Salivary Cortisol Reactivity in Preterm Infants in Neonatal Intensive Care: An Integrative Review
Abstract
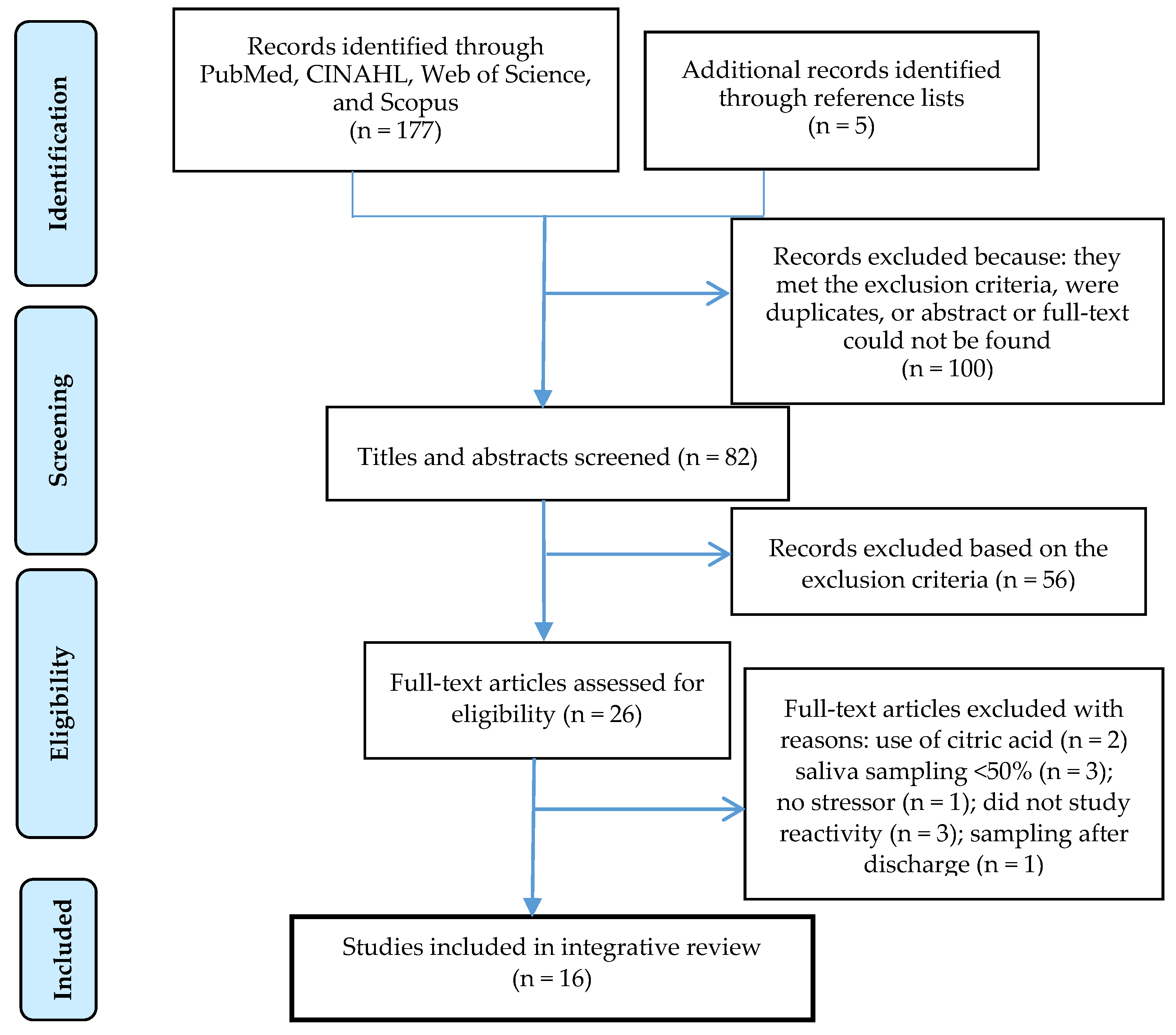
:1. Introduction
- -
- What interventions lead to an increase in salivary cortisol levels from the baseline to response in preterm infants in the NICU?
