Hemoglobin Status and Externalizing Behavioral Problems in Children
Abstract
:1. Introduction
2. Methods
2.1. Study Site and Participants
2.2. Blood Hb Levels at Age 4 Years
2.3. Behavioral Assessment at Age 6 Years
2.4. Social Adversity
2.5. Statistical Analysis
3. Results
3.1. Effect of Low Hb
3.2. Moderator Effect: Social Adversity-by-Hb Level Interactions
3.3. Moderator Effect: Sex-by-Hb Level Interactions
3.4. Social Adversity-by-Hb Level Interactions in Girls
4. Discussion
Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Mason, J.; Bailes, A.; Beda-Andourou, M.; Copeland, N.; Curtis, T.; Deitchler, M.; Foster, L.; Hensley, M.; Horjus, P.; Johnson, C.; et al. Recent trends in malnutrition in developing regions: Vitamin A deficiency, anemia, iodine deficiency, and child underweight. Food Nutr. Bull. 2005, 26, 59–108. [Google Scholar] [CrossRef] [PubMed]
- Milman, N. Anemia—Still a major health problem in many parts of the world! Ann. Hematol. 2011, 90, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Bhutta, Z.A.; Salam, R.A. Global nutrition epidemiology and trends. Ann. Nutr. Metab. 2012, 61, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liao, Q. Prevalence of iron deficiency in children aged 7 months to 7 years in China. Chin. J. Pediatr. 2004, 42, 886–891. [Google Scholar]
- Jorgenson, L.A.; Wobken, J.D.; Georgieff, M.K. Perinatal iron deficiency alters apical dendritic growth in hippocampal CA1 pyramidal neurons. Dev. Neurosci. Baseline 2003, 25, 412–420. [Google Scholar] [CrossRef]
- Rao, R.; Tkac, I.; Townsend, E.L.; Gruetter, R.; Georgieff, M.K. Perinatal iron deficiency alters the neurochemical profile of the developing rat hippocampus. J. Nutr. 2003, 133, 3215–3221. [Google Scholar] [PubMed]
- Uematsu, A.; Matsui, M.; Tanaka, C.; Takahashi, T.; Noguchi, K.; Suzuki, M.; Nishijo, H. Developmental trajectories of amygdala and hippocampus from infancy to early adulthood in healthy individuals. PLoS ONE 2012, 7, e46970. [Google Scholar] [CrossRef] [PubMed]
- Youdim, M. Brain iron deficiency and excess; cognitive impairment and neurodegeneration with involvement of striatum and hippocampus. Neurotox. Res. 2008, 14, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Singh, M. Role of micronutrients for physical growth and mental development. Indian J. Pediatr. 2004, 71, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Raine, A. The effect of childhood malnutrition on externalizing behavior. Curr. Opin. Pediatr. 2006, 18, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B. Early iron deficiency has brain and behavior effects consistent with dopaminergic dysfunction. J. Nutr. 2011, 141, 740S–746S. [Google Scholar] [CrossRef] [PubMed]
- Grantham-McGregor, S.; Ani, C. A review of studies on the effect of iron deficiency on cognitive development in children. J. Nutr. 2001, 131, 649S–666S. [Google Scholar] [PubMed]
