Occupational Disparities in the Association between Self-Reported Salt-Eating Habit and Hypertension in Older Adults in Xiamen, China
Abstract
:1. Introduction
2. Methods
2.1. Study Population
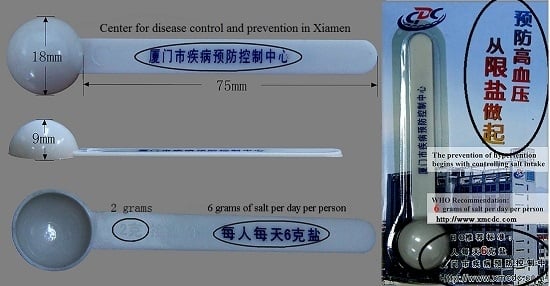
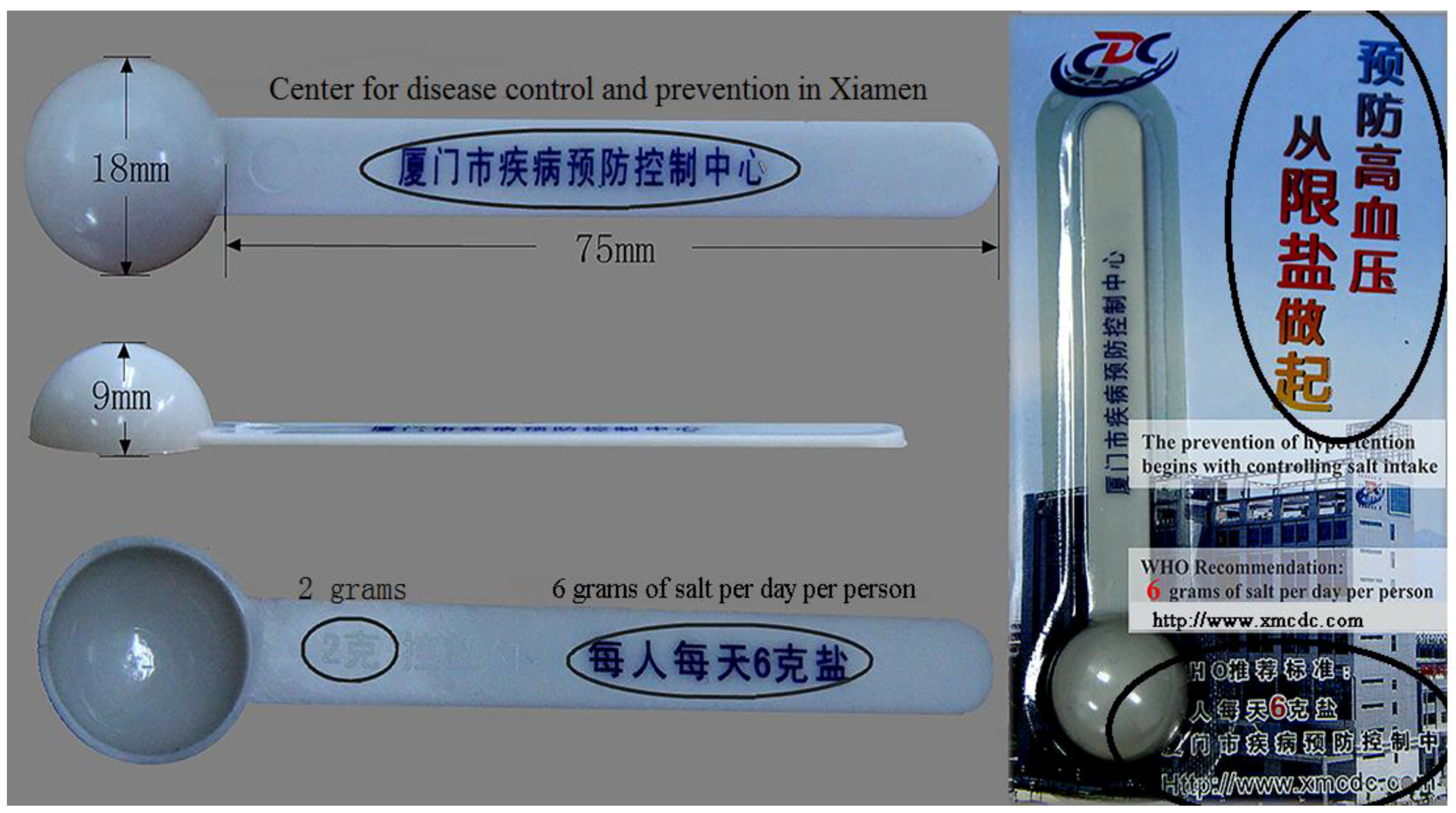
2.2. Measurements
2.3. Statistical Analysis
3. Results
3.1. Hypertension Prevalence
| Characteristic | Normotension | Hypertension | Prevalence a (%) | p b |
|---|---|---|---|---|
| Total, N | 9586 | 4152 | 30.22 | |
| Sex, N (%) | 0.044 | |||
| Female | 5033 (52.5) | 2258 (54.38) | 30.97 | |
| Male | 4553 (47.5) | 1894 (45.62) | 29.38 | |
| Age, mean(SD)/years | 70.92 (8.34) | 72.91 (8.21) | - | <0.001 |
| Age level, N (%) | <0.001 | |||
| 60~ | 2751 (28.7) | 790 (19.03) | 22.31 | |
| 65~ | 2211 (23.06) | 858 (20.66) | 27.96 | |
| 70~ | 1508 (15.73) | 759 (18.28) | 33.48 | |
| 75~ | 1416 (14.77) | 786 (18.93) | 35.69 | |
| 80~ | 961 (10.03) | 575 (13.85) | 37.43 | |
| 85~ | 739 (7.71) | 384 (9.25) | 34.19 | |
| Salt-eating habit, N (%) | <0.001/0.976 d | |||
| Low | 4283 (44.68) | 1976 (47.59) | 31.57 | |
| Medium | 4584 (47.82) | 1839 (44.29) | 28.63 | |
| High | 719 (7.5) | 337 (8.12) | 31.91 | |
| Occupation, N (%) | <0.001 | |||
| Indoor labor | 4591 (47.89) | 2393 (57.63) | 34.26 | |
| Outdoor labor | 4995 (52.11) | 1759 (42.37) | 26.04 | |
| Education, N (%) | <0.001 | |||
| Illiterate | 3230 (33.69) | 1295 (31.19) | 28.62 | |
| Primary | 3033 (31.64) | 1169 (28.16) | 27.82 | |
| Junior high school | 1763 (18.39) | 838 (20.18) | 32.22 | |
