Effects of Long-Term In Vivo Exposure to Di-2-Ethylhexylphthalate on Thyroid Hormones and the TSH/TSHR Signaling Pathways in Wistar Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Animals and Treatments
2.3. Sample Collection and Preparation
2.4. Iodine Level Detection
2.5. Haematoxilin-Eosin (HE) Staining and Histological Evaluation
2.6. Radioimmunoassay RIA
2.7. Enzyme-Linked Immunoabsorbent Assay
2.8. RNA Extraction and Real-Time Quantitative PCR
2.9. Western Blot Analysis
2.10. Statistical Analysis
3. Results
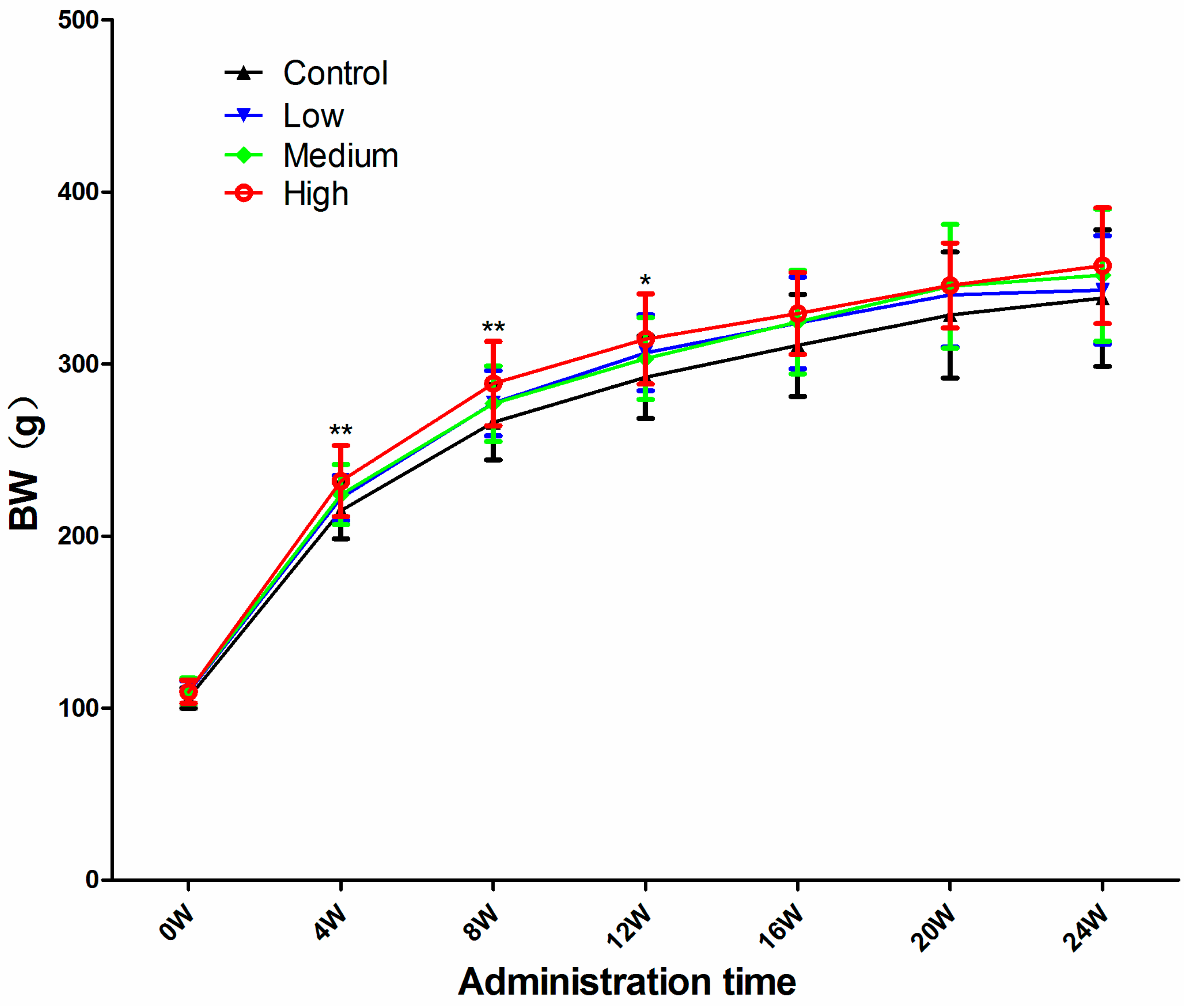
3.1. Body Weight (BW)
3.2. DEHP Elevated Relative Thyroid Weights and Reduced Urine Iodine Levels of Treated Rats
3.3. Histological Changes in the Thyroid and Liver
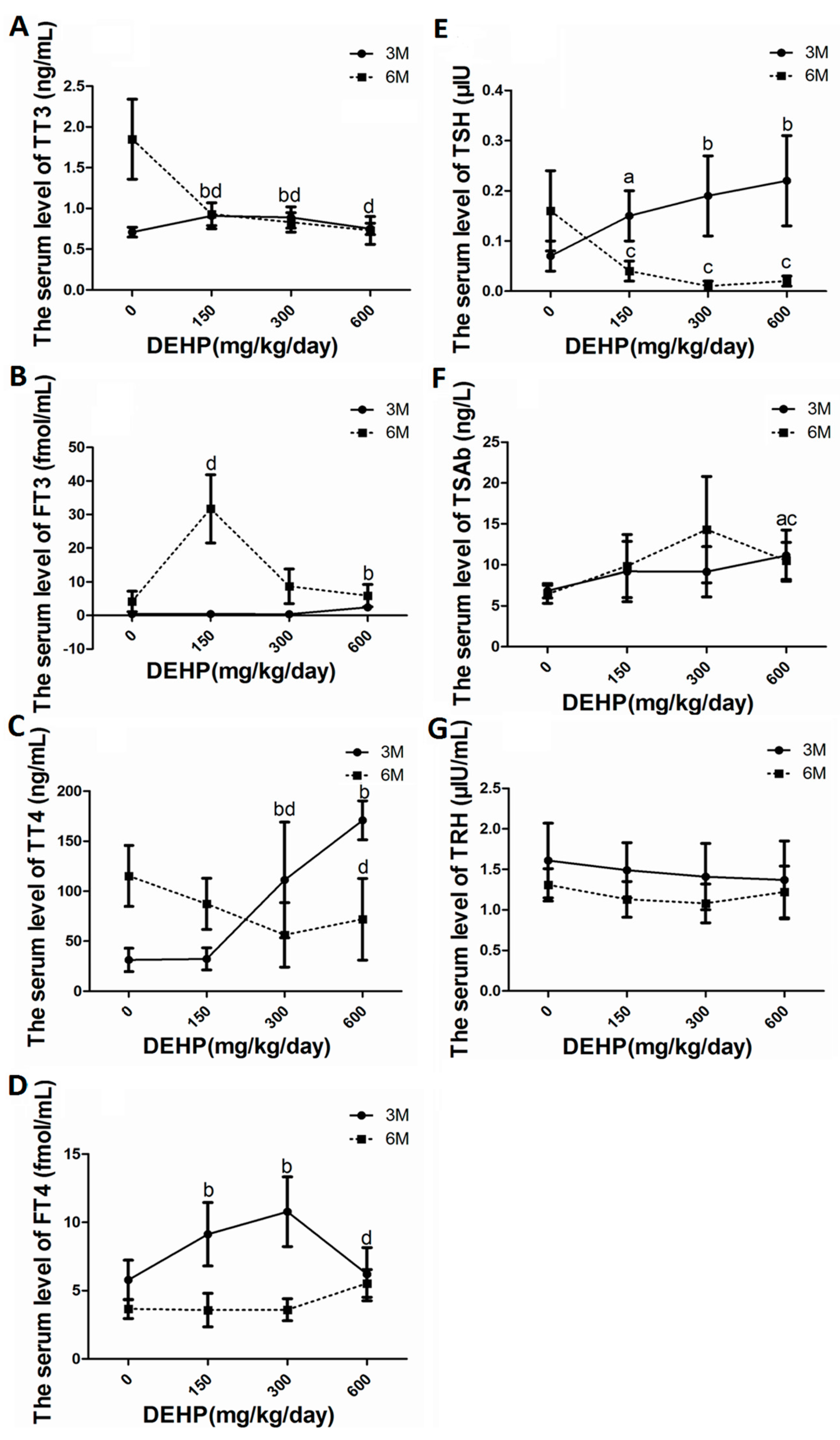
3.4. DEHP Altered Serum Levels of Thyroid Hormones
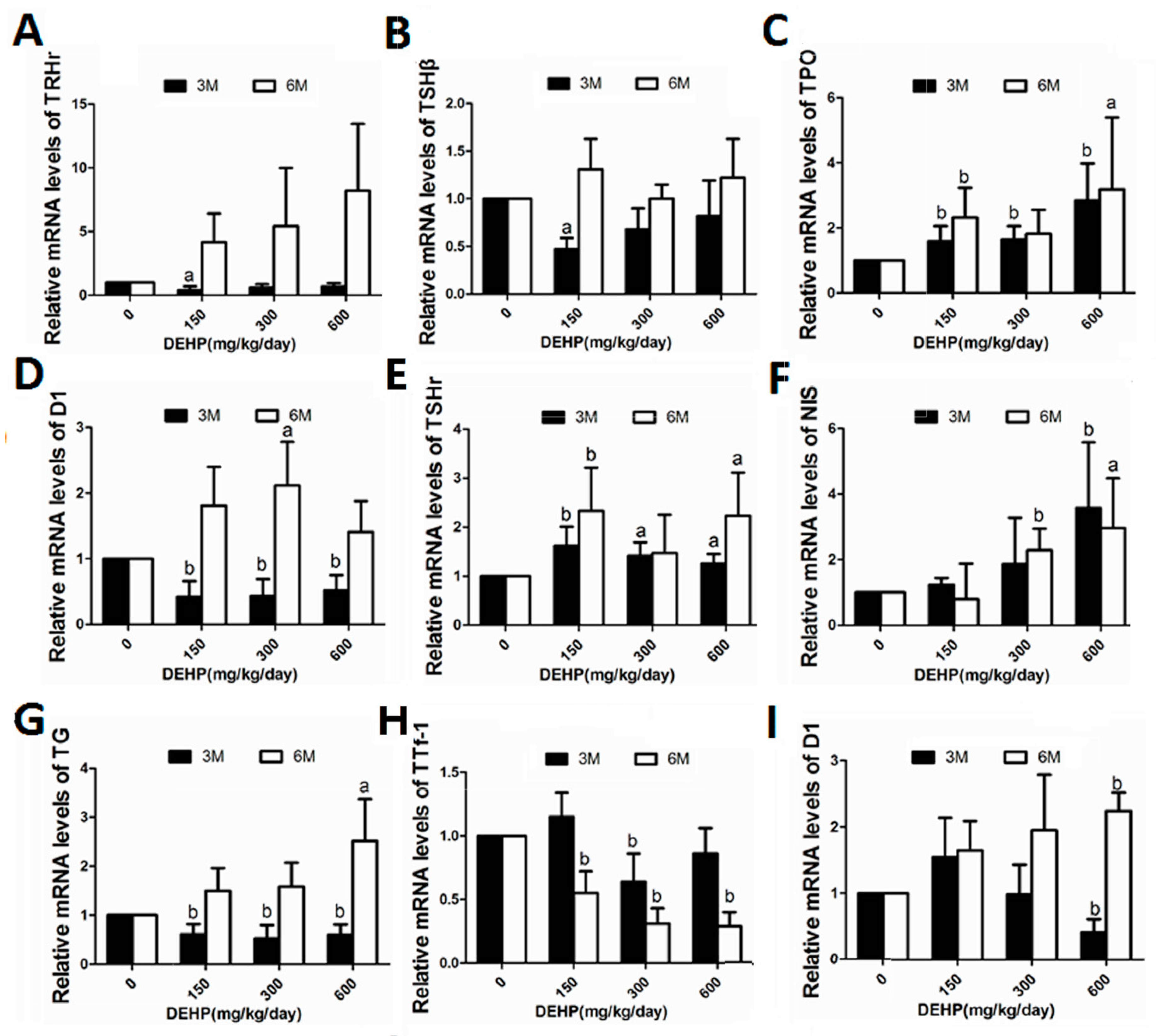
3.5. DEHP Influenced mRNA Expressions of Hypothalamus TRHr, Pituitary TSHβ, Thyroid TPO, TSHr, NIS, Tg, TTf-1, D1 and Liver Deiodinase 1
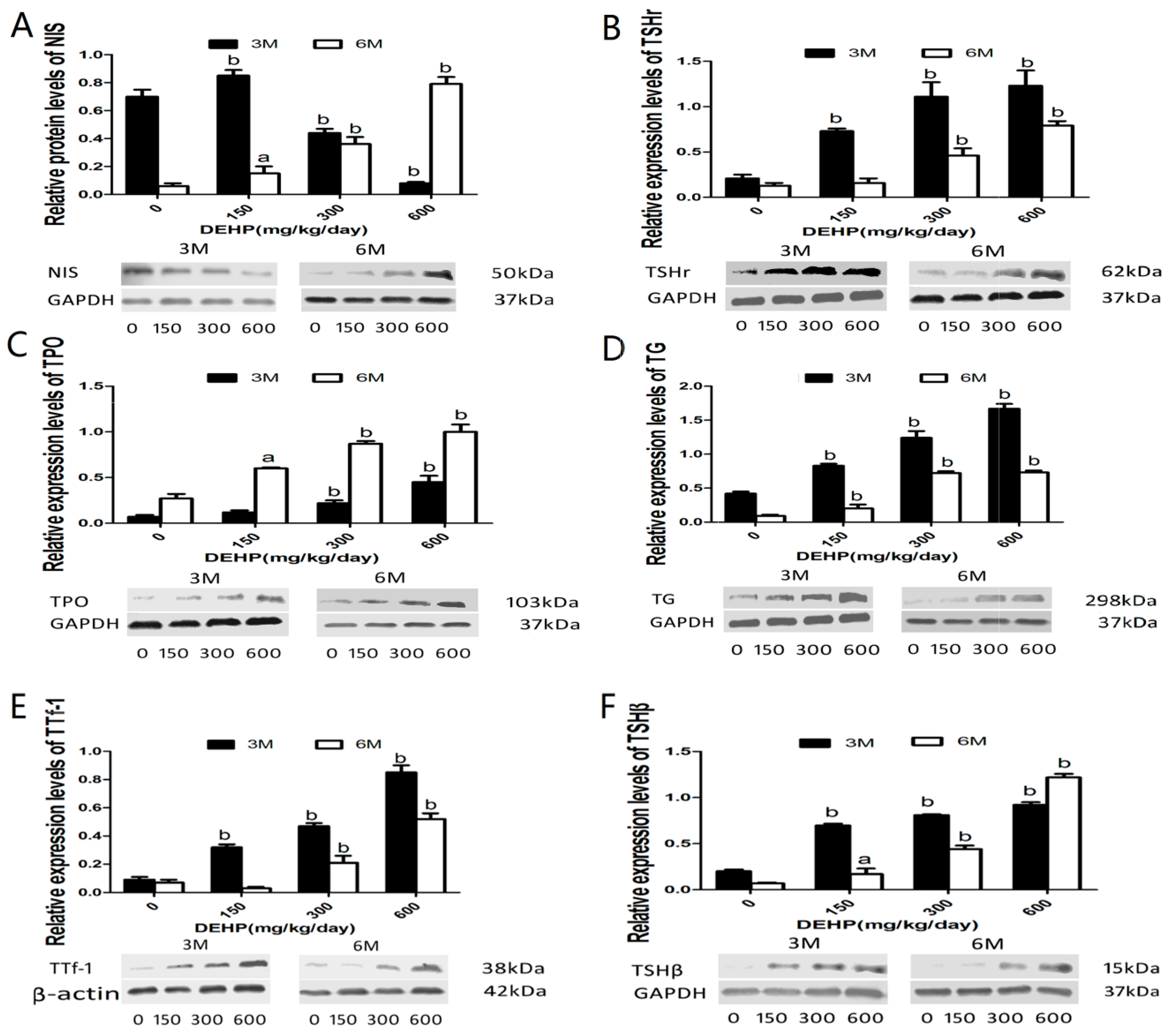
3.6. DEHP Influenced Protein Expressions of NIS, TSHr, TPO, TG, TTf-1 and TSHβ
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wartofsky, L.; Burman, K.D. Alterations in thyroid function in patients with systemic illness: The “euthyroid sick syndrome”. Endocr. Rev. 1982, 3, 164–217. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.C.; Kuo, P.L.; Guo, Y.L.; Liao, P.C.; Lee, C.C. Associations between urinary phthalate monoesters and thyroid hormones in pregnant women. Hum. Reprod. 2007, 22, 2715–2722. [Google Scholar] [CrossRef] [PubMed]