- -
- What interventions lead to a decrease in salivary cortisol levels from the baseline to response in preterm infants in the NICU?
2. Materials and Methods
2.1. Design
2.2. Study Selection
2.3. Search Strategy
2.4. Analysis Method
3. Results
3.1. Characteristics and Quality of Included Studies
3.2. Interventions Leading to a Change in Salivary Cortisol from Baseline
3.3. Summary of Painful, Handling, and Pleasant Interventions
3.3.1. Painful Interventions
3.3.2. Handling Interventions
3.3.3. Pleasant Interventions
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACTH | Adrenocorticotropic hormone |
| CV | Coefficient of variation |
| GA | Gestational age |
| HPA | Hypothalamic-pituitary-adrenal |
| NICU | Neonatal intensive care unit |
| NIDCAP | Newborn individualized developmental care and assessment program |
| SSC | Skin-to-skin contact |
References
- Levine, S. Developmental determinants of sensitivity and resistance to stress. Psychoneuroendocrinology 2005, 30, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Gitau, R.; Fisk, N.M.; Glover, V. Human fetal and maternal corticotrophin releasing hormone responses to acute stress. Arch. Dis Child. Fetal Neonatal Ed. 2004, 89, F29–F32. [Google Scholar] [CrossRef] [PubMed]
- Feldman, R.; Singer, M.; Zagoory, O. Touch attenuates infants’ physiological reactivity to stress. Dev. Sci. 2010, 13, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Bolt, R.J.; Van Weissenbruch, M.M.; Popp-Snijders, C.; Sweep, F.G.; Lafeber, H.N.; Delemarre-van de Waal, H.A. Maturity of the adrenal cortex in very preterm infants is related to gestational age. Pediatr. Res. 2002, 52, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Murphy, B.E. Cortisol and cortisone in human fetal development. J. Steroid. Biochem. 1979, 11, 509–513. [Google Scholar] [CrossRef]
- Hillman, N.H.; Kallapur, S.G.; Jobe, A.H. Physiology of transition from intrauterine to extrauterine life. Clin. Perinatol. 2012, 39, 769–783. [Google Scholar] [CrossRef] [PubMed]
- Ivars, K.; Nelson, N.; Theodorsson, A.; Theodorsson, E.; Strom, J.O.; Mörelius, E. Development of salivary cortisol circadian rhythm and reference intervals in full-term infants. PloS ONE 2015, 10, e0129502. [Google Scholar] [CrossRef] [PubMed]
- Watterberg, K.L.; Scott, S.M. Evidence of early adrenal insufficiency in babies who develop bronchopulmonary dysplasia. Pediatrics 1995, 95, 120–125. [Google Scholar] [PubMed]
- Fernandez, E.F.; Watterberg, K.L. Relative adrenal insufficiency in the preterm and term infant. J. Perinatol. 2009, 29 (Suppl. 2), S44–S49. [Google Scholar] [CrossRef] [PubMed]
- Grunau, R.E.; Whitfield, M.F.; Petrie-Thomas, J.; Synnes, A.R.; Cepeda, I.L.; Keidar, A.; Rogers, M.; Mackay, M.; Hubber-Richard, P.; Johannesen, D. Neonatal pain, parenting stress and interaction, in relation to cognitive and motor development at 8 and 18 months in preterm infants. Pain 2009, 143, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Lombroso, P.J.; Sapolsky, R. Development of the cerebral cortex: XII Stress and brain development: I. J. Am. Acad. Child Adolesc. Psychiatry 1998, 37, 1337–1339. [Google Scholar] [CrossRef] [PubMed]