| Senior high school and beyond | 1560 (16.27) | 850 (20.47) | 35.27 | |
| BMI (kg/m2) c, N (%) | 0.132 | |||
| Underweight (<18.5) | 477 (4.98) | 189 (4.55) | 28.38 | |
| Normal (18.5–22.9) | 5733 (59.81) | 2453 (59.08) | 29.97 | |
| Overweight (23.0–26.9) | 2735 (28.53) | 1239 (29.84) | 31.18 | |
| Obese (≥27) | 641 (6.69) | 271 (6.53) | 29.71 | |
| Marriage, N (%) | 0.079 | |||
| In-marriage | 6730 (70.21) | 2826 (68.06) | 29.57 | |
| Single | 98 (1.02) | 42 (1.01) | 30.00 | |
| Divorced | 96 (1) | 49 (1.18) | 33.79 | |
| Widowed | 2662 (27.77) | 1235 (29.74) | 31.69 | |
| Number of Children, N (%) | 0.003 | |||
| 0 | 246 (2.57) | 84 (2.02) | 25.45 | |
| 1 | 1482 (15.46) | 578 (13.92) | 28.06 | |
| 2 | 2518 (26.27) | 1112 (26.78) | 30.63 | |
| ≥3 | 5340 (55.71) | 2378 (57.27) | 30.81 | |
| Smoking, N (%) | <0.001 | |||
| Never | 6024 (62.84) | 2832 (68.21) | 31.98 | |
| Sometimes | 1365 (14.24) | 489 (11.78) | 26.38 | |
| Often | 1660 (17.32) | 526 (12.67) | 24.06 | |
| Quit | 537 (5.6) | 305 (7.35) | 36.22 | |
| Alcohol Drinking, N (%) | <0.001 | |||
| Never | 6469 (67.48) | 2896 (69.75) | 30.92 | |
| Sometimes | 2279 (23.77) | 861 (20.74) | 27.42 | |
| Often | 491 (5.12) | 193 (4.65) | 28.22 | |
| Quit | 347 (3.62) | 202 (4.87) | 36.79 | |
| Exercise, N (%) | 0.057 | |||
| Never | 3602 (37.58) | 1472 (35.45) | 29.01 | |
| Sometimes | 3505 (36.56) | 1581 (38.08) | 31.09 | |
| Often | 2479 (25.86) | 1099 (26.47) | 30.72 |
| Salt-Eating Habit | Occupation, N (%) | p a | |
|---|---|---|---|
| Indoor | Outdoor | ||