- Khairy, G.A. Solitary thyroid nodule: The risk of cancer and the extent of surgical therapy. East Afr. Med. J. 2004, 81, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Are, C.; Shaha, A.R. Anaplastic thyroid carcinoma: Biology, pathogenesis, prognostic factors, and treatment approaches. Ann. Surg. Oncol. 2006, 13, 453–464. [Google Scholar] [CrossRef] [PubMed]
- Nix, P.; Nicolaides, A.; Coatesworth, A.P. Thyroid cancer review 1: Presentation and investigation of thyroid cancer. Int. J. Clin. Pract. 2005, 59, 1340–1344. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhao, L.; Wei, L.; Li, L. Dehp reduces thyroid hormones via interacting with hormone synthesis-related proteins, deiodinases, transthyretin, receptors, and hepatic enzymes in rats. Environ. Sci. Pollut. Res. Int. 2015, 22, 12711–12719. [Google Scholar] [CrossRef] [PubMed]
- Lottrup, G.; Andersson, A.M.; Leffers, H.; Mortensen, G.K.; Toppari, J.; Skakkebaek, N.E.; Main, K.M. Possible impact of phthalates on infant reproductive health. Int. J. Androl. 2006, 29, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.A.; Henttu, P.; Parker, M.G.; Sumpter, J.P. The estrogenic activity of phthalate esters in vitro. Environ. Health Perspect. 1997, 105, 802–811. [Google Scholar] [CrossRef] [PubMed]
- Ema, M.; Miyawaki, E. Adverse effects on development of the reproductive system in male offspring of rats given monobutyl phthalate, a metabolite of dibutyl phthalate, during late pregnancy. Reprod. Toxicol. 2001, 15, 189–194. [Google Scholar] [CrossRef]