- Haley, D.W.; Weinberg, J.; Grunau, R.E. Cortisol, contingency learning, and memory in preterm and full-term infants. Psychoneuroendocrinology 2006, 31, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Quesada, A.A.; Tristao, R.M.; Pratesi, R.; Wolf, O.T. Hyper-responsiveness to acute stress, emotional problems and poorer memory in former preterm children. Stress 2014, 17, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Wadsby, M.; Nelson, N.; Ingemansson, F.; Samuelsson, S.; Leijon, I. Behaviour problems and cortisol levels in very-low-birth-weight children. Nord. J. Psychiatry 2014, 68, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.; Barrington, K.J.; Taddio, A.; Carbajal, R.; Filion, F. Pain in Canadian NICUS: Have we improved over the past 12 years? Clin. J. Pain 2011, 27, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Grunau, R.E.; Haley, D.W.; Whitfield, M.F.; Weinberg, J.; Yu, W.; Thiessen, P. Altered basal cortisol levels at 3, 6, 8 and 18 months in infants born at extremely low gestational age. J. Pediatr. 2007, 150, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Brummelte, S.; Chau, C.M.; Cepeda, I.L.; Degenhardt, A.; Weinberg, J.; Synnes, A.R.; Grunau, R.E. Cortisol levels in former preterm children at school age are predicted by neonatal procedural pain-related stress. Psychoneuroendocrinology 2015, 51, 151–163. [Google Scholar] [CrossRef] [PubMed]
- Grunau, R.E.; Weinberg, J.; Whitfield, M.F. Neonatal procedural pain and preterm infant cortisol response to novelty at 8 months. Pediatrics 2004, 114, e77–e84. [Google Scholar] [CrossRef] [PubMed]
- Holsti, L.; Weinberg, J.; Whitfield, M.F.; Grunau, R.E. Relationships between adrenocorticotropic hormone and cortisol are altered during clustered nursing care in preterm infants born at extremely low gestational age. Early Hum. Dev. 2007, 83, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Grunau, R.E.; Holsti, L.; Haley, D.W.; Oberlander, T.; Weinberg, J.; Solimano, A.; Whitfield, M.F.; Fitzgerald, C.; Yu, W. Neonatal procedural pain exposure predicts lower cortisol and behavioral reactivity in preterm infants in the NICU. Pain 2005, 113, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Anand, K.J. Clinical importance of pain and stress in preterm neonates. Biol. Neonate 1998, 73, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Owens, R. Intraventricular hemorrhage in the premature neonate. Neonatal Netw. 2005, 24, 55–71. [Google Scholar] [CrossRef] [PubMed]
- McEwen, B.S.; Wingfield, J.C. The concept of allostasis in biology and biomedicine. Horm. Behav. 2003, 43, 2–15. [Google Scholar] [CrossRef]
- Gunnar, M.R.; Talge, N.M.; Herrera, A. Stressor paradigms in developmental studies: What does and does not work to produce mean increases in salivary cortisol. Psychoneuroendocrinology 2009, 34, 953–967. [Google Scholar] [CrossRef] [PubMed]
- Comaru, T.; Miura, E. Postural support improves distress and pain during diaper change in preterm infants. J. Perinatol. 2009, 29, 504–507. [Google Scholar] [CrossRef] [PubMed]
- Lyngstad, L.T.; Tandberg, B.S.; Storm, H.; Ekeberg, B.L.; Moen, A. Does skin-to-skin contact reduce stress during diaper change in preterm infants? Early Hum. Dev. 2014, 90, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Pokela, M.L. Pain relief can reduce hypoxemia in distressed neonates during routine treatment procedures. Pediatrics 1994, 93, 379–383. [Google Scholar] [PubMed]
- Yung-Weng, W.; Ying-Ju, C. A preliminary study of bottom care effects on premature infants’ heart rate and oxygen saturation. J. Nurs Res. 2004, 12, 161–168. [Google Scholar] [PubMed]