| Male (N = 6447) | <0.001 | ||
| Low | 1201 (45.46) | 1377 (36.19) | |
| Medium | 1179 (44.63) | 2085 (54.80) | |
| High | 262 (9.92) | 343 (9.01) | |
| Female (N = 7291) | <0.001 | ||
| Low | 2281 (52.53) | 1400 (47.47) | |
| Medium | 1784 (41.09) | 1375 (46.63) | |
| High | 277 (6.38) | 174 (5.90) | |
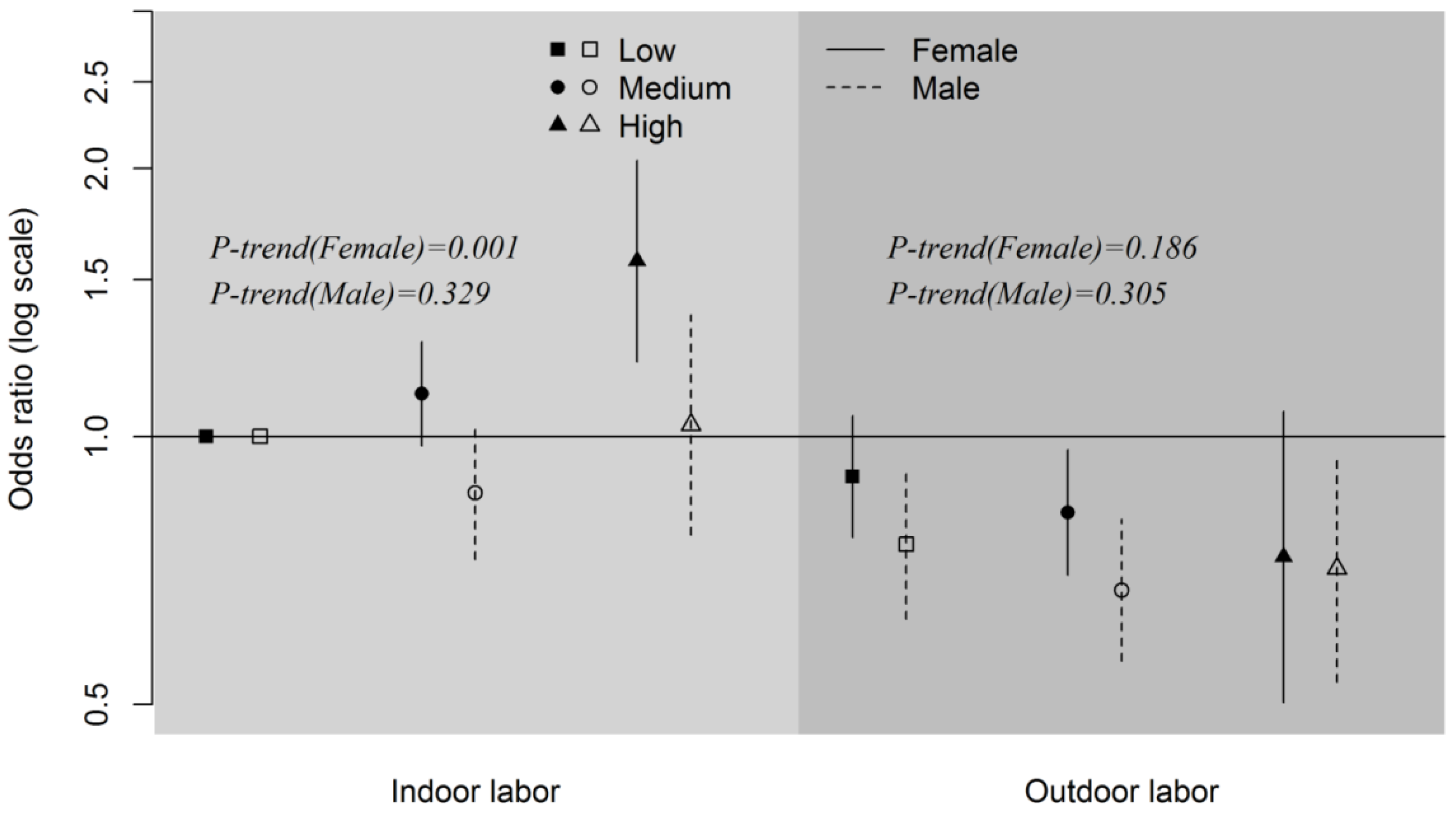
3.2. Interactions of Occupation and Self-Reported Salt-Eating Habit on Hypertension
| Occupation | Low | Medium | High | p-trend b | |||
|---|---|---|---|---|---|---|---|
| OR | 95% CI | OR | 95% CI | OR | 95% CI | ||
| Indoor labor | 1 | - | 1.01 | 0.91–1.13 | 1.32 | 1.09–1.59 | 0.048 |
| Outdoor labor | 0.84 | 0.75–0.95 | 0.75 | 0.67-0.85 | 0.75 | 0.60-0.94 | 0.091 |
4. Discussion
5. Limitations of the Study
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- He, J.; Gu, D.; Chen, J.; Wu, X.; Kelly, T.N.; Huang, J.F.; Chen, J.C.; Chen, C.S.; Bazzano, L.A.; Reynolds, K.; et al. Premature deaths attributable to blood pressure in China: A prospective cohort study. Lancet 2009, 374, 1765–1772. [Google Scholar] [CrossRef]
- Lawes, C.M.; Vander Hoorn, S.; Rodgers, A.; International Society of Hypertension. Global burden of blood-pressure-related disease, 2001. Lancet 2008, 371, 1513–1518. [Google Scholar] [CrossRef]
- He, J.; Gu, D.; Wu, X.; Reynolds, K.; Duan, X.; Yao, C.; Wang, J.; Chen, C.S.; Chen, J.; Wildman, R.P.; et al. Major causes of death among men and women in China. N. Engl. J. Med. 2005, 353, 1124–1134. [Google Scholar] [CrossRef] [PubMed]