- Hermoso, M.; Vucic, V.; Vollhardt, C.; Arsic, A.; Roman-Viñas, B.; Iglesia-Altaba, I.; Koletzko, B. The effect of iron on cognitive development and function in infants, children and adolescents: A systematic review. Ann. Nutr. Metab. 2011, 59, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Raine, A.; Venables, P.H.; Dalais, C.; Mednick, S.A. Malnutrition at age 3 years and lower cognitive ability at age 11 years: Independence from psychosocial adversity. Arch. Pediatr. Adolesc. Med. 2003, 157, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.H.; Su, T.P.; Chen, Y.S.; Hsu, J.W.; Huang, K.L.; Chang, W.H.; Bai, Y.M. Association between psychiatric disorders and iron deficiency anemia among children and adolescents: A nationwide population-based study. BMC Psychiatry 2013, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Konofal, E.; Lecendreux, M.; Deron, J.; Marchand, M.; Cortese, S.; Zaim, M.; Mouren, M.C.; Arnulf, I. Effects of iron supplementation on attention deficit hyperactivity disorder in children. Pediatr. Neurol. 2008, 38, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Jimenez, E.; Hagen, J.; Mollen, E.; Wolf, A.W. Poorer behavioral and developmental outcome more than 10 years after treatment for iron deficiency in infancy. Pediatrics 2000, 105, E51. [Google Scholar] [CrossRef] [PubMed]
- Walter, T.; Kovalskys, J.; Stekel, A. Effect of mild iron deficiency on infant mental development scores. J. Pediatr. 1983, 102, 519–522. [Google Scholar] [CrossRef]
- Walter, T.; De Andraca, I.; Chadud, P.; Perales, C.G. Iron deficiency anemia: Adverse effects on infant psychomotor development. Pediatrics 1989, 84, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Klein, N.K.; Prabucki, K.M. Iron-deficient anemic infants at play. J. Dev. Behav. Pediatr. 1986, 7, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Yehuda, S.; Rabinovitz, S.; Carasso, R.; Mostofsky, D. Long-lasting cognitive, physiological and hematological effects in rehabilitated, early dietary iron-deficiency adult rats, and improvement by treatment with a mixture of essential fatty acids. Nutr. Neurosci. 2008, 11, 167–171. [Google Scholar] [CrossRef] [PubMed]
- Lozoff, B.; Klein, N.K.; Nelson, E.C.; McClish, D.K.; Manuel, M.; Chacon, M.E. Behavior of infants with iron-deficiency anemia. Child Dev. 1998, 69, 24–36. [Google Scholar] [CrossRef] [PubMed]
- Dommergues, J.P.; Archambeaud, B.; Ducot, Y. Iron deficiency and psychomotor development scores: A longitudinal study between ages 10 months and 4 years. Arch. Fr. Pediatr. 1989, 46, 487–490. [Google Scholar] [PubMed]
- Lozoff, B.; Corapci, F.; Burden, M.J.; Kaciroti, N.; Angulo-Barroso, R.; Sazawal, S.; Black, M. Preschool-aged children with iron deficiency anemia show altered affect and behavior. J. Nutr. 2007, 137, 683–689. [Google Scholar] [PubMed]
