A Meta-Analysis of the Efficacy of Interferon Monotherapy or Combined with Different Nucleos(t)ide Analogues for Chronic Hepatitis B
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion and Exclusion Criteria
- (i)
- randomized controlled trials including HBeAg-positive and/or negative adult CHB patients;
- (ii)
- the trial drugs included IFN combination with NAs (LAM, ADV or ETV) and IFN mono-therapy; and
- (iii)
- the therapy was administered for at least 24 weeks; and
- (iv)
- initial combination therapy was needed.
- (i)
- inclusion of patients co-infected with human immune deficiency virus, hepatitis C virus, or hepatitis D virus;
- (ii)
- the study did not evaluate efficacy.
2.3. Definitions of Efficacy
2.4. Data Extraction
2.5. Statistical Analysis
3. Results
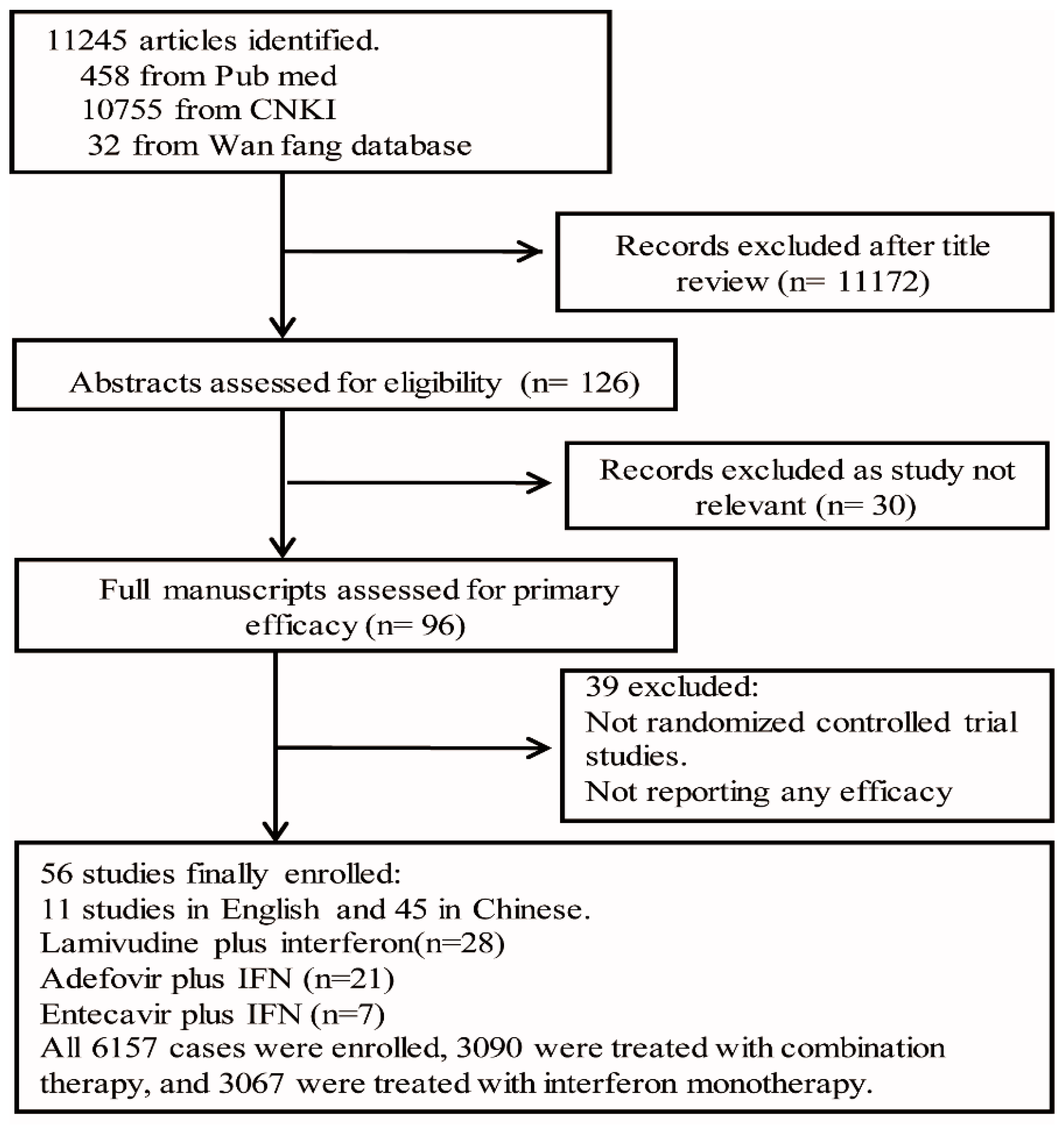
3.1. Selection and Characteristics of Studies
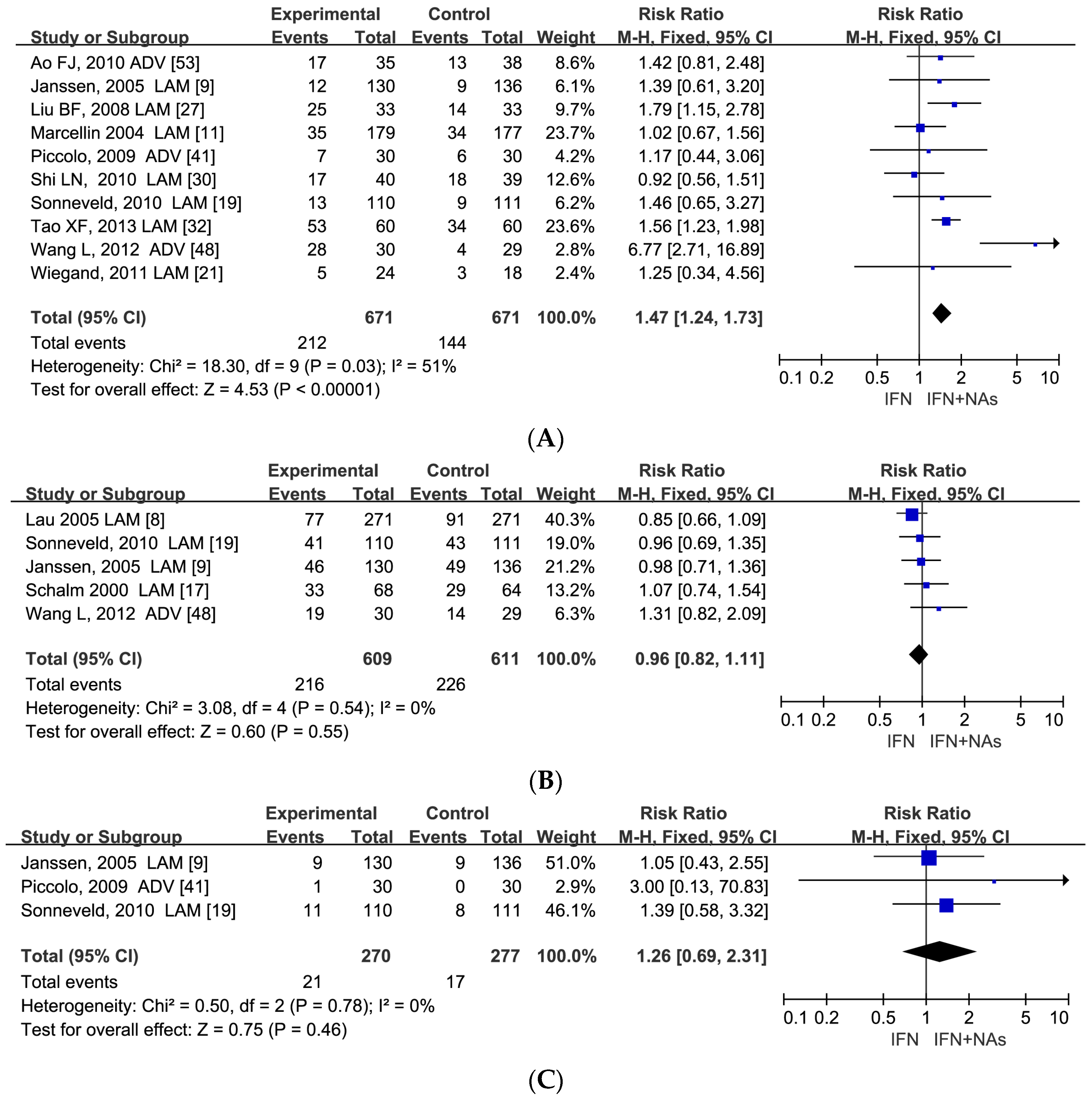
3.2. Better Efficacy of Combination Therapy in Virological and Serological Responses at Week 24 and 48 of Treatment
3.3. Similar Virological and Serological Responses in Different NAs Combination Therapies at Week 24 and 48 of Treatment
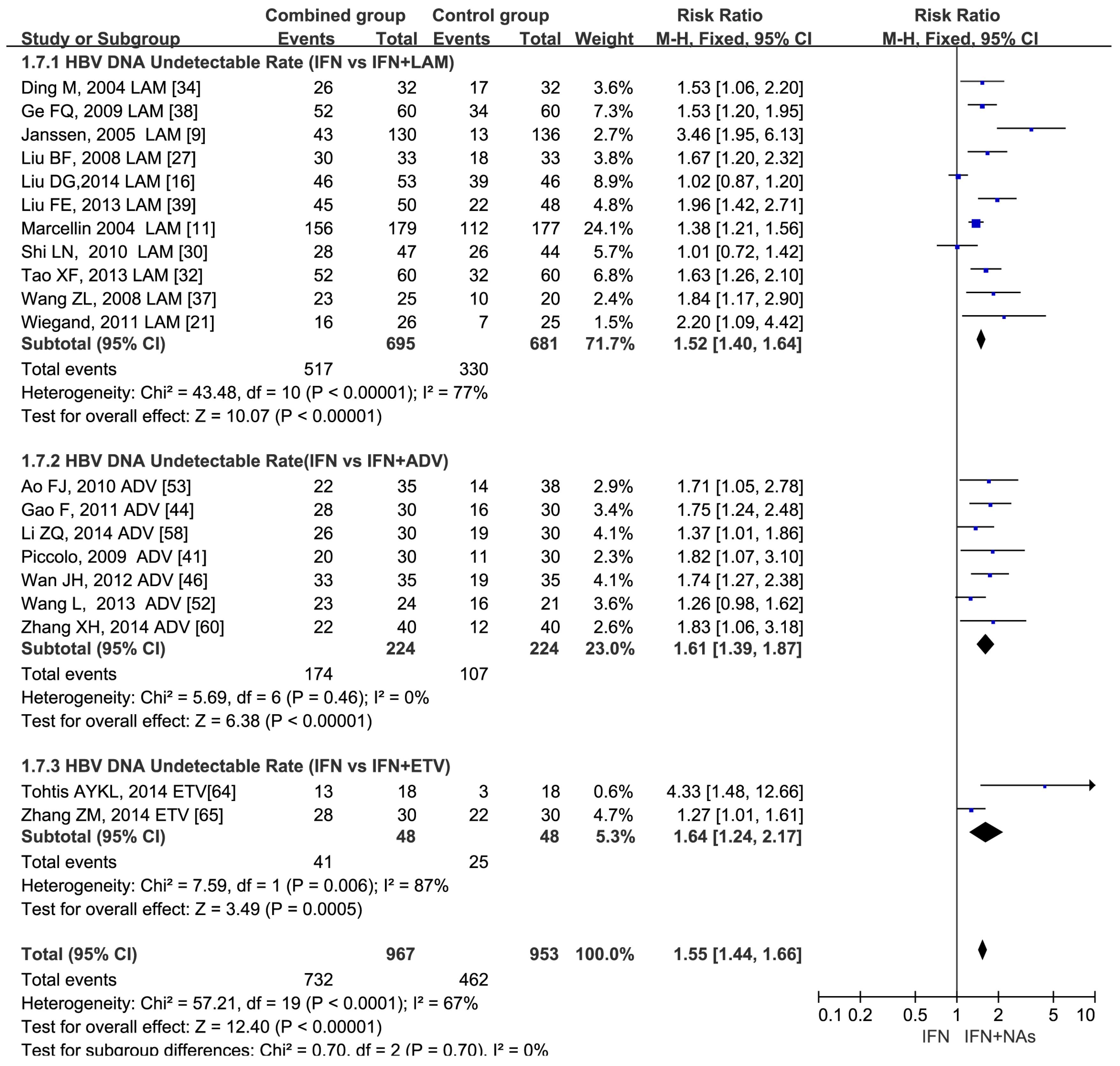
3.4. Higher HBV DNA Undetectable Rate in NAs Combined with IFN Therapy at the End of Follow-Up of 24 –52 Weeks
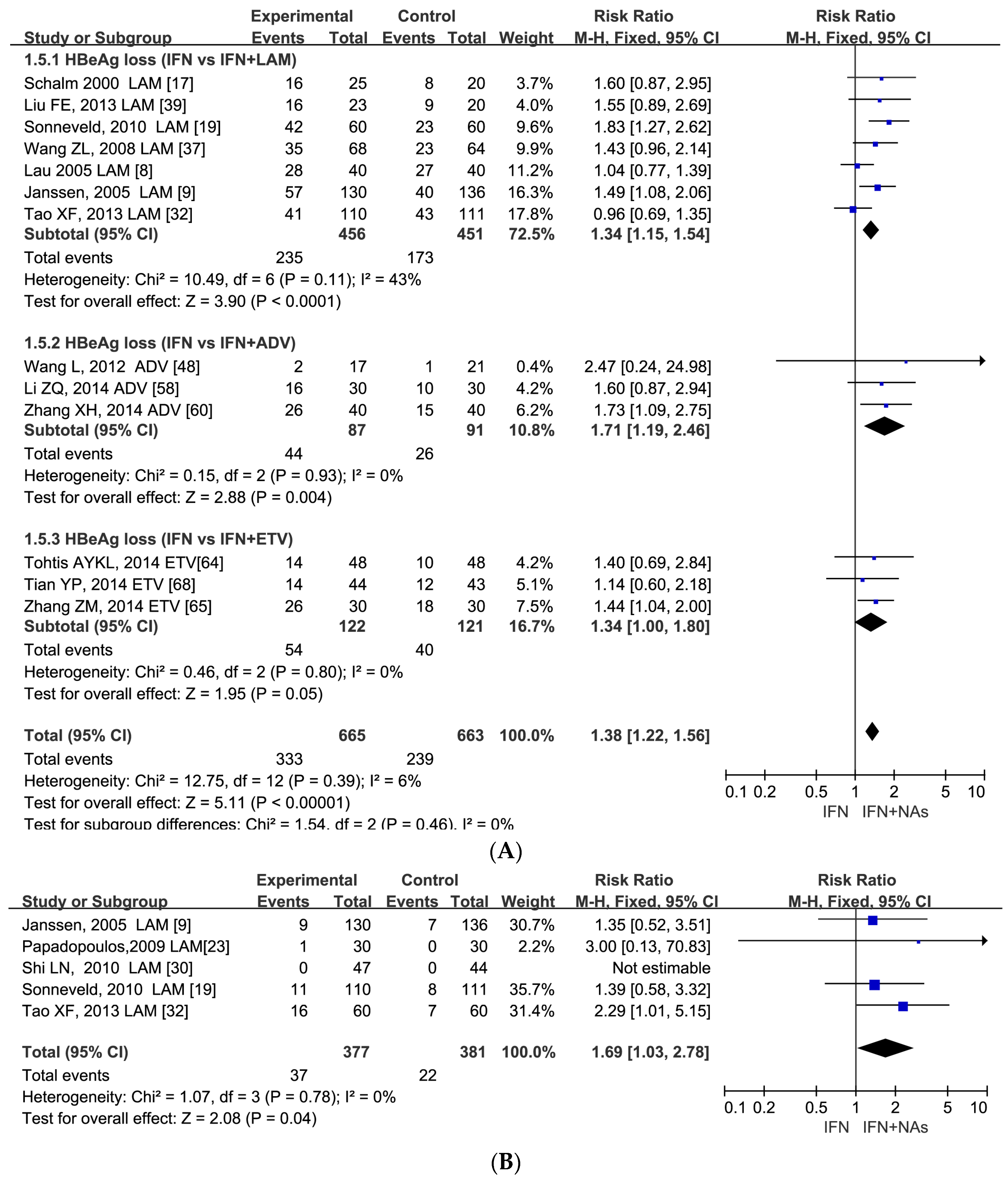
3.5. Similar Serological Response between IFN Monotherapy and Combination Therapy at the Follow-Up 24–52 Weeks
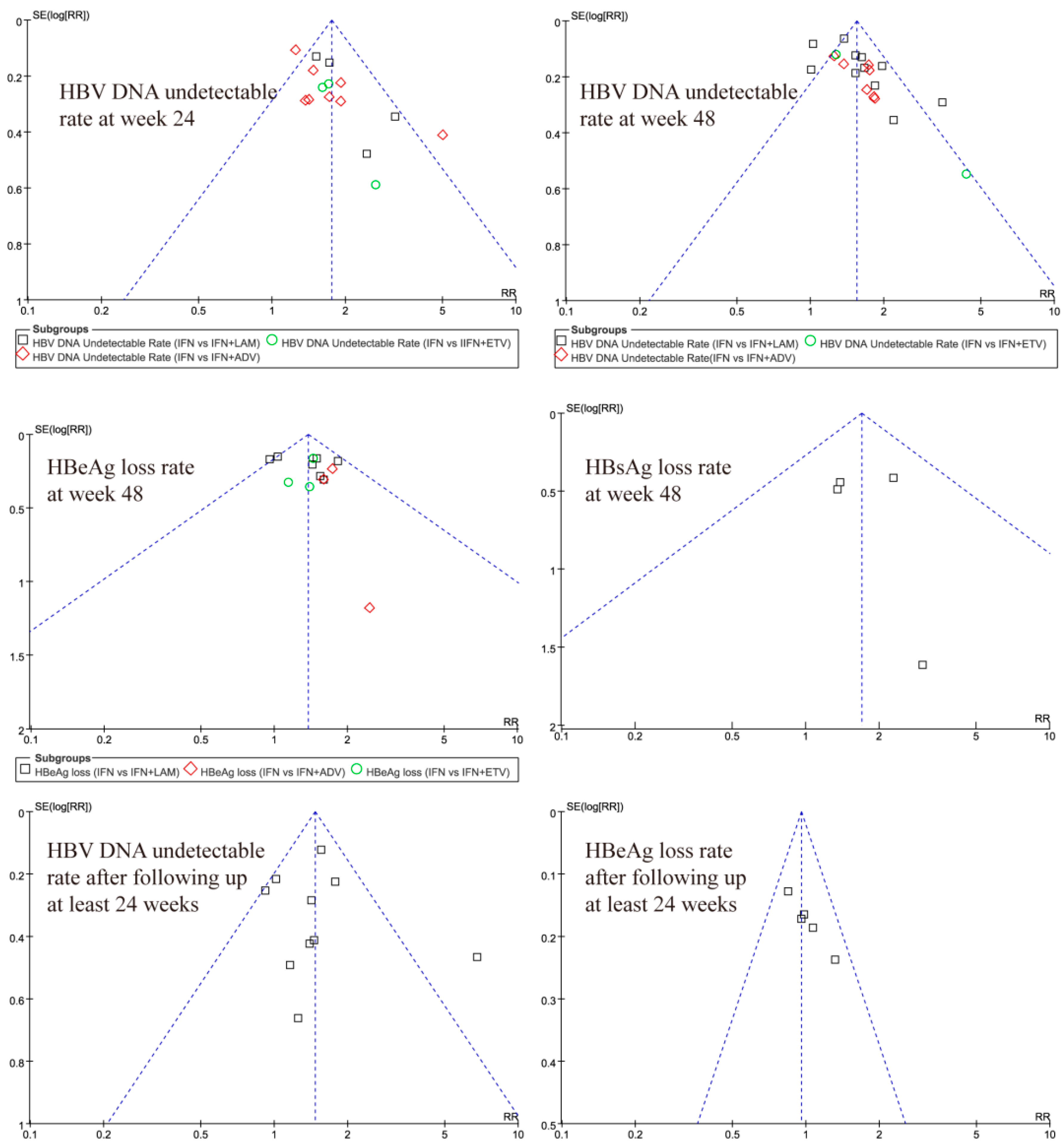
3.6. Publication Bias Test
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Barbaro, G.; Zechini, F.; Pellicelli, A.M.; Francavilla, R.; Scotto, G.; Bacca, D.; Bruno, M.; Babudieri, S.; Annese, M.; Matarazzo, F.; et al. Long-term efficacy of interferon alpha-2b and lamivudine in combination compared to lamivudine mono-therapy in patients with chronic hepatitis B. An Italian multicenter, randomized trial. J. Hepatol. 2001, 35, 406–411. [Google Scholar] [CrossRef]
- Chan, H.L.; Hui, A.Y.; Wong, V.W.; Chim, A.M.; Wong, M.L.; Sung, J.J. Long-term follow-up of peginterferon and lamivudine combination treatment in HBeAg-positive chronic hepatitis B. Hepatology 2005, 41, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.K.; Chung, Y.H.; Choi, M.H.; Kim, J.A.; Ryu, S.H.; Shin, J.W.; Kim, I.S.; Park, N.H.; Lee, H.C.; Lee, Y.S.; et al. Combination of alpha interferon with lamivudine reduces viral breakthrough during long-term therapy. J. Gastroenterol. Hepatol. 2004, 19, 1363–1368. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Hagiwara, S.; Nishida, N.; Kudo, M. Antiviral therapy for chronic hepatitis B: Combination of nucleoside analogs and interferon. World J. Hepatol. 2015, 7, 2427–2431. [Google Scholar] [CrossRef] [PubMed]
