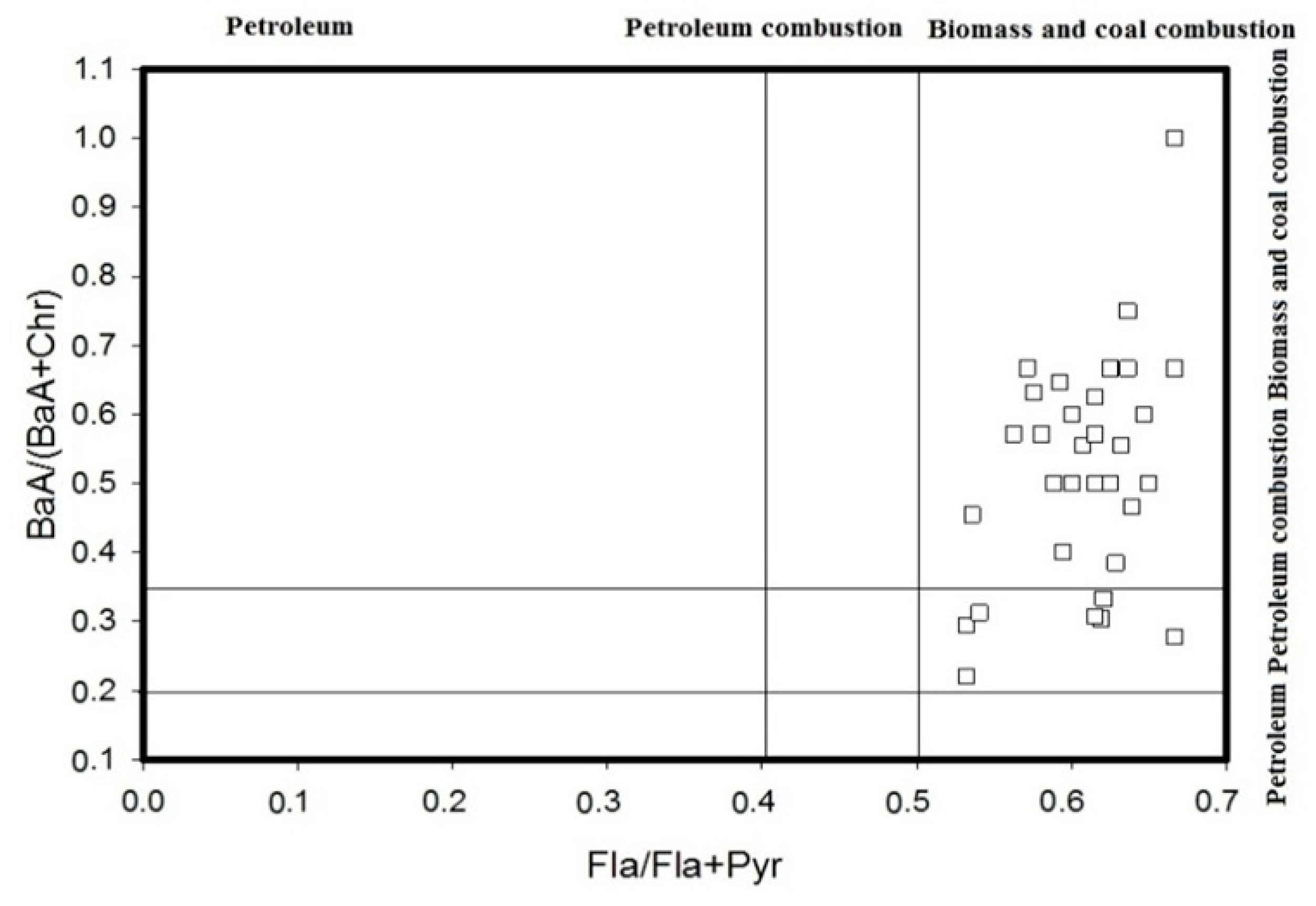
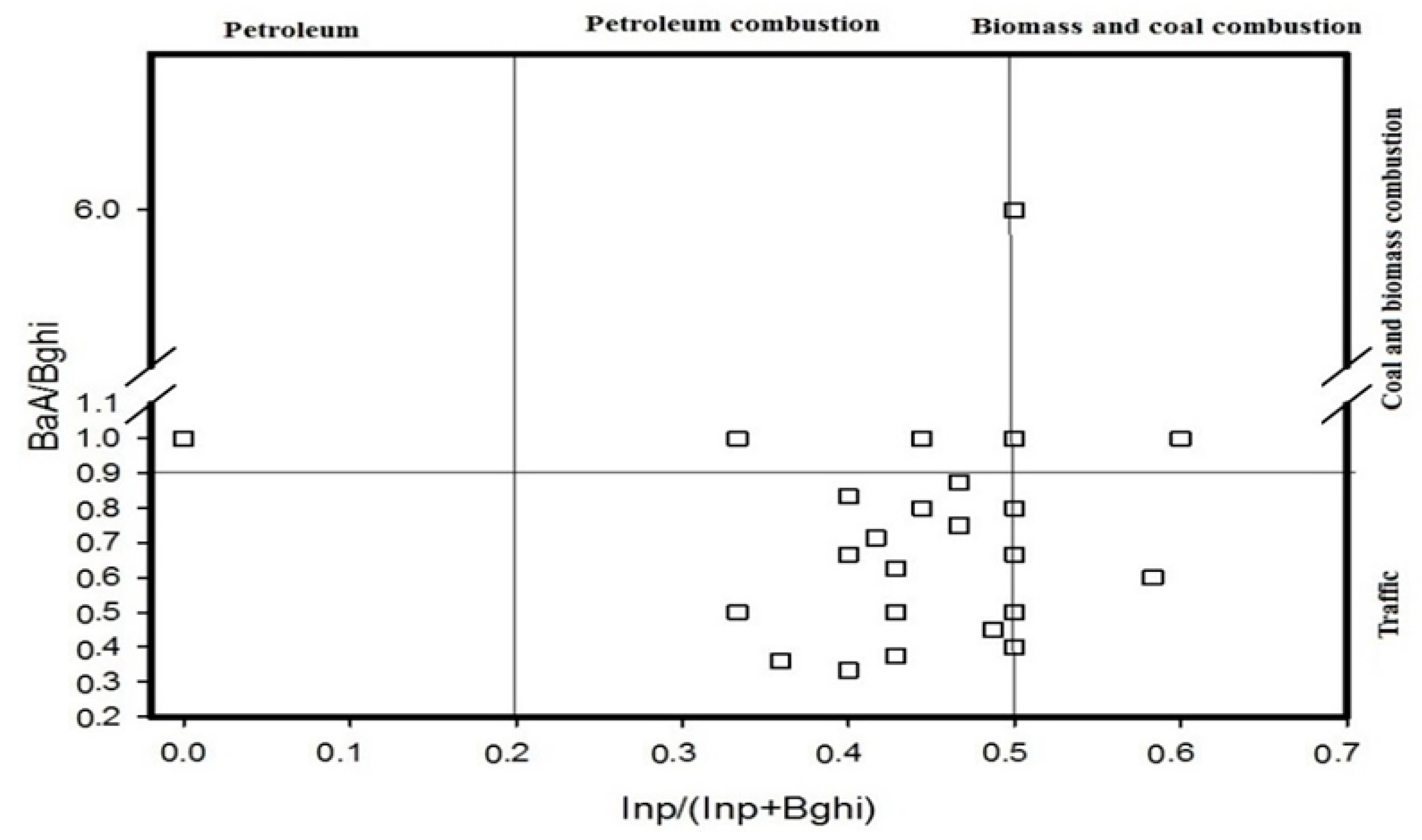
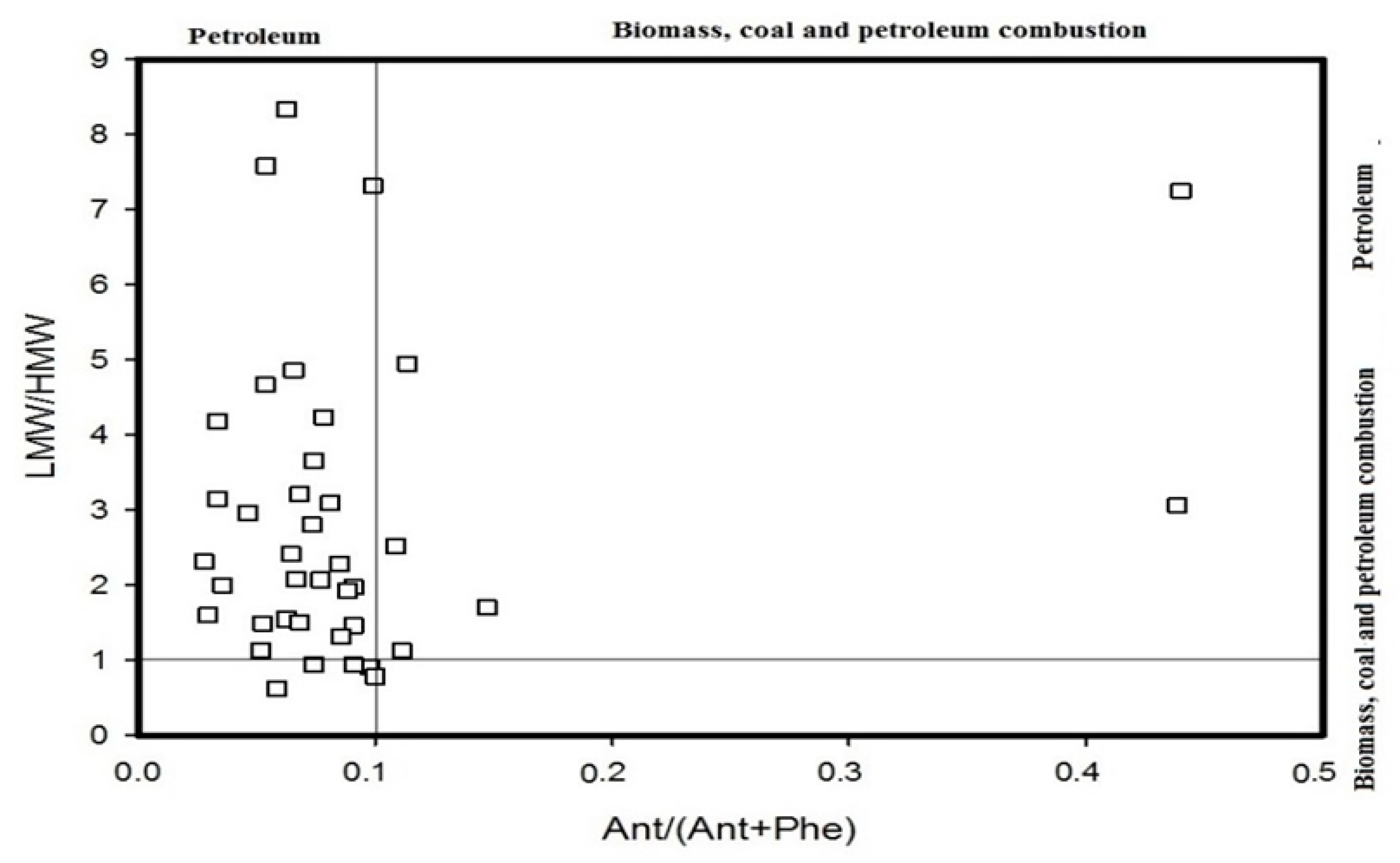
4.1. Diagnostic Ratio Method
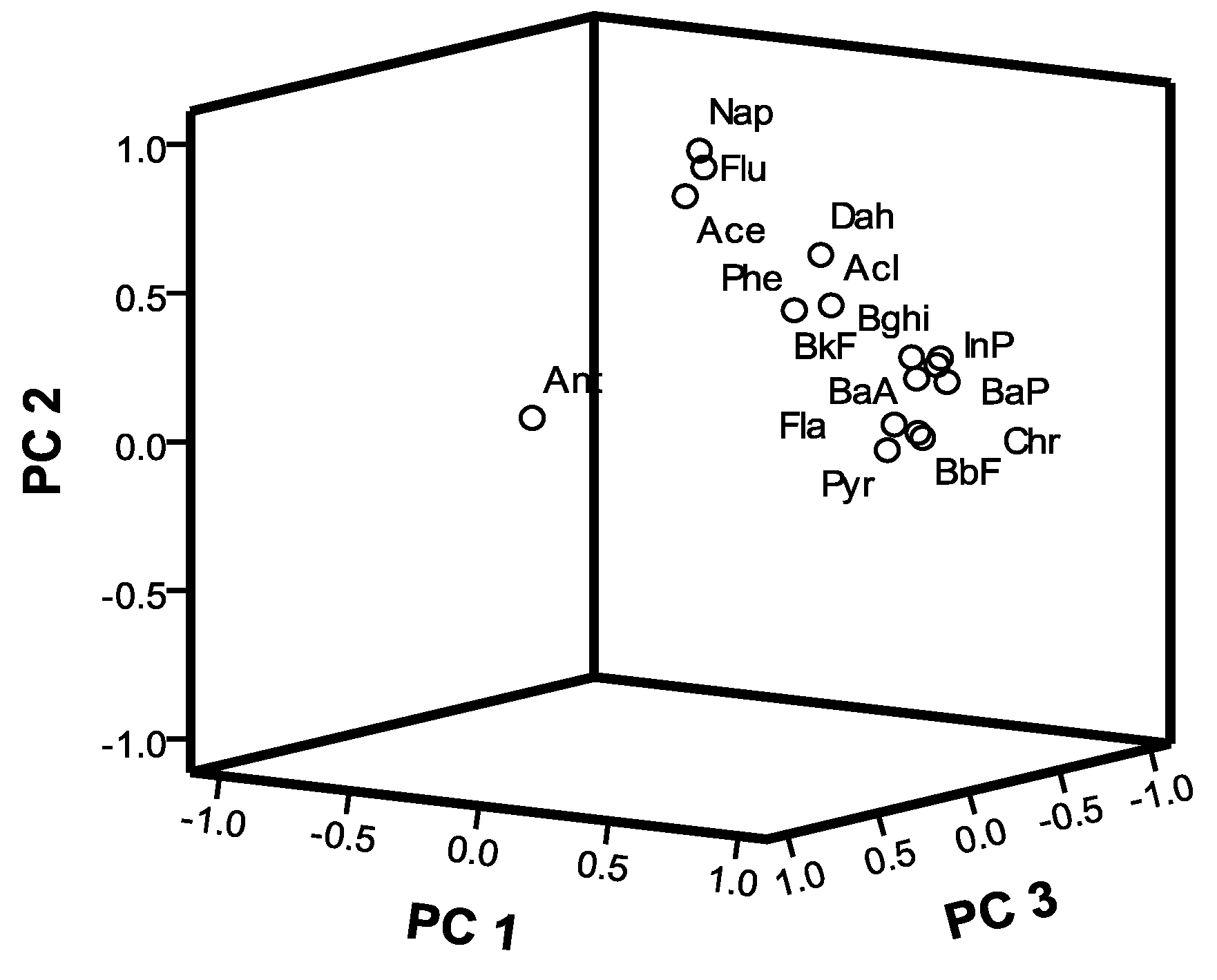
4.2. PCA-MLR
5. Pollution Assessment and Ecology Risk Assessment of PAHs in Soils of the Momoge Wetland