- Kearney, P.M.; Whelton, M.; Reynolds, K.; Muntner, P.; Whelton, P.K.; He, J. Global burden of hypertension: Analysis of worldwide data. Lancet 2005, 365, 217–223. [Google Scholar] [CrossRef]
- Sheng, C.S.; Liu, M.; Kang, Y.Y.; Wei, F.F.; Zhang, L.; Li, G.L.; Dong, Q.; Huang, Q.F.; Li, Y.; Wang, J.G. Prevalence, awareness, treatment and control of hypertension in elderly Chinese. Hypertens. Res. 2013, 36, 824–828. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Deshmukh, P.R.; Garg, B.S. Prevalence and correlates of sustained hypertension in adolescents of rural Wardha, central India. Indian J. Pediatr. 2012, 79, 1206–1212. [Google Scholar] [CrossRef] [PubMed]
- Dalai, N.; Cui, H.; Yan, M.; Rile, G.; Li, S.; Su, X. Risk factors for the development of essential hypertension in a mongolian population of China: A case-control study. Genet. Mol. Res. 2014, 13, 3283–3291. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.R.; Kumanyika, S.K.; Cutler, J.A. Effect of change in sodium excretion on change in blood pressure corrected for measurement error. The trials of hypertension prevention, phase I. Am. J. Epidemiol. 1998, 148, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R., 3rd; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the dietary approaches to stop hypertension (Dash) diet. Dash-sodium collaborative research group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture; U.S. Department of Health and Human Services. Dietary Guidelines for Americans, 2010, 7th ed.U.S. Government Printing Office: Washington, DC, USA, 2010.