- Galler, J.R.; Bryce, C.P.; Waber, D.P.; Medford, G.; Eaglesfield, G.D.; Fitzmaurice, G. Early malnutrition predicts parent reports of externalizing behaviors at ages 9–17. Nutr. Neurosci. 2011, 14, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Achenbach, T.M. Manual for the Child Behavior Checklist/4–18 and 1991 Profile; Department of Psychiatry, University of Vermont: Burlington, VT, USA, 1991. [Google Scholar]
- Galler, J.R.; Bryce, C.P.; Zichlin, M.L.; Fitzmaurice, G.; Eaglesfield, G.D.; Waber, D.P. Infant malnutrition is associated with persisting attention deficits in middle adulthood. J. Nutr. 2012, 142, 788–794. [Google Scholar] [CrossRef] [PubMed]
- Raine, A. Annotation: The role of prefrontal deficits, low autonomic arousal, and early health factors in the development of antisocial and aggressive behavior in children. J. Child Psychol. Psychiatry 2002, 43, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wuerker, A. Biosocial bases of aggressive and violent behavior—Implications for nursing studies. Int. J. Nurs Stud. 2005, 42, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Moffitt, T.E. Adolescence-limited and life-course-persistent antisocial behavior: A developmental taxonomy. Psychol. Rev. 1993, 100, 674–701. [Google Scholar] [CrossRef] [PubMed]
- Liu, J. Early health risk factors for violence: Conceptualization, review of the evidence, and implications. Aggress. Violent Behav. 2011, 16, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Ttofi, M.M.; Farrington, D.P. Risk and protective factors, longitudinal research, and bullying prevention. New Dir. Youth Dev. 2012, 2012, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hanlon, A.; Ma, C.; Zhao, S.R.; Cao, S.; Compher, C. Low blood zinc, iron, and other sociodemographic factors associated with behavior problems in preschoolers. Nutrients 2014, 6, 530–545. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cao, S.; Chen, Z.; Raine, A.; Hanlon, A.; Ai, Y.; Zhou, G.; Yan, C.; Leung, P.; McCauley, L.; et al. Cohort profile update: The China Jintan child cohort study. Int. J. Epidemiol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; McCauley, L.A.; Zhao, Y.; Zhang, H.; Pinto-Martin, J. Cohort profile: The China Jintan child cohort study. Int. J. Epidemiol. 2010, 39, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Achenbach, T.; Rescorla, L. Manual for the ASEBA Preschool Forms & Profiles; Research Center for Children, Youth, & Familly, University of Vermont: Burlington, VT, USA, 2001. [Google Scholar]
- Liu, J.; McCauley, L.; Leung, P.; Wang, B.; Yan, C.; Shen, X.; Pinto-Martin, J.; Jintan Cohort Group. Community-based participatory research (CBPR) approach to study children’s health in China: Experiences and reflections. Int. J. Nurs. Stud. 2011, 48, 904–913. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Cheng, H.; Leung, P.W. The application of the preschool child behavior checklist and the caregiver-teacher report form to mainland Chinese children: Syndrome structure, gender differences, country effects, and inter-informant agreement. J. Abnorm. Child Psychol. 2011, 39, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Rutter, M. Family, area and school influences in the genesis of conduct disorders. Book Suppl. J. Child Psychol. Psychiatr. 1978, 1, 95–113. [Google Scholar] [PubMed]