- Enomoto, M.; Tamori, A.; Nishiguchi, S.; Kawada, N. Combination therapy with a nucleos(t)ide analogue and interferon for chronic hepatitis B: Simultaneous or sequential. J. Gastroenterol. 2013, 48, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.L.; Wong, V.W.; Chan, H.L. Combination therapy of interferon and nucleotide/nucleoside analogues for chronic hepatitis B. J. Viral Hepat. 2014, 21, 825–834. [Google Scholar] [CrossRef] [PubMed]
- Lau, G.K.; Piratvisuth, T.; Luo, K.X.; Marcellin, P.; Thongsawat, S.; Cooksley, G.; Gane, E.; Fried, M.W.; Chow, W.C.; Paik, S.W.; et al. Peg interferon alfa-2a, lamivudine, and the combination for HBeAg-positive chronic hepatitis B. N. Engl. J. Med. 2005, 352, 2682–2695. [Google Scholar] [CrossRef] [PubMed]
- Janssen, H.L.; van Zonneveld, M.; Senturk, H.; Zeuzem, S.; Akarca, U.S.; Cakaloglu, Y.; Simon, C.; So, T.M.; Gerken, G.; de Man, R.A.; et al. Pegylated interferon alfa-2b alone or in combination with lamivudine for HBeAg-positive chronic hepatitis B: A randomized trial. Lancet 2004, 365, 123–129. [Google Scholar] [CrossRef]
- Marcellin, P.; Bonino, F.; Lau, G.K.; Farci, P.; Yurdaydin, C.; Piratvisuth, T.; Jin, R.; Gurel, S.; Lu, Z.M.; Wu, J.; et al. Sustained response of hepatitis B e antigen-negative patients 3 years after treatment with peg-interferon alfa-2a. Gastroenterology 2009, 136, 2169–2179. [Google Scholar] [CrossRef] [PubMed]
- Marcellin, P.; Lau, G.K.; Bonino, F.; Farci, P.; Hadziyannis, S.; Jin, R.; Lu, Z.M.; Piratvisuth, T.; Germanidis, G.; Yurdaydin, C.; et al. Peg interferon alfa-2a alone, lamivudine alone, and the two in combination in patients with HBeAg-negative chronic hepatitis B. N. Engl. J. Med. 2004, 351, 1206–1217. [Google Scholar] [CrossRef] [PubMed]
- Piratvisuth, T.; Lau, G.; Chao, Y.C.; Jin, R.; Chutaputti, A.; Zhang, Q.B.; Tanwandee, T.; Button, P.; Popescu, M. Sustained response to peg-interferon alfa-2a (40 kD) with or without lamivudine in Asian patients with HBeAg-positive and HBeAg-negative chronic hepatitis B. Hepatol. Int. 2008, 2, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Hao, Y.; Zhang, J.; Wu, C. Interferon-alpha plus adefovir combination therapy versus interferon-alpha mono-therapy for chronic hepatitis B treatment: A meta-analysis. Hepatol. Res. 2013, 43, 1040–1051. [Google Scholar] [PubMed]