- Evert, A.B.; Boucher, J.L.; Cypress, M.; Dunbar, S.A.; Franz, M.J.; Mayer-Davis, E.J.; Neumiller, J.J.; Nwankwo, R.; Verdi, C.L.; Urbanski, P.; et al. Nutrition therapy recommendations for the management of adults with diabetes. Diabetes Care 2013, 36, 3821–3842. [Google Scholar] [CrossRef] [PubMed]
- Graudal, N.; Jurgens, G.; Baslund, B.; Alderman, M.H. Compared with usual sodium intake, low- and excessive-sodium diets are associated with increased mortality: A meta-analysis. Am. J. Hypertens. 2014, 27, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Whelton, P.K.; Appel, L.J. Sodium and cardiovascular disease: What the data show. Am. J. Hypertens. 2014, 27, 1143–1145. [Google Scholar] [CrossRef] [PubMed]
- Alderman, M.H.; Cohen, H.W. The iom report fails to detect evidence to support dietary sodium guidelines. Am. J. Hypertens. 2013, 26, 1198–1200. [Google Scholar] [CrossRef] [PubMed]
- Alderman, M.H.; Cohen, H.W. Dietary sodium intake and cardiovascular mortality: Controversy resolved? Curr. Hypertens. Rep. 2012, 14, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Graudal, N.A.; Hubeck-Graudal, T.; Jurgens, G. Effects of low sodium diet versus high sodium diet on blood pressure, renin, aldosterone, catecholamines, cholesterol, and triglyceride. Cochrane Database Syst. Rev. 2011, CD004022. [Google Scholar] [CrossRef]
- Alderman, M.H. Reducing dietary sodium: The case for caution. JAMA 2010, 303, 448–449. [Google Scholar] [CrossRef] [PubMed]
- Lelong, H.; Galan, P.; Kesse-Guyot, E.; Fezeu, L.; Hercberg, S.; Blacher, J. Relationship between nutrition and blood pressure: A cross-sectional analysis from the nutrinet-sante study, a french web-based cohort study. Am. J. Hypertens. 2015, 28, 362–371. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Yusuf, S.; Mente, A.; Gao, P.; Mann, J.F.; Teo, K.; McQueen, M.; Sleight, P.; Sharma, A.M.; Dans, A.; et al. Urinary sodium and potassium excretion and risk of cardiovascular events. JAMA 2011, 306, 2229–2238. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.M.; Flack, J.M. Salt sensitivity and hypertension in african Americans: Implications for cardiovascular nurses. Prog. Cardiovasc. Nurs. 2000, 15, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Haddy, F.J. Salt-sensitive hypertension. N. Engl. J. Med. 2002, 347, 448–449. [Google Scholar] [PubMed]
- Luft, F.C.; Grim, C.E.; Fineberg, N.; Weinberger, M.C. Effects of volume expansion and contraction in normotensive whites, blacks, and subjects of different ages. Circulation 1979, 59, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Wright, J.T., Jr.; Rahman, M.; Scarpa, A.; Fatholahi, M.; Griffin, V.; Jean-Baptiste, R.; Islam, M.; Eissa, M.; White, S.; Douglas, J.G. Determinants of salt sensitivity in black and white normotensive and hypertensive women. Hypertension 2003, 42, 1087–1092. [Google Scholar] [CrossRef] [PubMed]
- Bates, G.P.; Miller, V.S. Sweat rate and sodium loss during work in the heat. J. Occup. Med. Toxicol. 2008, 3, 4. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.W.; Bailey, A.P.; Tan, W.; Shparago, M.; Young, E. Long-term high salt diet causes hypertension and decreases renal expression of vascular endothelial growth factor in sprague-dawley rats. J. Am. Soc. Hypertens. 2008, 2, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Takase, H.; Sugiura, T.; Kimura, G.; Ohte, N.; Dohi, Y. Dietary sodium consumption predicts future blood pressure and incident hypertension in the Japanese normotensive general population. J. Am. Heart Assoc. 2015, 4, e001959. [Google Scholar] [CrossRef] [PubMed]