- Mörelius, E.; Hellstrom-Westas, L.; Carlen, C.; Norman, E.; Nelson, N. Is a nappy change stressful to neonates? Early Hum. Dev. 2006, 82, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Neu, M.; Pan, Z.; Workman, R.; Marcheggiani-Howard, C.; Furuta, G.; Laudenslager, M.L. Benefits of massage therapy for infants with symptoms of gastroesophageal reflux disease. Bio. Res. Nurs. 2014, 16, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Kurihara, H.; Chiba, H.; Shimizu, Y.; Yanaihara, T.; Takeda, M.; Kawakami, K.; Takai-Kawakami, K. Behavioral and adrenocortical responses to stress in neonates and the stabilizing effects of maternal heartbeat on them. Early Hum. Dev. 1996, 46, 117–127. [Google Scholar] [CrossRef]
- Mörelius, E.; Theodorsson, E.; Nelson, N. Stress at three-month immunization: Parents’ and infants’ salivary cortisol response in relation to the use of pacifier and oral glucose. Eur. J. Pain. 2009, 13, 202–208. [Google Scholar] [CrossRef] [PubMed]
- South, M.M.; Strauss, R.A.; South, A.P.; Boggess, J.F.; Thorp, J.M. The use of non-nutritive sucking to decrease the physiologic pain response during neonatal circumcision: A randomized controlled trial. Am. J. Obstet. Gynecol. 2005, 193, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Mörelius, E.; Ortenstrand, A.; Theodorsson, E.; Frostell, A. A randomised trial of continuous skin-to-skin contact after preterm birth and the effects on salivary cortisol, parental stress, depression, and breastfeeding. Early Hum. Dev. 2015, 91, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Neu, M.; Hazel, N.A.; Robinson, J.; Schmiege, S.J.; Laudenslager, M. Effect of holding on co-regulation in preterm infants: A randomized controlled trial. Early Hum. Dev. 2014, 90, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Feldman, R.; Rosenthal, Z.; Eidelman, A.I. Maternal-preterm skin-to-skin contact enhances child physiologic organization and cognitive control across the first 10 years of life. Biol. Psychiatr. 2014, 75, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.; Fisk, N.M.; Glover, V. Mode of delivery and subsequent stress response. Lancet 2000, 355, 120. [Google Scholar] [CrossRef]
- Schuller, C.; Kanel, N.; Muller, O.; Kind, A.B.; Tinner, E.M.; Hosli, I.; Zimmermann, R.; Surbek, D. Stress and pain response of neonates after spontaneous birth and vacuum-assisted and cesarean delivery. Am. J. Obstet. Gynecol. 2012, 207, e411–e416. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, T.G.; Bergman, K.; Sarkar, P.; Glover, V. Prenatal cortisol exposure predicts infant cortisol response to acute stress. Dev. Psychobiol. 2013, 55, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Dickerson, S.S.; Kemeny, M.E. Acute stressors and cortisol responses: A theoretical integration and synthesis of laboratory research. Psychol. Bull. 2004, 130, 355–391. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.; Beijers, R.; Riksen-Walraven, M.; de Weerth, C. Cortisol reactivity in young infants. Psychoneuroendocrinology 2010, 35, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.J.; Walker, R.F.; Riad-Fahmy, D.; Hughes, D.; Murphy, J.F.; Gray, O.P. Assessment of adrenocortical activity in term newborn infants using salivary cortisol determinations. J. Pediatr. 1987, 111, 129–133. [Google Scholar] [CrossRef]
- Schwartz, E.B.; Granger, D.A.; Susman, E.J.; Gunnar, M.R.; Laird, B. Assessing salivary cortisol in studies of child development. Child. Dev. 1998, 69, 1503–1513. [Google Scholar] [CrossRef] [PubMed]