- The CNKI Database. Available online: http://www.cnki.net/ (accessed on 19 May 2015). (In Chinese)
- The Wan Fang Database. Available online: http://www.wanfangdata.com.cn/ (accessed on 28 May 2015). (In Chinese)
- Liu, D.G. Efficacy of antiviral therapy in patients with chronic hepatitis B. Guide China Med. 2014, 12, 134–135. (In Chinese) [Google Scholar]
- Schalm, S.W.; Heathcote, J.; Cianciara, J.; Farrell, G.; Sherman, M.; Willems, B.; Dhillon, A.; Moorat, A.; Barber, J.; Gray, D.F. Lamivudine and alpha interferon combination treatment of patients with chronic hepatitis B infection: A randomized trial. Gut 2000, 46, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Rudin, D.; Shah, S.M.; Kiss, A.; Wetz, R.V.; Sottile, V.M. Interferon and lamivudine vs. interferon for hepatitis B e antigen positive hepatitis B treatment: Meta-analysis of randomized controlled trials. Liver Int. 2007, 27, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Sonneveld, M.J.; Rijckborst, V.; Boucher, C.A.; Hansen, B.E.; Janssen, H.L. Prediction of sustained response to peginterferon alfa-2b for hepatitis B e antigen-positive chronic hepatitis B using on-treatment hepatitis B surface antigen decline. Hepatology 2010, 52, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Kaymakoglu, S.; Oguz, D.; Gur, G.; Gurel, S.; Tankurt, E.; Ersöz, G.; Ozenirler, S.; Kalayci, C.; Poturoglu, S.; Cakaloglu, Y.; et al. Pegylated interferon Alfa-2b mono-therapy and pegylated interferon alfa-2b plus lamivudine combination therapy for patients with hepatitis B virus E antigen-negative chronic hepatitis B. Antimicrob. Agents Chemother. 2007, 51, 3020–3022. [Google Scholar] [CrossRef] [PubMed]
- Wiegand, J.; Brosteanu, O.; Kullig, U.; Wiese, M.; Berr, F.; Maier, M.; Tillmann, H.L.; Schiefke, I. Quantification of HBsAg and HBV-DNA During therapy with peg-interferon alpha-2b plus lamivudine alpha-2b plus lamivudine and peg-interferon alpha-2b alone in a German chronic hepatitis B cohort. Z. Gastroenterol. 2011, 49, 1463–1469. [Google Scholar] [CrossRef] [PubMed]
- Ayaz, C.; Celen, M.K.; Colak, H.; Hosoglu, S.; Geyik, M.F. Comparison of lamivudine and alpha-interferon combination with alpha-interferon alone in the treatment of HBeAg-positive chronic hepatitis B. Indian J. Gastroenterol. 2006, 25, 71–73. [Google Scholar] [PubMed]