- Takachi, R.; Ishihara, J.; Iwasaki, M.; Ishii, Y.; Tsugane, S. Self-reported taste preference can be a proxy for daily sodium intake in middle-aged Japanese adults. J. Acad. Nutr. Diet. 2014, 114, 781–787. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Cho, H.J.; Bae, E.; Kim, Y.C.; Kim, S.; Chin, H.J. Not salt taste perception but self-reported salt eating habit predicts actual salt intake. J. Korean Med. Sci. 2014, 29, S91–S96. [Google Scholar] [CrossRef] [PubMed]
- Okura, Y.; Urban, L.H.; Mahoney, D.W.; Jacobsen, S.J.; Rodeheffer, R.J. Agreement between self-report questionnaires and medical record data was substantial for diabetes, hypertension, myocardial infarction and stroke but not for heart failure. J. Clin. Epidemiol. 2004, 57, 1096–1103. [Google Scholar] [CrossRef] [PubMed]
- Vargas, C.M.; Burt, V.L.; Gillum, R.F.; Pamuk, E.R. Validity of self-reported hypertension in the national health and nutrition examination survey III, 1988–1991. Prev. Med. 1997, 26, 678–685. [Google Scholar] [CrossRef] [PubMed]
- Wildman, R.P.; Gu, D.; Reynolds, K.; Duan, X.; He, J. Appropriate body mass index and waist circumference cutoffs for categorization of overweight and central adiposity among chinese adults. Am. J. Clin. Nutr. 2004, 80, 1129–1136. [Google Scholar] [PubMed]
- Thrift, A.G.; Evans, R.G.; Kalyanram, K.; Kartik, K.; Fitzgerald, S.M.; Srikanth, V. Gender-specific effects of caste and salt on hypertension in poverty: A population-based study. J. Hypertens. 2011, 29, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, T.; Suyama, A.; Igarashi, A.; Osaki, Y.; Okamoto, M.; Yamamoto, T.; Nanba, E.; Kurosawa, Y.; Fukumoto, S. Angiotensinogen gene variation and hypertension in a cohort study in Japanese. J. Epidemiol. 2001, 11, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Verwoert, G.C.; Hofland, J.; Amin, N.; Mattace-Raso, F.U.; Sijbrands, E.J.; Hofman, A.; van den Meiracker, A.H.; Uitterlinden, A.G.; van Duijn, C.M.; de Jong, F.H.; et al. Expression and gene variation studies deny association of human HSD3B1 gene with aldosterone production or blood pressure. Am. J. Hypertens. 2015, 28, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.W.; Hall, J.E. Racial and ethnic differences in blood pressure: Biology and sociology. Circulation 2006, 114, 2757–2759. [Google Scholar] [CrossRef] [PubMed]
- Naidu, B.R.; Ngeow, Y.F.; Pang, T. Ethnic distribution of chlamydophila pneumoniae antibodies in a Malaysian population and possible correlation with coronary heart disease. Eur. J. Epidemiol. 2003, 18, 135–137. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Gu, D.; Chen, J.; Jaquish, C.E.; Rao, D.C.; Hixson, J.E.; Chen, J.C.; Duan, X.; Huang, J.F.; Chen, C.S.; et al. Gender difference in blood pressure responses to dietary sodium intervention in the gensalt study. J. Hypertens. 2009, 27, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Wesseling, S.; Koeners, M.P.; Joles, J.A. Salt sensitivity of blood pressure: Developmental and sex-related effects. Am. J. Clin. Nutr. 2011, 94, 1928S–1932S. [Google Scholar] [CrossRef] [PubMed]