- Moffitt, T.E. The neuropsychology of delinquency: A critical review. In Crime and Justice: A Review of Research; Tonry, M., Ed.; University of Chicago Press: Chicago, IL, USA, 1990; pp. 99–169. [Google Scholar]
- Siberry, G.K.; Iannone, R. The Harriet Lane Handbook: A Manual for Pediatric House Officers, 15th ed.; Mosby Year Book: St. Louis, MO, USA, 2000. [Google Scholar]
- Lozoff, B.; Beard, J.; Connor, J.; Felt, B.; Georgieff, M.; Schallert, T. Long-lasting neural and behavioral effects of iron deficiency in infancy. Nutr. Rev. 2006, 64, S34–S43. [Google Scholar] [CrossRef] [PubMed]
- Sherriff, A.; Emond, A.; Bell, J.; Golding, J. Should infants be screened for anaemia? A prospective study investigating the relation between haemoglobin at 8, 12, and 18 months and development at 18 months. Arch. Dis. Child 2001, 84, 480–485. [Google Scholar] [CrossRef] [PubMed]
- Fuglestad, A.J.; Georgieff, M.K.; Iverson, S.L.; Miller, B.S.; Petryk, A.; Johnson, D.E.; Kroupina, M.G. Iron deficiency after arrival is associated with general cognitive and behavioral impairment in post-institutionalized children adopted from Eastern Europe. Matern. Child Health J. 2013, 17, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Black, M.M.; Quigg, A.M.; Hurley, K.M.; Pepper, M.R. Iron deficiency and iron-deficiency anemia in the first two years of life: Strategies to prevent loss of developmental potential. Nutr. Rev. 2011, 69, S64–S70. [Google Scholar] [CrossRef] [PubMed]
- Galler, J.; Ramsey, F.; Archer, E. A follow-up study of the influence of early malnutrition on subsequent development. Home and classroom behavior. J. Am. Acad. Child Adolesc. Psychiatry 1989, 28, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Galler, J.R.; Barrett, L.R. Children and famine: Long-term effects on behavioral development. Ambul. Child Health 2002, 7, 85–95. [Google Scholar] [CrossRef]
- Algarin, C.; Peirano, P.; Garrido, M.; Pizarro, F.; Lozoff, B. Iron deficiency anemia in infancy: Long-lasting effects on auditory and visual system functioning. Pediatr. Res. 2003, 53, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Madan, N.; Rusia, U.; Sikka, M.; Sharma, S.; Shankar, N. Developmental and neurophysiologic deficits in iron deficiency in children. Indian J. Pediatr. 2011, 78, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Jellen, L.C.; Lu, L.; Wang, X.; Unger, E.L.; Earley, C.J.; Allen, R.P.; Jones, B.C. Iron deficiency alters expression of dopamine-related genes in the ventral midbrain in mice. Neuroscience 2013, 252, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Feinman, S.; Roberts, D.; Hsieh, K.F.; Sawyer, D.; Swanson, D. A Critical Review of Social Referencing in Infancy; Plenum Press: New York, NY, USA, 1992; pp. 15–54. [Google Scholar]