- Papadopoulos, V.P.; Chrysagis, D.N.; Protopapas, A.N.; Goulis, I.G.; Dimitriadis, G.T.; Mimidis, K.P. Peg-interferon alfa-2b as mono-therapy or in combination with lamivudine in patients with HBeAg-negative chronic hepatitis B: A randomized study. Med. Sci. Monit. 2009, 15, CR56–CR61. [Google Scholar] [PubMed]
- Yang, J.; Zhang, L.S.; Zhao, X.K. Effect of single-using or combined-using lamivudine/interferon alpha-1b on serum markers in patients with hepatitis B. Chin. J. Hepatol. 2003, 11, 109–110. (In Chinese) [Google Scholar]
- Ding, X.H.; Zhuang, L.; Sun, J.; Dong, J.L.; Chen, W.; Yang, Z.L.; Ge, H.J.; Yuan, H.X. Study on dynamic changes of hepatitis B virus during combination therapy of chronic hepatitis B with lamivudine and IFN. Chin. J. Clin. Hepatol. 2003, 19, 33–35. (In Chinese) [Google Scholar]
- Peng, J.; Yang, J.W. Efficacy of interferon-α combined with lamivudine in the treatment of chronic hepatitis B. Mod. Med. Health 2007, 23, 1909–1910. (In Chinese) [Google Scholar]
- Liu, B.F.; Tang, M.X.; Fan, B.C.; Wang, L.H. Clinical evaluation of lamivudine in combination with interferon a for chronic hepatitis B. Chin. J. Gen. Pract. 2008, 6, 946–947. (In Chinese) [Google Scholar]
- Ding, N.L.; Zhu, X.; Wang, Y.P. Clinical observation on lamivudine combination interferon treatment patients with chronic hepatitis B. China Med. Her. 2010, 7, 47–48. (In Chinese) [Google Scholar]
- Guo, B.Y.; Li, J.Z. Effect of lamivudine combined with alpha interferon with chronic HBeAg-positive hepatitis. Med. J. Qilu 2010, 2, 246–250. (In Chinese) [Google Scholar]
- Shi, L.N. Comparison between pegylated interferon mono-therapy and pegylated interferon plus lamivudine combination therapy for efficacy and safety in HBeAg-negative patients. Chin. Gen. Pract. 2010, 13, 3867–3869. (In Chinese) [Google Scholar]
- Huang, J.F.; Jin, R.; Liu, Z.H.; Li, H.W.; Xie, Y.M. The effect of recombinant human interferon α-1b combined with lamivudine on treatment HBeAg positive chronic hepatitis B. Mod. Med. J. China 2012, 14, 18–21. (In Chinese) [Google Scholar]
- Tao, X.F.; Luo, X.Y.; Lou, L.Q.; Zhu, J.H.; Chen, Y.X.; Ye, W.W.; Wu, S.; Chen, X.Y. Efficacy of combination therapy with Peg IFNα-2a and lamivudine for treating HBeAg positive chronic hepatitis B patients. Chin. J. Microecol. 2013, 25, 1330–1332. (In Chinese) [Google Scholar]