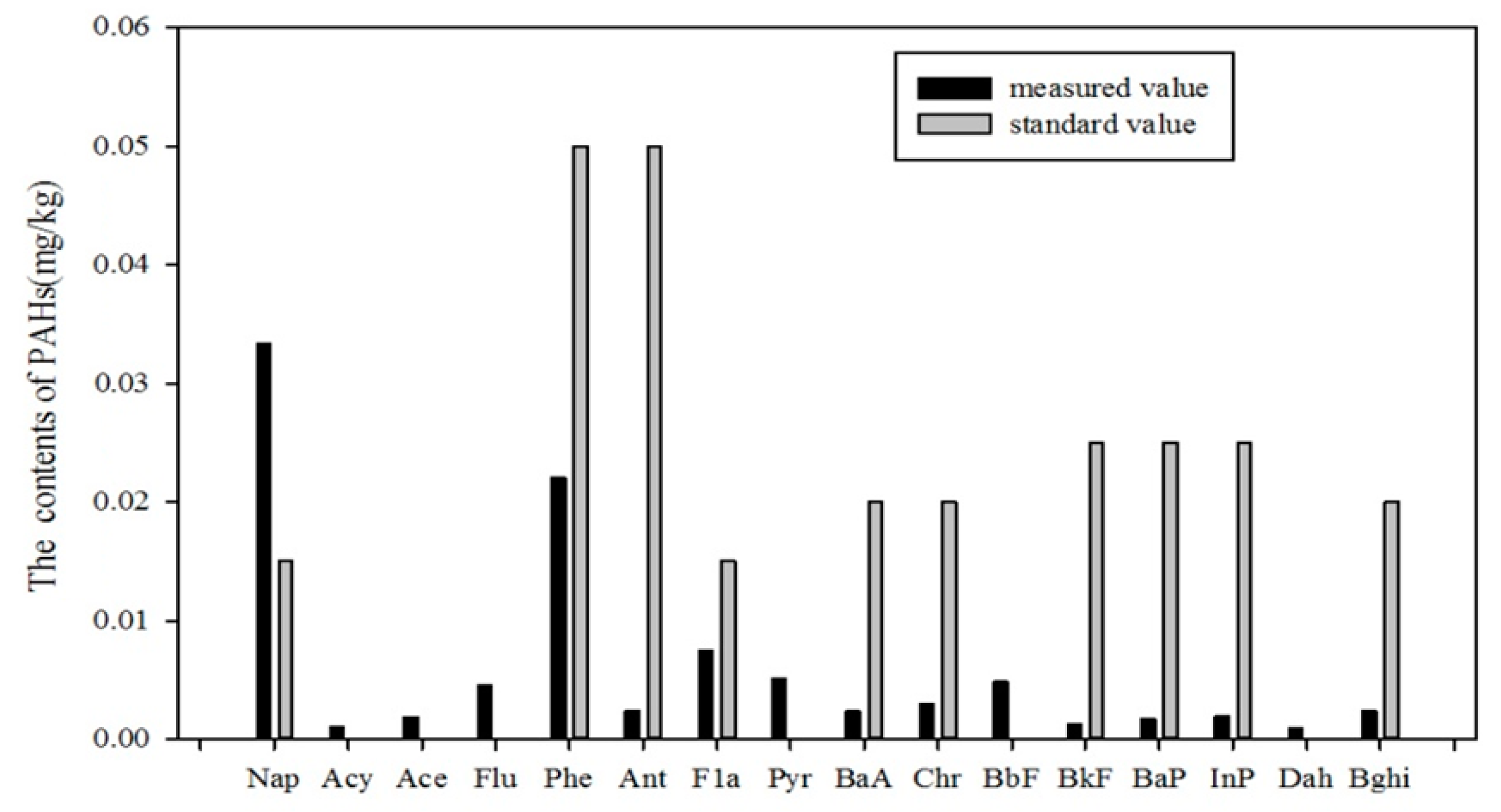
5.1. Pollution Assessment of PAHs in Soils of the Momoge Wetland
5.1.1. Standard Index Method
5.1.2. Pollution Interval Value Method
5.2. Ecological Risk Assessment of PAHs in Soils in the Momoge Wetland