- Stachenfeld, N.S.; Taylor, H.S. Effects of estrogen and progesterone administration on extracellular fluid. J. Appl. Physiol. 2004, 96, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Stachenfeld, N.S.; DiPietro, L.; Palter, S.F.; Nadel, E.R. Estrogen influences osmotic secretion of AVP and body water balance in postmenopausal women. Am. J. Physiol. 1998, 274, R187–R195. [Google Scholar] [PubMed]
- Tamaki, J.; Kikuchi, Y.; Yoshita, K.; Takebayashi, T.; Chiba, N.; Tanaka, T.; Okamura, T.; Kasagi, F.; Minai, J.; Ueshima, H.; et al. Stages of change for salt intake and urinary salt excretion: Baseline results from the high-risk and population strategy for occupational health promotion (HIPOP-OHP) study. Hypertens. Res. 2004, 27, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Wiernik, E.; Nabi, H.; Pannier, B.; Czernichow, S.; Hanon, O.; Simon, T.; Simon, J.M.; Thomas, F.; Ducolombier, C.; Danchin, N.; et al. Perceived stress, sex and occupational status interact to increase the risk of future high blood pressure: The ipc cohort study. J. Hypertens. 2014, 32, 1979–1986, discussion 1986. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.; Kang, M.Y.; Paek, D.; Min, B.; Cho, S.I. Effect of long working hours on self-reported hypertension among middle-aged and older wage workers. Ann. Occup. Environ. Med. 2014, 26, 25. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Kim, T.H.; Lee, H.H.; Lee, S.H.; Wang, T. Postmenopausal hypertension and sodium sensitivity. J. Menopausal Med. 2014, 20, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Yanes, L.L.; Romero, D.G.; Iliescu, R.; Zhang, H.; Davis, D.; Reckelhoff, J.F. Postmenopausal hypertension: Role of the renin-angiotensin system. Hypertension 2010, 56, 359–363. [Google Scholar] [CrossRef] [PubMed]
- Matthews, K.A.; Abrams, B.; Crawford, S.; Miles, T.; Neer, R.; Powell, L.H.; Wesley, D. Body mass index in mid-life women: Relative influence of menopause, hormone use, and ethnicity. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Steiner, M.; Dunn, E.; Born, L. Hormones and mood: From menarche to menopause and beyond. J. Affect. Disord. 2003, 74, 67–83. [Google Scholar] [CrossRef]
- Lambert, E.; Dawood, T.; Straznicky, N.; Sari, C.; Schlaich, M.; Esler, M.; Lambert, G. Association between the sympathetic firing pattern and anxiety level in patients with the metabolic syndrome and elevated blood pressure. J. Hypertens. 2010, 28, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Laughlin, G.A.; Barrett-Connor, E.; Kritz-Silverstein, D.; von Muhlen, D. Hysterectomy, oophorectomy, and endogenous sex hormone levels in older women: The Rancho Bernardo study. J. Clin. Endocrinol. Metab. 2000, 85, 645–651. [Google Scholar] [PubMed]
- Coylewright, M.; Reckelhoff, J.F.; Ouyang, P. Menopause and hypertension: An age-old debate. Hypertension 2008, 51, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Dubey, R.K.; Oparil, S.; Imthurn, B.; Jackson, E.K. Sex hormones and hypertension. Cardiovasc. Res. 2002, 53, 688–708. [Google Scholar] [CrossRef]
- Karppanen, H. Minerals and blood pressure. Ann. Med. 1991, 23, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Fogari, R.; Zoppi, A.; Corradi, L.; Marasi, G.; Vanasia, A.; Zanchetti, A. Transient but not sustained blood pressure increments by occupational noise. An ambulatory blood pressure measurement study. J. Hypertension 2001, 19, 1021–1027. [Google Scholar] [CrossRef]