- Chen, A.P. Clinical efficiency analysis of lamivudine combination with recombinant human interferon α-2b in the treatment of 144 cases of patients with chronic hepatitis B. J. Hainan Med. Univ. 2013, 19, 209–211. (In Chinese) [Google Scholar]
- Ding, M.; Jiang, S.Z.; Wang, J.H. The efficacy of lamivudine combined with interferon treatment of chronic hepatitis B. Zhejiang Prev. Med. 2003, 15, 67–68. (In Chinese) [Google Scholar]
- Zhang, Z.L.; Yang, Z.G.; Liu, J.F. Clinical observation of 50 cases of a-interferon combined with lamivudine of chronic hepatitis B. Ningxia Med. J. 2005, 27, 199–200. (In Chinese) [Google Scholar]
- Liu, Z.G.; Cao, M.G. The efficacy of lamivudine combination with interferon therapy in chronic hepatitis B. Mod. J. Integr. Tradit. Chin. West. Med. 2006, 15, 720–721. (In Chinese) [Google Scholar]
- Wang, Z.L.; Jia, S.J.; Chen, J.H. Observation of lamivudine combined with interferon treatment of HBeAg-positive chronic hepatitis B patients. Mod. J. Integr. Tradit. Chin. West. Med. 2008, 17, 522–523. (In Chinese) [Google Scholar]
- Ge, F.Q.; Xu, J.; Geng, J.H.; Huang, G.Q. Comparison of the efficacy of lamivudine in combination with or interferon alone in the treatment of chronic hepatitis B. Chin. J. Misdiagnosis 2009, 9, 8346–8347. (In Chinese) [Google Scholar]
- Liu, F.E.; Xie, Y.L.; Li, X.J.; Lu, J.; Liu, C.M.; Mo, M. Clinical study of lamivudine combined with interferon-l b treatment of chronic hepatitis B. J. Chin. Phys. 2013, 15, 920–923. (In Chinese) [Google Scholar]
- Wu, J.S. Comparing the early efficacy and safety of Pegasys alone with Pegasys in combination with lamivudine in HBeAg-negative chronic hepatitis B patients. Seek Med. Ask Med. 2013, 11, 290–291. (In Chinese) [Google Scholar]
- Piccolo, P.; Lenci, I.; Demelia, L.; Bandiera, F.; Piras, M.R.; Antonucci, G.; Nosotti, L.; Mari, T.; De Santis, A.; Ponti, M.L.; et al. A randomized controlled trial of pegylated interferon-alpha2a plus adefovir dipivoxil for hepatitis B e antigen-negative chronic hepatitis B. Antivir. Ther. 2009, 14, 1165–1174. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.B.; Ma, G.Q.; Xu, L.; Zhang, X.Q.; Sui, H.L.; Ding, L. The clinical efficacy of peg-IFN α-2a combined with adefovir dipivoxil for HBeAg-positive chronic hepatitis B. Chin. J. Clin. Infect. Dis. 2008, 1, 168–170. (In Chinese) [Google Scholar]