5.2.1. Biological Toxicity Assessment
5.2.2. Normalization of Organic Carbon
6. Conclusions
- (1)
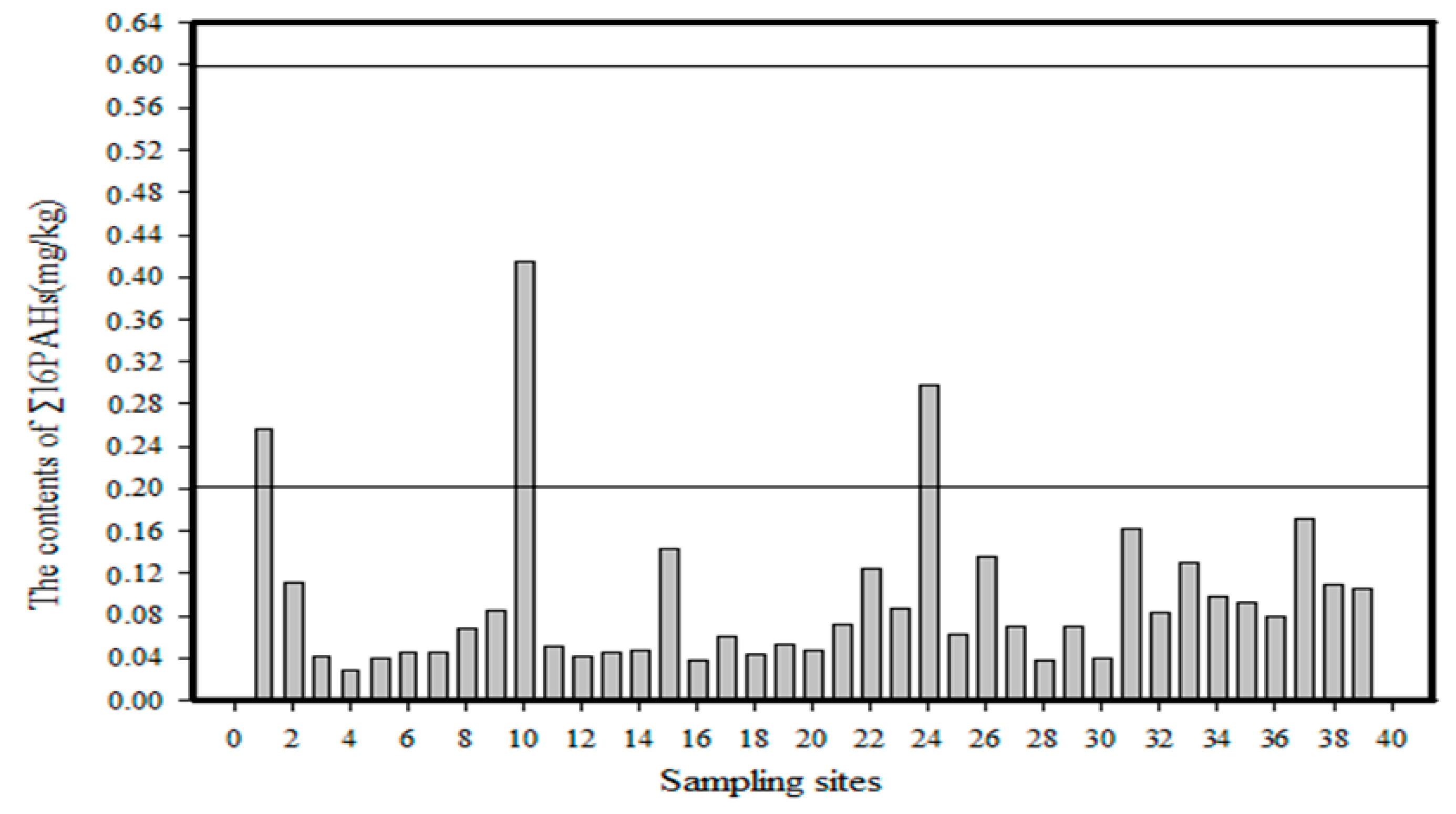
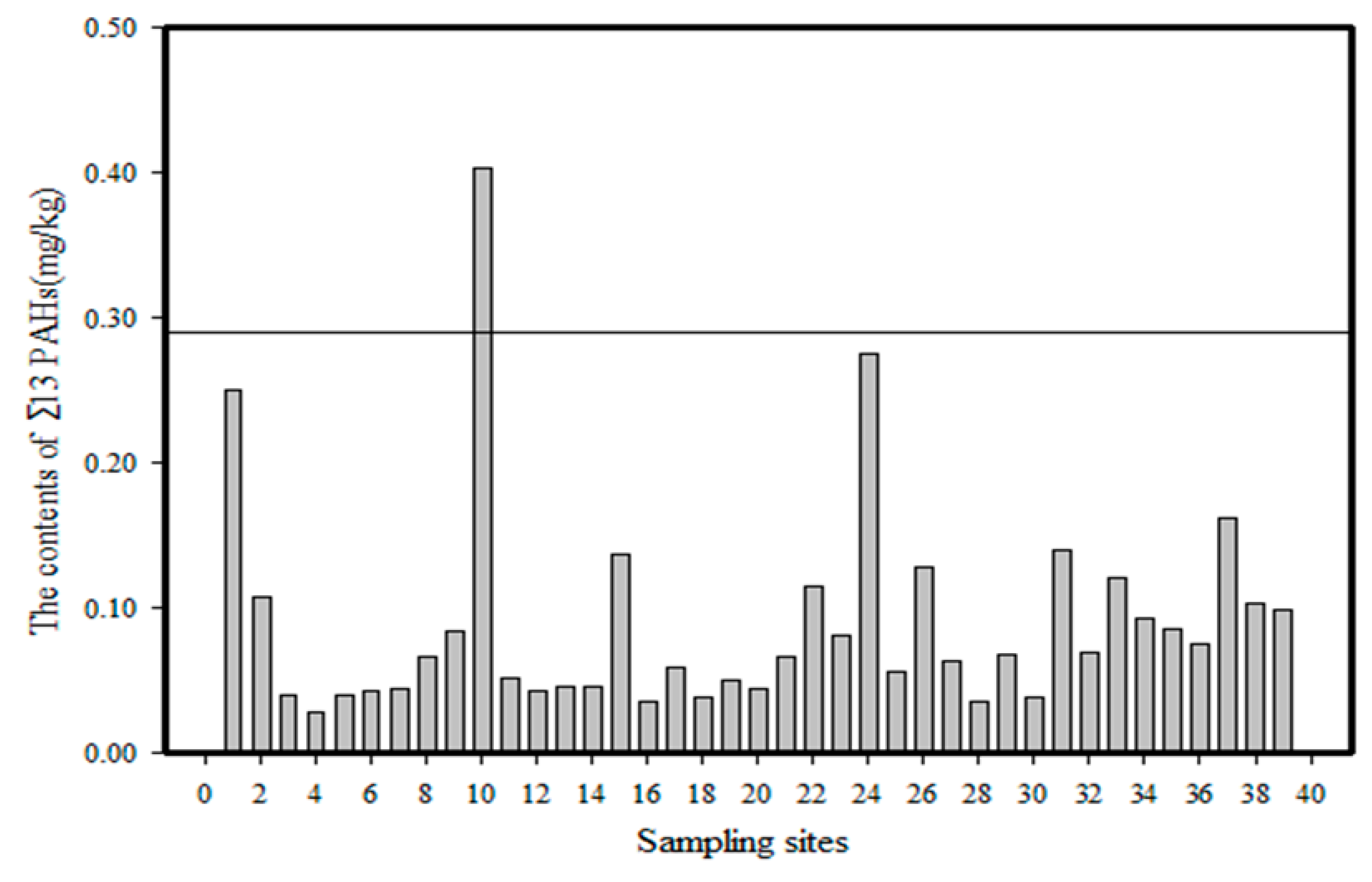
- The summarised PAH content (Σ16 PAHs) in the Momoge wetland soil samples was in the range (0.029–0.4152) mg/kg. The average content was 0.096 mg/kg. PAHs in wetland soil were primarily 2–3-rings PAHs. The content of Σ16 PAHs in the No. 10 soil sample was the highest, and that in the No. 4 soil sample was the lowest.
- (2)
- The diagnostic ratio and PCA-MLR model methods were utilized to conduct source identifications. The results showed that PAHs in the Momoge Wetland soil have multiple sources: petroleum source, combustion of petroleum and combustion of coal. The summation of the combustion of petroleum and coal sources account for 38.0% and 59.3%, respectively.