- Yoshimura, T. Occupational health. J. Epidemiol. 1996, 6, S115–S120. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Schnall, P.L.; Jauregui, M.; Su, T.C.; Baker, D. Work hours and self-reported hypertension among working people in California. Hypertension 2006, 48, 744–750. [Google Scholar] [CrossRef] [PubMed]
- Grotto, I.; Huerta, M.; Sharabi, Y. Hypertension and socioeconomic status. Curr. Opin. Cardiol. 2008, 23, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.B.; Sharma, J.P.; Rastogi, V.; Niaz, M.A.; Singh, N.K. Prevalence and determinants of hypertension in the Indian social class and heart survey. J. Hum. Hypertens. 1997, 11, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Ordunez, P.; Munoz, J.L.; Espinosa-Brito, A.; Silva, L.C.; Cooper, R.S. Ethnicity, education, and blood pressure in Cuba. Am. J. Epidemiol. 2005, 162, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Fernald, L.C.; Adler, N.E. Blood pressure and socioeconomic status in low-income women in Mexico: A reverse gradient? J. Epidemiol. Community Health 2008, 62, e8. [Google Scholar] [CrossRef] [PubMed]
- Mendez, M.A.; Cooper, R.; Wilks, R.; Luke, A.; Forrester, T. Income, education, and blood pressure in adults in Jamaica, a middle-income developing country. Int. J. Epidemiol. 2003, 32, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.A.; Appel, L.J.; Okuda, N.; Brown, I.J.; Chan, Q.; Zhao, L.; Ueshima, H.; Kesteloot, H.; Miura, K.; Curb, J.D.; et al. Dietary sources of sodium in China, Japan, the United Kingdom, and the United States, women and men aged 40 to 59 years: The intermap study. J. Am. Diet. Assoc. 2010, 110, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, X.L.; Seo, D.C.; Xu, A.Q.; Xun, P.C.; Ma, J.X.; Shi, X.M.; Li, N.; Yan, L.X.; Li, Y.; et al. Inaccuracy of self-reported low sodium diet among Chinese: Findings from baseline survey for Shandong & Ministry of Health Action on Salt and Hypertension (SMASH) project. Biomed. Environ. Sci. 2015, 28, 161–167. [Google Scholar] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, M.; Chen, W.; Teng, B.; Fang, Y. Occupational Disparities in the Association between Self-Reported Salt-Eating Habit and Hypertension in Older Adults in Xiamen, China. Int. J. Environ. Res. Public Health 2016, 13, 148. https://doi.org/10.3390/ijerph13010148
Yuan M, Chen W, Teng B, Fang Y. Occupational Disparities in the Association between Self-Reported Salt-Eating Habit and Hypertension in Older Adults in Xiamen, China. International Journal of Environmental Research and Public Health. 2016; 13(1):148. https://doi.org/10.3390/ijerph13010148
Chicago/Turabian StyleYuan, Manqiong, Wei Chen, Bogang Teng, and Ya Fang. 2016. "Occupational Disparities in the Association between Self-Reported Salt-Eating Habit and Hypertension in Older Adults in Xiamen, China" International Journal of Environmental Research and Public Health 13, no. 1: 148. https://doi.org/10.3390/ijerph13010148
APA StyleYuan, M., Chen, W., Teng, B., & Fang, Y. (2016). Occupational Disparities in the Association between Self-Reported Salt-Eating Habit and Hypertension in Older Adults in Xiamen, China. International Journal of Environmental Research and Public Health, 13(1), 148. https://doi.org/10.3390/ijerph13010148