- Ding, W.M. The efficacy of Peg-IFN combined with adefovir dipivoxil treatment for 22 cases of chronic hepatitis B. Chin. J. Mod. Drug Appl. 2010, 4, 164–165. (In Chinese) [Google Scholar]
- Gao, F.; Zhao, L.F. The efficacy of interferon a-2b combination adefovir or alone in HBeAg-positive chronic hepatitis B patients. Natl. Med. Front. Chin. 2011, 6, 28–29. (In Chinese) [Google Scholar]
- Li, Z. The efficacy of alfa-2a 180 ug combined with adefovir dipivoxil in chronic hepatitis B. World Latest Med. Inf. 2012, 12, 267–268. (In Chinese) [Google Scholar]
- Wan, J.H.; Liu, F.X. The efficacy of peg-interferon a-2a combined with adefovir dipivoxil in HBeAg-positive chronic hepatitis B patients. Chin. Health Care Nutr. 2012, 22, 4351–4352. (In Chinese) [Google Scholar]
- Wu, S.H. The clinical studies of adefovir dipivoxil combined with recombinant human interferon a-2b in chronic hepatitis B treatment. Med. Innov. Chin. 2010, 7, 65–66. (In Chinese) [Google Scholar]
- Wang, L.; Zhang, X.Y.; Yu, H.Y.; Yang, J.M.; Cao, X.G.; Ding, Q.Y.; Pan, J. Clinical observation of interferon α-2b combined with adefovir dipivoxil in HBeAg positive chronic hepatitis B. Chin. J. Biochem. Pharm. 2012, 33, 849–851. (In Chinese) [Google Scholar]
- Lai, D.Q. Clinical observation of interferon plus adefovir dipivoxil therapy in chronic hepatitis B. Med. Forum 2013, 17, 1297–1298. (In Chinese) [Google Scholar]
- Zhang, Q. Investigate the effect of 50 cases interferon α-2a combined with adefovir dipivoxil chronic hepatitis B. Med. Inf. 2013, 26, 149–150. (In Chinese) [Google Scholar]
- Ji, M.F. The research of combination therapy with adefovir dpivoxil. Northwest Pharm. J. 2013, 28, 635–636. (In Chinese) [Google Scholar]
- Wang, L.; Zhang, X.Y.; Tian, Y.L.; Zang, Z.D.; Zhu, X.Y.; Yin, W.W.; Fu, X.L. The efficacy of peg-interferon combined with adefovir dipivoxil for HBeAg-negative chronic hepatitis B. Chin. Hepatol. 2013, 18, 839–841. (In Chinese) [Google Scholar]
- Wang, J.P. Clinical observation of 38 cases of interferon in combination with adefovir dipivoxil treatment chronic hepatitis B. Chin. J. Mod. Drug Appl. 2014, 8, 146–146. (In Chinese) [Google Scholar]
- Hu, Y.S.; Li, H.Y. The analysis of the influence of combining adefovir dipivoxil with-IFN on the hepatitis B markers. Chin. Pract. Med. 2009, 4, 45–46. (In Chinese) [Google Scholar]