- Ghisari, M.; Bonefeld-Jorgensen, E.C. Effects of plasticizers and their mixtures on estrogen receptor and thyroid hormone functions. Toxicol. Lett. 2009, 189, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Weir, S.M.; Wooten, K.J.; Smith, P.N.; Salice, C.J. Phthalate ester leachates in aquatic mesocosms: Implications for ecotoxicity studies of endocrine disrupting compounds. Chemosphere 2014, 103, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Wenzel, A.; Franz, C.; Breous, E.; Loos, U. Modulation of iodide uptake by dialkyl phthalate plasticisers in frtl-5 rat thyroid follicular cells. Mol. Cell. Endocrinol. 2005, 244, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Hinton, R.H.; Mitchell, F.E.; Mann, A.; Chescoe, D.; Price, S.C.; Nunn, A.; Grasso, P.; Bridges, J.W. Effects of phthalic acid esters on the liver and thyroid. Environ. Health Perspect. 1986, 70, 195–210. [Google Scholar] [CrossRef] [PubMed]
- Price, S.C.; Chescoe, D.; Grasso, P.; Wright, M.; Hinton, R.H. Alterations in the thyroids of rats treated for long periods with di-(2-ethylhexyl) phthalate or with hypolipidaemic agents. Toxicol. Lett. 1988, 40, 37–46. [Google Scholar] [CrossRef]
- Poon, R.; Lecavalier, P.; Mueller, R.; Valli, V.E.; Procter, B.G.; Chu, I. Subchronic oral toxicity of di-n-octyl phthalate and di(2-ethylhexyl) phthalate in the rat. Food Chem. Toxicol. 1997, 35, 225–239. [Google Scholar] [CrossRef]