- Hornik, R.; Gunnar, M.R. A descriptive analysis of infant social referencing. Child Dev. 1988, 59, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Mireault, G.C.; Crockenberg, S.C.; Sparrow, J.E.; Pettinato, C.A.; Woodard, K.C.; Malzac, K. Social looking, social referencing and humor perception in 6-and-12-month-old infants. Infant Behav. Dev. 2014, 37, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Pelaez, M.; Virues-Ortega, J.; Gewirtz, J.L. Acquisition of social referencing via discrimination training in infants. J. Appl. Behav. Anal. 2012, 45, 23–36. [Google Scholar] [CrossRef] [PubMed]
- Corapci, F.; Radan, A.E.; Lozoff, B. Iron deficiency in infancy and mother-child interaction at 5 years. J. Dev. Behav. Pediatr. 2006, 27, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Raine, A.; Venables, P.H.; Mednick, S.A. Malnutrition at age 3 years and externalizing behavior problems at ages 8, 11, and 17 years. Am. J. Psychiatry 2004, 161, 2005–2013. [Google Scholar] [CrossRef] [PubMed]
| Variable | Total Group (N = 98) | Boys (N = 57) | Girls (N = 41) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Range | Mean | SD | Range | Mean | SD | Range | |
| Blood Hb-level b (g/dL) | 12.65 | 0.91 | 10.00–15.40 | 12.55 | 0.95 | 10–15.40 | 12.78 | 0.83 | 11–14.20 |
| CBCL Attention Score c | 3.07 | 1.71 | 0–7 | 3.42 | 1.70 | 0–7 | 2.74 | 1.79 | 0–6 |
| CBCL Aggression Score c | 9.90 | 5.73 | 0–30 | 10.93 | 5.93 | 0–30 | 9.07 | 5.77 | 0–21 |
| CBCL Externalizing Behaviors Score c | 12.97 | 6.90 | 0–37 | 14.35 | 7.08 | 0–37 | 11.81 | 7.04 | 0–26 |
| Social adversity d | 3.04 | 2.06 | 0–11 | 2.96 | 2.18 | 0–11 | 3.21 | 1.90 | 0–8 |
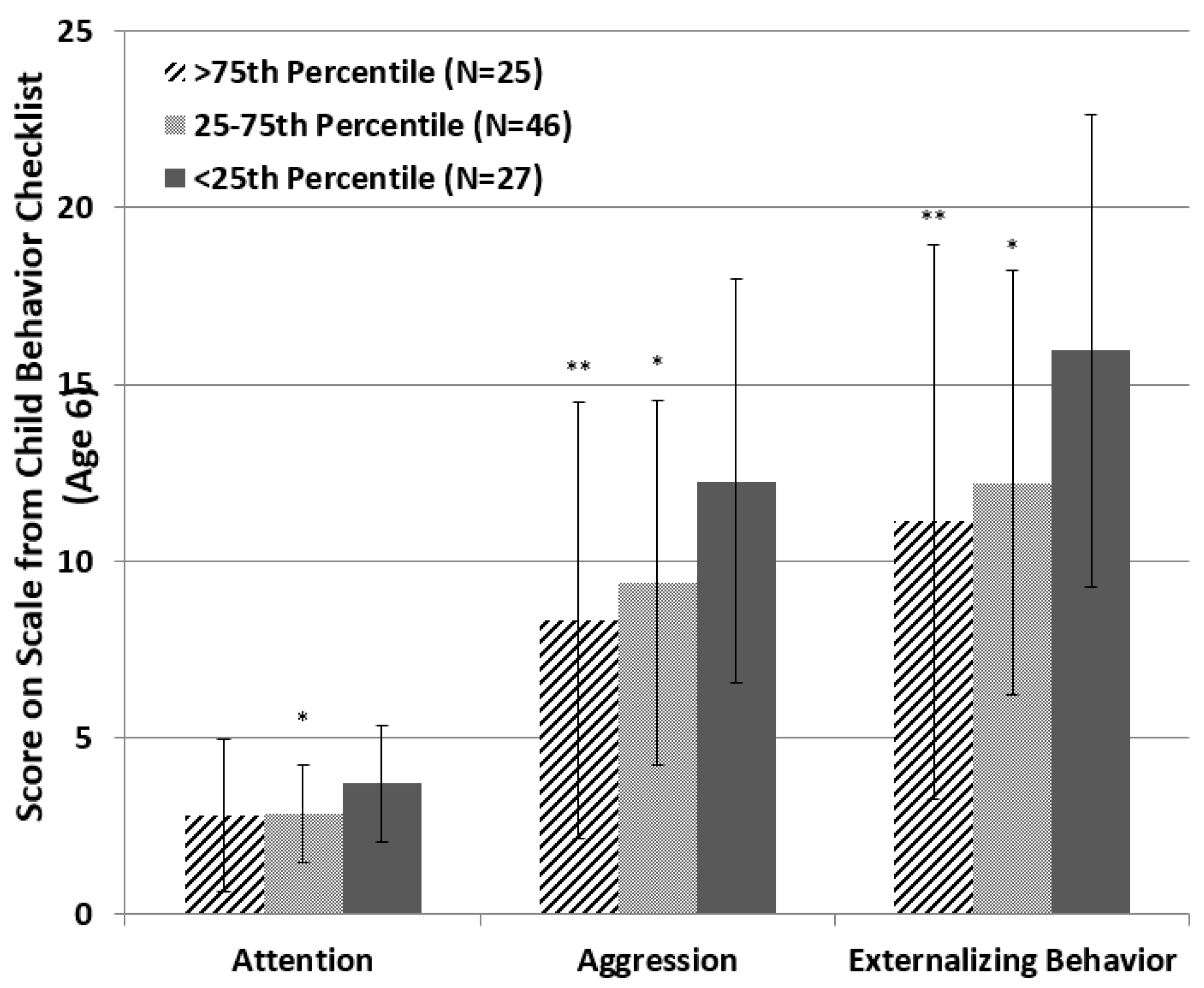
| Demographic Variable | <25th Percentile (N = 27) | 25–75th Percentile (N = 48) | >75th Percentile (N = 25) | Analysis | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % of Sex Group c | Mean | SD | % of Sex Group c | Mean | SD | % of Sex Group c | Mean | SD | X2 | F | df | p | |
| Sex | 5.723 | 2 | 0.057 | ||||||||||
| Male | 20.2 | 26.0 | 10.6 | ||||||||||
| Female | 7.69 | 20.2 | 15.4 | ||||||||||
| Social adversity b | 2.70 | 1.61 | 3.04 | 2.04 | 3.40 | 2.48 | 0.741 | 95, 2 | 0.479 | ||||
| N = 98 | Main Effect: Hemoglobin | Effect Size | Moderator Effects: | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sex Interaction | Social Adversity c Interaction | |||||||||||
| F | df | p | F | df | p | Effect Size | F | df | p | Effect Size | ||
| Multivariate | 2.016 | 4, 190 | 0.094 * | 0.041 | 2.298 | 4, 184 | 0.061 * | 0.048 | 1.496 | 4, 184 | 0.205 | 0.031 |
| Univariate | ||||||||||||