- Cignacco, E.; Denhaerynck, K.; Nelle, M.; Buhrer, C.; Engberg, S. Variability in pain response to a non-pharmacological intervention across repeated routine pain exposure in preterm infants: A feasibility study. Acta Paediatr. 2009, 98, 842–846. [Google Scholar] [CrossRef] [PubMed]
- Cong, X.; Ludington-Hoe, S.M.; Walsh, S. Randomized crossover trial of kangaroo care to reduce biobehavioral pain responses in preterm infants: A pilot study. Biol. Res. Nurs. 2011, 13, 204–216. [Google Scholar] [CrossRef] [PubMed]
- Badiee, Z.; Asghari, M.; Mohammadizadeh, M. The calming effect of maternal breast milk odor on premature infants. Pediatr. Neonatol. 2013, 54, 322–325. [Google Scholar] [CrossRef] [PubMed]
- Badiee, Z.; Nassiri, Z.; Armanian, A. Cobedding of twin premature infants: Calming effects on pain responses. Pediatr. Neonatol. 2014, 55, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Campbell-Yeo, M.L.; Johnston, C.C.; Joseph, K.S.; Feeley, N.; Chambers, C.T.; Barrington, K.J.; Walker, C.D. Co-bedding between preterm twins attenuates stress response after heel lance: Results of a randomized trial. Clin. J. Pain 2014, 30, 598–604. [Google Scholar] [PubMed]
- Kleberg, A.; Warren, I.; Norman, E.; Morelius, E.; Berg, A.C.; Mat-Ali, E.; Holm, K.; Fielder, A.; Nelson, N.; Hellstrom-Westas, L. Lower stress responses after newborn individualized developmental care and assessment program care during eye screening examinations for retinopathy of prematurity: A randomized study. Pediatrics 2008, 121, e1267–e1278. [Google Scholar] [CrossRef] [PubMed]
- Ivars, K.; Nelson, N.; Finnstrom, O.; Morelius, E. Nasopharyngeal suctioning does not produce a salivary cortisol reaction in preterm infants. Acta Paediatr. 2012, 101, 1206–1210. [Google Scholar] [CrossRef] [PubMed]
- Boyer, K.; Johnston, C.; Walker, C.D.; Filion, F.; Sherrard, A. Does sucrose analgesia promote physiologic stability in preterm neonates? Biol. Neonate 2004, 85, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Magnano, C.L.; Gardner, J.M.; Karmel, B.Z. Differences in salivary cortisol levels in cocaine-exposed and noncocaine-exposed NICU infants. Dev. Psychobiol. 1992, 25, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.P.; Townsend, E.L.; Gunnar, M.R.; Georgieff, M.K.; Guiang, S.F.; Ciffuentes, R.F.; Lussky, R.C. Effects of prenatal betamethasone exposure on regulation of stress physiology in healthy premature infants. Psychoneuroendocrinology 2004, 29, 1028–1036. [Google Scholar] [CrossRef] [PubMed]
- Mörelius, E.; Brostrom, E.B.; Westrup, B.; Sarman, I.; Ortenstrand, A. The Stockholm neonatal family-centered care study: Effects on salivary cortisol in infants and their mothers. Early Hum. Dev. 2012, 88, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Candia, M.F.; Osaku, E.F.; Leite, M.A.; Toccolini, B.; Costa, N.L.; Teixeira, S.N.; Costa, C.R.; Piana, P.A.; Cristovam, M.A.; Osaku, N.O. Influence of prone positioning on premature newborn infant stress assessed by means of salivary cortisol measurement: Pilot study. Rev. Bras. Ter. Intensiva 2014, 26, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Mörelius, E.; Theodorsson, E.; Nelson, N. Salivary cortisol and mood and pain profiles during skin-to-skin care for an unselected group of mothers and infants in neonatal intensive care. Pediatrics 2005, 116, 1105–1113. [Google Scholar] [CrossRef] [PubMed]
- Dorn, F.; Wirth, L.; Gorbey, S.; Wege, M.; Zemlin, M.; Maier, R.F.; Lemmer, B. Influence of acoustic stimulation on the circadian and ultradian rhythm of premature infants. Chronobiol. Int. 2014, 31, 1062–1074. [Google Scholar] [CrossRef] [PubMed]