- (3)
- By comparing local data with international standards and pollution range methods, we could reach the conclusion that the Nap content in the Momoge wetland soil is excessive, and that there are some sampling sites that exhibit low-grade contamination. The result of biotoxicity and ecological assessments shows that there are two sampling sites with occasional ecological toxic hazards. The result of the organic carbon normalization method shows that only at the 10th sample point does ecological risk currently exist.
- (4)
- The sources of PAHs in the Momoge Wetland soil are multiple. It should be noted that, in the future, the soil PAHs should be monitored more often and associated with stronger protective measures.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wu, Y.; Hao, J.; Li, P. Spatial distribution and change of metal elements in soil in Mount Beigu wetland. J. Jiangsu Univ. (Nat. Sci. Ed.) 2005, 26, 340–344. (In Chinese) [Google Scholar]
- State Forestry Administration Bureau of China. 2nd National Wetland Resource Survey. Available online: http://www.forestry.gov.cn/main/65/content-758154.html (accessed on 16 November 2016).
- Zhu, X.; Sun, Q. Review of Wetland Research. J. Jiangxi For. Technol. 2007, 3, 47–49. (In Chinese) [Google Scholar]
- He, C.; Zhao, K.; Yu, G.; Zhao, Z.; Wang, G. Research Progress of Wetland Ecological Process. Adv. Earth Sci. 2000, 15, 164–171. [Google Scholar]
- Liu, Q.; Xie, W.; You, J.; Zhao, X.; Lu, Z. Environmentally Chemical Behaviors of Heavy Metals in Wetland Sediments: A Review. Soils 2013, 45, 8–16. (In Chinese) [Google Scholar]
- Xu, X.; Huang, S. Advances in studies on migration and transformation of polycyclic aromatic hydrocarbons in surface water. Environ. Sci. Technol. 2011, 34, 26–33. [Google Scholar]
- Wise, S.A.; Sander, L.C.; Schantz, M.M. Analytical Methods for Determination of Polycyclic Aromatic Hydrocarbons (PAHs)—A Historical Perspective on the 16 U.S. EPA Priority Pollutant PAHs. Polycycl. Aromat. Compd. 2015, 35, 109–134. [Google Scholar] [CrossRef]
- Ma, J.; Zhai, Y.; Wang, D. PAHs Enrichment and Influence in the Songhua River water environment on ecological environment. Environ. Sci. Manag. 2006, 31, 91–92. [Google Scholar]
- Dong, J. Modelling Environmental Fate and Risk Assessment of Polycyclic Aromatic Hydrocarbons in the Lanzhou Area. Doctoral Thesis, Lanzhou University, Lanzhou, China, May 2010. [Google Scholar]
- Cao, W.; Qiao, M.; Zhang, Y.; Liu, B. Removal of Polycyclic Aromatic Hydrocarbons (PAHs) and Their Derivatives in Typical Sewage Treatment Plants and Health Risk of Reclaimed Water. Asian J. Ecotoxicol. 2016, 11, 173–179. [Google Scholar]
- Momoge National Natural Reserve of Jilin Province. Available online: http://mmgnr.forestry.gov.cn/publicfiles/business/htmlfiles/mmgbhq/stzy/201305/16530.html (accessed on 16 November 2016).
- Pan, X.; Zhang, D.; Quan, L. Interactive factors leading to dying-off Carextato in Momoge wetland polluted by crude oil, Western Jilin, China. Chemosphere 2006, 65, 1772–1777. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Zhang, N.; Cao, Y. Protection and development of tourism resource in Momoge national natural reserve. J. Northeast Norm. Univ. (Nat. Sci. Ed.) 2005, 37, 109–114. (In Chinese) [Google Scholar]
- U.S. EPA. Method 3570: Microscale Solvent Extraction from Solids. Washington, DC: US Environmental Protection Agency; 2002. Available online: http://www.epa.gov/epaoswer/hazwaste/test/pdfs/3570.pdf (assessed on 17 November 2016). [Google Scholar]
- U.S. EPA. Method 8270D: Semivolatile Organic Compounds by Gas Chromatography/Mass Spectrometry (GC/MS). Washington, DC: US Environmental Protection Agency; 1996. Available online: http://www.epa.gov/epawaste/hazard/testmethods/sw846/pdfs/8270d.pdf (assessed on 17 November 2016). [Google Scholar]
- Zhao, L.; Hou, H.; Shangguan, Y.; Cheng, B.; Xu, Y.; Zhao, R.; Zhang, Y.; Hua, X. Occurrence, sources, and potential human health risks of polycyclic aromatic hydrocarbons in agricultural soils of the coal production area surrounding Xinzhou, China. Ecotoxicol. Environ. Saf. 2014, 108, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Soclo, H.H.; Garrigues, P.; Ewald, M. Origin of polycyclic aromatic hydrocarbons (PAHs) in coastal marine sediments: Case studies in Cotonou (Benin) and Aquitaine (France) areas. Mar. Pollut. Bull. 2000, 40, 387–396. [Google Scholar] [CrossRef]
- Yunker, M.B.; Macdonald, R.W.; Goyette, D.; Paton, D.W.; Fowler, B.R.; Sullivan, D.; Boyd, J. Natural and anthropogenic inputs of hydrocarbons to the Strait of Georgia. Sci. Total Environ. 1999, 225, 181–209. [Google Scholar] [CrossRef]
- Sofowote, U.M.; Mccarry, B.E.; Marvin, C.H. Source apportionment of PAH in Hamilton Harbour suspended sediments: Comparison of two factor analysis methods. Environ. Sci. Technol. 2008, 42, 6007–6014. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Shen, Y.; Zhang, S.; Ye, Y.; Shen, Q.; Hu, J.; Wang, X. Application of spatial analysis and multivariate analysis techniques in distribution and source study of polycyclic aromatic hydrocarbons in the topsoil of Beijing, China. Environ. Geol. 2009, 56, 1041–1050. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, L.; Niu, J.; Wang, J.; Shen, Z. Pollution assessment and source identifications of polycyclic aromatic hydrocarbons in sediments of the Yellow River Delta, a newly born wetland in China. Environ. Monit. Assess. 2009, 158, 561–571. [Google Scholar] [CrossRef] [PubMed]
- Dutch Ministry of Housing, Spatial Planning and Environment. Dutch Target and Intervention Values; ANNEXES Circular on Target Values and Intervention Values for Soil Remediation. Available online: http://159.226.251.229/videoplayer/annexS_I2000Dutch%20Environmental%20Standards.pdf?ich_u_r_i=954de0071dbc21ae8bf19c80e0e18d9d&ich_s_t_a_r_t=0&ich_e_n_d=0&ich_k_e_y=1745018916750263292454&ich_t_y_p_e=1&ich_d_i_s_k_i_d=6&ich_u_n_i_t=1 (accessed on 17 November 2016).