- Howarth, J.A.; Price, S.C.; Dobrota, M.; Kentish, P.A.; Hinton, R.H. Effects on male rats of di-(2-ethylhexyl) phthalate and di-n-hexylphthalate administered alone or in combination. Toxicol. Lett. 2001, 121, 35–43. [Google Scholar] [CrossRef]
- Mitchell, F.E.; Price, S.C.; Hinton, R.H.; Grasso, P.; Bridges, J.W. Time and dose-response study of the effects on rats of the plasticizer di(2-ethylhexyl) phthalate. Toxicol. Appl. Pharmacol. 1985, 81, 371–392. [Google Scholar] [CrossRef]
- Meeker, J.D.; Ferguson, K.K. Relationship between urinary phthalate and bisphenol a concentrations and serum thyroid measures in U.S. Adults and adolescents from the national health and nutrition examination survey (NHANES) 2007–2008. Environ. Health Perspect. 2011, 119, 1396–1402. [Google Scholar] [CrossRef] [PubMed]
- Boas, M.; Feldt-Rasmussen, U.; Skakkebaek, N.E.; Main, K.M. Environmental chemicals and thyroid function. Eur. J. Endocrinol./Eur. Fed. Endocr. Soc. 2006, 154, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.T.; Wu, C.F.; Chen, B.H.; Chen, E.K.; Chen, Y.L.; Shiea, J.; Lee, W.T.; Chao, M.C.; Wu, J.R. Intake of phthalate-tainted foods alters thyroid functions in Taiwanese children. PLoS ONE 2013, 8, e55005. [Google Scholar] [CrossRef] [PubMed]
- David, R.M.; Moore, M.R.; Finney, D.C.; Guest, D. Chronic toxicity of di(2-ethylhexyl)phthalate in rats. Toxicol. Sci. 2000, 55, 433–443. [Google Scholar] [CrossRef] [PubMed]