| Attention | 2.651 | 2, 95 | 0.076 * | 0.053 | 3.346 | 2, 92 | 0.040 ** | 0.068 | 1.594 | 2, 92 | 0.209 | 0.033 |
| Aggression | 3.617 | 2, 95 | 0.031 ** | 0.071 | 1.221 | 2, 92 | 0.300 | 0.026 | 1.393 | 2, 92 | 0.253 | 0.029 |
| Sex, Type of Analysis, and Behavior Variable | Main Effect: Hemoglobin | Effect Size | Moderator Effects: | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Social Adversity c Interaction | ||||||||||
| Mean (SD) | F | df | p | Mean (SD) | F | df | p | Effect Size | ||
| Boys (N = 57) | 12.55 (0.95) | 2.96 (2.18) | ||||||||
| Multivariate | 1.780 | 4, 108 | 0.138 | 0.062 | 0.981 | 4, 102 | 0.422 | 0.037 | ||
| Univariate | ||||||||||
| Attention | 3.387 | 2, 54 | 0.041 ** | 0.111 | 0.340 | 2, 51 | 0.713 | 0.013 | ||
| Aggression | 0.745 | 2, 54 | 0.480 | 0.027 | 1.209 | 2, 51 | 0.307 | 0.045 | ||
| Girls (N = 41) | 12.78 (0.83) | 3.21 (1.90) | ||||||||
| Multivariate | 2.200 | 4, 76 | 0.077 * | 0.104 | 2.061 | 4, 40 | 0.095 * | 0.105 | ||
| Univariate | ||||||||||
| Attention | 1.617 | 2, 38 | 0.212 | 0.078 | 3.283 | 2, 35 | 0.049 ** | 0.158 | ||
| Aggression | 3.601 | 2, 38 | 0.037 ** | 0.159 | 2.797 | 2, 35 | 0.075 * | 0.138 | ||
| Social Adversity, Type of Analysis, and Behavior Variable | Mean (SD) | Main Effect: Hemoglobin | Effect Size | ||
|---|---|---|---|---|---|
| F | df | p | |||
| Low Social Adversity (N = 24) | 12.65 (0.81) | ||||
| Multivariate | 3.936 | 4, 42 | 0.008 ** | 0.273 | |
| Univariate | |||||
| Attention | 5.495 | 2, 21 | 0.012 ** | 0.344 | |
| Aggression | 5.404 | 2, 21 | 0.013 ** | 0.340 | |
| High Social Adversity (N = 17) | 13.04 (0.81) | ||||
| Multivariate | 0.357 | 4, 28 | 0.837 | 0.049 | |
| Univariate | |||||
| Attention | 0.448 | 2, 14 | 0.647 | 0.060 | |
| Aggression | 0.474 | 2, 14 | 0.632 | 0.063 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Su, J.; Cui, N.; Zhou, G.; Ai, Y.; Sun, G.; Zhao, S.R.; Liu, J. Hemoglobin Status and Externalizing Behavioral Problems in Children. Int. J. Environ. Res. Public Health 2016, 13, 758. https://doi.org/10.3390/ijerph13080758
Su J, Cui N, Zhou G, Ai Y, Sun G, Zhao SR, Liu J. Hemoglobin Status and Externalizing Behavioral Problems in Children. International Journal of Environmental Research and Public Health. 2016; 13(8):758. https://doi.org/10.3390/ijerph13080758
Chicago/Turabian StyleSu, Jianhua, Naixue Cui, Guoping Zhou, Yuexian Ai, Guiju Sun, Sophie R. Zhao, and Jianghong Liu. 2016. "Hemoglobin Status and Externalizing Behavioral Problems in Children" International Journal of Environmental Research and Public Health 13, no. 8: 758. https://doi.org/10.3390/ijerph13080758
APA StyleSu, J., Cui, N., Zhou, G., Ai, Y., Sun, G., Zhao, S. R., & Liu, J. (2016). Hemoglobin Status and Externalizing Behavioral Problems in Children. International Journal of Environmental Research and Public Health, 13(8), 758. https://doi.org/10.3390/ijerph13080758