- Ao, F.J.; Ma, W.M.; Zhou, P.P.; Zhou, D.Q.; Hu, Y.W.; He, Q.; Dai, W.; Xu, B.; Peng, Y.Z.; Fan, Q.; et al. Comparison of efficacy and safety of pegylated interferon alfa-2a or adefovir dipivoxil mono-therapy with combination therapy in HBeAg positive chronic hepatitis B patients. Chin. J. Infect. Dis. 2010, 28, 214–217. (In Chinese) [Google Scholar]
- Xu, T.M.; Tong, X.C. The short-term efficacy and safety of interferon alfa-2b in combination with adefovir dipivoxil in treatment of patients with HBeAg positive chronic hepatitis B. J. Clin. Hepatol. 2012, 15, 506–507. (In Chinese) [Google Scholar]
- Yang, Y.R.; Li, H.; Jia, T.; Shen, L. The curative effect of polyethylene glycol (peg) Interferonalpha-2a combined with adefovir on HBeAg positive chronic hepatitis B. J. Kunming Med. Univ. 2012, 22, 106–108. (In Chinese) [Google Scholar]
- Li, Z.Q.; Zhen, J.Q.; Zhao, M.C. Clinical efficacy of interferon combined with adefovir dipivoxil in treatment of chronic hepatitis B. J. Bethune Med. Sci. 2014, 12, 114–115. (In Chinese) [Google Scholar]
- Peng, F.J.; Zhang, T.X.; Yuan, M.R.; Chen, F.S. Clinical observation of Interferon combined with adefovir dipivoxil in the treatment of patients with HBeAg (+) chronic hepatitis B. Chin. Phamacist 2014, 17, 984–986. (In Chinese) [Google Scholar]
- Zhang, X.H.; Yi, C.G. Clinical observation of adefovir dipivoxil and polyethylene glycol interferon alfa-2a in the treatment of chronic hepatitis B. J. Mod. Med. Health 2014, 30, 1637–1638. (In Chinese) [Google Scholar]
- Li, W.B.; Ding, J.G.; Sun, Q.F.; Hong, L.; Fu, R.Q. Efficacy of pegylated interferon alpha-2a combined with adefovir dipivoxil in treatment of HBeAg positive chronic hepatitis B. Chin. J. Nosocomiol. 2013, 23, 1250–1251. (In Chinese) [Google Scholar]
- Chen, Y.S.; Fei, X.Y.; Liu, W.T.; Shu, Q. The efficacy of 33 cases of interferon alpha-1b in combination with entecavir on HBeAg positive chronic hepatitis B patients. J. Chin. Intern. Med. 2013, 30, 42–43. (In Chinese) [Google Scholar]
- Zeng, W.; Yuan, J.; Liu, Y.X.; Zhang, Y.; Li, S.Q.; Yao, S.M.; Lin, Y.M.; Chen, C.T.; Zhao, M.F.; Zhou, B.T.; et al. Efficacy of peg-interferon alpha-2a combined with entecavir on HBeAg positive chronic hepatitis B patients with high serum hepatitis B viral loads. Chin. J. Exp. Clin. Virol. 2013, 27, 115–118. (In Chinese) [Google Scholar]
- Tohti, A.Y.K.L. Pegylated interferon alpha-2a combined with entecavir cure analysis of 18 cases of chronic hepatitis B. World Latest Med. Inf. 2014, 14, 150–151. (In Chinese) [Google Scholar]