- Schwilling, D.; Vogeser, M.; Kirchhoff, F.; Schwaiblmair, F.; Boulesteix, A.L.; Schulze, A.; Flemmer, A.W. Live music reduces stress levels in very low-birthweight infants. Acta Paediatr. 2015, 104, 360–367. [Google Scholar] [CrossRef] [PubMed]
- Gunnar, M.R. Studies of the human infant’s adrenocortical response to potentially stressful events. New Dir. Child. Dev. 1989, 3–18. [Google Scholar] [CrossRef]
- Hanrahan, K.; McCarthy, A.M.; Kleiber, C.; Lutgendorf, S.; Tsalikian, E. Strategies for salivary cortisol collection and analysis in research with children. Appl. Nurs. Res. 2006, 19, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Thompson, R.F.; Spencer, W.A. Habituation: A model phenomenon for the study of neuronal substrates of behavior. Psychol. Rev. 1966, 73, 16–43. [Google Scholar] [CrossRef] [PubMed]
- Gunnar, M.R.; Hertsgaard, L.; Larson, M.; Rigatuso, J. Cortisol and behavioral responses to repeated stressors in the human newborn. Dev. Psychobiol. 1991, 24, 487–505. [Google Scholar] [CrossRef] [PubMed]
- Cignacco, E.; Hamers, J.P.; Stoffel, L.; van Lingen, R.A.; Gessler, P.; McDougall, J.; Nelle, M. The efficacy of non-pharmacological interventions in the management of procedural pain in preterm and term neonates. A systematic literature review. Eur. J. Pain 2007, 11, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Bueno, M.; Yamada, J.; Harrison, D.; Khan, S.; Ohlsson, A.; Adams-Webber, T.; Beyene, J.; Stevens, B. A systematic review and meta-analyses of nonsucrose sweet solutions for pain relief in neonates. Pain Res. Manag. 2013, 18, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Stevens, B.; Yamada, J.; Lee, G.Y.; Ohlsson, A. Sucrose for analgesia in newborn infants undergoing painful procedures. Cochrane Database Syst. Rev. 2013, 1, CD001069. [Google Scholar] [PubMed]
- Field, T.; Goldson, E. Pacifying effects of nonnutritive sucking on term and preterm neonates during heelstick procedures. Pediatrics 1984, 74, 1012–1015. [Google Scholar] [PubMed]
- Schaffer, L.; Luzi, F.; Burkhardt, T.; Rauh, M.; Beinder, E. Antenatal betamethasone administration alters stress physiology in healthy neonates. Obstet. Gynecol. 2009, 113, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Champagne, F.; Meaney, M.J. Like mother, like daughter: Evidence for non-genomic transmission of parental behavior and stress responsivity. Prog Brain Res. 2001, 133, 287–302. [Google Scholar] [PubMed]
- Linden, W.; Earle, T.L.; Gerin, W.; Christenfeld, N. Physiological stress reactivity and recovery: Conceptual siblings separated at birth? J. Psychosom. Res. 1997, 42, 117–135. [Google Scholar] [CrossRef]
- Wilder, J. Adrenalin and the law of initial value: A critical survey. Exp. Med. Surg. 1957, 15, 47–67. [Google Scholar] [PubMed]
- Mörelius, E.; Nelson, N.; Gustafsson, P.A. Salivary cortisol response in mother-infant dyads at high psychosocial risk. Child Care Health Dev. 2007, 33, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Mörelius, E.; Theodorsson, E.; Nelson, N. Sample volume matters when sampling saliva in paediatric clinical analysis. Acta Paediatr. 2013, 102, e389. [Google Scholar] [CrossRef] [PubMed]
- Mörelius, E.; Nelson, N.; Theodorsson, E. Salivary cortisol and administration of concentrated oral glucose in newborn infants: Improved detection limit and smaller sample volumes without glucose interference. Scand. J. Clin. Lab. Investig. 2004, 64, 113–118. [Google Scholar] [CrossRef]