- Ge, R.S.; Chen, G.R.; Tanrikut, C.; Hardy, M.P. Phthalate ester toxicity in leydig cells: Developmental timing and dosage considerations. Reprod. Toxicol. 2007, 23, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Massart, F.; Massai, G.; Placidi, G.; Saggese, G. Child thyroid disruption by environmental chemicals. Minerva Pediatr. 2006, 58, 47–53. [Google Scholar] [PubMed]
- McIntosh, G.H.; Jones, G.B.; Howard, D.A.; Belling, G.B.; Potter, B.J.; Hetzel, B.S. Low-iodine diet for producing iodine deficiency in rats. Aust. J. Biol. Sci. 1980, 33, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Dohan, O.; De la Vieja, A.; Paroder, V.; Riedel, C.; Artani, M.; Reed, M.; Ginter, C.S.; Carrasco, N. The sodium/iodide symporter (NIS): Characterization, regulation, and medical significance. Endocr. Rev. 2003, 24, 48–77. [Google Scholar] [CrossRef] [PubMed]
- Yen, P.M. Physiological and molecular basis of thyroid hormone action. Physiol. Rev. 2001, 81, 1097–1142. [Google Scholar] [PubMed]
- Weiss, S.J.; Philp, N.J.; Ambesi-Impiombato, F.S.; Grollman, E.F. Thyrotropin-stimulated iodide transport mediated by adenosine 3′,5′-monophosphate and dependent on protein synthesis. Endocrinology 1984, 114, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Riesco-Eizaguirre, G.; Santisteban, P. A perspective view of sodium iodide symporter research and its clinical implications. Eur. J. Endocrinol./Eur. Fed. Endocr. Soc. 2006, 155, 495–512. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Shi, Y.; Lin, L.; Li, X.; Li, L.; Lin, X.; Xu, D. Intracellular iodinated compounds affect sodium iodide symporter expression through tsh-mediated signaling pathways. Mol. Med. Rep. 2011, 4, 77–80. [Google Scholar] [PubMed]