- Zhang, Z.M.; Ji, B.Y.; Li, L. Effect of entecavir combined with α-2b interferon on treatment of chronic hepatitis B. J. Bengbu Med. Coll. 2014, 39, 621–623. (In Chinese) [Google Scholar]
- Li, Y. The efficacy of α-2b interferon combined with entecavir in patients with chronic hepatitis B. China Pract. Med. 2011, 6, 136–137. (In Chinese) [Google Scholar]
- Li, J. The efficacy of early treatment of chronic hepatitis B of interferon combined with entecavir. Chin. Hepatol. 2012, 17, 714–715. (In Chinese) [Google Scholar]
- Tian, Y.P. Clinical observation of IFN- and entecavir in treating chronic hepatitis B at the initial stage. China Mod. Dr. 2014, 52, 22–24. (In Chinese) [Google Scholar]
- Xie, Q.L.; Zhu, Y.; Wu, L.H.; Fu, L.L.; Xiang, Y. The efficacy and safety of entecavir and interferon combination therapy for chronic hepatitis B virus infection: A meta-analysis. PLoS ONE 2015, 1, e0132219. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Guidelines for the Prevention, Care and Treatment of Persons with Chronic Hepatitis B Infection; WHO: Geneva, Switzerland, 2015. [Google Scholar]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, J.; Wu, X.; Wei, W.; You, H.; Jia, J.; Kong, Y. A Meta-Analysis of the Efficacy of Interferon Monotherapy or Combined with Different Nucleos(t)ide Analogues for Chronic Hepatitis B. Int. J. Environ. Res. Public Health 2016, 13, 730. https://doi.org/10.3390/ijerph13070730
Zhou J, Wu X, Wei W, You H, Jia J, Kong Y. A Meta-Analysis of the Efficacy of Interferon Monotherapy or Combined with Different Nucleos(t)ide Analogues for Chronic Hepatitis B. International Journal of Environmental Research and Public Health. 2016; 13(7):730. https://doi.org/10.3390/ijerph13070730
Chicago/Turabian StyleZhou, Jialing, Xiaoning Wu, Wei Wei, Hong You, Jidong Jia, and Yuanyuan Kong. 2016. "A Meta-Analysis of the Efficacy of Interferon Monotherapy or Combined with Different Nucleos(t)ide Analogues for Chronic Hepatitis B" International Journal of Environmental Research and Public Health 13, no. 7: 730. https://doi.org/10.3390/ijerph13070730
APA StyleZhou, J., Wu, X., Wei, W., You, H., Jia, J., & Kong, Y. (2016). A Meta-Analysis of the Efficacy of Interferon Monotherapy or Combined with Different Nucleos(t)ide Analogues for Chronic Hepatitis B. International Journal of Environmental Research and Public Health, 13(7), 730. https://doi.org/10.3390/ijerph13070730