- Mörelius, E.; Nelson, N.; Theodorsson, E. Saliva collection using cotton buds with wooden sticks: A note of caution. Scand. J. Clin. Lab. Investig. 2006, 66, 15–18. [Google Scholar] [CrossRef] [PubMed]
| Author Year Country | GA Age + | Post–Natal Age, Days | Design | Study Groups | n | Intervention | Sampling Times | Successful Saliva Sampling | Storage Temperature (°C) | Analyze Method | Intra/Inter CV | Cortisol Response |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Painful | ||||||||||||
| Cignacco et al. 2009 Switzerland [44] | 28–31 | <14 | Single group | Sucrose | 9 | Heel lance | –25, +30 | 99% | Not stated | RIA | Not stated | Not significant |
| Cong et al. 2011 USA [45] | 30–32 | 2–9 | Randomized crossover | Incubator care vs. SSC 30 min or 80 min | 18 + 10 | Heel lance | 0, +20 | * | −70 | Not stated | 4.8/6.3 | Not stated |
| Badiee et al. 2013 Iran [46] | 32–37 | 1–30 | Randomized clinical study | Formula odor vs. breast milk odor | 25 + 25 | Heel lance | 0, +20 | Not stated | Not stated | ELISA | Not stated | Not stated |
| Badiee et al. 2014 Iran [47] | 26–34 | <20 | Randomized clinical study | Standard care vs. Co-bedding | 50 + 50 | Heel lance | 0, +20 | Not stated | Not stated | ELISA | Not stated | Not stated ## |
| Campbell–Yeo et al. 2014 Canada [48] | 28–36 | Mean 12.0 and 18.96 | Randomized clinical study | Standard care vs. Co-bedding | 62 + 72 | Heel lance | 0, +20 | 57% | −20 | EIA | 2.24/2.47 | Decrease ## |
| Kleberg et al. 2008 Sweden [49] | 23–31 | <35 | Randomized crossover | Standard care vs NIDCAP | 36 | Eye-screening exam. | 0, +30, +60, +4 h | 75% | −22 | RIA | /6.0 –12.0 | Increase # |
| Ivars et al. 2012 Sweden [50] | 27–33 | 4–86 | Randomized crossover | Control vs oral glucose | 11 | Nasopharyngeal suctioning | 0, +30 | 98% | −22 | RIA | /6.0 –12.0 | Not significant |
| Boyer et al. 2004 Canada [51] | <31 | 1–7 | Randomized controlled study | Water vs. Sucrose for one week | 105 | Different painful procedures | 0, +30 | 54% | −10 | Not stated | Not stated | Not significant |
| Painful + Handling | ||||||||||||
| Magnano et al. 1992 USA [52] | 30–37 | 5–53 | Two group comparative design | Control vs. Cocaine exposed infants | 47 + 11 | Heel lance and Physical exam. | 0, +30 | 79% | Not stated | RIA | 3.3/10.1 | Increase # and Increase # |
| Davis et al. 2004 USA [53] | 33–34 | 3–6 | Two group comparative design | Control vs. Antenatal Betam-ethasone | 9 + 9 | Heel lance and Physical exam. | 0, +20–25, +40–45 | 98% | −20 | DELFIA | 4.3/11.99 | Decrease ## and Increase # |
| Handling | ||||||||||||
| Mörelius et al. 2006 Sweden [29] | 23–38 | 2–7 and 10–18 | Two group comparative design | Full-term vs preterm | 39 + 30 | Diaper change | 0, +30 | 90% | −22 | RIA | /6.0–12.0 | Not significant |
| Mörelius et al. 2012 Sweden [54] | 24–37 | ≤112 | Randomized clinical study | Standard care vs. family care | 137 + 152 | Diaper change | 0, +30 | 97% | −22 | RIA | /6.0–12.0 | Not significant |
| Candia et al. 2014 Brazil [55] | 26–36 | 1–33 | Single group | Lateral/supine position vs. Prone position | 21 | Prone position | 0, +30 | 76% | Not stated | ECL | Not stated | Decrease ## |
| Pleasant | ||||||||||||
| Mörelius et al. 2005 Sweden [56] | 25–33 | 2–21 and 4–26 | Crossover | Incubator care vs. SSC | 17 | SSC | 0, +30 | 89% | −22 | RIA | /6.0–12.0 | Not significant |