- Kogai, T.; Endo, T.; Saito, T.; Miyazaki, A.; Kawaguchi, A.; Onaya, T. Regulation by thyroid-stimulating hormone of sodium/iodide symporter gene expression and protein levels in frtl-5 cells. Endocrinology 1997, 138, 2227–2232. [Google Scholar] [CrossRef] [PubMed]
- Shimura, H.; Shimura, Y.; Ohmori, M.; Ikuyama, S.; Kohn, L.D. Single strand DNA-binding proteins and thyroid transcription factor-1 conjointly regulate thyrotropin receptor gene expression. Mol. Endocrinol. 1995, 9, 527–539. [Google Scholar] [PubMed]
- Zimmermann, M.B.; Adou, P.; Torresani, T.; Zeder, C.; Hurrell, R.F. Effect of oral iodized oil on thyroid size and thyroid hormone metabolism in children with concurrent selenium and iodine deficiency. Eur. J. Clin. Nutr. 2000, 54, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J. Factors altering thyroid hormone metabolism. Environ. Health Perspect. 1981, 38, 65–70. [Google Scholar] [CrossRef] [PubMed]

| Primer | Type | Primer Sequence | GenBank |
|---|---|---|---|
| TSHβ | Forward | 5‘-TACTGCCTGACCATCAACACC-3’ | NM_013116.2 |
| Reverse | 5’-GGTAGGAGAAATAAGGAGCAACAT-3’ | ||
| NIS | Forward | 5’-CAGTTCTGGAATGGACACGG-3’ | NM_052983.2 |
| Reverse | 5’-TCTTGGTCACAGCAGGGATG-3’ | ||
| TSHr | Forward | 5’-GTGGGAATAAGCAGCTACGC-3’ | NM_012888.1 |
| Reverse | 5’-GGATTTCGGACGGTGATGT-3’ | ||
| TTF-1 | Forward | 5’-GCACTTGGAGTAAGGCAGAAA-3’ | XM_006224320.2 |
| Reverse | 5’-ACCCCACGATACACGAACC-3’ | ||