- Barbara, M.K. Polycyclic aromatic hydrocarbons in agricultural soils in Poland: Preliminary proposals for criteria to evaluate the level of soil contamination. Appl. Geochem. 1996, 11, 121–127. [Google Scholar]
- Long, E.R.; Macdonald, D.D.; Smith, S.L.; Calder, F.D. Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuary sediments. Environ. Manag. 1995, 19, 81–97. [Google Scholar] [CrossRef]
- Swartz, R.C. Consensus sediment quality guidelines for polycyclic aromatic hydrocarbons mixtures. Environ. Toxicol. Chem. 1999, 18, 780–787. [Google Scholar] [CrossRef]
- Zhou, H.; Zhao, J.; Lu, J.; Wang, Y.; Yuan, H.; Wan, X. Distribution, sources and ecological risk assessment of polycyclic aromatic hydrocarbons in sediments from Baiyangdian wetland. Asian J. Ecotoxicol. 2008, 3, 291–299. (In Chinese) [Google Scholar]
- Wang, Z.; Liu, Z.; Yang, Y.; Liu, M. Distribution of PAHs in tissues of wetland plants and the surrounding sediments in the Chongming wetland, Shanghai, China. Chemosphere 2012, 89, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Lang, Y.; Li, G.; Liu, W. Distribution of polycyclic aromatic hydrocarbons (PAHs) and health risk assessment in Jiaozhou bay wetland. Environ. Chem. 2013, 10, 1989–1990. (In Chinese) [Google Scholar]
- Liao, S.; Lang, Y.; Wang, Y. Distribution and sources of PAHs in soil from Liaohe Estuarine wetland. Environ. Sci. 2011, 32, 1094–1100. [Google Scholar]
- Yuan, H.; Ye, S.; Gao, M.; He, X. Distribution and Ecological Risk Assessment of Polycyclic Aromatic Hydrocarbons (PAHs) in the Surface Soil of Wetlands in the Southern Yellow River. Mar. Geol. Front. 2011, 27, 24–28. (In Chinese) [Google Scholar]
- Yuan, H.; Zhao, G.; Pang, S.; Gao, G.; Ye, S. Polycylic aromatic hydrocarbons (PAHs) exposure and their source analysis in the northern wetland of the yellow river delta. Mar. Geol. Quat. Geol. 2008, 26, 57–62. (In Chinese) [Google Scholar]
- Zhang, Y.; Liu, M.; Chen, H.; Hou, G. Source identification of polycyclic aromatic hydrocarbons in different ecological wetland components of the Qinkenpao Wetland in Northeast China. Ecotoxicol. Environ. Saf. 2014, 102, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. The Distribution and Source Analysis of PAHs in the Typical Wetlands of Sanjiang Plain. Master’s Thesis, Jilin University, Changchun, China, June 2013. [Google Scholar]
- Liu, J.; Qi, S.; Zhang, G.; Zhang, W. Polycyclic aromatic hydrocarbons in Lalu wetland of Lasa city and their source. Geophys. Geochem. Explor. 2003, 27, 490–492. (In Chinese) [Google Scholar]
- R, X.; Bai, J.; Wang, J.; Lu, Q.; Zhao, Q.; Cui, B.; Liu, X. Polycyclic aromatic hydrocarbons (PAHs) in wetland soils under different land uses in a coastal estuary: Toxic levels, sources and relationships with soil organic matter and water-stable aggregates. Chemosphere 2014, 110, 8–16. [Google Scholar]
- Kimbrough, K.L.; Dickhut, R.M. Assessment of polycyclic aromatic hydrocarbon input to urban wetlands in relation to adjacent land use. Mar. Pollut. Bull. 2006, 52, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Sartori, F.; Wade, T.L.; Sericano, J.T.; Mohanty, B.P.; Smith, K.A. Polycyclic aromatic hydrocarbons in soil of the Canadian river floodplain in Oklahoma. J. Environ. Manag. 2010, 39, 568–579. [Google Scholar] [CrossRef] [PubMed]
| PAHs | Rate of Recovery (%) | PAHs | Rate of Recovery (%) |
|---|---|---|---|
| Nap | 89–109 | BaA | 103–125 |
| Acl | 107–116 | Chr | 87–106 |
| Ace | 92–113 | BbF | 110–116 |
| Flu | 100–125 | BkF | 101–109 |
| Phe | 93–116 | BaP | 90–115 |
| Ant | 99–123 | Ind | 98–109 |
| F1a | 105–129 | Dah | 90–91 |
| Pyr | 112–101 | Bghi | 104–106 |
| District | Range of the Contents of 16 PAHs (mg/kg) |
|---|---|
| Baiyangdian Wetland [26] | 0.325–1.739 * |