| Dorn et al. 2014 Germany [57] | 30–37 | Mean 4.9 | Randomized clinical study | Control vs. Music vs. Maternal voice | 22 + 20 + 20 | Music and Maternal voice | −10, +10 | 65% | −20 | ELISA | Not stated | Not significant |
| Schwilling et al. 2015 Germany [58] | 23–33 | <21 | Single group | Day 1 Day 2 Day 3 | 20 | Music | 0, +25, +4 h | 91% | −20 | Mass- spec-trometry | Not stated | Decrease ## |
| Author Year | Intervention | Results |
|---|---|---|
| Magnano 1992 [52] | Heel lance | Cocaine-exposed as well as non-cocaine-exposed infants showed a significant increase in salivary cortisol 30 min after heel lance. |
| Physical examination | Non-cocaine-exposed infants showed a significant increase in salivary cortisol 30 min after physical examination. | |
| Davis 2004 [53] | Heel lance | Infants not exposed to antenatal betamethasone showed a significant increase in salivary cortisol 20 min after heel lance. |
| Kleberg 2008 [49] | Eye-screening | NIDCAP treated as well as non-NIDCAP treated infants showed a significant increase in salivary cortisol from the baseline to 30 and 60 min after eye-screening examination for retinopathy of the premature eye. |
| Author Year | Intervention | Results |
|---|---|---|
| Davis 2004 [53] | Heel lance | Infants exposed to antenatal betamethasone showed a significant decrease in salivary cortisol from the baseline to 20 and 40 min after heel lance. |
| Kleberg 2008 [49] | Eye-screening examination | NIDCAP-treated infants showed a significant decrease in salivary cortisol from 30 to 60 min after eye-screening for retinopathy of the premature eye. |
| Campbell-Yeo 2014 [48] | Heel lance | Salivary cortisol was significantly lower than the baseline 20 min after heel lance in infants randomized to co-bedding but not in the control group. |
| Candia 2014 [55] | Prone position | Salivary cortisol was significantly lower than the baseline 30 min after changing the position from the lateral/supine position in the incubator to the prone position in the incubator. |
| Schwilling 2015 [58] | Music | Salivary cortisol was significantly lower than the baseline 25 min after exposure to 15 min of live harp music during days 1 and 3 but not on day 2. Salivary cortisol was significantly lower than the baseline 4 h after exposure to 15 min of live music during day 1 but not on days 2 and 3. |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mörelius, E.; He, H.-G.; Shorey, S. Salivary Cortisol Reactivity in Preterm Infants in Neonatal Intensive Care: An Integrative Review. Int. J. Environ. Res. Public Health 2016, 13, 337. https://doi.org/10.3390/ijerph13030337
Mörelius E, He H-G, Shorey S. Salivary Cortisol Reactivity in Preterm Infants in Neonatal Intensive Care: An Integrative Review. International Journal of Environmental Research and Public Health. 2016; 13(3):337. https://doi.org/10.3390/ijerph13030337
Chicago/Turabian StyleMörelius, Evalotte, Hong-Gu He, and Shefaly Shorey. 2016. "Salivary Cortisol Reactivity in Preterm Infants in Neonatal Intensive Care: An Integrative Review" International Journal of Environmental Research and Public Health 13, no. 3: 337. https://doi.org/10.3390/ijerph13030337
APA StyleMörelius, E., He, H.-G., & Shorey, S. (2016). Salivary Cortisol Reactivity in Preterm Infants in Neonatal Intensive Care: An Integrative Review. International Journal of Environmental Research and Public Health, 13(3), 337. https://doi.org/10.3390/ijerph13030337