| TPO | Forward | 5’-ATGAGGCTGTGACTGAAGATGA-3’ | NM_019353.2 |
| Reverse | 5’-GTGGTCCGTGAGGAGTTTGA-3’ | ||
| TG | Forward | 5’-GCCCTAACTCATCCGTCCA-3’ | NM_030988.2 |
| Reverse | 5’-TGTTGATAAGCCCATCGTCCT-3’ | ||
| D1 | Forward | 5’-GCAGACCCCTGGTGTTGAA-3’ | NM_021653.3 |
| Reverse | 5’-GGTCCTGGAGGCTTCGGT-3’ | ||
| TRHr | Forward | 5’-AGGAGTCAGACCGCTTTAGCA-3’ | NM_013047.3 |
| Reverse | 5’-GAACTGGGTCCATTCTTCTCG-3’ | ||
| β-actin | Forward | 5’-CCGTAAAGACCTCTATGCCAACA-3’ | NM_013116.2 |
| Reverse | 5’-GGGGCCGGACTCATCGTA-3’ |
| Primary Antibodies | Primary Source | Dilution | Brand | Second Source | Dilution |
|---|---|---|---|---|---|
| GAPDH | Rabbit | 1:800 | Santa Cruz, USA | Anti-Rabbit | 1:800 |
| NIS | Goat | 1:800 | Santa Cruz, USA | Anti-Goat | 1:800 |
| TSHr | Goat | 1:200 | Santa Cruz, USA | Anti-Goat | 1:800 |
| TTf-1 | Rabbit | 1:500 | Santa Cruz, USA | Anti-Rabbit | 1:800 |
| TPO | Rabbit | 1:200 | Abcam, USA | Anti-Rabbit | 1:800 |
| TG | Goat | 1:500 | Santa Cruz, USA | Anti-Goat | 1:800 |
| TSHβ | Goat | 1:250 | R&D Systems, USA | Anti-Mouse | 1:800 |
| Group | Liver/Weight (g/100 g) | Thyroid/Weight (g/100 g) | Urine Iodine (μg/L) | |||
|---|---|---|---|---|---|---|
| 3 M | 6 M | 3 M | 6 M | 3 M | 6 M | |
| C | 2.29 ± 0.73 | 2.46 ± 0.17 | 8.19 ± 1.61 | 8.27 ± 2.63 | 160.82 ± 43.56 | 95.73 ± 14.47 |
| L | 2.69 ± 0.32 a | 2.55 ± 0.34 | 8.51 ± 1.46 | 10.48 ± 2.85 | 147.90 ± 31.79 | 70.92 ± 17.27 b |
| M | 2.79 ± 0.17 b | 2.71 ± 0.25 a | 8.55 ± 1.76 | 10.59 ± 2.00 | 133.36 ± 24.58 | 72.29 ± 18.97 b |