| Chongming Wetland in Shanghai [27] | 0.039–0.136 |
| Jiaozhou Bay Wetland [28] | 0.176–0.563 |
| Liaohe Estuary Wetland [29] | 0.293–1.937 |
| south of Yellow River Delta Wetland [30] | 0.071–1.826 |
| north of Yellow River Delta Wetland [31] | 0.027–0.129 |
| Qinkenpa Wetland in Daqing [32] | 0.023–0.250 |
| typical wetland in Three-river-plain [33] | 2.909–5.645 |
| Lalu Wetland in Tibet [34] | 0.082–0.195 |
| Zhujiang Estuary Wetland [35] | 0.427–1.019 |
| estuary wetland in Elizabeth [36] | 1.200–22.200 |
| floodplains wetland in Canada [37] | 0.016–12.000 |
| This study | 0.029–0.415 |
| PAHs | Principal Component | ||
|---|---|---|---|
| 1 | 2 | 3 | |
| Nap | 0.431 | 0.812 | −0.170 |
| Acl | 0.781 | 0.194 | 0.046 |
| Ace | 0.469 | 0.708 | 0.060 |
| Flu | 0.553 | 0.770 | 0.017 |
| Phe | 0.802 | 0.242 | 0.265 |
| Ant | 0.064 | 0.276 | 0.782 |
| Fla | 0.918 | −0.235 | 0.217 |
| Pyr | 0.879 | −0.301 | 0.259 |
| BaA | 0.804 | −0.083 | −0.187 |
| Chr | 0.906 | −0.298 | 0.105 |
| BbF | 0.930 | −0.317 | 0.127 |
| BkF | 0.907 | −0.141 | −0.017 |
| BaP | 0.818 | −0.145 | −0.270 |
| Ind | 0.923 | −0.198 | −0.135 |
| Dah | 0.538 | 0.328 | −0.359 |
| Bghi | 0.873 | −0.124 | −0.228 |
| Eigenvalues | 9.313 | 2.470 | 1.165 |
| Variance % | 58.204 | 15.441 | 7.281 |
| Cumulative variance % | 58.204 | 73.645 | 80.926 |
| Principal Component | PC1 | PC2 | PC3 |
|---|---|---|---|
| Source of PAHs | Combustion source of petroleum and coal | Petroleum source | Other source |
| Standardized regression coefficient | 0.81 | 0.519 | −0.037 |
| Contribution ratio | 59.3% | 38.0% | 2.7% |
| Conditional probability | 0.000 | 0.000 | 0.384 |
| Coefficient of determination | 0.942 | ||
| PAHs | ERL (mg/kg) | ERM (mg/kg) | Range of Concentration (mg/kg) | Number of Samples with a Concentration Less than ERL |
|---|---|---|---|---|
| Nap | 0.1600 | 2.1000 | 0.0041–0.2955 | 37 |
| Acl | 0.0440 | 0.6400 | 0.0000–0.0036 | 39 |
| Ace | 0.0160 | 0.5000 | 0.0000–0.0047 | 39 |
| Flu | 0.0190 | 0.5400 | 0.0020–0.0156 | 39 |
| Phe | 0.2400 | 1.5000 | 0.0122–0.0741 | 39 |
| Ant | 0.0853 | 1.1000 | 0.0005–0.0169 | 39 |
| Fla | 0.6000 | 5.1000 | 0.0020–0.0465 | 39 |
| Pyr | 0.6650 | 2.6000 | 0.0010–0.0409 | 39 |
| BaA | 0.2610 | 1.6000 | 0.0010–0.0062 | 39 |
| Chr | 0.3840 | 2.8000 | 0.0000–0.0199 | 39 |
| BbF | 0.0000–0.0388 | - | ||
| BkF | 0.0000–0.0051 | - | ||
| BaP | 0.4300 | 1.6000 | 0.0005–0.0046 | 39 |
| Ind | 0.0000–0.0097 | - | ||
| Dah | 0.0634 | 0.2600 | 0.0000–0.0066 | 39 |
| Bghi | 0.0000–0.0127 | - | ||
| Σ16 PAHs | 4.0220 | 44.792 | 0.0290–0.4152 | 39 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, J.; Wang, H.; Sheng, L.; Liu, X.; Zheng, X. Distribution Characteristics and Risk Assessment of Polycyclic Aromatic Hydrocarbons in the Momoge Wetland, China. Int. J. Environ. Res. Public Health 2017, 14, 85. https://doi.org/10.3390/ijerph14010085
Xu J, Wang H, Sheng L, Liu X, Zheng X. Distribution Characteristics and Risk Assessment of Polycyclic Aromatic Hydrocarbons in the Momoge Wetland, China. International Journal of Environmental Research and Public Health. 2017; 14(1):85. https://doi.org/10.3390/ijerph14010085
Chicago/Turabian StyleXu, Jianling, Hanxi Wang, Lianxi Sheng, Xuejun Liu, and Xiaoxue Zheng. 2017. "Distribution Characteristics and Risk Assessment of Polycyclic Aromatic Hydrocarbons in the Momoge Wetland, China" International Journal of Environmental Research and Public Health 14, no. 1: 85. https://doi.org/10.3390/ijerph14010085
APA StyleXu, J., Wang, H., Sheng, L., Liu, X., & Zheng, X. (2017). Distribution Characteristics and Risk Assessment of Polycyclic Aromatic Hydrocarbons in the Momoge Wetland, China. International Journal of Environmental Research and Public Health, 14(1), 85. https://doi.org/10.3390/ijerph14010085