| H | 3.78 ± 0.46 b | 3.00 ± 0.30 b | 10.87 ± 2.63 b | 12.50 ± 6.65 | 79.04 ± 15.46 b | 71.24 ± 13.90 b |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dong, X.; Dong, J.; Zhao, Y.; Guo, J.; Wang, Z.; Liu, M.; Zhang, Y.; Na, X. Effects of Long-Term In Vivo Exposure to Di-2-Ethylhexylphthalate on Thyroid Hormones and the TSH/TSHR Signaling Pathways in Wistar Rats. Int. J. Environ. Res. Public Health 2017, 14, 44. https://doi.org/10.3390/ijerph14010044
Dong X, Dong J, Zhao Y, Guo J, Wang Z, Liu M, Zhang Y, Na X. Effects of Long-Term In Vivo Exposure to Di-2-Ethylhexylphthalate on Thyroid Hormones and the TSH/TSHR Signaling Pathways in Wistar Rats. International Journal of Environmental Research and Public Health. 2017; 14(1):44. https://doi.org/10.3390/ijerph14010044
Chicago/Turabian StyleDong, Xinwen, Jin Dong, Yue Zhao, Jipeng Guo, Zhanju Wang, Mingqi Liu, Yunbo Zhang, and Xiaolin Na. 2017. "Effects of Long-Term In Vivo Exposure to Di-2-Ethylhexylphthalate on Thyroid Hormones and the TSH/TSHR Signaling Pathways in Wistar Rats" International Journal of Environmental Research and Public Health 14, no. 1: 44. https://doi.org/10.3390/ijerph14010044
APA StyleDong, X., Dong, J., Zhao, Y., Guo, J., Wang, Z., Liu, M., Zhang, Y., & Na, X. (2017). Effects of Long-Term In Vivo Exposure to Di-2-Ethylhexylphthalate on Thyroid Hormones and the TSH/TSHR Signaling Pathways in Wistar Rats. International Journal of Environmental Research and Public Health, 14(1), 44. https://doi.org/10.3390/ijerph14010044





